Abstract
The benefits of stem cell therapy for patients with chronic symptomatic systolic heart failure due to ischemic and nonischemic cardiomyopathy (ICM and NICM, respectively) are unclear. We performed a systematic review of major published and ongoing trials of stem cell therapy for systolic heart failure and compared measured clinical outcomes for both types of cardiomyopathy. The majority of the 29 published studies demonstrated clinical benefits of autologous bone marrow-derived mesenchymal stem cells (BM-MSCs). Left ventricular ejection fraction (LVEF) was improved in the majority of trials after therapy. Cell delivery combined with coronary artery bypass grafting was associated with the greatest improvement in LVEF. Left ventricular end-systolic volume (or diameter), New York Heart Association functional classification, quality of life, and exercise capacity were also improved in most studies after cell therapy. Most ICM trials demonstrated a significant improvement in perfusion defects, infarct size, and myocardial viability. Several larger clinical trials that are in progress employ alternative delivery modes, cell types, and longer follow-up periods. Stem cells are a promising therapeutic modality for patients with heart failure due to ICM or NICM. More data are required from larger blinded trials to determine which combination of cell type and delivery mode will yield the most benefit with avoidance of harm in these patient populations.
Keywords
Introduction
Over the past decade, and more particularly in the past 2 years, there has been an upsurge in clinical trials investigating the effects of stem cells in patients with chronic systolic heart failure. In an attempt to determine the best strategy to improve myocardial function, several types of cells and delivery methods have been studied in patients with ischemic and nonischemic cardiomyopathy (ICM and NICM, respectively). The results of these clinical trials have been conflicting, with some showing substantial clinical improvement and others demonstrating little benefit. The heterogeneity of the reported clinical benefits seems to be related to the type of study (open-label or blinded trial) and the underlying mechanism of heart failure (ICM or NICM). Hence, in this article, we review and compare clinical outcomes of major published trials of stem cells for patients with chronic, symptomatic systolic heart failure due to ICM and NICM. We focus on variables that are of clinical importance for patients with heart failure. Additionally, we present and discuss major ongoing clinical trials of stem cell therapy for chronic heart failure.
Literature Review
A systematic review of the literature was performed. PubMed, OVID, and Cochrane Library were searched through October 16, 2015. The following key words were included in the search: heart failure, cardiomyopathy, stem cells, progenitor cells, and bone marrow. The search was completed using references manually extracted from retrieved articles. We included clinical trials in adults (18 years or older) for which any type of stem cell was given for chronic severe and symptomatic systolic heart failure from either ICM or NICM. The results were further refined to include only studies that contained patients with a left ventricular ejection fraction (LVEF) ≤45% and New York Heart Association (NYHA) functional classification II to IV. Patients with ICM had to be enrolled at least 1 month after a myocardial infarction. Only articles written in English, French, or Spanish were reviewed. There was no restriction on publication year. In order to better evaluate the effect of stem cell therapy, trials without a control group that received standard therapy alone were excluded. Also excluded were abstracts, reports from scientific sessions, and publications limited to preliminary results. For each trial, if a parameter failed to demonstrate a sustained significant improvement during the trial follow-up period, that parameter was considered as unchanged by cell therapy in our analyses. A separate search was performed using clinicaltrials.gov for ongoing and unpublished trials using the above-defined criteria. Trials that were terminated or withdrawn prior to enrollment were not included.
Search Results
The initial search of the three databases identified a total of 7,232 original articles. After excluding the duplicates and reviewing the title, abstract, or the complete article, 29 published trials that met our inclusion criteria were identified. The IMPACT-DCM and Catheter-DCM trials were analyzed and reported together in a single publication 1 . For the purpose of our analysis, we considered them as two separate trials and presented the aggregate results of the NICM patients in Table 1 and the ICM patients in Table 2. The trial by Perin et al. 2 enrolled patients with both NICM and ICM, but since 85% of the study patients had ICM, the trial results were added to Table 3 only.
Trials in Nonischemic Cardiomyopathy

For cell type, all cells used are autologous unless specified. 6MWT, 6-min walk distance test; BMCs, bone marrow cells; BMMCs, bone marrow mononuclear cells; BNP, brain natriuretic peptide; IC, intracoronary; LVEDD, left ventricule end-diastolic diameter; LVEF, left ventricular ejection fraction; LVEDV, left ventricule end-diastolic volume; LVESD, left ventricule end-systolic diameter; LVESV, left ventricule end-systolic volume; MACE, major adverse cardiovascular events; MRI, magnetic resonance imaging; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; NYHA, New York Heart Association; PBSCs, peripheral blood stem cells; TEn, transendocardial; TEp, transepicardial; TTE; transthoracic echocardiography; VO2max, maximal oxygen consumption.
ns: The results did not meet statistical significance for that variable.
–This variable was not studied/reported.
Significant difference (p < 0.05) from baseline (pretreatment) to posttreatment.
Significant difference (p < 0.05) between treatment and control group.
Only the results for patients with nonischemic cardiomyopathy are presented in this table.
Open-Label Trials in Ischemic Cardiomyopathy

For cell type, all cells used are autologous unless specified. LV, left ventricule; PET, position emission tomography; SPECT, single-photon emission computerized tomography; SMBs, skeletal myoblasts. See Table 1 for other abbreviations.
ns: The results did not meet statistical significance for that variable.
–This variable was not studied/reported.
Significant difference (p < 0.05) from baseline (pretreatment) to posttreatment.
Significant difference (p < 0.05) between treatment and control group.
Only the results for patients with ischemic cardiomyopathy from these two trials are presented in this table
Different treatment doses were used in the trial.
Blinded Trials in Ischemic Cardiomyopathy

For cell type, all cells used are autologous unless specified. BMMSCs, bone marrow mesenchymal stem cells; MUGA, multi gated acquisition scan. See Tables 1 and 2 for other abbreviations.
ns: The results did not meet statistical significance for that variable.
–This variable was not studied/reported.
Significant difference (p < 0.05) from baseline (pretreatment) to posttreatment.
Significant difference (p < 0.05) between treatment and control group.
Different treatment doses were used in the trial.
Seven patients enrolled in this trial had NICM.
Trials in which coronary artery bypass grafting surgery was performed.
Nonischemic Cardiomyopathy Trials: Open-Label and Blinded Trials
Eight trials included patients with NICM (Table 1). Six studies were randomized, open-label trials1,3–7, 1 was single blinded 8 , and 1 was double blinded 9 . Five studies used autologous bone marrow or bone marrow-derived mesenchymal stem cells (BM-MSCs)1,4,7–9, and 3 used autologous peripheral blood stem cells (PBSCs)3,5,6. A total of 261 patients received stem cells, and 229 were used as controls. The preferred delivery route was intracoronary (IC) (6 out of 8 trials), and the administered number of stem cells ranged from 35 to 295 × 106 cells. The mean endpoint assessment time was 13.4 months. Four trials demonstrated a significant improvement in LVEF when compared to the control group, with a mean LVEF change of 5.8% (range 4.6% to 7.0%) in the treated patients, and −1.3% (range −2.7% to 0.4%) in the controls (Fig. 1A). Left ventricular end-systolic volume (LVESV) was improved in only 2 trials after therapy. Quality of life was significantly improved in the majority of trials. The NYHA classification and brain natriuretic peptide (BNP) level were both improved in half of the NICM trials. Only 2 studies were able to demonstrate a significant reduction in mortality after stem cell therapy. There were only 2 trials that utilized blinding with mixed results as detailed in Table 1.
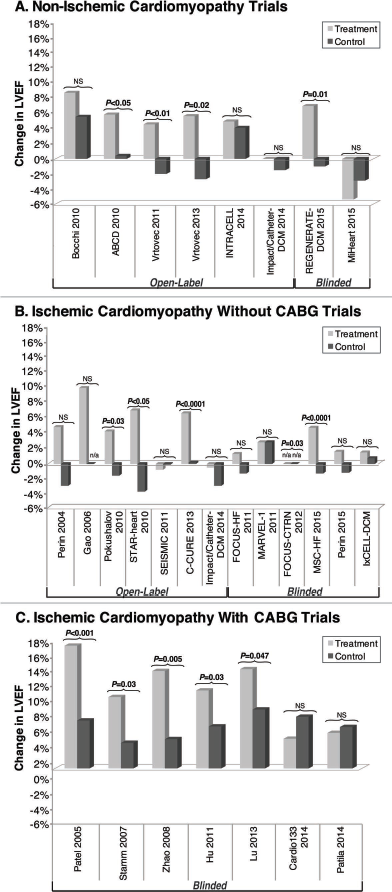
Changes in left ventricular ejection fraction after stem cell therapy. (A) For each nonischemic cardiomyopathy trial, the reported mean change in left ventricular ejection fraction (LVEF) is shown for patients who received stem cell therapy (treatment group) and for the corresponding control group. Similarly, the ischemic cardiomyopathy trials that administered stem cell therapy alone are presented (B), and the ones that administered stem cell therapy in combination with coronary artery bypass grafting (CABG) are shown (C).
Ischemic Cardiomyopathy Trials: Open-Label Trials
There were 15 trials that included patients with ICM, 8 of which were open label (Table 2) and 11 of which were single or double blinded (Table 3). As presented in Table 2, 6 out of 8 open-label trials were randomized. Autologous bone marrow or bone marrow-derived cells were used in 6 ICM open-label trials1,10–15, and skeletal myoblasts were administered in the other 2 studies16,17. A total of 354 patients were treated and compared to 324 controls. The delivery mode was mixed: 5 trials utilized a transendocardial (TEn) approach, 2 trials used IC delivery, and 1 trial performed TEn or transepicardial (TEp) injections. Between 4 and 733 × 106 cells were administered in these ICM trials. Endpoints were assessed at a mean time of 8.9 months. Three of the 7 ICM studies demonstrated a significant improvement in LVEF after therapy when compared to the control group, with a mean LVEF change of 6.3% in the treatment group and −1.7% in the control group (Fig. 1B). LVESV improved significantly after stem cell injections in 5 out of 7 ICM open-label trials. Quality of life was significantly improved in 3 out of 5 studies in which it was reported. Significant improvement in maximal oxygen consumption (VO2max), NYHA classification, and 6-min walk distance test (6MWDT) was observed in almost all trials. Despite having low reported adverse event rates, a significant reduction in mortality was demonstrated in 4 out of 7 open-label studies. Moreover, all of the open-label ICM trials demonstrated an improvement in perfusion defects, myocardial viability, and/or infarct size following stem cell therapy.
Ischemic Cardiomyopathy Trials: Blinded Trials
There were 13 randomized, blinded clinical trials of patients with ICM (Table 3). Patients received autologous bone marrow cells in 10 trials18–27, skeletal myoblasts in 1 trial 28 , allogeneic bone marrow mesenchymal precursor cells in 1 trial 2 , and ixmyelocel-T (ex vivo expanded autologous bone marrow cells) in 1 trial 29 . The delivery mode for these blinded trials was TEp in 5, TEn in 6, and intragraft in the remaining 2 trials. The study patients received 5.1 to 840 × 106 cells, and endpoints were assessed at a mean follow-up time of 7.3 months. When comparing 415 treated subjects to 305 controls, 5 studies demonstrated a significant improvement in LVEF when compared to the control group. Change in LVEF was assessed by transthoracic echocardiography (TTE) alone or in combination with another modality in 9 trials. Magnetic resonance imaging (MRI) alone was used for LVEF assessment in 3 trials, while computed tomography (CT) or MRI was used in 1. When MRI was used, 3 out of 5 ICM studies were able to demonstrate a significant change in LVEF. The mean change in LVEF for the blinded ICM trials that administered stem cell injections alone was 3.2% in the treatment group and −1.3% in the controls (Fig. 1B). In blinded ICM trials where concurrent coronary artery bypass grafting (CABG) and cell injection were performed, a significant improvement in LVEF was noted when compared to the controls (12.8% vs. 5.5%) (Fig. 1C). LVESV was significantly reduced in only 3 out of 11 trials after therapy, while an improvement in NYHA functional classification was reported in 5 trials. Quality of life was improved in 3 studies. However, none of these blinded trials demonstrated a significant improvement in VO2max or mortality in the treated patients. Of the 10 trials that measured perfusion defects or infarct size, 7 reported a significant improvement.
Ongoing Trials
We identified 13 ongoing clinical trials that are enrolling similar patients with heart failure (Table 4). All trials are randomized, 10 are double blind, and the majority utilizes a sham procedure in the control group. Nine are administering autologous cells, while the others are giving allogeneic cells [cardiosphere-derived cells in 2, mesenchymal stem cell precursors in 1, BM-MSCs in 1, and umbilical cord-derived mesenchymal stem cells (UCB-MSCs) in 1]. Even though two new delivery modes are being studied (intravenously and retrograde injections through the coronary sinus), the IC and TEn delivery modes are still used in most trials. MRI and TTE remain the favored method for left ventricular function (LVEF) and dimension assessment. Clinical outcomes such as heart failure exacerbation, recurrent hospital admission for heart failure-related symptoms, and duration of hospital stay are monitored in almost all of these studies and even constitute the primary outcome in several trials. Overall, these ongoing trials are enrolling more patients than the previous trials, have longer follow-up periods, and are mainly conducted in North America. This differs from the previously published trials that were, for the most part, performed outside of the US.
Ongoing Clinical Trials in Patients With Chronic and Symptomatic Systolic Heart Failure With LVEF ≥45%

For cell type, all cells used are autologous unless specified. ASCs, adipose-derived stem cells; CABG, coronary artery bypass grafting; CDCs, cardiosphere-derived cells; CS, coronary sinus; CT, computed tomography; HF, heart failure; IV, intravenous; MSCs, mesenchymal stem cells; OMT, optimal medical therapy. See Tables 1–2 for other abbreviations.
n/a: nonavailable.
As of October 16, 2015.
Stands for BNP or NT-proBNP.
Partial results have been published.
Some patients enrolled will be NYHA class I.
Discussion
When compared to previously published reviews30–33, this systematic review provides a broad contemporary perspective of the role of stem cells in patients with heart failure due to ICM or NICM. Stem cell therapy has been evaluated in a wide spectrum of cardiac disease, including acute ST-elevation myocardial infarction, ICM, and NICM. Prior reviews have often focused only on a part of that spectrum, such as stem cell therapy in patients with ICM and/or myocardial infarction. This review is unique in that it broadens the scope of analysis to include trials of both ICM as well as NICM. In terms of data reported, reviews have often focused specifically on left ventricular function (LVEF and LVESV) and mortality post procedure. Our review includes additional parameters that are important in the management of chronic heart failure, including quality of life, NYHA functional classification, BNP level, and exercise capacity. These parameters are usually reported in clinical trials, but are often underrepresented in systematic reviews.
Impact on Cell Therapy on Left Ventricular Function and Dimension
There was a significant improvement in LVEF from pretreatment values in cell therapy-treated patients in 16 of the 28 trials presented3–8,12–15,18–20,22–24,27. In 14 of the 28 trials, there was a significant improvement in LVEF when compared to the control group (4 out of 8 NICM studied4–6,8, and 10 out of 20 ICM studies13–15,18–20,22–24,27). This difference could have resulted from some of the trials being underpowered to demonstrate significance between group differences. Patients who received stem cells in combination with CABG for ICM had the greatest improvement in mean LVEF compared to the control group (12.8%) followed by NICM patients (5.8%), and ICM patients who received cell therapy alone (without CABG) (5.1%).
LVESV or diameter reduction was observed in more than half of the trials, suggesting that cell therapy may help reverse cardiac remodeling in these patients with heart failure. Additionally, a significant change in LVESV or diameter after cell injection correlated with an improvement in LVEF in most trials. This association was especially noticeable for ICM studies in which 13 out of 17 reported concordant results for both variables1,12–15,20,21,24–27. However, left ventricular end-diastolic volume (LVEDV) or diameter (LVEDD) did not appear to be as useful as indicators of therapy response, as they remained unchanged in the majority of trials (reduced in 1 out of 8 NICM trials 5 , and 6 out of 19 ICM trials14,15,18,20,22,24), and had poor correlation with a change in LVEF after therapy.
Impact of Cell Therapy on Subjective and Objective Markers of Heart Failure
Overall, NYHA functional classification and quality of life were improved in the majority of trials after cell therapy. Open-label trials were more likely to demonstrate an improvement in NYHA classification after treatment compared to blinded trials. This could be due to a “placebo effect” in the open-label-treated group 34 . The confounding effect of a “placebo effect” should be less pronounced in the ongoing trials given their use of a single- or double-blind design (with many employing a sham-treated control group) (Table 4).
The objective changes in exercise capacity, measured by the 6MWDT and VO2max, were also more likely to be improved in the open-label studies. These two variables were only reported together in three trials9,15,25, and only one trial was able to demonstrate similar results for both parameters; both remained unchanged after therapy. The 6MWDT was more often improved when compared to the control group than the VO2max was after cell therapy, suggesting that the 6MWDT may be a better marker of improvement following cell therapy. The change in the 6MWDT and VO2max did not differ between NICM and ICM patients.
Although the BNP level was significantly improved in half of the open-label trials, this was observed in only 2 of the blinded trials8,22. This discordance could be due to the fact that some of the trials were underpowered to demonstrate significant changes in BNP level. An additional possibility is that cell therapy did not change the volume status of these patients.
Impact of Cell Therapy on Myocardial Perfusion Imaging
Perfusion defect size, infarct size, and myocardial viability were only assessed in ICM trials. Overall, these parameters were improved after stem cell therapy. Several assessment methods were used. The more commonly adopted one was single-photon emission computed tomography (SPECT), followed by MRI, and both yielded similar results for detection of perfusion imaging change. Myocardial viability assessed by SPECT, NOGA® mapping, or position emission tomography (PET) was reported to be significantly improved in 3 of the 5 trials in which it was measured10,12,16. Perfusion defect size, infarct size, and myocardial viability were all assessed within 12–36 months of therapy. There is only 1 trial that reported data beyond 1 year, and therefore the benefits of stem cells on myocardial perfusion after that time remain unclear. More data are needed to confirm these benefits and to determine which imaging modality is most accurate at identifying such perfusion changes.
Stem Cell Type and Number
Most of the trials presented here administered autologous BM-MSCs with mixed results. All 3 trials that employed PBSCs (CD34+)3,5,6 as well as the C-CURE trial 15 , which used cardiopoietic cells that were grown from bone marrow cells, demonstrated overall beneficial results. However, the 3 trials that administered skeletal myoblasts16,17,28 were small studies that yielded overall negative results. There was no clear association between the number of cells administered and the improvement in clinical outcome. At this time, it remains unclear which cell type and what cell number are most beneficial for patients with chronic systolic heart failure. Ongoing trials should help clarify these important points.
Delivery Mode
The three preferred delivery modes were TEn, IC, and TEp. While IC was mainly used for NICM patients, TEn and TEp and intragraft were favored for ICM. Patients who had cells infused via an IC or intragraft routes were more likely to have an increased LVEF compared to the other techniques. This observation differs from existing data suggesting that TEn would be superior to IC delivery in terms of myocardial retention rates, improvement in LVEF, BNP level, and exercise capacity 35 . Of the 8 trials that administered cells by IC, 4 did not use a flow occlusion technique3,5,6,9. In the remaining trials, 3 had simultaneous proximal balloon inflation during infusion8,12,14 and 1 had coronary sinus balloon inflation for distal flow interruption 4 . The only IC trial that failed to show a significant improvement in LVEF did not use a flow-obstruction technique 9 , suggesting that a flow-occlusion technique might be favorable to cell retention and local effect.
Limitations
The interpretation of these trials' results is limited by the fact that cell therapy for heart failure is relatively new, and as such, most trials are either pilot studies or early phase II trials, with a small sample size. The heterogeneity and overall small number of trials in this review limit our ability to perform detailed subgroup analyses that could provide clinically important information. For example, it is certainly possible that cell preparation techniques used in some of the trials may have impacted results; however, the small number of these studies, the heterogeneity of preparation techniques, and insufficient information on the details of cell preparation preclude drawing meaningful conclusions on this subject. The follow-up time of these studies also tended to be short, especially for the blinded ICM trials. A small group of trials registered on clinicaltrials.gov was terminated early due to lack of funding or administrative problems, and thus their results were not published. Additionally, our results could be limited by “publication bias,” in which negative studies were not published. These missing negative trials could exaggerate the perceived benefits of cell therapy for patients with heart failure. Our search for ongoing trials was limited to trials listed on clinicaltrials.gov. Hence, it is possible that there are other pertinent ongoing trials not listed on clinicaltrials.gov website. This may be the case for trials performed outside the US.
Conclusions
This review suggests that stem cells are a promising therapeutic modality for patients with severe symptomatic heart failure due to ICM and NICM. However, larger randomized, double-blind clinical trials with longer follow-up periods are needed to determine which combination of cell type, delivery mode, and subset of patients would most optimally demonstrate safety and efficacy. Future studies will also clarify which outcome variables are most appropriate to assess efficacy of these novel therapies. The surge of new trials should help clarify these questions.
Footnotes
Acknowledgment
Dr. Schaer serves on the steering committee of the ixCELL trial. Otherwise, the other authors declare no conflicts of interest.
