Abstract
Intrauterine inflammation is a significant cause of injury to the developing fetal brain. Using a preterm fetal sheep model of in utero infection, we asked whether human amnion epithelial cells (hAECs) were able to reduce inflammation-induced fetal brain injury. Surgery was undertaken on pregnant sheep at ~105 days gestation (term is 147 days) for implantation of vascular catheters. Lipopolysaccharide (LPS; 150 ng/kg bolus) or saline was administered IV at 109, 110, and 111 days. Sixty million fluorescent-labeled hAECs were administered at 110, 111, and 112 days gestation via the brachial artery catheter. Brains were collected at 114 days for histological assessment. hAECs were observed within the cortex, white matter, and hippocampus. Compared to control lambs, LPS administration was associated with significant and widespread fetal brain inflammation and injury as evidenced by increased number of activated microglia in the periventricular white matter (p = 0.02), increased pyknosis, cell degeneration (p = 0.01), and a nonsignificant trend of fewer oligodendrocytes in the subcortical and periventricular white matter. Administration of hAECs to LPS-treated animals was associated with a significant mitigation in both inflammation and injury as evidenced by fewer activated microglia (p = 0.03) and pyknotic cells (p = 0.03), significantly more oligodendrocytes in the subcortical and periventricular white matter (p = 0.01 and 0.02, respectively), and more myelin basic protein-positive cells within the periventricular white matter (p = 0.02). hAEC administration to fetal sheep exposed to multiple doses of LPS dampens the resultant fetal inflammatory response and mitigates associated brain injury.
Keywords
Introduction
Intrauterine infection, such as chorioamnionitis, is a leading cause of preterm birth 1 and subsequent neurological morbidity and cerebral palsy2,3. In particular, cerebral white matter abnormalities, including lesions to the periventricular white matter (PVWM) known as periventricular leukomalacia (PVL), are common sequelae in those preterm infants who survive severe chorioamnionitis. PVL includes both focal and diffuse necrosis, gliosis, and, in severe cases, cystic lesions in the white matter adjacent to the lateral ventricles 4 . A significant proportion of preterm babies with this form of white matter injury subsequently develop cerebral palsy 5 .
There are complex interactions between intrauterine infection, preterm birth, and white matter brain injury that are dependent on the timing of injury in pregnancy, the type of insult, and the duration and severity. In particular, disturbances to normal oligodendrocyte glial cell lineage development are central to any resulting preterm white matter injury and PVL 6 . Mature oligodendrocytes ensheath axons with myelin. Injury to these cells can irreversibly impair myelination and white matter development. Oligodendrocyte precursors are predominate in the human brain between 24 and 32 weeks gestation. Infection at this stage of pregnancy is associated with apparently increased oligodendrocyte precursor vulnerability to injury 7 . The precise mechanisms that contribute to oligodendrocyte precursor cell injury are not fully understood. However, proinflammatory cytokines, upregulated as part of the fetal inflammatory response, are likely culprits 8 . Cytokines can directly induce oligodendrocyte cell death 9 . Cytokines also mediate indirect effects on the developing brain such as increasing the permeability of the blood-brain barrier (BBB), allowing the increased influx of circulating inflammatory cells that in turn activate microglia, and astrocyte proliferation and the further production of other cytokines 10 . There is no current treatment that might interrupt this cascade of injury.
Human amnion epithelial cells (hAECs) are stem-like cells obtained from the amniotic membrane. They possess a number of important properties that make them of interest as a candidate in neuroprotective therapy. They have low immunogenicity 11 and can prevent the activation of both innate and adaptive immune systems12,13. hAECs suppress proinflammatory cytokines 14 , regulate macrophage recruitment and function 15 , and secrete factors that inhibit the chemotactic activity of neutrophils and macrophages 12 . In vitro, they are neuroprotective in an adult model of ischemic and oxidative stress injury 16 and have the ability to differentiate into neurons 17 . In vivo, we have also recently shown that hAECs decrease gene expression of proinflammatory cytokines and reduce inflammatory cell infiltration in both mouse and sheep models of acute lung injury15,18,19.
Accordingly, in this study we aimed to examine whether hAECs are able to protect oligodendrocytes and white matter development when administered to preterm fetal sheep after initiation of an inflammatory response induced by lipopolysaccharide (LPS). We have previously shown that hAECs reduced brain astrogliosis and inflammation and improved BBB function when administered at the same time as a fetal inflammatory stimulus induced by LPS 20 . The current study utilized a clinically relevant experimental design where LPS was administered to the fetus on 3 consecutive days to mimic an ongoing subclinical fetal inflammatory response, while hAEC treatment commenced 24 h after the first LPS exposure. We hypothesized that hAEC administration, following induction of inflammation, would protect white matter development in the preterm brain by reducing brain inflammation and protecting the developing oligodendrocytes.
Materials and Methods
Animals and Surgical Preparation
All experimental procedures received prior approval from the appropriate institutional animal ethics committee and conformed to the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (MMCA 2010/04) and Monash Health Research Ethics Committee (#12223B). Twenty-four singleton-bearing ewes of known gestational age were used. During the study, ewes were kept in individual cages with free access to food and water under a 12-h light/dark cycle (lights on at 07:00 h).
At 105 days gestation (term is 147 days), surgery was performed under isoflurane (Isoflo; Abbott, Sydney, N.S.W., Australia) general anesthesia for implantation of polyvinyl catheters to the fetus (Dural Plastics, Silver-water, Australia). Under aseptic conditions, the fetus was exposed, and catheters were inserted into a fetal brachial artery, femoral vein, and amniotic cavity. The brachial artery catheter was used to infuse hAECs so that they passed to the head and brain before entering the systemic and placental circulations, as previously investigated 21 . The fetus was returned to the uterus, and catheters were exteriorized through an incision in the ewe's flank. A maternal jugular vein catheter was also inserted at surgery. Antibiotics were given for 3 days after surgery by maternal intravenous (IV) injection (0.1 mg/kg oxytetra-cycline; Engemycin©; MSA Animal Health, Wellington, New Zealand) and into the amniotic sac (500 mg of ampicillin; Austrapen; CSL Ltd., Parkville, Australia).
Experimental Design
At 109 days gestation (experimental day 1), each fetus was assigned randomly to one of four groups (Fig. 1): (1) control: fetuses administered an IV injection of saline solution on days 1, 2, and 3 (n = 6); (2) LPS: fetuses administered a 150 ng/kg IV injection of LPS derived from Escherichia coli (055:B5; Sigma-Aldrich, St. Louis, MO, USA) on days 1, 2, and 3 (n = 6); (3) LPS + hAECs: fetuses administered LPS as above, plus 60 million hAECs via the fetal brachial artery on days 2, 3, and 4 (n = 5); and (4) hAECs: fetuses received an IV injection of saline on days 1, 2, and 3, plus 60 million hAECs into the fetal brachial artery on days 2, 3, and 4 (n = 5). The hAECs were administered concurrently with LPS at 09:00 h. Fetal arterial blood samples were collected at time 0, 6, 12, and 24 h in respect to each LPS administration and at postmortem for collection of plasma and for assessment of fetal pH, oxygen saturation (SaO2), partial pressure of oxygen (PaO2), partial pressure of carbon dioxide (PaCO2), glucose, and lactate concentration using an ABL 700 blood gas analyzer (Radiometer, Copenhagen, Demark).
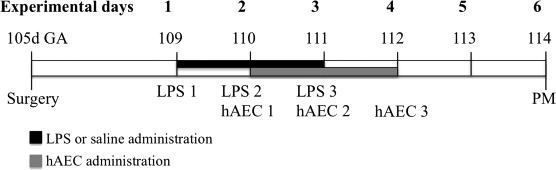
The experimental timeline in days (d). GA, gestational age; hAECs, human amnion epithelial cells; LPS, lipopolysaccharide; PM, postmortem.
The ewe and fetus were euthanized on day 6 (114 days gestation) with a maternal IV overdose of pentobarbital (Lethabarb Virbac Pty Ltd., Peakhurst, Australia). The fetal brain was immediately removed from the skull, weighed, and cut sagittally. The right hemisphere was placed in a custom-made mold shaped to fit the fetal sheep brain, and coronal sections were obtained by cutting through the hemisphere at 0.5-cm intervals. These slices were fixed by immersion in 4% paraformaldehyde (PFA; ProSci Tech, Thuringowa, Australia) for 48 h prior to embedding in paraffin. Subsequently, 10-μm sections were cut for histological analysis.
Human Amnion Epithelial Cell Preparation and Injection
Placentae were collected from healthy women with a healthy singleton pregnancy undergoing an elective repeat cesarean section at term following written informed consent and with the approval of Monash Health Human Research Ethics Committee. Cells were isolated from the placentae as previously described 19 . Cell isolates with a minimum of 80% viability were used. Before injection, hAECs were passively labeled with carboxyfluorescein succinimidyl ester (CFSE; Abcam, Melbourne, Australia) to enable identification in tissues 22 and resuspended in sterile saline at a concentration of 6 million cells/ml for administration to fetal sheep as previously described 20 .
Brain Pathology
Six fields of view over two duplicate slides per brain region were examined and averaged for each animal. Brain areas of interest were the hippocampus (CA1 and CA3), cortex, subcortical white matter (SCWM), PVWM, and striatum (internal capsule and external capsule). Personnel were blinded to the experimental group during image capture and analysis.
Microglia were identified using rabbit anti-ionized calcium-binding adaptor molecule 1 (Iba-1) antibody (Wako Pure Chemical Industries, Ltd., Osaka, Japan), raised against synthetic peptide corresponding to the C-terminal of Iba-1. The antibody was diluted 1:500 in phosphate-buffered saline (PBS) solution (0.1 mol/L, pH 7.4). Myelin basic protein (rabbit anti-human MBP; 1:500; Millipore, Billerica, MA, USA) was used to count the number of mature, myelin-producing oligodendrocytes, and oligodendrocyte transcription factor 2 (rabbit anti-Olig-2; 1:1,000; Millipore), a marker for oligodendrocytes at all stages of their lineage, was used to count total oligodendrocyte quantity. A mouse anti-CNPase (1:200; Sigma-Aldrich) was used to identify the integrity of myelin by measuring the density of myelinated axons. Rabbit anti-sheep albumin (1:1,000; Accurate Chemical and Scientific Corp., Westbury, NY, USA) was used to detect blood protein extravasation. All sections were treated with a secondary antibody [1:200; biotiny-lated anti-rabbit or anti-mouse immunoglobulin G (IgG) antibody; Vector Laboratories, Burlingame, CA, USA], and staining was revealed using 3,3-diaminobenzidine (DAB; Pierce Biotechnology, Rockford, IL, USA).
Statistical Analysis
Data are shown as mean ± standard error of the mean (SEM). Statistical analysis was performed with Sigma Stat software (Systat Software, San Jose, CA, USA) and/or GraphPad Prism (GraphPad Software, San Diego, CA, USA). Fetal arterial blood parameters were analyzed by two-way repeated-measures analysis of variance (ANOVA). One-way ANOVA was used to analyze fetal weight and histological data, with post hoc least significant difference as required. Differences were considered significant at p < 0.05.
Results
Fetal Outcomes
Intravenous injection of LPS alone resulted in 21% (three fetuses) fetal mortality rate within 24 h. hAECs given with or without LPS did not result in any fetal mortality. Fetal body weight was not different at postmortem between groups (control, 2.1 ± 0.1 kg; LPS, 2.2 ± 0.1 kg; LPS + hAECs, 2.3 ± 0.1 kg; hAECs, 2.3 ± 0.2 kg). In addition, there was no significant difference in fetal brain weight between groups (control, 34.9 ± 1.0 g; LPS, 37.4 ± 1.2 g; LPS + hAECs, 36.3 ± 1.2 g; hAECs, 35.5 ± 2.3 g).
Table 1 summarizes blood gases for all animals. Arterial pH, SaO2, PaO2, and glucose did not change in control animals over the experimental period. The administration of LPS was associated with transient hypoxemia and hypercapnia. Within the LPS + hAECs group, fetuses did not show signs of hypoxemia until 12-24 h following the second LPS administration with a significant increase in PaCO2 and a significant decrease in pH values, and at 12 h following the third LPS administration, there was also a significant decrease in SaO2 and an increase in glucose levels. Fetuses in the hAECs-only group had significantly higher PaCO2 values before cell treatment, which remained elevated until the end of the experiment. This increase also caused fetal acidemia by the end of the experiment, while SaO2, PaO2, and glucose were not different to basal values.
Fetal Arterial Blood Gases and pH Before, During, and After LPS and hAECs Treatment
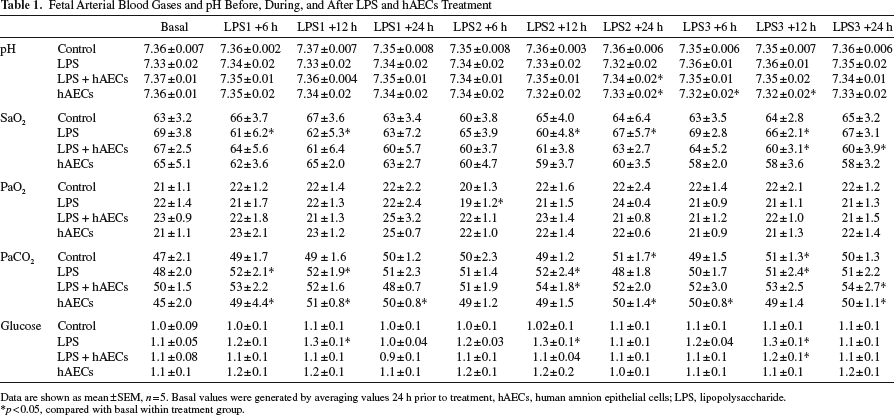
Data are shown as mean±SEM, n = 5. Basal values were generated by averaging values 24 h prior to treatment, hAECs, human amnion epithelial cells; LPS, lipopolysaccharide.
p < 0.05, compared with basal within treatment group.
Brain Histology and Immunohistochemistry
The brains of all control animals were normal with no evidence of injury. Cystic SCWM injury was observed in one of six animals in the LPS group (Fig. 2b), and five of six LPS animals exhibited evidence of widespread white matter (subcortical and subventricular) rarefaction (Fig. 2c).

(a) The number of pyknotic cells in the cortex, subcortical white matter (SCWM), periventricular white matter (PVWM), external capsule (EC), internal capsule (IC), and CA1 and CA3 regions of the hippocampus of control, lipopolysaccharide (LPS)-, human amnion epithelial cell (hAEC)-, and LPS + hAEC-treated animals. No pyknotic cells were found in control and hAEC-treated animals. Comparisons are made within the same brain region and not across regions. Each bar represents the mean ± standard error of the mean (SEM). *p < 0.05, significant difference between groups. Representative photomicrographs at 114 days gestation of the PVWM. Brain sections stained with cresyl violet and acid fuchsin (CV and AF; b-e) and albumin (f-i). Cystic cerebellar white matter injury was observed in one out of six animals in the LPS group (b). Five out of six LPS animals exhibited evidence of white matter rarefaction (c). Pyknotic cells were identified as shrunken cells with small, densely stained nuclei and acidophilic cytoplasm (d and e; e, magnified image from d). An intense staining of endogenous ovine plasma albumin was seen adjacent to blood vessels in the LPS-treated animals (g) but not in the control (f), LPS + hAEC (h)-, and hAEC (i)-treated animals. Scale bars: 50 μm.
Pyknotic Cell Death
Pyknotic degenerating cells were identified using acid fuchsin and cresyl violet stain as cells that demonstrated condensed morphology with small, densely stained nuclei and acidophilic cytoplasm (Fig. 2a-e). Pyknotic cells were rarely observed in the brains of the control group and hAECs-alone group (Fig. 2a). In LPS animals, an increase in pyknotic cell counts was observed within the PVWM (p = 0.01) (Fig. 2a), compared to controls. Administration of hAECs ameliorated cellular pyknosis in LPS fetuses, such that there was a significant reduction (p = 0.03) in pyknotic cell numbers in the PVWM of LPS + hAECs fetuses compared to LPS fetuses (Fig. 2a).
Albumin
The absence of immunoreactive albumin within the brain is indicative of a functional BBB, and albumin was not observed within control fetal brains (Fig. 2f), with the exception of some albumin extravasation in the internal capsule in two of six animals. In LPS-alone brains, intracellular immunoreactive albumin was observed in all brain regions in 30% of animals (Table 2). The occurrence of albumin extravasation from blood vessels was also observed in all brain regions of LPS-treated animals (Table 2). Where albumin was present in LPS brains, intense staining was predominantly observed in brain parenchyma adjacent to blood vessels (Fig. 2g). Intracellular albumin immunoreactivity was not observed in any of the LPS + hAEC-treated fetuses; albumin extravasation was only present in one of five fetuses within the cortex, two of five fetuses within the SCWM and PVWM, and none in the internal and external capsule. There was no albumin present in any of the hAECs-alone-treated fetuses (Fig. 2i).
The Incidence of Albumin in Fetal Sheep Brain Following LPS and hAECs Treatment
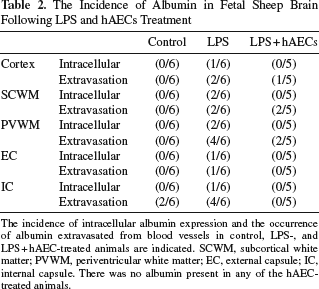
The incidence of intracellular albumin expression and the occurrence of albumin extravasated from blood vessels in control, LPS-, and LPS + hAEC-treated animals are indicated. SCWM, subcortical white matter; PVWM, periventricular white matter; EC, external capsule; IC, internal capsule. There was no albumin present in any of the hAEC-treated animals.
Inflammation
Cells that stained positively for Iba-1 (Fig. 3a) had the appearance of resting microglia (ramified with small cell bodies and long branching processes) (Fig. 3d, white arrowheads) and activated microglia (ameboid with large cell bodies) (Fig. 3e, black arrows). Activated microglia were observed in all treatment groups including the control animals (Fig. 3d, black arrow). In fetuses that received LPS alone, there was a significant increase in the number of activated microglia in the PVWM (p = 0.02 vs. control), and while activated microglia counts were also increased in the SCWM, external capsule, and hippocampus, values were not significantly different from control values (p > 0.05) (Fig. 3a). The administration of hAECs reduced the number of activated microglia cells after LPS exposure with no difference in the number of Iba-1+ cells between the LPS + hAECs and control brains across any brain region examined (Fig. 3a). Activated microglia cells were observed following hAEC administration alone, but levels were not significantly different from those in the control group (Fig. 3a and g). A significant positive correlation was observed between the number of activated Iba-1-immunoreactive cells and the number of pyknotic cells across all brain regions (R2 = 0.06, p = 0.003).

(a) The number of activated microglia [ionized calcium-binding adapter molecule 1-positive (Iba-1+) cells] in the cortex, subcortical white matter (SCWM), periventricular white matter (PVWM), external capsule (EC), internal capsule (IC), and CA1 and CA3 regions of the hippocampus of control (white bars), lipopolysaccharide (LPS; black bars)-, human amnion epithelial cell (hAEC; hatch bars)-, and LPS + hAEC-treated (gray bars) animals. The number (b) and density (c) of glial fibrillary acidic proteinpositive (GFAP+) cells in the SCWM and PVWM of control, LPS-, LPS + hAEC-, and hAEC-treated animals. Comparisons are made within the same brain region and not across regions. Each bar represents the mean ± standard error of the mean (SEM). *p < 0.05, significant difference between groups. Representative photomicrographs at 114 days gestation of the PVWM. Brain sections stained with Iba-1 (d-g) and GFAP (h-k) in control (d and h), LPS (e and i)-, LPS + hAEC (f and j)-, and hAEC (g and k)-treated animals. Iba-1-labeled cells demonstrated the morphology of activated/ameboid microglia (black arrows) or resting/ramified microglia with small cell bodies and long branching processes (white arrows). The increased number of activated microglia was associated with an increase in the number of astrocytes (GFAP+) in the LPS-treated animals (i). Scale bars: 50 μm.
Astrocytes
The number and morphology of astrocytes were examined by immunostaining for glial fibrillary acidic protein (GFAP) (Fig. 3). The number of GFAP+ cells was significantly increased in the SCWM (p = 0.02) of the LPS-treated fetuses compared to controls (Fig. 3b and c). Morphologically, astrocytes in the LPS treatment group alone were distinct from normal resting astrocytes observed within control brains, showing a reactive gliosis characterized by increased staining intensity of the cell soma and increased number and thickness of processes (Fig. 3i). Treatment with hAECs (LPS + hAECs) significantly decreased the number of astrocytes and the incidence of astrogliosis, compared to LPS-alone animals, within the subcortical and PVWM (p = 0.01 and p = 0.03 respectively). Astrocyte cell number and morphology were not different between the hAECs-alone group and the control group.
Oligodendrocytes
Olig-2+ oligodendrocytes were present in the white and gray matter of control and LPS-exposed fetal brains. The number of Olig-2+ cells was reduced in the SCWM (p = 0.08) and PVWM (p = 0.05) of the LPS-treated fetuses compared to controls. Following hAEC treatment, the number of Olig-2+ cells in the LPS + hAECs group was significantly increased in the SCWM (p = 0.006) and PVWM (p = 0.038) compared to LPS-alone group (Fig. 4a and d-g). The number of Olig-2+ cells was not changed in any hippocampal regions (Fig. 4a). MBP staining was present in the white matter of control and LPS-exposed fetal brains (Fig. 4b). The number of MBP+ cell bodies was reduced in the PVWM of the LPS-treated fetuses compared to controls, but this decrease did not quite reach significance (p = 0.09). Similarly, the number of MBP+ cells in the PVWM in the LPS + hAEC group was significantly increased (p = 0.016) compared to the LPS-alone group (Fig. 4b and h-k), while a significant negative correlation was found between the number of Iba-1-immunoreactive cells and the number of Olig-2+ cells across all brain regions (R2 = 0.21, p < 0.0001).

(a) The number of Olig-2+ cells in the cortex, subcortical white matter (SCWM), periventricular white matter (PVWM), external capsule (EC), internal capsule (IC), and CA1 and CA3 regions of the hippocampus of control, lipopolysaccharide (LPS)-, human amnion epithelial cell (hAEC)-, and LPS + hAEC-treated animals. The number of myelin basic protein-positive (MBP+) cells (b) and the density of myelinated axons (CNPase+; c) in the cortex, SCWM, PVWM, EC, and IC of control, LPS-, LPS + hAEC-, and hAEC-treated animals. Comparisons are made within the same brain region and not across regions. Each bar represents the mean ± standard error of the mean (SEM). *p < 0.05, significant difference between groups. Representative photomicrographs at 114 days gestation of the PVWM. Brain sections stained with Olig-2 (d-g), MBP (h-k), and CNPase (l-o) in control (d, h, and l), LPS (e, I, and m)-, LPS + hAEC (f, j, and n)-, and hAEC (g, k, and o)-treated animals. The arrows indicate MBP+ cells. Scale bars: 50 μm (d-g), 50 μm (h-k), 100 μm (l-o).
CNPase immunostaining revealed that myelin density was not affected across the treatment groups (Fig. 4c). However, we observed well-organized, densely distributed myelinated white matter tracts in the PVWM in control brains (Fig. 4l), while in LPS brains, white matter tracts were fragmented and disorganized (Fig. 4m), and LPS + hAECs fetuses appeared to have restored myelin morphology that was well organized with dense myelin (Fig. 4o).
The presence and location of hAECs within LPS + hAEC-treated brains were determined by visualizing the CFSE green fluorescent label incorporated into hAECs. Fluorescent cells were counted at 4× magnification under a dual fluorescent filter to rule out any nonspecific signal. hAECs were found within the brain of three of six LPS + hAEC-treated animals and in three of four hAECs-only animals at postmortem. In these six LPS-exposed animals, hAECs were diversely distributed throughout the brain in gray and white matter regions (Fig. 5). Within the gray matter, 5.0 ± 1.6 and 8.5 ± 0.8 cells/mm2 labeled cells were observed in the LPS + hAECs- and the hAECs-treated animals, respectively. Within the white matter, 2.7 ± 2.1 and 5.2 ± 1.3 cells/mm2 labeled cells were observed in the LPS + hAECs- and hAECs-treated animals, respectively.

Representative photomicrographs show immunohistochemistry of 4′,6-diamidino-2-pheylindole (DAPI; nuclear stain, blue) and human amnion epithelial cell [hAECs; carboxyfluorescein succinimidyl ester (CFSE) labeled, green] of the cortex, periventricular white matter (PVWM), and hippocampus in lipopolysaccharide (LPS) + hAEC-treated animal at 114 days gestation. Scale bars: 100 μm.
Discussion
In this study, we report the neuroprotective actions of hAECs to prevent cerebral white matter preterm brain injury secondary to an inflammatory insult induced by LPS. This is clinically significant since fetoplacental inflammation and preterm birth are principal contributors to perinatal brain injury (reviewed by Hagberg et al. 23 ), but there are currently no therapies aimed at reducing per inatal brain injury in high-risk preterm infants. Systemic administration of the potent inflammatory agent LPS caused widespread necrotic lesions resulting in white matter brain injury in preterm fetuses, consistent with PVL in preterm human infants 8 . Indeed, we observed characteristic cerebral white matter injury in response to LPS, with a decrease in the number of oligodendrocyte lineage cells, inflammatory cell activation, loss of BBB integrity, and astrogliosis. These findings were most commonly observed within the periventricular and SCWM, typical of preterm white matter regional vulnerability 24 . The neuroprotective actions of hAECs appear to be principally mediated by an anti-inflammatory role due to the decreased number of activated microglia present following hAEC administration.
LPS was administered directly to the fetus on 3 consecutive days to mimic an ongoing subclinical fetal inflammation with a relatively standardized pathological response, while hAEC treatment commenced 24 h after the first LPS exposure. This experimental design was chosen to clinically align with the potential application of hAECs to treat preterm brain injury, in which cells would be administered after birth to infants identified as high risk for brain injury. We specifically aimed to induce white matter brain damage mediated by a fetal inflammatory response, equivalent to the time of greatest vulnerability of the brain to damage, ~30 weeks gestation of human development (<110 days gestation in fetal sheep). Clinically, cell administration would likely occur in response to signs of inflammation rather than prophylactically, and it is clinically feasible that cell administration could occur at 24 h after birth in preterm infants that were identified as being exposed to intrauterine infection/chorioamnionitis. Repeated LPS administration caused physiological responses in the fetus following each injection, including hypoxemia and hypercapnia. However, these responses were attenuated with recurrent LPS exposure, while the degree of mild hyperglycemia was similar following each LPS treatment. Previous studies have described these transient and mild physiological responses to LPS, which are presumed to be due to altered placental permeability and substrate transfer25,26. The administration of hAECs did not attenuate the hypoxemic or metabolic responses to LPS, suggesting that hAECs do not protect placental function in response to fetal LPS.
At 5 days after the first LPS exposure, fetal neuropathology was evident. Principally, we found LPS to be more injurious to the cerebral white matter (periventricular and subcortical) than to the gray matter, which supports previous experimental studies using LPS in preterm animal models and clinical observations of PVL pathology 27 29 . There is a complex interplay between white and gray matter injury in preterm infants (reviewed by Volpe 31 ), wherein neuronal injury could occur secondary to damage to white matter tracts. Delayed administration of hAECs to the fetus preserved white matter development, as judged by multiple hAEC actions, including the protection of oligodendrocyte quantity, reduced neuroinflammation and astrogliosis, and maintenance of the integrity of the BBB. This is the first study to show that hAECs are protective to the developing brain following an inflammatory insult and extends our previous work showing that hAECs are neuroprotective when administered concurrently with the initiation of an inflammatory insult20,30. The observation that hAECs confer brain protection when administered 24 h after the initiation of an inflammatory insult is important, since white matter injury to the preterm brain is likely to result from progressive cerebral ischemia and inflammation 31 without a detectable point of onset.
Within white matter regions, but not gray matter, we observed LPS-induced microglial activation (Iba-1+ cells) by 6 days after the first LPS exposure. This type of injury is commonly seen in the clinic as periventricular damage and is the most common pathology observed in human preterm infants32,33. The administration of hAECs in response to LPS prevented this neuroinflammation, with no difference observed between the number of activated microglia in LPS + hAECs and control fetal brains. There was a significant correlation between the number of activated microglia and the loss of oligodendrocytes, indicating that activated microglia play a critical role in oligodendrocyte cell death. Indeed, an increase in activated microglial cells following LPS is a primary feature of brain neuropathology and has been previously correlated with oligodendrocyte cell death in response to fetal inflammation or hypoxia-ischemia20,34. Back and Rosenberg 35 report that injury to the white matter is distinguished by the presence of reactive microglia within the PVWM. Although reactive astrocytes might not be the predominant feature of early injury, they are associated with mediating toxic edema, provoking inflammation, releasing cytotoxins, and forming scars that inhibit axonal regeneration 36 . While the etiology of white matter injury in the human preterm brain is complex, it is appreciated that neuroinflammation is a major contributing factor and that microglial activation is the mechanism by which it manifests 37 . Therefore, preventing microglial activation with a repeat course of hAECs appears to be a beneficial neuroprotective action.
In the current study, we show that LPS caused breakdown of the BBB within selected regions of the white matter, but not the cortex. We also found that hAEC administration preserved BBB integrity, which may be triggered by an increase in proinflammatory mediators within the brain. Proinflammatory cytokines can readily cross the BBB under conditions of brain compromise 38 . This, in turn, results in an increased number of infiltrating inflammatory cells, activation of resident microglia, increased oxidative stress, and subsequent gliosis within the white matter 39 41 .
Leviton and Paneth 5 show evidence that between 28 and 32 weeks of gestation, when the risk of white matter damage is especially high, the most visually obvious expression of disturbed or damaged myelinogenesis is necrosis of premyelin and other glial cells in the white matter. In the current study, fetal LPS exposure induced a considerably high level of cellular pyknosis within the PVWM region, with no evidence of neuronal death, either via pyknosis in gray matter regions or reduced neuronal cell number (NeuN cell count; data not shown). The administration of hAECs to the fetus reduced pyknotic cells within the PVWM after LPS exposure. The decrease in oligodendrocyte numbers within the PVWM after LPS administration confirms that it is glial cells that are vulnerable to inflammatory insult, causing a significant injury to the white matter. In human infants, there is evidence to show that diffuse injury within the white matter in infants born at less than 33 weeks gestation is clinically associated with poor cognitive function at 19 years of age 42 . The pattern of white matter injury we observed in our study is similar to that described in response to inflammation in other animal studies43,44 and is consistent with that observed in infants with PVL associated with higher systemic cytokine expression 45 .
In the control animals treated with hAECs, we observed an unexpected, albeit nonsignificant, change in the number of inflammatory and MBP+ cells within the white matter. The brain at this developmental stage is in a highly dynamic state, and the balance of cell proliferation and cell death is critical. hAECs may modify these events, potentially by shifting the brain toward an anti-inflammatory state. We have previously shown 15 that hAECs can directly influence macrophage behavior in a proreparative manner and are able to mediate these effects independent of other immune cell types. Therefore, it would be of interest to further phenotype the inflammatory cells of treated and nontreated animals to determine the role of hAECs during brain development. It is, however, worth mentioning that in the setting of preterm white matter brain injury induced by in utero inflammation, we do not propose that hAECs should be administered prophylactically. We suggest that hAECs could be used after birth for high-risk infants with confirmed intrauterine infection/chorioamnionitis, and therefore “control” healthy infants would not be treated with hAECs.
Forty-eight hours after the last cell administration, a small number of fluorescent-labeled hAECs were found within the white matter, cortex, and hippocampus. This finding is consistent with our earlier work in fetal sheep18,20,30. Previous studies in models of adult lung disease have demonstrated that the actions of hAECs were independent of cell differentiation and, most likely, were due to paracrine factors released by hAECs rather than through direct cell replacement by hAECs15,18,19. The role of hAECs in immunomodulation appears multifactorial, characterized by suppression of proinflammatory cytokines 46 and secretion of factors that inhibit the chemotactic activity of neutrophils and macrophages 12 . Further, recent work indicates that nanosized vesicles, called exosomes, are released by hAECs and exert reparative effects by activating endogenous repair mechanisms 47 . Amniotic exosomes act directly on immune cells to reduce T-cell proliferation and, later, macrophage activities in a manner similar to that reported for hAECs 47 . Given that hAECs do not engraft as part of their reparative process, but rather exert their effects through paracrine means, we suggest that exploring exosomal contents warrants further investigation.
Our current findings demonstrate that hAECs administered 24 h after fetal inflammation act via anti-inflammatory and antiapoptotic actions to protect the developing brain. Further, we believe that the neuroprotective and neurorestorative properties of hAECs are mediated via their ability to control immune cell infiltration into the brain, thus reducing inflammation via secretion of various trophic and angiogenic factors that stimulate angiogenesis and cell survival and thereby act to decrease apoptotic cell death. That hAECs may be capable of repairing established injury makes them an appropriate candidate for repairing damage to the developing brain, which is known to be mediated by inflammatory events and is often only revealed once injury has occurred. We do however acknowledge that delayed cell treatment 5 or more days after an insult would provide insight into the reparative role of hAECs once brain injury is already established. Further, it is important to note that an insight on protein and mRNA levels of all the markers of brain injury would confirm the pathological outcome. However, because of regional specific injury, this would likely be undetected. Hence, we believe that the cellular data that we present in this study in the form of immunohistochemical changes are a better cellular-level representation of regional specific injury. Nevertheless, these findings support the use of hAECs as a therapy following inflammatory-induced brain injury.
In summary, this study demonstrated that hAECs mitigate fetal brain inflammation and reduce white matter injury in response to inflammation caused by multiple IV LPS administration. The likely mechanism of protective action of hAECs on the fetal brain is via anti-inflammatory effects, by reducing the number of activated microglial cells in the white matter. Activation of microglia, loss of oligodendrocytes, and BBB leakiness were prominent pathological features associated with brain injury after LPS exposure. hAECs administered via the brachial artery protected the developing brain when administered 24 h after the establishment of an inflammatory response. These findings support the use of hAECs in the treatment of high-risk preterm infants where the injury/insult may have already occurred in utero.
Footnotes
Acknowledgments
The authors acknowledge funding support from the NHMRC Australia, an ARC Future Fellowship to S.M., and the Victorian Government's Operational Infrastructure Support Program. The authors also thank Ms. Era Anwar and Dr. Yen Pham for their technical assistance. The authors declare no conflicts of interest.
