Abstract
There are some limitations to the therapeutic effects of mesenchymal stem cells (MSCs) on acute respiratory distress syndrome (ARDS) due to their low engraftment and differentiation rates in lungs. We found previously that noncanonical Wnt5a signaling promoted the differentiation of mouse MSCs (mMSCs) into type II alveolar epithelial cells (AT II cells), conferred resistance to oxidative stress, and promoted migration of MSCs in vitro. As receptor tyrosine kinase-like orphan receptor 2 (ROR2) is an essential receptor for Wnt5a, it was reasonable to deduce that ROR2 might be one of the key molecules for the therapeutic effect of MSCs in ARDS. The mMSCs that stably overexpressed ROR2 or the green fluorescent protein (GFP) control were transplanted intratracheally into the ARDS mice [induced by intratracheal injection of lipopolysaccharide (LPS)]. The results showed that ROR2-overexpressing mMSCs led to more significant effects than the GFP controls, including the retention of the mMSCs in the lung, differentiation into AT II cells, improvement of alveolar epithelial permeability, improvement of acute LPS-induced pulmonary inflammation, and, finally, reduction of the pathological impairment of the lung tissue. In conclusion, MSCs that overexpress ROR2 could further improve MSC-mediated protection against epithelial impairment in ARDS.
Introduction
Despite extensive research into the pathogenesis of acute respiratory distress syndrome (ARDS), a major cause of acute respiratory failure in critically ill patients, there are currently no effective therapies to reverse or retard the course of ARDS. Mortality due to ARDS is still high at approximately 40% annually1,2. The physiological hallmark of ARDS is disruption of the alveolar–capillary membrane barrier. Both alveolar epithelial and endothelial cell injury and/or death have been implicated in the pathogenesis of ARDS 3 . The repair and regeneration of alveolar epithelium are therefore critical for recovery from ARDS 4 .
Mesenchymal stem cells (MSCs) are multipotent adult stem cells found in the bone marrow (BM) and other tissues that have the ability to differentiate into multiple cell types5,6. Recent studies have demonstrated that MSCs can engraft into the injured lung7,8 and even differentiate into lung epithelial cells in vivo7,9,10. However, the engraftment and differentiation rate of MSCs in injured lungs are limited 7 , which inhibits the beneficial effects of MSCs as a therapeutic agent for ARDS therapy. Therefore, it is essential to clarify the mechanisms underlying MSC function in epithelial repair in ARDS for the improvement of cellular retention in injured lung tissue, and differentiation of MSCs into alveolar epithelial cells.
The Wnt signaling pathway is one of the fundamental pathways in cell proliferation and motility, cell fate determination, cell polarity during embryonic development, and adult tissue homeostasis 11 . It can be divided into the canonical and noncanonical Wnt pathways according to their downstream signaling, which is either dependent or not dependent on the accumulation of β-catenin. The activation of the noncanonical Wnt pathway through the binding of Wnt ligands like Wnt5a or Wnt11 to receptor tyrosine kinase-like orphan receptor 2 (ROR2) depends on the phosphorylation of downstream calmodulin-dependent protein kinase (CaMK), protein kinase C (PKC), or c-Jun N-terminal kinase (JNK). It is currently believed that Wnt5a is a representative noncanonical Wnt ligand that can activate noncanonical Wnt signaling, and ROR2 acts as a receptor or coreceptor for Wnt5a to mediate various cellular functions including cell migration, polarity, invasion, or differentiation12–16 and is the most studied molecule in the noncanonical Wnt pathway. Several recent studies have shown that noncanonical Wnt5a/ROR2 signaling has critical effects on the differentiation of MSCs, which express a number of ligands, receptors, and inhibitors important for this pathway13,17,18. In our previous study, we found that Wnt5a, through the noncanonical Wnt pathway, Wnt/JNK signaling alone, or both Wnt/JNK and Wnt/PKC signaling promoted the differentiation of mouse MSCs (mMSCs) into type II alveolar epithelial cells (AT II cells) and the migration of mMSCs. Additionally, through Wnt/PKC signaling, Wnt5a increased the survival of mMSCs in vitro after exposure to hydrogen peroxide (H2O2) 19 . We also found that Wnt5a was increased in the lung tissue of ARDS mice after intratracheal (IT) lipopolysaccharide (LPS) administration (supplemental Fig. S, available at http://a4.qpic.cn/psb?/V12TR5ms0Vh2Ap/zXHF4.5GHW72mtdJ2Ec.rgS2a0EE7dvn.qaLCUw229s!/b/dG8AAAAAAAAA&ek=1&kp=1&pt=0&bo=PQOAAgAAAAAFB5g!&sce=0-12-12&rf=viewer_4) 19 , as ROR2 is considered the key signaling regulator of the Wnt5a/ROR2 pathway. We therefore hypothesized that the Wnt5a/ROR2 pathway may play an important role in the fate and therapeutic effect of MSCs on ARDS in vivo. Because of the more complicated circumstances and regulatory mechanisms of the in vivo microenvironment, which is drastically different than the specific and limited cultural conditions of in vitro differentiation that affect MSCs, a long-term and stable mMSC line that overexpresses ROR2 was created using a lentiviral vector in our previous study 20 . The overexpression of ROR2 also was confirmed to be able to mediate the activation of the Wnt5a/JNK and Wnt5a/PKC pathways and regulate the proliferation, migration, and differentiation of mMSCs. Therefore, long-term and stable mMSC lines that overexpress ROR2 are applicable for in vivo investigations 20 . The aim of our study was to identify the effects of overexpression of ROR2 on the retention and differentiation of type II alveolar epithelial cells, and the reparative abilities of mMSCs on injured alveolar epithelium in LPS-induced ARDS mice.
Materials and Methods
MSC Transfection and Culture
mMSCs were derived from the BM of 4- to 6-week-old male C57BL/6 mice, (Cyagen Biosciences Inc., Guangzhou, China). The details of the transfection of MSCs, mediated by lentiviral vectors, were described in our previous work 20 . Briefly, the recombinant lentivirus vector overexpressing the ROR2 gene was constructed using Gateway cloning technology (Gateway BP Clonase II Enzyme Mix; Invitrogen, Life Technologies, Carlsbad, CA, USA) by using an elongation factor 1α (EF-1α) promoter-dependent lentiviral expression vector; an empty EF-1α-enhanced green fluorescent protein (EGFP) vector was used as a control. The lentivirus was packaged in 293FT cells (Cyagen Biosciences) with the aid of three packaging plasmids, and then a high titer of the recombinant lentivirus was obtained and used to transfect the mMSCs. mMSCs carrying empty vectors and EGFP (mMSC control) or mMSCs carrying both the ROR2 gene and EGFP (ROR2-mMSCs) were harvested after selection using G418 for 7–14 days. Subsequently, mMSC control or ROR2-mMSCs were cultured in a 1:1 mix of Dulbecco's modified Eagle's medium/nutrient mixture F12 (DMEM/F12) (Wisent Inc., Saint-Bruno, QC, Canada) containing 10% fetal bovine serum (FBS; Wisent Inc.) and 1% streptomycin and penicillin (Wisent Inc.), and incubated at 37°C in a humidified atmosphere of 5% CO2. The cells at passages 6–10 were used for in vivo experiments.
Animals
Male C57BL/6 mice, aged 8–12 weeks and weighing 20–25 g, were obtained from the Laboratory Animal Center at the Academy of Military Medical Sciences (Beijing, China). All animal experiments performed in this study conformed to the Guide for the Care and Use of Laboratory Animals [National Institutes of Health (NIH), Bethesda, MD, USA] and were approved by the Institutional Animal Care and Use Committee (IACUC) of Southeast University. Animals were maintained under artificial day–night cycles [12-h light–dark cycles; 23 ± 1°C room temperature (RT), 30–60% environment humidity], allowed free access to standard rodent chow and drinking water, and allowed to adapt to laboratory conditions for at least 3 days.
Murine Model of LPS-Induced ARDS
The mice were first anesthetized with a dose of 50 mg/kg pentobarbital (Sigma-Aldrich, St. Louis, MO, USA) by intraperitoneal (IP) injection and received a single, IT delivered dose of LPS (100 μg) from Escherichia coli serotype 0111:B4 (Sigma-Aldrich) in 50 μl of sterile normal saline (NS) 21 . The mice were then allowed to recover in a 100% oxygen chamber until fully awake. The control mice received 0.9% NS instead of LPS.
Experimental Protocol
Mice were randomly divided into four groups according to a random number table (n = 36 for each group):
the NS + PBS group, in which mice received 30 μl of phosphate-buffered saline (PBS; Gibco, Grand Island, NY, USA) IT 4 h after IT administration of 0.9% NS;
the LPS + PBS group, in which mice received 30 μl of PBS IT 4 h after induction of ARDS; (3) the LPS + mMSC control group, in which mice received the control mMSCs (500,000 cells in 30 μl of PBS) IT 4 h after induction of ARDS; and (4) the LPS + ROR2-mMSC group, in which mice received ROR2-mMSCs (500,000 cells in 30 μl of PBS) IT 4 h after induction of ARDS. Mice were sacrificed at the end of either 3, 7, or 14 days, and samples were collected from each mouse for assessment of lung injury, biochemical analysis, and histology.
Hematoxylin and Eosin Staining and Lung Injury Scoring
The right upper lobe of the lung was embedded in paraffin (Thermo Fisher Scientific, Waltham, MA, USA) and sliced at 5 μm sagittally. The sections were stained with hematoxylin and eosin (H&E; Beyotime, Shanghai, China). The severity of lung injury was determined using a previously described method 22 . Briefly, the severity of lung injury was quantified on the basis of the findings in 10 randomly selected high-power fields (×400). Lung injury was graded from 0 (normal) to 4 (severe) for a number of different symptoms (described below), and the total lung injury score per mouse was determined as sum of the scores. Edema, alveolar and interstitial inflammation and hemorrhage, atelectasis, necrosis, and hyaline membrane formation were each scored using the 0–4 point scale: 0 = no injury; 1 = injury in 25% of the field; 2 = injury in 50%; 3 = injury in 75%; and 4 = injury throughout the field of vision. The lung injury was finally scored according to the sum of these scores. Ten randomly selected high-power fields (400×) in each slide were analyzed by two investigators who were blinded to the treatment groups.
Labeling and Tracking of mMSCs
The cultured control mMSCs and ROR2-mMSCs were harvested and washed with basic culture media (DMEM/F12; Wisent Inc.) and labeled with CellVue NIR815 dye (eBioscience Inc., San Diego, CA, USA) following the manufacturer's instructions. NIR815-labeled cells (5 × 105) were directly administered IT to the LPS + mMSC control and LPS + ROR2-mMSC groups. Three lungs of subjects in each group were imaged, ex vivo, at three time points: 3, 7, and 14 days posttransplantation, using a Maestro In-Vivo Optical Imaging System (excitation = 786 nm, emission = 814 nm, exposition time = 4,000 ms; Caliper Life Sciences, Hopkinton, MA, USA) 23 . The autofluorescence spectra were then unmixed on the basis of their spectral patterns using Maestro 2.4 software (Caliper Life Sciences). The fluorescence intensity of the lungs was measured by placing the regions of interest (ROIs) on the organ, and the average signals were normalized by the exposure time and the area of ROI (scaled counts/s).
Immunofluorescence Staining
Immunofluorescence staining for the detection of engraftment of mMSCs in vivo in order to evaluate the homing and differentiation ability of the cells was performed as previously described 24 . Briefly, the left lung tissue samples from the LPS + mMSC control group and the LPS + ROR2-mMSC group were perfused with NS, snapfrozen in liquid nitrogen, and then stored at −80°C until use. The tissue was embedded in optimal cutting temperature compound (OCT; Thermo Fisher Scientific, Bremen, Germany) and cut at a thickness of 10 μm. The slides were fixed in acetone (Spectrum Chemical Mfg. Corp., Shanghai, China) at 4°C for 15 min, and then blocked by 3% bovine serum albumin (BSA) in PBS/0.3% Triton X-100 (Spectrum Chemical Mfg. Corp.) for 30 min at RT. After washing and draining, the slides were incubated overnight at 4°C with green fluorescent protein (GFP) primary antibody (1:100; Abcam, Cambridge, MA, USA) either alone or with the same volume of GFP and prosurfactant protein C (SP-C) primary antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, CA, USA). After three washes with PBS, the slides were incubated with secondary antibody goat anti-mouse Alexa Fluor 488 and goat anti-rabbit Alexa Fluor 647 (Abcam) at a 1:200 dilution in 2% BSA for 1 h at 37°C in dark. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich) for 5 min. The images were captured using a fluorescence microscope (Olympus, Tokyo, Japan). The retention or differentiation of transplanted MSCs was quantified on the basis of the count of GFP-positive MSCs or the ratio of the count of GFP-positive cells to the count of SP-C-positive MSCs in randomly selected high-power fields (×400) for each slide by histopathologists blinded to the protocol design.
Western Immunoblot Analysis
Total protein (TP) was extracted by using radio-immunoprecipitation assay (RIPA) lysis buffer (Beyotime). Protein was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and electrotransferred to polyvinylidene fluoride (PVDF) membranes (Millipore, Bedford, MA, USA). The membranes were blocked in Tris-buffered saline (TBS) (pH 7.4) containing 0.1% Tween 20 (Shanghai Chemical Reagent Company of China Pharmaceutical Group, Shanghai, China) and 5% BSA for 1 h at RT, and then incubated at 4°C overnight with primary antibodies recognizing SP-C (1:100; Santa-Cruz Biotechnology), β-actin (1:500; Santa Cruz Biotechnology), and occludin (1:200; Abcam). Subsequently, membranes were incubated for 1 h at RT with a goat anti-rabbit immunoglobulin G (IgG)-horseradish peroxidase (HRP)-conjugated secondary antibody (1:10,000; Zhongshan Golden Bridge Biotechnology Co. Ltd., Beijing, China). Immunoreactive bands were detected with Pierce ECL Western Blotting Substrate (Thermo Fisher Scientific).
Evaluation of the Lung Edema
Lung edema was measured using the ratio of lung wet weight to body weight (LWW/BW) as previously described 25 . Briefly, the whole lung was removed and cleared of all extrapulmonary tissues, and the LWW/BW was calculated on the basis of the values of LWW and BW. The results were expressed in mg/g.
Bronchoalveolar Lavage Fluid (BALF) Cytokine and Protein Measurements
BALF was measured after sacrifice of the mice; 1-ml aliquots of ice-cold PBS were flushed back and forth three times through a tracheal cannula. BALF was collected and centrifuged at 800 × g for 10 min, and the concentrations of interleukin-1β (IL-1β), IL-6, and IL-10 proteins in the supernatant were measured with murine cytokine-specific enzyme-linked immunosorbent assay (ELISA) kits (Shanghai ExCell Biological Products, Shanghai, China) strictly according to the manufacturer's instructions. TP and albumin (ALB) in BALF were measured from all experimental groups as a marker of epithelial permeability with ELISA kits (Cusabio Biotech, Wuhan, China). In addition, the level of keratinocyte growth factor (KGF) in BALF was measured.
Masson's Trichrome Staining and Fibrosis Scoring
The lung sections were stained sequentially with Weigert's iron hematoxylin solution (Bogoo, Shanghai, China), Biebrich scarlet acid fuchsin solution (Bogoo), and aniline blue solution (Bogoo); a blue signal indicated positive staining for collagen. Lung fibrosis was quantified on the basis of the findings in 10 randomly selected high-power fields (400×) for each slide by a histopathologist blinded to the protocol design. The fibrotic grade of each field was assessed using the criteria of Ashcroft et al. 26 and graded from 0 to 5 as follows: 0 = normal lung; 1 = minimal fibrous thickening of the alveolar or bronchial walls; 2 = moderate thickening of the walls without obvious damage to the lung architecture; 3 = increased fibrosis with definite damage to the lung structure and the formation of fibrous bands or small fibrous masses; 4 = severe distortion of the structure and large fibrous areas (honeycomb lung); and 5 = total fibrous obliteration in the field. The lung fibrosis was finally scored according to the average of the above scores.
Statistical Analysis
The data were presented as the mean ± standard deviation (SD). Statistical analyses were performed using SPSS 16.0 software package (IBM, Armonk, NY, USA). Comparisons among multiple groups were performed by one-way analysis of variance (ANOVA) followed by Bonferroni's post hoc test. A value of p < 0.05 was considered to be statistically significant.
Results
Effect of ROR2-Overexpressing MSCs on Pulmonary Histopathology
An increased thickening of the alveolar wall, alveolar and interstitial inflammatory cell infiltration, hemorrhage, alveolar exudation, and edema were found in the lung tissue of mice after LPS-induced lung injury, and the Smith score 22 for quantification of the lung injury was also increased compared with the NS + PBS group (p < 0.05). However, these histopathological characteristics and the Smith score were alleviated at 3, 7, and 14 days in the LPS + mMSC control and LPS + ROR2-mMSC groups compared with the LPS + PBS group (p < 0.05). The effect was greater in the LPS + ROR2-mMSC group than in the LPS + mMSC control group (p < 0.05) (Fig. 1A and B).

Effect of control mouse mesenchymal stem cells (mMSCs) or receptor tyrosine kinase-like orphan receptor 2 (ROR2)-overexpressing mMSCs on the histopathology of lipopolysaccharide (LPS)-induced lung injury and survival over 14 days. (A) Histopathological analysis of lung tissues from mice of all the experimental groups was performed at days 3, 7, and 14 after the LPS challenge [hematoxylin and eosin (H&E) staining, magnification: ×200, scale bars: 20 μm]. (B) Quantification of lung injury showed a significant reduction in the severity of lung injury in the LPS + mMSC control and LPS + ROR2-mMSC mice. The change was more significant in the LPS + ROR2-mMSCs group than in the LPS + mMSC control group (n = 6 at each time point for each group). The results are expressed as mean ± standard deviation (SD). n = 6; *p < 0.05 versus NS + PBS group; #p < 0.05 versus LPS + PBS group; &p < 0.05 versus LPS + mMSC control group.
Overexpression of ROR2 Increases the Retention of MSCs in the Lung After LPS Challenge
Ex vivo near infrared region (NIR) imaging was performed on the lungs from LPS + mMSC control and LPS + ROR2-mMSC mice 3, 7, and 14 days after MSC administration to track the intrapulmonary mMSCs. Fluorescence images indicated that the signals in the LPS + ROR2-mMSC group were stronger than those in the LPS + mMSC control group at the end of days 3, 7, and 14 after the LPS challenge. For each group, the signal gradually decreased after day 3. Although the signal decreased significantly on day 14 after the LPS challenge, it could still be detected (Fig. 2A). Similar results were also observed using an immunofluorescence staining assay to detect the MSCs in lung tissue (Fig. 2B).

Effect of ROR2 overexpression on the retention of mMSCs in the lung after LPS challenge. (A) Ex vivo near infrared region (NIR) imaging of the lungs. Representative photographs of fluorescence images (scale bar: 5 mm) are shown from three mouse lungs obtained 3, 7, and 14 days after mMSC administration (from the LPS + mMSC control and LPS + ROR2-mMSC groups). (B) Immunofluorescence staining to detect mMSC engraftment in lung tissue is shown as green fluorescent protein (GFP) positive (white arrows point to the GFP-positive cells). The nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI). Representative photographs are shown from six mouse lungs 3, 7, and 14 days after mMSC administration (from the LPS + mMSC control and LPS + ROR2-mMSC groups). All microphotographs were taken at ×400 magnification (scale bar: 20 μm). The count of GFP-positive MSCs in randomly selected high-power fields (count/HP) is presented as mean ± SD. n = 6; &p < 0.05 versus LPS + mMSC control group.
Overexpression of ROR2 Promotes the Differentiation of MSCs Into AT II Cells In Vivo
The total expression of SP-C protein in the lung tissue 14 days after MSC administration was evaluated using Western blot analysis. As compared with the normal control group, the SP-C protein in the lung tissue was decreased after LPS-induced lung injury whether or not mice were treated with control mMSCs or ROR2-mMSCs (p < 0.05). The results showed that SP-C protein was upregulated in the LPS + mMSC control group and the LPS + ROR2-mMSC group compared with the LPS + PBS group (p < 0.05), and the increase in the LPS + ROR2-mMSC group was more significant than the increase in the LPS + mMSC control group (p < 0.05) (Fig. 3A). Moreover, differentiation of the MSCs into AT II cells was evaluated 14 days after the MSC administration by analyzing the expression of SP-C, a specific type II alveolar epithelial cell marker, in the engrafted MSCs by immunofluorescence staining. Colocalization of SP-C and MSCs in the lung tissue (identified by arrows) could be seen in both the LPS + ROR2-mMSC and LPS + mMSC control groups; however, ROR2-mMSC treatment led to a higher differentiation efficiency of the MSCs into AT II cells than the mMSC control treatment (p < 0.05) (Fig. 3B).
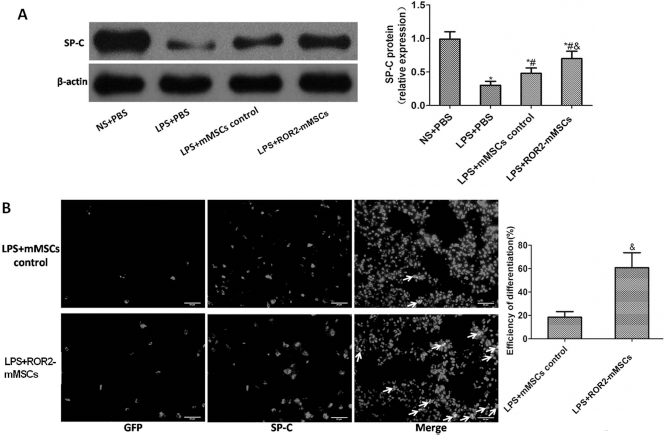
Effect of ROR2 overexpression on the differentiation of mMSCs into type II alveolar epithelial cells (AT II cells) in vivo. (A) The expression of the SP-C protein in the lung tissue on day 14 after mMSC administration was evaluated using Western blot analysis. β-Actin was used as an internal control, and the results are presented as mean ± SD. n = 6; *p < 0.05 versus NS + PBS group; #p < 0.05 versus LPS + PBS group; &p < 0.05 versus LPS + mMSC control group. (B) The differentiation of mMSCs into AT II cells was detected by immunofluorescence staining 14 days after mMSC administration in mice from the LPS + mMSC control and LPS + ROR2-mMSC groups. mMSC engraftment in lung tissue appears GFP positive (the first column), a specific type II alveolar epithelial cell marker SP-C positive (the second column), and colocalization of the GFP-positive and SP-C positive cells are indicated by the white arrows (the third column). The nuclei were stained with DAPI. All microphotographs were taken at ×400 magnification (scale bars: 20 μm). The ratio of the count of GFP-positive to the count of SP-C-positive MSCs in randomly selected high-power fields is presented as mean ± SD. n = 6; &p < 0.05 versus LPS + mMSC control group.
ROR2-Overexpressing MSCs Improved LPS-Induced Lung Permeability
The LWW/BW ratio was calculated to evaluate lung edema. LWW/BW was significantly reduced in the LPS + ROR2-mMSC group compared with the LPS + PBS group at days 3, 7, and 14 (p < 0.05) and in the LPS + mMSC control group at days 3 and 14 (p < 0.05). At day 14, LWW/BW was almost restored to normal levels in the LPS + ROR2-mMSC group (Fig. 4A).

Effect of control mMSCs or ROR2-overexpressing mMSCs on LPS-induced lung permeability. (A) Lung edema was measured using the ratio of lung wet weight to body weight (LWW/BW). The results are shown for samples taken 3, 7, and 14 days after LPS exposure. (B) Total protein (TP) and (C) albumin (ALB) concentration in bronchoalveolar lavage fluid (BALF) was measured using mouse-specific enzyme-linked immunosorbent assays (ELISAs) to evaluate the epithelial permeability of the lung. (D) Expression of the occludin protein in the lung tissue on day 14 after mMSC administration was evaluated using Western blot analysis. β-Actin was used as an internal control. (E) Keratinocyte growth factor (KGF) concentration in BALF was measured using a mouse-specific ELISA. The data are presented as mean ± SD of six animals at each time point per group. *p < 0.05 versus NS + PBS group; #p < 0.05 versus LPS + PBS group; &p < 0.05 versus LPS + mMSC control group.
To evaluate the effect of ROR2-overexpressing MSCs on epithelial permeability of the lung, the TP and ALB concentrations in the BALF were measured using mouse-specific ELISAs. TP and ALB were significantly increased in BALF after LPS-induced lung injury whether mice were treated with mMSCs or ROR2-mMSCs when compared with the normal control NS + PBS group (p < 0.05). TP and ALB were significantly reduced in the LPS + ROR2-mMSC group compared with the LPS + PBS group (p < 0.05) and the LPS + mMSC control group on days 3, 7, and 14 (p < 0.05). Significant decreases in TP and ALB were also observed in the LPS + mMSC control group compared with the LPS + PBS group on days 3 and 7 (p < 0.05), while no difference was observed on day 14 (Fig. 4B and C).
Additionally, to analyze the tight junctions of the pulmonary epithelial cells after MSC administration, occludin protein expression was evaluated in the lung tissue 14 days after MSC administration using Western blotting analysis. The results showed that occludin protein was upregulated in the LPS + mMSC control and LPS + ROR2-mMSC groups compared with the LPS + PBS group (p < 0.05), and the increase observed in the LPS + ROR2-mMSC group was more significant than that in the LPS + mMSC control group (p < 0.05) (Fig. 4D).
The concentration of KGF, an important cytokine for the improvement of lung permeability, was measured in BALF using a mouse-specific ELISA. KGF was significantly increased in the LPS + PBS, LPS + mMSC control, and LPS + mMSC-ROR2 groups compared with the NS + PBS group on days 3, 7, and 14 (p < 0.05). Both control mMSC and ROR2-mMSC administration had decreased expression of KGF on day 3 compared with LPS + PBS treatment (p < 0.05). On day 14, KGF was higher in the LPS + ROR2-mMSC group than in the LPS + PBS (p < 0.05) and LPS + mMSC control groups (p < 0.05) (Fig. 4E).
ROR2-Overexpressing MSCs Attenuated Acute LPS-Induced Pulmonary Inflammation
The levels of the proinflammatory cytokines IL-1β and IL-6 and the anti-inflammatory cytokine IL-10 were measured in the BALF of mice 3 days after LPS treatment using ELISA. All three cytokines were significantly higher in the LPS + PBS group than in the NS + PBS group (p < 0.05). IL-1β and IL-6 were reduced in the LPS + mMSC control and LPS + ROR2-mMSC groups compared with the LPS + PBS group (Fig. 5A and B, respectively), while IL-10 was increased (p < 0.05) (Fig. 5C). Comparatively, the decrease in IL-1β and increase in IL-10 observed in the LPS + ROR2-mMSC group were more significant than the changes observed in the LPS + mMSC control group (p < 0.05).

Effect of control mMSCs or mMSCs overexpressing ROR2 on acute LPS-induced pulmonary inflammation. Levels of the proinflammatory cytokines (A) IL-1β, (B) IL-6, and anti-inflammatory cytokine (C) IL-10 in BALF in mice receiving MSCs at 3 days after LPS-induced ARDS were measured using ELISA. Data are expressed as mean ± SD. n = 6; *p < 0.05 versus NS + PBS group; #p < 0.05 versus LPS + PBS group; &p < 0.05 versus LPS + mMSC control group.
ROR2-Overexpressing MSCs Inhibited Lung Fibrosis
The deposition of collagen in lung tissue after 14 days of LPS exposure was evaluated by Masson's trichrome staining and was markedly increased in the LPS + PBS, LPS + mMSC control, and LPS + ROR2-mMSC groups compared with the NS + PBS group (p < 0.05). Reduced deposition of collagen was observed after intervention with either GFP-transfected MSCs or ROR2-transfected MSCs when compared with LPS + PBS (p < 0.05), but the decrease observed in the LPS + ROR2-mMSC group was more significant than that observed in the LPS + mMSC control group (p < 0.05) (Fig. 6).

Effect of control mMSCs or ROR2-overexpressing mMSCs on lung fibrosis. (A) Lung fibrosis was evaluated by Masson's trichrome staining 14 days after LPS exposure (×200, scale bars: 50 μm). (B) The quantification of lung fibrosis is shown as arbitrary units. n = 6; *p < 0.05 versus NS + PBS group; #p < 0.05 versus LPS + PBS group; &p < 0.05 versus LPS + mMSC control group.
Discussion
MSC therapy may be a promising and novel treatment for ARDS 27 because of the ability of MSCs to migrate to and engraft in the injured lung and differentiate into lung epithelial cells in vivo7–10. However, the engraftment and differentiation rates of MSCs in the injured lungs have been relatively insufficient7,28,29, which has limited their beneficial effects for ARDS therapy. There is growing evidence suggesting that specific signaling pathways are involved in regulating the biological behavior of MSCs, and intervention in these signaling pathways may further improve the therapeutic potential of these cells30,31. In our previous in vitro study, we found that activation of the noncanonical Wnt5a/JNK and/or Wnt5a/PKC pathway(s) promotes differentiation of mMSCs into type II alveolar epithelial cells, confers resistance to oxidative stress, and promotes their migration to injured lung tissue 19 . This indicated that the noncanonical Wnt pathway may contribute to the improvement of the therapeutic effects of MSCs in ARDS. In the present study, we confirmed this positive effect of the noncanoncial pathway in LPS-induced ARDS mice using MSCs stably transfected with the ROR2 gene.
There are about nine pathways in the transduction of noncanonical Wnt signaling that have been disclosed, but the Wnt/planar cell polarity (PCP) (also referred to as Wnt/JNK) and Wnt/Ca2+ signaling are the two most studied molecular signaling pathways. The Wnt/JNK pathway is initiated by the activation of frizzled (FZD) and ROR2 by Wnts32,33, which is critical for many biological behaviors, including cell differentiation, proliferation, and apoptosis34–36. In the Wnt/Ca2+ pathway, the binding of the Wnts to their receptors increases an intracellular release of Ca2+ or an extracellular Ca2+ influx, which then activates PKC and CaMK II. It was found that PKC could regulate cell adhesion and tissue development37,38. Wnt5a, through binding to its receptor, ROR2, regulates various cellular functions, including cell migration, polarity, invasion, or differentiation12–16, and is the most studied molecule in the noncanonical Wnt pathway. Several studies have found that ROR2 is the key receptor for the activation of the noncanonical Wnt pathway. Therefore, we selected ROR2 as the target gene for the regulation of the noncanonical Wnt5a/ROR2 pathways. To obtain MSCs with stable, long-term transgene expression of ROR2, lentiviral vectors were used, and the transduction efficiencies were as high as 95% even 20 passages after transduction. We also confirmed that the overexpression of ROR2 increased the activation of the Wnt5a/JNK and Wnt5a/PKC signaling in mMSCs in our previous study.
This construction of stable, long-term mMSC lines with ROR2 gene alteration was also confirmed in our previous study by changes in cellular phenotype as well as altered biological behaviors including proliferation, differentiation, and migration of mMSCs 20 . These characteristics suggested that the gene modification might facilitate ARDS therapy, and the observation in the present study that the administration of ROR2-overexpressing mMSCs improved both the histopathological morphology and survival of ARDS mice compared with mMSCs alone confirmed the benefits of using ROR2-overexpressing mMSCs for ARDS.
The ability of MSCs to differentiate into specialized cell types, mediating the repair of injured tissue, is based on their concentration and localization in the injured sites. Several studies have observed enhanced recruitment of transplanted MSCs to the injured lung tissue in ARDS mice compared to normal control mice8,39. Our previous study also found that the migration of mMSCs to injured lung tissue through Transwell chambers was significantly increased when the activation of the noncanonical PKC or JNK pathway was stimulated by Wnt5a 19 . Our other previous study found that ROR2 overexpression promoted mMSC horizontal and vertical migration ability in vitro 20 . In agreement with these studies, our present study showed that the overexpression of ROR2 increased the retention of mMSCs in the lungs of ARDS mice after LPS challenge.
Besides migration, the proliferation and survival of MSCs in the injured tissues after transplantation also played an important role in their retention. Low survival of grafted MSCs limits their transdifferentiation and positive effects on tissue repair 40 . In our previous study, we found that Wnt5a supplementation, through Wnt/PKC signaling, increased the survival of mMSCs after being treated with H2O2; this resulted in the production of oxidants, which occurs in inflamed lung tissue and also causes lung injury 41 in vitro 19 . In another previous study, we found that overexpression of ROR2 in mMSCs significantly increases their proliferation 20 . All of these mechanisms may contribute to the elevated retention of mMSCs in the lungs after LPS challenge.
MSCs are multipotent adult stem cells that have the capability of differentiating into multiple cell types5,6. Many recent studies have demonstrated that MSCs can differentiate into lung epithelial cells in vivo7,9,10. We also found in this study that MSCs could differentiate into AT II cells in IT LPS-induced ARDS mice and that overexpression of ROR2 increased the differentiation rate. AT II cells are considered critical for the repair of injured lung tissue in ARDS patients42,43. Many investigations, including our previous study, have revealed that MSCs could differentiate into AT II cells in ARDS animals and in some in vitro conditions and that these cells were involved in the repair of the alveolar epithelium in ARDS19,44–48. However, the differentiation rate of MSCs in injured lungs was limited7,46; for instance, the differentiation rate of MSCs into type II alveolar epithelial cells is only about 26% following bleomycin-induced lung injury in mice 46 . The role of the noncanonical Wnt pathway in the differentiation of mMSCs into AT II cells has been elucidated in our previous in vitro study 19 ; however, its effect on the differentiation of mMSCs into AT II cells in mice with LPS-induced lung injuries has not been thoroughly explored. Our results showed that the differentiation rate of MSCs into type II alveolar epithelial is about 22% following LPS-induced lung injury in mice, which is comparable with previous studies. Consistent with our preliminary in vitro study 20 , our results demonstrated that overexpression of ROR2 promotes the differentiation of MSCs into AT II cells in vivo. ROR2, which is indispensable for mouse development and morphogenesis, plays a critical role in the differentiation of MSCs 49 . Recent evidence suggests that through the simulation of Wnt5a, ROR2 is required for the osteoblastogenesis of MSCs13,16. Several investigators observed that ROR2 initiated commitment of MSCs to the osteoblastic lineage and promoted differentiation at both the early and late stages of osteoblastogenesis. Interestingly, other studies found that ROR2 overexpression promoted osteogenic differentiation while inhibiting the adipogenic differentiation of human MSCs16,50. Some other studies also found that Wnt5a/ROR2 promotes MSC differentiation into osteoblasts or chondrocytes13,18. In our previous in vitro study, we found that ROR2 gene overexpression promoted mMSC differentiation into osteoblasts while inhibiting the adipogenic differentiation of mMSCs 20 . In addition, we found that the overexpression of ROR2 in mMSCs, stimulated by Wnt5a, upregulated the downstream signaling of ROR2 including JNK and PKC in the noncanonical Wnt pathway 20 . Our previous study also found that Wnt5a was increased in the lung tissue of ARDS mice after LPS (IT) administration, and Wnt5a, either through Wnt/JNK signaling alone or through the combination of Wnt/JNK and Wnt/PKC signaling, promoted the differentiation of mMSCs into AT II cells 19 . AT II cells are characterized by their ability to synthesize and secrete alveolar surfactant that reduces surface tension and prevents collapse of the alveoli and by their ability to differentiate into type I alveolar epithelial cells (AT I cells) that serve as progenitor cells for reepithelialization of impaired alveoli, which are significant in both physiological and pathological conditions 43 . Our present study found that overexpression of ROR2 in mMSCs significantly increased the differentiation of MSCs into type II alveolar epithelial cells in the lungs after LPS challenge. From these results, we speculate that noncanonical Wnt5a/ROR2 signaling plays a critical role in the differentiation of mMSCs into pneumonocytes in vivo. It is worth noting that the increased SP-C protein in lungs shown in our results might be attributed not only to differentiation of MSCs but also to AT II cell survival or proliferation, and even may be attributed to the reepithelization of the local progenitor cells induced by MSCs, so further research is needed.
Injury of the alveolar epithelium in ARDS is considered to be a key factor in the leakage of protein-rich edema fluid into the interstitium and alveolar space 51 . Many studies have found that MSC treatment significantly reduces the lung wet-to-dry ratio, the amount of excess lung fluid, and the level of BALF protein, a marker of endothelial and epithelial permeability, in experimental ARDS28,52. In our study, we demonstrated that the ROR2-overexpressing mMSCs also significantly improved epithelial permeability, as evident from LWW/BW, and BALF ALB and TP.
The permeability barrier in the terminal airspace of the lung is largely due to tight junctions between alveolar epithelial cells 53 . Occludin is one of the well-characterized tight junction proteins and provides most of the barrier function of tight junctions 54 . Functional opening of the tight junction barrier and downregulation of occludin protein expression have been observed in acute lung injury in adult animals 55 . Our results presented here showed that the expression of occludin protein in the lung tissue was downregulated after LPS challenge and that mMSC treatment inhibited the repression of occludin protein. Moreover, the overexpression of ROR2 in mMSCs may provide additional benefits.
KGF, which is capable of stimulating proliferation and migration in AT II cells, leading to the propagation of alveolar epithelial restitution 56 and reduction of lung edema and inflammation, was mechanistically implicated in the beneficial effect of MSCs on alveolar fluid clearance in ARDS. In our study, the results showed that treatment with ROR2-overexpressing mMSCs markedly increased BALF KGF compared with treatment with mMSCs alone at day 14 after LPS administration. These results indicated that the further improvement in lung permeability observed after treatment with ROR2-overexpressing mMSCs may contribute to the repair of tight junctions and the increased paracrine signaling of KGF from the mMSCs.
MSCs have a role in immunological regulation through influencing the activity of a broad range of immune cells57,58. Several investigations including our present study found that there was a significant decrease in systemic concentrations in the levels of LPS-induced proinflammatory cytokines interferon-γ (IFN-γ), IL-1β, IL-6, and increased anti-inflammatory cytokine IL-10 after infusion of MSCs in mice28,39. Moreover, in the present study, we found that overexpression of ROR2 in mMSCs amplified this effect. However, the mechanisms underlying these effects remain unclear and should be explored in the future.
In addition to the alveolar epithelial cells, engrafted MSCs in the lungs could also differentiate into lung fibroblasts, myofibroblasts, and interstitial monocytes, which may participate in pulmonary fibrosis 46 . We evaluated whether the ROR2-overexpressing mMSCs or control mMSCs alone increased the risk of pulmonary fibrosis. The results of the present study showed that lung fibrosis decreased after treatment with control mMSCs alone and that this effect was more significant after the engraftment of ROR2-overexpressing mMSCs.
In conclusion, overexpression of ROR2 increased the retention of mMSCs in the lung, promoted the differentiation of mMSCs into AT II cells, further improved the lung epithelial permeability, attenuated acute pulmonary inflammation, and inhibited lung fibrosis compared with mMSCs alone. The activation of the noncanonical Wnt pathway by overexpressing ROR2 could further improve the protection of mMSCs against epithelial impairment and contribute to the therapeutic effects of mMSCs in ARDS mice.
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 81070049, 81170057, 81201489, 81372093, 81300060, and 81300043), the Natural Science Foundation of Jiangsu Province of China (Nos. BK2011600 and BK20131302), and the Graduate Innovation Project in Jiangsu Province of China (Nos. CXLX13_123 and CXCL_151). The authors declare no conflicts of interest.
