Abstract
Mesenchymal stem cells (MSCs) are multipotent cells characterized by immunomodulatory properties and are therefore considered a promising tool for the treatment of autoimmune diseases. One functional B-cell subset, regulatory B cells (Bregs), has recently been shown to restrain excessive inflammatory responses in autoimmune diseases. In the present study, we investigated the impact of human adipose-derived MSCs on Bregs and their therapeutic effect in an animal model of systemic lupus erythematosus (SLE). Coculture of human adipose-derived MSCs with splenocytes from C57BL/6 mice expanded the population of interleukin-10-producing B cells (B10 B cells). In vivo treatment with human adipose-derived MSCs reduced serum anti-double-stranded antibody levels and improved renal pathology of lupus mice (Roquinsan/san mice). MSCs decreased ICOS+CD44+ follicular helper T cells, Th1 cells and Th17 cells, in spleens of Roquinsan/san mice. In contrast, MSCs increased Foxp3-expressing regulatory T cells. MSCs also decreased the size and number of germinal centers and effector B cells. As expected, in vivo treatment with MSCs expanded the population of Bregs in spleens of Roquinsan/san mice. Our results indicate that human adipose-derived MSCs induce the expansion of Bregs and ameliorate autoimmunity in a murine model of SLE. These findings suggest that human adipose-derived MSCs may be a promising therapeutic strategy targeting B-cell-mediated autoimmune diseases such as SLE.
Keywords
Introduction
B Cells are a major cell type involved in adaptive immune responses, as they are specialized in antibody production. Therefore, they are the central contributors to the development of certain autoimmune diseases, such as systemic lupus erythematosus (SLE). In addition to antibody production, B cells perform a variety of immunological functions, including antigen presentation and the production of multiple cytokines. One functional B-cell subset, regulatory B cells (Bregs), has been identified recently. Bregs contribute to the maintenance of the fine equilibrium required for tolerance. Moreover, they restrain excessive inflammatory responses that occur in autoimmune diseases (20,21). The central player of Bregs function is interleukin (IL)-10, which inhibits the production of proinflammatory cytokines and supports regulatory T-cell (Treg) differentiation (3).
Mesenchymal stem cells (MSCs) are multipotent stromal cells that can differentiate into a variety of cell types, including osteoblasts, chondrocytes, and adipocytes under controlled culture conditions (12,23). They are characterized by their adherence to plastic, the expression of surface markers, such as CD73, CD90, and CD105, and the absence of typical hematopoietic markers, such as CD45 and CD11b. It is well known that MSCs are hypoimmunogenic due to low expression of HLA and costimulatory molecules. In addition, they exert potent immunosuppressive actions (13) and thus have been considered a new therapeutic option for the treatment of autoimmune diseases like SLE and rheumatoid arthritis (RA) (2,8).
Given the immunomodulatory properties of MSCs, their effects on a variety of immune cells, including T cells (9), dendritic cells (27), and natural killer (NK) cells (28), have been studied widely. However, the interaction between MSCs and B cells, particularly Bregs, remains to be determined.
In this study, we investigated the impact of human adipose-derived MSCs on Bregs from mice in vitro. We also examined whether in vivo treatment with human adipose-derived MSCs has therapeutic potential in an animal model of SLE, Roquinsan/san mice, in which the sanroque mutation disrupts a repressor of ICOS, an essential costimulatory molecule for follicular helper T cells (Tfh cells), resulting in excessive formation of Tfh cells and germinal centers (GC) (17,31). We finally determined the effect of in vivo treatment with human adipose-derived MSCs on the populations of various T cells including Th1 cells, Th17 cells, Tfh cells, and regulatory T-cell and B-cell subsets, including Bregs, follicular B (FOB) cells, marginal zone B (MZB) cells, and plasma cells in Roquinsan/san mice.
Materials and Methods
Mice
Female Roquinsan/san C57BL/6 mice between 15 and 20 weeks of age were purchased from the Jackson Laboratory (Bar Harbor, ME, USA) and subsequently bred under specific pathogen-free conditions in an animal facility with controlled humidity (55 ± 5%), light (12 h/12 h light/dark), and temperature (22 ± 1°C). The air in the facility was passed through a HEPA filter system designed to exclude bacteria and viruses. Animals were fed mouse chow and tap water ad libitum. The protocols used in this study were approved by the Animal Care and Use Committee of The Catholic University of Korea.
Isolation and Culture of MSCs
Mesenchymal stem cells used in this study were derived from a surplus of frozen cell stock for stem cell banking (RNLBio, Seoul, Korea) and no longer needed for that purpose. Fat samples were obtained from donors with written informed consent for research use. In brief, human abdominal subcutaneous fat tissues were obtained by simple liposuction from two male donors, 59 and 77 years old. MSCs were isolated from the fat stromal vascular fraction (SVF) by their adherence to plastic and were culture expanded. Cryopreserved stem cells, stored in liquid nitrogen vapor, were thawed and recultured in growth medium (RKCM; RNLBio) based on the injection schedule under Good Manufacturing Practice conditions (RNLBio). Harvested cells were anonymized and transported to the preclinical investigator under cold storage conditions (4,24).
Cell Cultures
Splenocytes isolated from C57BL/6 (wild type) or Roquinsan/san mice were cultured in RPMI-1640 medium (Gibco, Grand Island, NY, USA) containing 20 mM HEPES (Gibco), 2 mM L-glutamine (Gibco), 5% heat-inactivated FBS (Gibco), 100 mM sodium pyruvate (Gibco), and 1% antibiotics [penicillin (100 U/ml)-streptomycin (100 mg/ml); Gibco] alone or with irradiated human adiposederived MSCs (20:1, 5:1, and 1:1 ratios, respectively) in the presence or absence of 10 μg/ml lipopolysaccharide (LPS; Sigma-Aldrich St. Louis, MO, USA) for 3 days. MSCs were irradiated (2,000 cGy) using a 137Cs source (Gammacell® 3000; MDS Nordion, Ottawa, ON, Canada) to inhibit mitosis.
Stem Cell Injection
Mice received lateral tail vein injections of 1 × 106 MSCs in 0.2 ml phosphate-buffered saline (PBS; Gibco). The negative control group received PBS instead of cells.
FACS Analysis
Mononuclear cells were immunostained with various combinations of the following fluorescence-conjugated antibodies (0.2 μg/106 cells): CD19, CD1d, CD5, CD21, CD23, CD4, CD25, CD44, CD95, CD138, GL-7, ICOS, and B220 (BD Biosciences, San Diego, CA, USA). These cells were also intracellularly stained with the following antibodies (0.25 μg/106 cells): IL-4 (BD Biosciences), IL-10 (Biolegend, San Diego, CA, USA), IL-17 (eBioscience, San Diego, CA, USA), and Foxp3 (eBioscience). Before intracellular staining, the cells were restimulated for 4 h with 25 ng/ml PMA (Sigma-Aldrich) and 250 ng/ml ionomycin (Sigma-Aldrich) in the presence of GolgiSTOP (BD Biosciences). Intracellular staining was conducted using an intracellular staining kit (eBioscience) according to the manufacturer's protocol. Flow cytometry was performed using a FACSCalibur cytometer (BD Biosciences).
Assessment of Histopathology Scoring of Nephritis
Kidneys were harvested and fixed in 10% neutral-buffered formalin (Sigma-Aldrich). Formalin-fixed tissues were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin (Sigma-Aldrich), eosin (Muto Pure Chemical Co., Ltd., Tokyo, Japan), and/or Periodic acid–Schiff (PAS; Sigma-Aldrich) for histological examination. The severity of the nephritis score was graded on a 0-4 scale as follows: 0, normal; 1, mild increase in mesangial cellularity and matrix; 2, moderate increase in mesangial cellularity and matrix with thickening of the glomerular basement membrane (GBM); 3, focal endocapillary hypercellularity with obliteration of capillary lumina and a substantial increase in the thickness and irregularity of the GBM; and 4, diffuse endocapillary hypercellularity, segmental necrosis, crescents, and hyalinized end-stage glomeruli (17).
Measurement of IgG Titers and Anti-dsDNA Antibodies
Serum levels of IgG and IgG1 antibodies were measured using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (Bethyl Laboratories, Montgomery, TX, USA). The IgG anti-dsDNA antibody was measured by ELISA using serum samples diluted at 1:500 and alkaline phosphatase (AP)-conjugated goat antimouse IgG, secondary antibody at a 1:1,000 dilution.
Confocal Staining
Spleen tissue was collected, snap frozen in liquid nitrogen, and stored at −80°C. Tissue cryosections (7 μm thick) were fixed in 4% paraformaldehyde (Sigma-Aldrich) and stained using PE-labeled anti-IFN-r, IL-4, IL-17, or Foxp3 antibody (1:100 dilution; eBioscience) or FITC-labeled anti-CD4 antibody (1:200 dilution; Biolegend). After overnight incubation at 4°C, the stained sections were analyzed using a confocal microscopy system (LSM 510 Meta; Zeiss, Gottingen, Germany).
Statistical Analysis
Statistical analyses were performed using SAS ver. 9 software (SAS Institute, Cary, NC, USA). The experimental values are presented as means ± standard deviations (SD). Data were analyzed by one-way ANOVA using Tukey's post hoc test in multiple groups and by a two-tailed unpaired Student's t-test in cases in which two groups were compared. Values of p < 0.05 were considered significant.
Results
Human Adipose-Derived MSCs Expand IL-10-Producing B Cells In Vitro
We first examined whether human adipose-derived MSCs had an effect on the population of Bregs mice in vitro. Splenocytes isolated from C57BL/6 mice were cultured in medium alone or graded doses of irradiated human adipose-derived MSCs in the presence or absence of LPS for 72 h. IL-10-producing B cells, which were considered to be Bregs, were identified by flow cytometry. As shown in Figure 1A, in vitro treatment with human adipose-derived MSCs expanded the IL-10-producing B cells in a dose-dependent manner. Cotreatment with LPS augmented the Breg-expanding effect of human adipose-derived MSCs. The percentage of IL-10-producing cells in B cells was also increased by human adipose-derived MSCs (Fig. 1B).

Human adipose-derived mesenchymal stem cells (MSCs) expand IL-10-producing B cells in a dose-dependent manner in vitro. (A) Splenocytes (1 × 106) isolated from C57BL/6 mice were cultured in medium alone or with irradiated human adipose-derived MSCs (20:1, 5:1, and 1:1 ratio, respectively) in the presence or absence of LPS for 72 h. IL-10-producing B cells (IL-10-producing CD19+ cells) were identified by flow cytometry as described in the Materials and Methods section. The representative result is shown. (B) The percentage of IL-10-producing cells in CD19+ B cells is shown. Data are expressed as the mean ± SD. *p < 0.05. (C) The population of CD1dhiCD5+ B cells obtained splenocytes from Roquinsan/san mice is shown in the left panel. Almost all B10 B cells expressed CD19, but only a small proportion of B10 B cells produced IL-10 without LPS stimulation (right panel). The representative results are shown. (D) Splenocytes (1 × 106) isolated from Roquinsan/san mice were cultured in medium alone or with irradiated human adipose-derived MSCs (20:1, 5:1, and 1:1 ratio, respectively) for 72 h. The percentage of CD1dhiCD5+ B cells in CD19+ B cells was determined by flow cytometry. *p < 0.05. (E) Splenocytes isolated from Roquinsan/san mice were cultured in medium alone or with irradiated human adipose-derived MSCs (20:1, 5:1, and 1:1 ratio, respectively) in the presence or absence of LPS for 72 h. IL-10-producing CD1dhiCD5+ B cells were determined by flow cytometry as described in the Materials and Methods section. The representative result is shown in the left panel. The percentage of IL-10-producing B10 B cells is shown in the right panel. Data are expressed as the mean ± SD. *p < 0.05.
Human Adipose-Derived MSCs Expand the Population of CD1dhiCD5+ B Cells In Vitro
Among various Breg phenotypes, CD1dhiCD5+ B cells are the most well-characterized Bregs in mice (21,32). Therefore, we investigated the effect of human adipose-derived MSCs on CD1dhiCD5+ B cells from Roquinsan/san mice in vitro. As shown in Figure 1C, the proportion of CD1dhiCD5+ B cells among CD19+ B cells from Roquinsan/san mice was small without stimulation, and a minority of CD1dhiCD5+ B cells produced IL-10. In vitro treatment with human adipose-derived MSCs increased the percentage of CD1dhiCD5+ B cells (Fig. 1D).
We also examined the effect of human adipose-derived MSCs on IL-10-producing CD1dhiCD5+ B cells in vitro. Splenocytes isolated from Roquinsan/san mice were cultured in medium alone or graded doses of irradiated human adipose-derived MSCs in the presence or absence of LPS for 72 h. The population of IL-10-producing CD1dhiCD5+ B cells was analyzed. The results showed that human adipose-derived MSCs expanded IL-10-producing CD1dhiCD5+ B cells in a dose-dependent manner in vitro (Fig. 1E).
Effect of In Vivo Cell Therapy with Human Adipose-Derived MSCs on Renal Histopathology and Autoantibody Production
We next tested whether in vivo treatment with human adipose-derived MSCs might have beneficial effects on renal histopathology and autoantibody production in the animal model of SLE (Roquinsan/san mice). Beginning at age 17 weeks, we initiated a therapeutic protocol consisting of intravenous infusion of 1 × 106 human adipose-derived MSCs or an equal volume of PBS, which was administered once weekly for 5 weeks. The following parameters were evaluated: histopathology of the kidney, serum levels of immunoglobulins (IgG, IgG1, IgM, and IgE), and IgG anti-dsDNA. The histopathologic analysis of the kidney revealed a significant improvement in mesangial proliferation and interstitial inflammatory cell infiltration in mice treated with human adipose-derived MSCs (Fig. 2A, B). Moreover, serum concentrations of immunoglobulins (IgG, IgG1, IgM, and IgE) and IgG anti-dsDNA decreased markedly following treatment with human adipose-derived MSCs (Fig. 2C, D). These results suggest that in vivo treatment with human adipose-derived MSCs ameliorate the lupus-like phenotype of Roquinsan/san mice.

In vivo treatment with human adipose-derived MSCs ameliorates the lupus-like phenotype of Roquinsan/san mice. (A) Representative images of kidney sections stained with hematoxylin and eosin (H&E) and Periodic acid–Schiff (PAS) from Roquinsan/san mice treated intravenously with 1 × 106 human adipose-derived MSCs once weekly for 5 weeks or phosphate-buffered saline (PBS). Scale bar: 50 μm. (B) Scores of nephritis severity in Roquinsan/san mice that received saline or MSCs. Data are represented as mean ± SD in bar graphs. (C and D) Serum levels of IgG, IgG1, IgM, IgE, and IgG anti-double-stranded DNA (anti-dsDNA) antibodies in Roquinsan/san mice treated with human adipose-derived MSCs or PBS, as determined by ELISA. The combined data from two independent experiments (n = 6 per group) are shown. Data are expressed as the mean ± SD. *p < 0.05.
In Vivo Treatment with Human Adipose-Derived MSCs Decreases Th1, Th17, and Tfh Cells but Increases Tregs in Roquinsan/san Mice
We also tested the effect of in vivo cell therapy with human adipose-derived MSCs on histopathology of the spleen in Roquinsan/san mice. We found that treatment with human adipose-derived MSCs significantly decreased the number of lymphoid follicles in spleens compared with those treated with PBS (Fig. 3A).
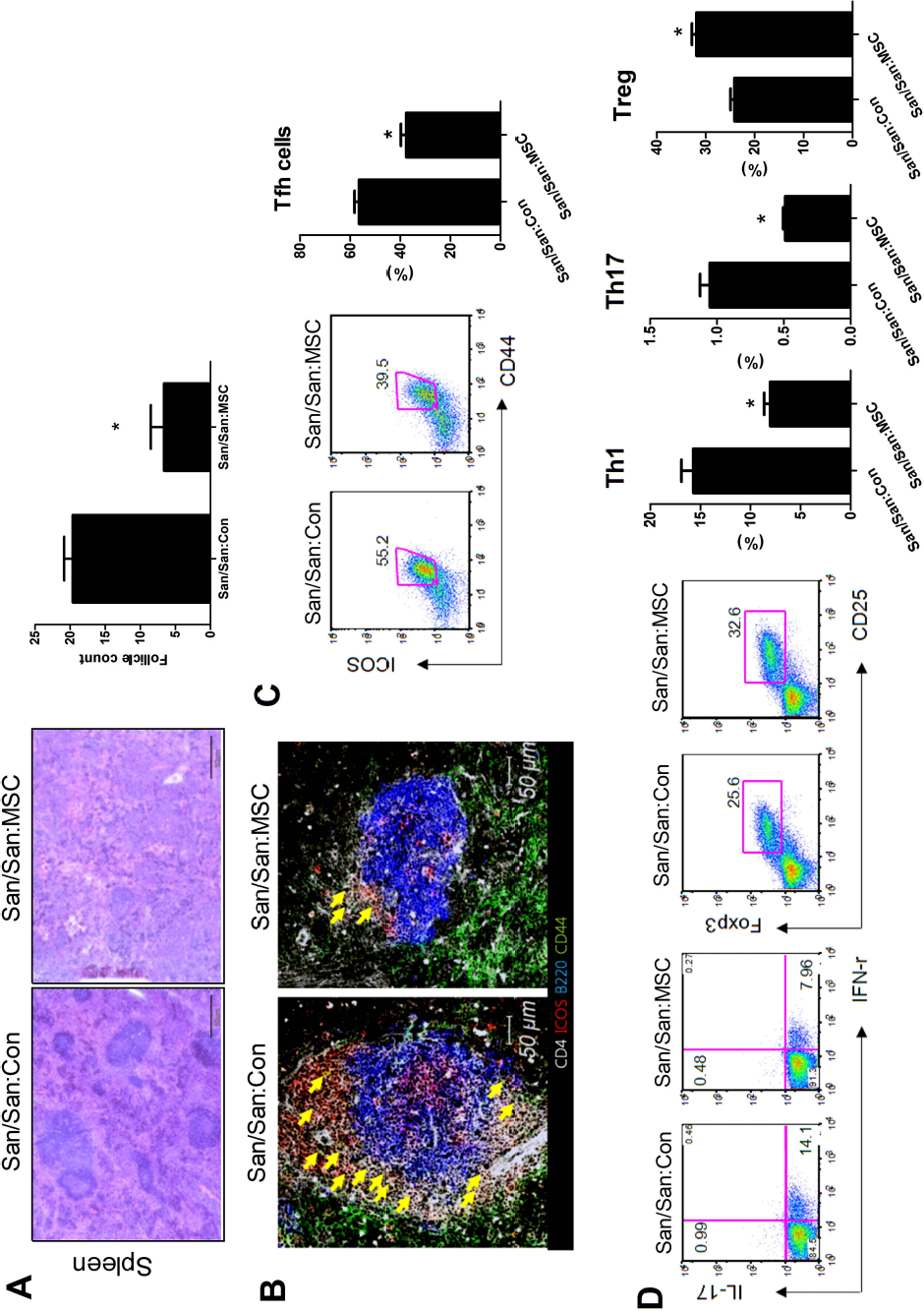
Human adipose-derived MSCs decrease Th1, Th17, and Tfh cells but increase regulatory T cells in Roquinsan/san mice. (A) Roquinsan/san mice (n = 6) were injected intravenously with 1 × 106 hMSCs every week for 5 weeks. Disease control mice (n = 6) received intravenous injections of an equal volume of PBS at the same time points. Representative images of spleen sections stained with H&E from each group of mice. Scale bar: 50 μm. Bar graph indicates the number of lymphoid follicles of spleens from each group of mice. The lymphoid follicles were enumerated visually at higher magnification (projected on a screen) by four individuals, and the mean values are presented in the form of histogram. (B) Representative confocal microscopic images of spleen sections from Roquinsan/san mice treated with human adipose-derived MSCs or PBS. Cells were stained with fluorescence-tagged antibodies to identify CD4 (white), ICOS (red), B220 (blue), and CD44 (green). CD44+ICOS+CD4+ cells, which are considered Tfh cells, are densely located near the B-cell zone (yellow arrows), especially in the spleen of the PBS-treated mice. Roquinsan/san mice treated with human adipose-derived MSCs showed markedly decreased size of the germinal center. (C) Representative flow cytometric contour plots and graphical analysis of CD4+ICOS+CD44+ T cells (follicular helper T cells, Tfh cells). Bar graph indicates the percentage of Tfh cells in CD4+ T cells in spleens from each group of mice (right panel). *p < 0.05. (D) Representative flow cytometric contour plots and graphical analysis of CD4+IFN-γ T cells (Th1 cells), CD4+IL-17+ T cells (Th17 cells), and CD4+CD25+Foxp3+ T cells (Tregs) (left panel). Bar graphs indicate the percentage of Th1 cells, Th17 cells, and Tregs in CD4+ T cells in spleens from each group of mice (right panel). The combined data from two independent experiments (n = 6 per group) are shown. *p < 0.05.
We analyzed the population of Th1 cells, Th17 cells, Tregs, and Tfh cells in spleens from mice treated with human adipose-derived MSCs or PBS by flow cytometry to determine the effect of in vivo cell therapy with human adipose-derived MSCs on various effector T helper cell subsets. Tfh cells, a recently discovered effector T helper cell subset, are deeply implicated in GC formation, which is an essential step in the humoral immune response (6,30).
A confocal microscopic analysis of spleen tissues revealed that mice treated with human adipose-derived MSCs showed markedly decreased GC size compared with mice treated with PBS (Fig. 3B). The population of CD4+ICOS+CD44+ T cells, which are considered Tfh cells, decreased profoundly in mice treated with human adipose-derived MSCs (Fig. 3C). The population of CD4+IFN-γ T cells (Th1 cells) and CD4+ IL-17+ T cells (Th17 cells) also decreased in mice treated with human adipose-derived MSCs. In contrast, spleen tissues from mice treated with human adipose-derived MSCs showed increased numbers of Foxp3+ Tregs, which suppress autoimmune processes and maintain peripheral tolerance (22), compared with that of mice treated with PBS (Fig. 3D).
Effect of In Vivo Cell Therapy with Human Adipose-Derived MSCs on B-cell Subsets in Roquinmn/san Mice
We investigated the effect of in vivo cell therapy with human adipose-derived MSCs on various B-cell subsets in Roquinsan/san mice. Confocal microscopic analysis of spleen tissues revealed that mice treated with human adipose-derived MSCs showed markedly decreased expression of GL-7, the major surface marker of GC B cells (Fig. 4A). The population of GC B cells (GL-7+ CD95+ B220+ B cells) decreased markedly in mice treated with human adipose-derived MSCs (Fig. 4B).

Human adipose-derived MSCs decrease germinal center (GC) B cells and plasma cells but increase Breg cells in Roquinsan/san mice. (A) Representative confocal microscopic images of spleen sections from Roquinsan/san mice treated with human adipose-derived MSCs or PBS. Cells were stained with fluorescence-tagged antibodies to identify GL-7 (green), CD138 (red), B220 (blue), and CD4 (white). Roquinsan/san mice treated with human adipose-derived MSCs showed markedly decreased numbers of GL-7+ B cells. (B) Flow cytometric analysis shows the proportion of GL-7+CD95+B220+ B cells (germinal center B cells, GC B cells) in the spleen tissues from each group of mice. The percentage of GC B cells in B220+ B cells from each group of mice is shown in the right panel. *p < 0.05. (C) Representative flow cytometric contour plots and graphical analysis of CD1dhiCD5+ B cells (B10 B cells). Bar graphs indicate the percentage of CD1dhiCD5+ B cells in CD19+ B cells of spleen from each group of mice (upper panel). IL-10-producing CD1dhiCD5+ B cells were determined by flow cytometry as described in the Materials and Methods section. Bar graphs indicate the percentage of IL-10-producing CD1dhiCD5+ B cells in CD19+ B cells in spleens from each group of mice (lower panel). *p < 0.05. (D) Representative flow cytometric contour plots and graphical analysis of B220+CD23highCD21low B cells (follicular B cells, FOB cells) and B220+CD23lowCD21high B cells (marginal zone B cells, MZB cells) from each group of mice are shown in the upper panel. Representative flow cytometric contour plots and graphical analysis of plasma cells (B220-CD138+IgD- cells) from each group of mice are shown in the lower panel. The combined data from two independent experiments (n = 6 per group) are shown.*p < 0.05.
To our surprise, spleen tissues from mice treated with human adipose-derived MSCs showed increased numbers of both CD1dhiCD5+ B cells and IL-10-producing CD1dhiCD5+ B cells compared with those of mice treated with PBS (Fig. 4C), suggesting that treatment with human adipose-derived MSCs expands the population of Bregs from mice both in vitro and in vivo.
We also analyzed the other B-cell subsets, including B220+CD23highCD21low B-cells (FOB cells), B220+ CD23lowCD21high B-cells (MZB cells), and plasma cells (B220-CD138+IgD- cells). The results revealed that mice treated with human adipose-derived MSCs had decreased numbers of FOB cells and plasma cells and increased numbers of MZB cells (Fig. 4D).
Discussion
The aim of the present study was to investigate the impact of human adipose-derived MSCs on the Bregs from mice and to examine whether in vivo treatment with human adipose-derived MSCs had therapeutic effects in an animal model of SLE, the prototype of B-cell-mediated autoimmune disease.
It is well known that MSCs modulate several effector immune functions by interacting both with innate and adaptive immune cells. Therefore, cell therapy utilizing ex vivo expanded MSCs may be an interesting approach for the treatment of autoimmune diseases (16,19). Bone marrow MSCs are a standard in the field of adult stem cell biology and clinical application. However, human adipose-derived MSCs, due to their minimal invasive accessibility and availability in the body, are becoming an attractive option for treating a variety of diseases (16).
Not many studies have investigated the effects of MSCs on B-cell proliferation, differentiation, and function (1,5,25). Unfortunately, the results are still controversial in contrast to the established immunosuppressive effects of MSCs on T cells (9). Moreover, the effects of MSCs on Bregs, a recently identified effector B-cell subset that inhibits the excessive inflammatory responses during the development of autoimmune diseases, remain undetermined. In this study, we demonstrated the effect of MSCs on Bregs for the first time. In vitro treatment with human adipose-derived MSCs expanded B10 B cells from mice (Fig. 1). Since the identification of Bregs, IL-10 has been regarded as a key trait in this population. Unfortunately, there is no unique surface marker that exclusively characterizes Bregs. Among various Bregs, CD1dhiCD5+ B cells are the most well-known Bregs in mice (32). Thus, we also showed that human adipose-derived MSCs increase not only B10 B cells but also IL-10-producing Bregs from the mice (Fig. 1). It is well known that in vitro treatment with LPS alone strongly induces the expansion of B10 B cells (33). In our study, LPS alone expanded the B10 B cells like previous reports. In addition, cotreatment with hMSCs augmented the Breg-expanding effect of LPS (Fig. 1).
SLE is a systemic autoimmune disease of unknown etiology, characterized by the production of diverse auto-antibodies and end organ damage from immune complex deposit. Based on the well-known immunomodulatory properties of MSCs, it can be speculated that cell therapy with MSCs might be a promising therapeutic strategy for treating SLE. However, discordant results exist regarding the therapeutic effects of MSCs on SLE in animal models (4,10,11,26,34,35). These discrepancies between studies might be partially explained by different sources of MSCs (bone marrow, umbilical cord, and adipose) or transplantation type (allo or xeno). Therefore, only a few clinical studies have been conducted to investigate the therapeutic effect of allogenic MSC transplantation in patients with severe and refractory SLE (15,29).
In our study, in vivo treatment with human adipose-derived MSCs improved renal pathology of Roquinsan/san mice, an animal model of SLE (Fig. 2A, B). It decreased serum levels of immunoglobulins (IgG, IgG1, IgM, and IgE) and autoantibodies (anti-dsDNA antibodies) (Fig. 2C, D). These findings suggest that human adipose-derived MSCs ameliorate the autoimmune phenotype of Roquinsan/san mice. The results of the present study are consistent with a recent report showing that serial human adipose-derived MSC transplantation has beneficial effects on the (NZB x NZW)F1 mice, an animal model of SLE different from Roquinsan/san mice (4).
Germinal centers (GCs) are sites within secondary lymphoid organs where mature B-lymphocytes proliferate, differentiate, and mutate their antibodies through somatic hypermutation and switch the class of their antibodies during the immune response, indicating GCs are the key part of the B-cell humoral immune response. Our results showed that in vivo treatment with human adipose-derived MSCs decreased the size and number of GCs in Roquinsan/san mice, characterized by excessive GC formation (Fig. 3B).
Many investigations have been conducted regarding the effects of MSCs on effector T-cell subsets (7,9). Our results showed that in vivo treatment with human adipose-derived MSCs decreased the numbers of Th1 cells and Th17 cells, which are proinflammatory T helper cell subsets that have been deeply implicated in the development of autoimmune diseases (18) but increased Foxp3-expressing Tregs, which suppress autoimmune processes and maintain peripheral tolerance (22) (Fig. 3D) in Roquinsan/san mice. These findings correspond well with those found in earlier studies with regard to the effects of MSCs on effector T cells (9). Additionally, we demonstrated that cell therapy with human adipose-derived MSCs decreased Tfh cells, which are a newly discovered effector T helper cell subset that is deeply implicated in GC formation, for the first time.
Finally, we evaluated the in vivo effect of human adipose-derived MSCs on Bregs and other B-cell subsets in Roquinsan/san mice. We showed that in vivo treatment with human adipose-derived MSCs increased not only B10 B cells but also IL-10-producing Bregs (Fig. 4C), verifying that human adipose-derived MSCs expand the population of Bregs from mice both in vitro and in vivo. The populations of marginal zone B cells, which are known to include B10 B cells with a regulatory phenotype (14), were also expanded by human adipose-derived MSCs. In constrast, in vivo treatment with human adipose-derived MSCs decreased GC B cells, FOB cells, and plasma cells, which are effector B-cell subsets deeply implicated in antibody production (Fig. 4D).
In summary, we demonstrated that human adipose-derived MSCs induced the expansion of regulatory B cells and ameliorated autoimmunity in a murine model of SLE. These findings suggest that human adipose-derived MSCs may be a promising therapeutic strategy targeting B-cell-mediated autoimmune diseases, such as SLE.
Footnotes
Acknowledgments
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MEST) (grant No. 2012M3A9C6049783). The authors declare no conflict of interest.
