Abstract
Liver fibrosis is characterized by excessive accumulation of extracellular matrix components in the liver parenchyma that distorts the normal architecture and hepatic function. Progressive fibrosis could end in the advanced stage known as cirrhosis, resulting in the need to resort to liver transplantation. Amniotic membrane (AM) has emerged as an innovative therapeutic approach for chronic liver diseases due to its anti-inflammatory, antiscarring, and wound-healing effects. We have recently shown that AM can be used as a patch on the liver surface at the same time of fibrosis induction, resulting in significantly reduced progression and severity of biliary fibrosis. Here we investigated the effects of human AM on the established rat model of liver fibrosis, induced by the bile duct ligation (BDL). We also explored the effect of AM on the expression of transforming growth factor-β1 (TGF-β1), the main profibrogenic factor in hepatic fibrosis, and the proinflammatory cytokines, tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and anti-inflammatory cytokine IL-10. Two weeks after BDL, the liver was covered with a fragment of AM or left untreated. Six weeks later, the fibrosis was first assessed by the semiquantitative Knodell and the METAVIR scoring systems and, thereafter, by CellProfiler digital image analysis to quantify the area occupied by collagen deposition, ductular reactions (DRs), activated myofibroblasts, and TGF-β1. The hepatic cytokines were determined by ELISA. AM-treated rats showed a significantly lower score compared to the control BDL rats (2.5 ± 0.9 vs. 3.5 ± 0.3, respectively; p < 0.05). The collagen deposition, DRs, number of activated myofibroblasts, and TGF-β1 were all reduced to about 50% of levels observed in untreated BDL rats. These findings suggest that AM, when applied as a patch onto the liver surface, is useful for treating well-established cholestatic fibrosis, and the mechanism was partly by means of downregulating the profibrotic factor TGF-β1 and IL-6.
Keywords
Introduction
Liver fibrosis is an excessive wound-healing response of the liver to repeated injury that occurs in most chronic liver diseases. It is characterized by an excessive production of extracellular matrix (ECM), mostly collagen type I, and scar formation. Myofibroblasts are the major effector cells during the development of liver fibrosis (3). They originate from liver-resident hepatic stellate cells (HSCs) and portal fibroblasts (PFs) following a process termed “activation.” This process is mostly trigged by profibrogenic cytokines including transforming growth factor-β (TGF-β) and interleukin-6 (IL-6) (8,13,49). If hepatic injury persists, the progressive fibrosis could end in the advanced stage known as cirrhosis, which is characterized by a distortion of the liver architecture with fibrotic bands surrounding regenerative nodules of hepatic parenchyma (32,39). If the hepatic injury is removed, regression of fibrosis is associated with a decrease in levels of fibrogenic cytokines and ECM production, increased collagenase activity, dissolution of the fibrous scar, and disappearance of activated myofibroblasts (3,19,49). Globally, progressive hepatic fibrosis and cirrhosis constitute a major health care burden and are closely linked epidemiologically to hepatocellular carcinoma risk, with a further increase in the relative mortality rate (37,39). In Brazil, between 2001 and 2010, cirrhosis was the leading cause not only of hospitalization but also the eighth leading cause of death (29).
Human amniotic membrane (hAM) is emerging as a potential source of stem/progenitor cells and as a whole tissue for the development of therapeutic strategies in regenerative medicine to restore damaged or diseased tissues and organs (10). hAM is the innermost layer of the fetal membranes, usually discarded after birth as part of the placenta, making it readily available in large amounts and free from ethical debate (30). The cells in hAM express markers of mesenchymal as well as embryonic stem cells (MSCs and ESCs, respectively) and are capable of differentiating, in vitro, toward tissues of all three germ layers (28,31), including the hepatogenic lineage in vitro and in vivo with the production of albumin and glycogen storage (42,43). Furthermore, hAM-derived cells have low immunogenicity, which explains the absence of acute rejection observed when cells or intact hAM was applied, in vivo, by using experimental xenogeneic (2,5,47) and allogeneic (5,17) transplantation models.
Preclinical studies using the toxin carbon tetrachloride (CCl4)-induced liver fibrosis model suggest that hAM-derived cells might be useful for restoring the functionality of hepatic tissues that have been compromised by disease or injury (26,50). In addition to the amniotic cells, the hAM itself can exert ameliorative effects. It has been reported for the first time as a biological dressing to heal skin wounds a century ago. Since then, several beneficial properties of hAM, including its antiangiogenic, immuno modulatory, anti-inflammatory, antiscarring, anti microbial, wound-healing, or regenerative properties, and pain-reducing effects (9,24,27,38) have raised interest in the possibility of using hAM in several clinical conditions, including dermal burns and ocular disorders (25), and as a biomaterial for reconstruction of oral mucosa and pharyngeal defects (18), and for abdominal wall (31) and bone defects (40).
These ongoing applications led to experimental studies that demonstrated the beneficial effects of hAM on inflammation and fibrosis in lungs and liver [reviewed in Manuelpillai et al. (27)]. In addition, using BDL-induced rat liver fibrosis, we have previously demonstrated that hAM used as a patch on the liver surface at the same time as fibrosis induction significantly reduces the progression and severity of biliary fibrosis. Considering these previous results and the fact that the treatment after the development of fibrosis, rather than at the initiation of the fibrotic stimulus, mimics human disease more closely, the aim of this study was to investigate the potential use of hAM in a BDL model of established fibrosis, thus applying the membrane 2 weeks after the BDL procedure. In an attempt to elucidate the potential mechanism(s) underlying the beneficial effects of AM, we also explored profibrotic TGF-β1 and pro- and anti-inflammatory cytokines [tumor necrosis factor-α (TNF-α), IL-6, and IL-10].
Materials and Methods
Animal and Experimental Groups
All experiments and procedures were approved by an Institutional Animal Care and Use Committee (IACUC) of the University of Vale do Paraíba under the protocol number A030/2011 in accordance with the Brazilian Federal law 11.794/2008. Twenty male Wistar rats, weighing 200–220 g, were obtained from the Multidisciplinary Center for Biological Investigation (CEMIB, UNICAMP, Campinas, Brazil) and randomly divided into two experimental groups of eight rats each. For the BDL group, rats were subjected to the BDL procedure, and for the BDL + hAM group, rats were subjected to the BDL procedure and treated with hAM 2 weeks after. At the moment of hAM application, the rats of the BDL group were subjected only to laparotomy and manipulation of the liver but without hAM application. All rats were euthanized at 6 weeks after BDL. In addition, four rats were also subjected to BDL and euthanized 2 weeks after this procedure in order to observe the degree of fibrosis at the moment of hAM application.
Human AM Collection and Processing
Human term placentas were obtained from cesarean sections with maternal consent and after testing negative for human immunodeficiency virus (HIV), hepatitis B and C, and syphilis in accordance with the Ethical Committee of the Hospital Municipal Dr. José de Carvalho Florence, in São José dos Campos, and approved by the Research Ethics Committee (CEP) of São José dos Campos Dental School (FOSJC), São Paulo State University (UNESP), under protocol number 063/2011. Fresh hAM was obtained and processed as previously described (35), except for the size of the hAM fragments, which were 9 cm × 9 cm, in order to cover the whole fibrotic liver.
Bile Duct Ligation Procedure
In order to induce liver fibrosis, bile duct ligation (BDL) was performed in rats as previously described (35). In animals of the hAM-treated group, the rats were also subjected to the BDL procedure. Two weeks after BDL, a hAM fragment was placed onto the liver with the stromal layer in contact with the liver surface, in such a way that the whole surface of the liver lobules was covered (Fig. 1). In the BDL group, 2 weeks after BDL induction, the rats underwent laparotomy and were subjected to the same manipulation procedure as the hAM-treated group, except for the AM application.
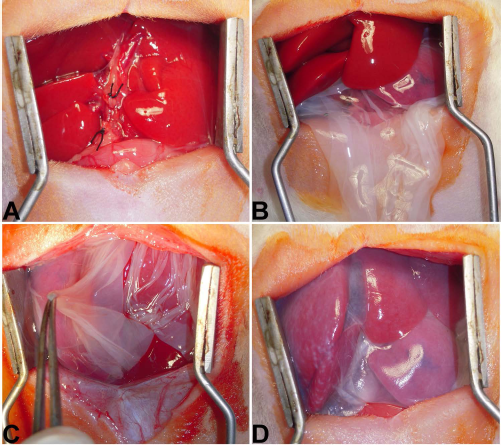
The procedures of transplanting human amniotic membrane (hAM) onto liver. Common bile duct was exposed, double ligated, and cut between the ligatures (A). AM fragment was inserted under the liver lobes (B), and the extremities were raised (C) and attached to each other to cover the whole liver surface (D) with its mesenchymal side in contact with the liver (epithelial side up).
Liver Explants and Histological Processing
Two or 6 weeks after BDL, animals from each group were euthanized with an excess of isoflurane (Vetflurano®; Virbac, São Paulo, Brazil). Liver samples were taken from the major left and median lobes (that comprise approximately 70% of the liver), fixed in 10% neutral-buffered formalin (Synth, São Paulo, Brazil) for 24 h, and embedded in paraffin (Paraplast; Oxford, St. Louis, MO, USA). At least four 4-μm-thick sections were used from each rat and stained with Sirius Red (EasyPath, São Paulo, Brazil) for fibrosis and collagen analysis, according to the manufacturer's instructions or placed on polysine-L-coated slides (Menzel-Glaser, Braunsch, Germany) for immunohistochemical stainings.
Semiquantitative Evaluation of Liver Fibrosis
The degree of liver fibrosis was examined semiquantitatively under a bright field Nikon Eclipse E200 microscope (Lupetec, São Paulo, Brazil) at 100× magnification by the Knodell scoring systems as previously described (36). In order to decrease the subjectivity of the semiquantitative analysis and increase its reproducibility, another classical system, METAVIR, was performed according to Ricci et al. (34). For each scoring system, the average score taken from 10 nonoverlapping random fields per section was used to generate a single score for each specimen, in each scoring system.
Immunohistochemistry and Quantitative Evaluation
Immunostainings for cytokeratin 19 (CK19) and α-smooth muscle actin (α-SMA) were performed to identify bile ducts and bile ductules [ductular reactions (DRs)] and activated myofibroblasts, respectively. The expression of the profibrogenic factor in hepatic tissue was examined by immunostaining for TGF-β1. Briefly, each section was deparaffinized and hydrated in graded ethanol and immersed into the preheated retrieval solution (citrate buffer 10 mM, pH 6.0; Synth) for 20 min at 98°C. A further digestion of 10 min in trypsin solution (Gibco®, Life Technologies, São Paulo, Brazil) at 37°C was necessary for staining with the anti-CK19 and anti-TGF-β1 antibodies. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide (Dinâmica, São Paulo, Brazil) for 15 min followed by three washes in phosphate-buffered saline (PBS) (Synth) containing 0.05% Tween 20 (Sigma-Aldrich, St. Louis, MO, USA), and unspecific labeling was reduced by 20-min incubation with normal horse serum (ImmPRESS Reagent Kit; Vector Laboratories, Burlingame, CA, USA). Then sections were incubated overnight in a humidity chamber at 4°C with primary mouse monoclonal antibodies against anti-human α-SMA (clone 1A4; dilution 1:600; Dako Corporation, Glostrup, Denmark) and against CK19 (Clone b170; dilution 1:100; Novocastra Laboratories, Newcastle, UK), and with primary rabbit polyclonal antibody against TGF-β1(sc-146; dilution 1:350; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). Sections were again washed and incubated with secondary antibody using the ImmPRESS Universal Reagent Anti-Mouse/Rabbit Ig Polymer Detection Kit (Vector Laboratories) according to the manufacturer's instructions. For visualization, 3,3′-diaminobenzidine (ImmPACT DAB; Vector Laboratories) was applied for 2 min. All sections were counterstained with hematoxylin and mounted with Permount® (Thermo Fisher Scientific, Fair Lawn, NJ, USA).
Sirius Red staining, CK19, α-SMA, and TGF-β1 immunostainings were then quantitatively evaluated by digital image analysis using the open-source CellProfiler cell image analysis software, previously configured for biliary fibrosis (36) and described previously (35).
Hepatic Cytokine Determinations
The cytokines were determined by enzyme-linked immunosorbent assay (ELISA) on liver homogenates. For liver homogenization, the rats were euthanized, and pieces of 250 mg of livers were snap frozen (in liquid nitrogen) and stored at −80°C until further analysis. The snap-frozen tissue pieces were ground using a precooled mortar and pestle. After the grinding process was completed, 2.5 ml of cold PBS containing 5% goat serum and 10 μl/ml of protease inhibitor cocktail (P8340; Sigma-Aldrich) was added. Homogenates were centrifuged three times at 10,621 × g for 30 min at 4°C. Then supernatants were kept at −80°C until cytokines analyses were performed. TNF-α, IL-6, and IL-10 concentrations were determined by ELISA sandwich according to the manufacturer's instructions (Invitrogen, Carlsbad, CA, USA). Samples were quantified against a standard curve, and the optical density was read at 450 nm on a microplate reader (uQuant; Biotek Instruments Inc., Winooski, VT, USA).
Statistical Analysis
Data are reported as mean ± standard deviation (SD). The two-tailed Student's t-test was used to evaluate the statistical significance of the difference among experimental groups. Data analysis was performed using the BioEstat software, version 5.3 (Mamirauá Institute, Tefé, Amazônia, Brazil). A value of p < 0.05 was considered statistically significant.
Results
Effects on Liver Fibrosis Degree
The degree of fibrosis was evaluated at 2 weeks post-BDL. Figure 2A demonstrates periductular collagen deposits along the enlarged portal tracts. These pathological alterations are characteristics of Knodell score 1 and confirmed the presence of fibrosis at the moment of hAM application on livers. Semiquantitative evaluation of fibrosis at 6 weeks from fibrosis induction by Knodell and METAVIR scoring systems in both BDL and BDL + AM groups is reported in Figure 2B. Applying Knodell scoring, we observed that rats from the hAM-treated group showed a significantly lower score compared to the untreated group (2.5 ± 0.9 × 3.6 ± 0.3; p < 0.05), respectively. Analysis performed by adopting the METAVIR scoring system confirmed the findings above (2.4 ± 0.9 × 3.4 ± 0.4; p < 0.05).

Semiquantitative evaluation of fibrosis. (A) Two weeks from fibrosis induction: collagen deposits can be recognized by red staining of fibers, whereas hepatic parenchyma is stained in pale yellow. Periductular fibrosis (thick arrow) is detected along enlarged portal tracts. CV, central vein; PT, portal tract; Sirius Red. (B) Six weeks from fibrosis induction: evaluation of fibrosis degree by Knodell and METAVIR scoring of experimental groups. Means ± standard deviation (SD) of eight independent experiments are represented as bars for bile duct ligation (BDL, black) and BDL + hAM (gray) groups. ∗p < 0.05 versus BDL group.
Effects on Collagen Deposition, DRs, Myofibroblasts, and TGF-β1
Figure 3 shows low-magnification microphotographs of histochemical and immunohistochemical staining for all parameters of biliary fibrosis in experimental groups. Bile duct ligation increases collagen deposits, DRs, myofibroblasts, and TGF-β1 (Fig. 3A, C, E, and G). Meanwhile, in the BDL + hAM group, all of these parameters were reduced into entire liver tissue, including areas near the hAM as well as in the most central areas of the liver lobe (Fig. 3B, D, F, and H).

Human amniotic membrane (hAM) attenuates all parameters of well-established cholestatic fibrosis. (A, C, E, G) Rat liver, bile duct ligation (BDL) group: representative microphotographs at 6 weeks. (B, D, F, H) Rat liver, BDL + AM group: representative microphotographs at 6 weeks. (A, B) Sirius Red staining for collagen. (C, D) Cytokeratin 19 (CK19) immunostaining for ductular reaction. (E, F) α-Smooth muscle actin (α-SMA) immunostaining for myofibroblasts. (G, H) The profibrotic transforming growth factor-β1 (TGF-β1) immunostaining. Scale bars: 500 μm.
The results of Sirius Red staining, CK19, SMA, and TGF immunostainings, and its quantitative evaluation by digital image analysis are demonstrated in Figures 4-7, respectively. At week 6, the area occupied by collagen deposition in BDL + hAM rats was only 14.71 ± 6.2%, while in BDL rats, the percentage of collagen deposition markedly increased (p < 0.01) involving 25.66 ± 5.3% of the liver parenchyma area (Fig. 4E). The CK19-positive stained cells represent the DR. In the BDL group, the mean area occupied by DRs was 24.33 ± 5.5%, while in the BDL + hAM group, the area was significantly lower, occupying only 11.42 ± 6.9% (p < 0.05) of the liver (Fig. 5E). Quantitative image analysis of α-SMA-positive stained cells revealed that, while in the BDL group the percentage of area occupied by myofibroblasts was 15.83 ± 5.2%, in rats treated with hAM, the area was significantly reduced (8.57 ± 4.9%; p < 0.05) (Fig. 6E). The same analysis showed that the percentage of the TGF-β1-positive area in rats treated with hAM was lower than that in the BDL-only group (10.14 ± 3.62 vs. 26.83 ± 4.22, respectively), and the difference between the groups was extremely significant (p < 0.001) (Fig. 7E).

Histochemical staining and quantitative evaluation of collagen deposits in experimental groups. (A) Bile duct ligation (BDL) group—intense periductular fibrosis (thick arrow) forming bridging septa between adjacent portal tracts and between portal tracts and the central vein. The interstitial collagen fibers (thin arrow) are evident in perisinusoidal spaces (or space of Disse) dividing the parenchyma in cirrhotic nodules (∗), leading to loss of lobule architecture. (B) BDL + human amniotic membrane (hAM) group—less robust periductular fibrosis (thick arrow) with less and thin fibrotic septa (short arrow), but without interstitial collagen fibers. The architecture of hepatic lobule is more preserved than in the BDL-only group. CV, central vein; PT, portal tract. Sirius Red. (C) Sample microphotograph taken before the image analysis procedure used to extrapolate the collagen staining from the image. (D) Sample image, same as in (C), representing a step of the image analysis procedure used: white areas are representative of red collagen staining. (E) Values of collagen deposition in different groups: means ± standard deviation (SD) of eight independent experiments are represented as bars for BDL (black) and BDL + hAM (gray) groups. ∗∗p < 0.01 versus BDL group. Scale bars: 200 μm.

Immunohistochemical and quantitative evaluation of cytokeratin 19 (CK19) staining in experimental groups. (A) Bile duct ligation (BDL) group—intense ductular reaction with CK19-positive biliary structures expanding to form thick septa that connect adjacent portal tracts and finally isolate small nodules or “islands” of hepatocytes (∗). CK19-positive small ductules arising within these nodules can be observed. (B) BDL + human amniotic membrane (hAM) group—CK19-positive biliary structures are lower than in the BDL group. (A) (BDL group) The septa connect adjacent portal tracts, and (B) (BDL + AM group) the septa were thin, and they did not connect adjacent portal tracts. CV, central vein; PT, portal tract. (C) Sample microphotograph taken before image analysis used to extrapolate the CK19 staining from the image. (D) Sample image, same as in (C), representing a step of the image analysis procedure used: white areas are representative of CK19 staining. (E) Values of CK19-immunostained cells in different groups: means ± standard deviation (SD) of eight independent experiments are represented as bars for BDL (black) and BDL + hAM (gray) groups. ∗p < 0.05 versus BDL group. Scale bars: 200 μm.
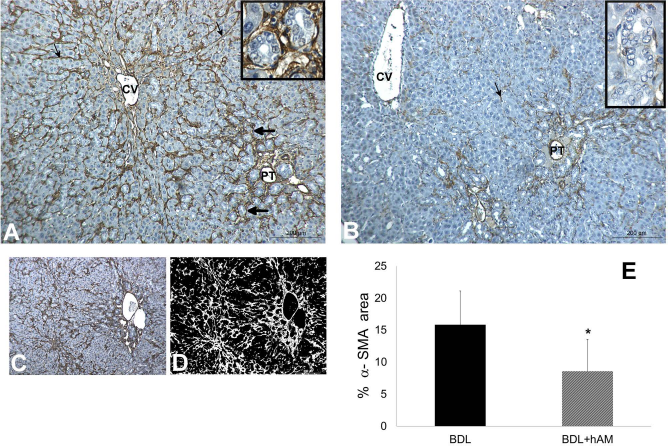
Immunohistochemical and quantitative evaluation of α-smooth muscle actin (α-SMA) staining in experimental groups. (A) Bile duct ligation (BDL) group—α-SMA-positive cells completely surround a large number of the biliary structures (thick arrow and in box) and are detected along perisinusoidal spaces (thin arrow). (B) BDL + human amniotic membrane (hAM) group—α-SMA-positive cells are scarcer than in the BDL group. Note that only a few α-SMA-positive cells are detected along perisinusoidal space (thin arrow), and not all biliary structures are surrounded by α-SMA-positive cells (in box). CV, central vein; PT, portal tract. (C) Sample microphotograph taken before the image analysis procedure used to extrapolate the α-SMA staining from the image. (D) Sample image, same as in (C), representing a step of the image analysis procedure used: white areas are representative of α-SMA staining. (E) Values of α-SMA-immunostained cells in different groups: means ± standard deviation (SD) of eight independent experiments are represented as bars for BDL (black) and BDL + hAM (gray) groups. ∗p < 0.05 versus BDL group. Scale bars: 200 μm.

Immunohistochemical and quantitative evaluation of transforming growth factor-β1 (TGF-β1) staining in experimental groups. (A) Bile duct ligation (BDL) group—strong expression of TGF-β1 in connective tissue of portal tracts (PT) and mainly around biliary structures from septa (thick arrow) that connect adjacent portal tracts and perisinusoidal spaces (thin arrow) distorting the hepatic parenchyma. (B) BDL + human amniotic membrane (hAM) group—weak expression of TGF-β1 in both around the biliary structures and in perisinusoidal locations with the integrity of hepatic parenchyma preserved. CV, central vein. (C) Sample microphotograph taken before the image analysis procedure used to extrapolate the TGF-β1 staining from the image. (D) Sample image, same as in (C), representing a step of the image analysis procedure used: white areas are representative of α-smooth muscle actin (α-SMA) staining. (E) Values of TGF-β1-immunostained cells in different groups: means ± standard deviation (SD) of eight independent experiments are represented as bars for BDL (black) and BDL + hAM (gray) groups. ∗∗∗p < 0.001 versus BDL group. Scale bars: 200 μm.
Effects on Hepatic Cytokines
Proinflammatory (IL-6 and TNF-α) and anti-inflammatory (IL-10) cytokines were determined by ELISA on liver homogenates in both experimental groups (Fig. 8). IL-6 cytokine levels were significantly lower in the group treated with hAM than the BDL-only group (138.32 ± 43.1 pg/ml vs. 217 ± 63 pg/ml; p < 0.05, respectively). IL-10 cytokine levels were higher in the BDL + hAM group (363.36 ± 165 pg/ml) than in the BDL group (323.29 ± 103.13 pg/ml). No significant differences were found in TNF-α between the two groups (BDL + hAM: 287.36 ± 68.4 pg/ml vs. BDL: 310.8 ± 72.4 pg/ml; p > 0.05).

Hepatic of proinflammatory [interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α)] and anti-inflammatory (IL-10) cytokine levels in rat liver 6 weeks from fibrosis induction. Means ± standard deviation (SD) of eight independent experiments are represented as bars for bile duct ligation (BDL, black) and BDL + human amniotic membrane (hAM, gray) groups. ∗p < 0.05 versus BDL group.
Discussion
We have previously demonstrated that the hAM, when applied as a patch onto the liver surface at the initiation of the fibrotic stimulus, does not prevent the formation of fibrosis but certainly delays progression to the end-stage disease (35).
Liver fibrosis and end-stage cirrhosis represent the common pathway of virtually all chronic liver diseases that often develops insidiously over many years, meaning that patients can present at an advanced stage of disease, with no guarantee that currently available treatments to remove the underlying cause will prove successful (39). The aim of the present study was to clarify if the beneficial effects of AM observed in a previous study (35) could also be applied in a more chronic status that more closely resembles human disease using a model that mimics more closely human liver fibrosis.
The common BDL model is a widely accepted experimental model in rodents. This model reproduces and allows for the study of the pathological features of biliary-type fibrosis that arise in a variety of human chronic liver diseases resulting from extrahepatic cholestasis such as extrahepatic, biliary atresia, and primary sclerosing cholangitis (20,21). The maneuver of BDL introduces biomechanical stress to biliary epithelium and initially triggers the compensatory proliferation and expansion of biliary epithelial cells (BECs) and looping and reduplication of ducts and ductules (11,12,21). Following this epithelial mitogenic phase, chronic obstruction of bile duct causes massive activation of myofibroblasts in the periductal region and ultimately results in biliary fibrosis/cirrhosis over a short period of time (4–6 weeks in rats) (48).
Combining a semiquantitative evaluation of the fibrotic process and the quantitative assessment of three main parameters specifically involved in the establishment and progression of biliary fibrosis (i.e., ductular reaction, myofibroblast activation, and collagen deposition), our findings indicate that the application of hAM, after fibrosis has developed, delays the cascade of events leading to liver fibrosis in the BDL model, resulting in a final reduction of biliary fibrosis.
Primarily, we have demonstrated that hAM application reduces the severity of liver fibrosis when evaluated using both the semiquantitative Knodell and METAVIR scoring systems, which allow a grading and staging of fibrosis, by considering the progressive expansion of ECM deposition (i.e., collagen deposition) from the portal tracts to gradually involving the hepatic parenchyma. Indeed, in the BDL group, fibrosis reaches advanced-stage cirrhosis within 6 weeks, with collagen accumulation around biliary structures and in the interstitial spaces. In the group treated with hAM, the fibrosis was confined to the portal/periportal area, thus preserving the architecture of liver parenchyma. Moreover, the quantitative image analysis confirmed the decrease in collagen deposition by about 50% in the treated group. These data indicate that treatment with hAM reduces both the degree or extent of fibrosis and also the amount of collagen in liver tissue.
Herein, we have shown that the application of human AM 2 weeks after fibrosis establishment reduced the area occupied by DRs. This is a very important finding, considering that the DR is one of the first pathophysiological events triggered by the BDL procedure and has a key role in the initiation and progression of biliary fibrosis (12,21). These results are in line with Chen et al. (6) and Al-Hayani (1), which demonstrated that chromium and selenium supplementation, respectively, attenuated biliary fibrosis, first evidenced by a decrease in the extension of DR. In addition, Beaussier et al. revealed that biliary-type liver fibrosis develops close to the DR, and the progression rates of fibrosis and of DR significantly correlate with each other (4). We have also demonstrated that the immunoexpression of α-SMA, which characterizes myofibroblasts, was markedly reduced by the application of hAM onto the liver surface. Importantly, not all periductular cells were α-SMA immunopositive. Considering that one of the mechanisms of fibrosis reduction is apoptosis/phenotype reversion in myofibroblasts (19,49) and that the AM induces apoptosis/phenotype reversion in these cells (15,22), it is possible that the reduced immunoexpression of α-SMA was an action of hAM in these cell death events. However, whether the hAM reduces the number of myofibroblasts through phenotype reversion or apoptosis remains to be tested in the BDL-induced fibrosis model. Taken together, our data indicate that the decrease in DR and in myofibroblast activation caused by the hAM may have contributed to the attenuation of collagen deposition in liver tissue, therefore reducing the fibrosis caused by BDL. These results are in parallel with previous studies that observed antifibrotic effects of cryopreserved or fresh hAM when applied immediately after BDL stimulus (34,35), indicating that, both at the beginning and 2 weeks after BDL, the hAM is able to reduce fibrosis and prevent the progression to cirrhosis.
In order to elucidate the potential mechanism(s) underlying the beneficial effects of hAM, specifically in the reduction of biliary fibrosis, our present work addressed some open questions that remained without answers in our previous results (35). Recent studies have demonstrated that mechanisms of liver fibrosis regression are characterized by decreased inflammatory and fibrogenic cytokine levels, increased collagenase activity, and the disappearance of myofibroblasts and fibrous scars (19,41). In the present study, we explored the action of hAM on the expression of the fibrogenic TGF-β1, and then the levels of pro- and anti-inflammatory cytokines (TNF-α, IL-6, and IL-10) in hepatic tissue.
Because of its central role during progression of fibrosis, TGF-β is considered a major therapeutic target for fibrosis (7). The reduction of TGF-β expression has been previously suggested to explain part of the mechanism of liver fibrosis reduction achieved by other approaches, such as hepatocyte growth factor (HGF) administration (48), curcumin (33), MSC transplantation (44), chromium supplementation (36), and the medical plant Artemisia iwayomogi Kitamura (14). According to Dooley and Dijke, TGF-β is a pleiotropic multifunctional cytokine that exerts effects on different cells, thus contributing to all stages of disease progression from initial liver injury through inflammation and fibrosis to cirrhosis and hepatocellular carcinoma (7). In our study, after 6 weeks from BDL, the immunolocalization of TGF-β1 was confirmed in different cells, including Kupffer and endothelial cells, fibroblasts of portal space, epithelium of bile ducts, and also necrotic hepatocytes (data not shown). These findings suggest a general response to chronic injury caused by the BDL procedure. During hepatic fibrogenesis, TGF-β has a pivotal role in the promotion and in the progression of differentiation of portal fibroblasts and HSCs into myofibroblasts via paracrine mechanisms and then to maintain the myofibroblastic phenotype by an autocrine mechanism. Concomitantly, with increased activity of TGF-β during fibrogenesis, myofibroblasts enhanced the production and deposition of collagen, leading to progressive scarring and loss of organ function. Thus, overexpression of TGF-β in the liver induces severe fibrosis (13). These reports explain the strong immunoexpression of TGF-β1 observed in our study in livers subjected to BDL, mainly around biliary structures and in the perisinusoidal cells, which are sites of myofibroblasts.
In addition, our results show that livers treated with hAM had a 62% reduction of TGF-β1 when compared to untreated controls. The immunoexpression of TGF-β1 was less intense and confined around DRs, which was also reduced. A very important finding in our study was the weak expression of TGF-β1 in cells of the perisinusoidal spaces, which demonstrated that hAM had a suppressive effect on this profibrogenic cytokine, preventing fibrosis from invading and fragmenting the hepatic parenchyma. Indeed, decreased expression of TGF-β protein was observed in CCl4-injured livers of mice receiving human amniotic epithelial cells (hAECs), coupled with an induction of collagen-degrading matrix metalloproteinases (MMPs) and a reduction of their inhibitors [tissue inhibitors of metalloproteinases (TIMPs)], thus reducing the progression of fibrosis to advanced stages (26). Additionally, the reduced TGF-β1-stained area observed in the hAM group may explain the reduced area occupied by myofibroblasts in our treated livers. This could be supported by the role of TGF on differentiation and maintenance of the myofibroblastic phenotype (7,13).
Considering the crucial role of the signals between biliary structures and portal fibroblasts in the progression of biliary fibrosis (8), we also evaluated the action of hAM on IL-6, which, besides TGF-β, is also involved in the communication between biliary structures and portal fibroblasts, leading to an explosive expansion of the mature BECs and periductal myofibroblasts (11). Among these proinflammatory cytokines, IL-6 was significantly reduced in livers treated with AM. This result is really important considering the critical role of IL-6 in biliary fibrosis. Additionally, rat liver myofibroblasts express IL-6, providing a mechanism for autocrine regulation as well (9). Therefore, the reduction in IL-6 observed in our study after hAM transplantation may serve to further reduce the development of fibrosis. Diminished IL-6 production is the mechanism by which the hAM exerts an antiscarring effect during fetal wound healing (23).
Regarding IL-10, our study demonstrated that the concentration of this cytokine was higher in the treated group but without statistically significant differences. However, hAM transplantation reduced all parameters of biliary fibrosis, including DRs, activated myofibroblasts, and collagen deposition, indicating that in the BDL model, the hAM exerted an antifibrotic effect on fibrosis independent of an increase in endogenous IL-10 in the hepatic tissue. This fact might be explained by the stromal hAM matrix acting as anti-inflammatory and not by induction of IL-10 in the hepatic tissue. This hypothesis could be supported by a recent in vitro study by Hodge et al., which demonstrated that hAEC-conditioned medium (CM) reduced the collagen production in activated, profibrotic HSCs, but without finding measurable IL-10 in medium. This suggests that the hAECs did not induce production of this cytokine in HSCs, and thus the positive effects of hAEC-CM on the reduction of collagen were independent of IL-10 (15).
The beneficial actions exerted by hAM patches in preclinical models of liver fibrosis cannot be attributed to cell replacement in the damaged tissue because no cells derived from AM were found in the liver parenchyma (36). Also, even when cryopreserved AM was used, the cell viability loss had not affected the ability of the membrane to exert beneficial, antifibrotic effects (34). It is also demonstrated, especially in applications for ocular disorders (16,25), that cell loss has not been shown to affect the ability of the AM to exert antifibrotic effects. These findings reinforce the notion that hAM seems to function primarily as a matrix and as a source of bioactive soluble factors rather than by virtue of its functional cells (27).
Taken together, our data regarding the antifibrotic mechanisms of hAM on biliary fibrosis are similar to those that have suggested beneficial effects of hAM transplantation in preventing scar formation during the repair process in studies of ocular disorders (9,16,46). Moreover, the antiscarring action of hAM does not merely result from its anti-inflammatory action (e.g., suppression of IL-1, IL-6, IL-8, and inflammatory cells) but is strongly produced by direct antiscarring actions of the hAM stroma (e.g., suppression of TGF-β) (45,46). Considering the fact that cirrhosis is the reason why many patients are on the waiting list for liver transplants, and that hAM reduces fibrosis, we suggest that hAM could be a possible therapeutic strategy against hepatic fibrosis that may delay the need for orthotropic liver transplant in some cases.
In conclusion, our study demonstrated that hAM, when applied onto the surface of the liver where fibrosis is already established, provided similar antifibrotic effects of those observed when AM was applied at the time of bile duct ligation. Moreover, one of the mechanisms by which transplanted hAM can reduce scarring is through reduced expression of the most profibrogenic factors, TGF-β1 and IL-6, which in turn prevents myofibroblast differentiation and thus the collagen deposition in hepatic tissue.
Footnotes
Acknowledgments
We thank Renata Mantovani, technical coordinator of the Nurse Department of Dr. José de Carvalho Florence Municipal Hospital, São José dos Campos, São Paulo, Brazil. We also thank Dr. Antonietta Silini for critically reviewing and for help in editing the manuscript. This study was supported by Grant No. 2011/879-8, São Paulo Research Foundation (FAPESP). The authors declare no conflicts of interest.
