Abstract
There is a growing interest in cell-based therapies in T2DM as β-cell failure is progressive and inexorable with the advancing duration of disease. This prospective, randomized, single-blinded placebo-controlled study evaluates the efficacy and safety of autologous bone marrow-derived stem cell transplantation (ABMSCT) in T2DM. Twenty-one patients with triple oral antidiabetic drug failure and requiring insulin ≥0.4 IU per kg per day with HbA1c <7.5% were randomly assigned to an intervention (n = 11) and control group (n = 10) and followed for 12 months. Patients in the intervention group received ABMSCT through a targeted approach, and after 12 weeks, a second dose of stem cells was administered through the antecubital vein after mobilization with G-CSF, while the control group underwent a sham procedure. The primary end point was a reduction in insulin requirement by ≥50% from baseline while maintaining HbA1c <7%. Nine out of the 11 (82%) patients in the intervention group achieved the primary end point, whereas none of the patients in the control group did over the study period (p = 0.002). The insulin requirement decreased by 66.7% in the intervention group from 42.0 (31.0-64.0) IU per day to 14.0 (0.0-30.0) IU per day (p = 0.011), while in controls it decreased by 32.1% from 40.5 (31.8-44.3) IU per day to 27.5 (23.5-33.3) IU per day (p = 0.008) at 12 months. The reduction in insulin requirement was significantly more in the intervention group compared to controls at both 6 (p = 0.001) and 12 months (p = 0.004). There was a modest but nonsignificant increase in HbA1c (%) in cases from 6.9% (6.4-7.2%) to 7.1% (6.6-7.5%) as well as in controls from 6.9% (6.2-7.0%) to 7.0% (6.9-7.5%). Ten out of 11 (91%) patients could maintain HbA1c <7% in the intervention group, whereas 6 out of 10 did (60%) in the control group (p = 0.167). The glucagon-stimulated C-peptide significantly increased in treated cases compared to controls (p = 0.036). The decrease in insulin requirement positively correlated with stimulated C-peptide (r = 0.8, p = 0.001). In conclusion, ABMSCT results in a significant decrease in the insulin dose requirement along with an improvement in the stimulated C-peptide levels in T2DM. However, a greater number of patients with a longer duration of follow-up are required to substantiate these observations.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a metabolic disorder characterized by insulin resistance and β-cell dysfunction (3,30). Currently available therapies for T2DM target either of the two defects. With advancing duration of disease, β-cell failure is progressive and inexorable, demanding a need for insulin therapy (8,17). Exogenous insulin therapy is associated with variable glycemic control, weight gain, hypoglycemia, and does not simulate physiologic insulin secretion (10). Therefore, there is a need for therapies targeting β-cell dysfunction and enhancing its neogenesis.
β-Cell regenerative therapies include various medications, pancreatic and islet cell transplantation, and stem cell therapy (6,34). Pioglitazone and glucagon-like peptide 1 (GLP-1) analogs in humans exhibit glycemic durability, have limited efficacy, and are associated with adverse events (16,22,23,29). Current limitations of islet transplantation include the restricted availability of cadaveric pancreata and the need for immunosuppression with its associated complications (27). The interest has shifted on stem cells that possess homing-in and transdifferentiation properties. Bone marrow is a rich source of stem cells, and its potential is being explored (19,24). There are relatively few studies available on the utility of bone marrow-derived stem cell transplantation for treatment of T2DM in humans. Previous studies by Estrada et al. and Wang et al. showed that combined use of autologous bone marrow-derived stem cell transplantation (ABMSCT) and hyperbaric oxygen (HBO) therapy resulted in a significant decrease in insulin doses and fasting plasma glucose; however, there were conflicting reports on the sustenance of C-peptide improvement at 12 months (13,33). These studies included heterogeneous population of patients with respect to antidiabetic therapy, poor glycemic control at inclusion, lack of control arm, high drop-out rate, and lastly whether the improvement in C-peptide in these patients was due to decrease in the glucotoxicity and/or to the stem cell therapy was not clear. Therefore, this prospective, randomized, single-blinded placebo-controlled study evaluates the efficacy and safety of ABMSCT in patients with T2DM with good glycemic control.
Materials and Methods
One hundred and twenty patients were screened between August 2009 and December 2009 at the outpatient department of the Post Graduate Institute of Medical Education and Research (PGIMER), Chandigarh, India. Informed consent was taken from all the subjects, and the study was approved by the institute's ethics committee. Out of these, 26 patients were eligible for the study, as per the inclusion criteria: patients with T2DM, age ranging from 30 to 70 years, having diabetes for ≥5 years with failure of triple oral antidiabetic drugs in optimal doses (metformin 2 g per day, glimepiride 4 mg per day, pioglitazone 15 mg per day) and requiring insulin (≥0.4 IU/kg per day) for at least 1 year to achieve optimal glycemic control. Before the randomization, patients were on stable doses of vildagliptin 100 mg per day + metformin 2 g per day (Galvus Met, Novartis India, Mumbai, India), pioglitazone 15 mg per day (Pioglar, Ranbaxy, Mumbai, India), and insulin (≥0.4 IU per kg per day) (Human Mixtard, Actrapid, or Insulatard, Novo-Nordisk, Bangalore, India) for at least 3 months with glycated hemoglobin (HbA1c) of <7.5%. Patients were excluded from the study if they had type 1 diabetes mellitus, glutamate decarboxylase 65 (GAD-65) seropositive status, serum creatinine >1.5 mg/dl, abnormal liver function tests, active infections, malignancy, or acute coronary syndrome in the previous 3 months. Out of these 26 patients, 21 patients were finally randomized into two groups: intervention (Group I) and placebo (Group II) (Fig. 1).

Plan of the study. G-CSF, granulocyte colony-stimulating factor.
Baseline Evaluation
All subjects underwent clinical assessment regarding glycemic control, micro-, and macrovascular complications. Evaluation at the time of enrollment included blood pressure (BP), body mass index (BMI), waist circumference (WC in cm), body fat estimation (Omron, HBF-302, Kyoto, Japan), fundus examination, ankle—brachial index, vibration perception threshold, electrocardiography, and echocardiography. HbA1c level was measured on automated high-performance liquid chromatography (HPLC)-based system using ion-exchange cartridge (D-10, Bio-Rad Laboratories, Inc., Hercules, CA, USA; normal range: 3.8-5.9%). Glucagon-stimulated C-peptide test was performed in fasting state after intravenous (IV) administration of glucagon (1 mg; GlucaGen, Novo Nordisk, Denmark), and blood samples were drawn at −15, 0, and 6 min after injection. C-peptide estimation was done by electrochemiluminescence immunoas-say (ECLIA) (Elecsys 2010, Roche Diagnostics GmbH, Mannheim, Germany; normal range: 1.1-4.4 ng/ml, assay sensitivity 0.01 ng/ml, intra- and interassay coefficient of variation 4.6% and 5.0%, respectively). GAD-65 antibody status was assessed by Immunoblot assay (DTEK, Mons, Belgium).
Fasting plasma insulin was assayed by ECLIA (assay sensitivity 0.2 μU/ml, intra- and interassay coefficients of variation 1.5% and 4.9%, respectively) after stopping the oral hypoglycemic drugs and insulin for 48 h. The homeostatic model assessment (HOMA) was applied for the assessment of HOMA-IR (insulin resistance) and HOMA-β (β-cell function). HOMA is a computer-based method that uses fasting plasma insulin and fasting plasma glucose levels to calculate insulin resistance and β-cell function. Patients were asked to monitor a five-point glucose profile (fasting, 2 h postbreakfast, postlunch, postdinner, and 4 am) using the Optium glu-cometer (Abbott, Mumbai, India) at least once per week with a target of fasting plasma glucose between 70 and 130 mg/dl and postprandial glucose ≤180 mg/dl. All the patients were advised physical activity in the form of a brisk walk for 150 min per week. The importance of physical activity was reinforced at each visit, and the exercise schedule was recorded at each visit.
Stem Cell Harvesting
Bone marrow (150-200 ml) was aspirated from the posterior superior iliac spine in Group I, under local anesthesia following aseptic precautions. The mononuclear cells (MNCs) were separated by ultracentrifugation after layering on density-gradient medium (Ficoll-Hypaque, Sigma-Aldrich, St. Louis, MO, USA) and were washed using phosphate-buffered saline (PBS; Himedia Laboratories Private Limited, Mumbai, India) and resuspended in normal saline with a final product volume of 8-10 ml. Aliquots (1 ml) were taken from the final product obtained from the bone marrow aspiration as well as from the peripheral blood harvesting procedure (which was performed 12 weeks later after stem cell transplantation), for total nucleated cell (TNC) count, MNC count, viability testing by trypan blue (Sigma-Aldrich) dye exclusion test, cluster of differentiation 34 positive (CD34+) and CD45+ cell counts (using antibodies from BD Bioscience, San Jose, CA, USA) by gated flow cytometry (FACS Aria II, BD Bioscience). Three to five milliliters of final wash supernatant was used for sterility testing using aerobic and anaerobic cultural vials (Bactec culture vial, BD Bioscience).
Stem Cell Transplantation
In the Group I subjects who underwent bone marrow aspiration and stem cell harvesting, a 5F catheter (Beacon® Tip Torcon NB® Advantage Catheter, Cook, Inc., Bloomington, IN, USA) was selectively navigated through a transfemoral route into the superior pancreaticoduodenal artery unless otherwise specified, and a superselective injection of stem cells was administered.
Postprocedure, the patients were monitored for any immediate complications for the next 24 h. After 12 weeks of targeted stem cell injection, a second dose of stem cells was administered through the antecubital vein after mobilization with granulocyte colony-stimulating factor (G-CSF; Filgastrim, Biocon India Limited, Bangalore, India) in these patients (Group I) at a dose of 10 μg per kg per day via the subcutaneous (SC) route for 5 days to achieve a target total leucocyte count (TLC) of 40,000-50,000 per mm3. After achieving the target TLC, these patients underwent leukopheresis on a cell separator machine (Cobe Spectra, Caridian BCT, Lakewood, CO, USA) utilizing the MNC program for the peripheral blood-harvesting procedure. The product obtained was subjected to ultracentrifugation after using the density-gradient medium (Ficoll Hypaque, Sigma) to separate the MNCs. After repeated washing with PBS, the cells were resuspended in normal saline to obtain the final product volume of 8-10 ml.
Patients in Group II (controls) underwent a sham procedure, which is briefly described as follows. Ten to 20 ml of bone marrow was aspirated from the posterior superior iliac spine, under local anesthesia following all the aseptic precautions. After 5 h of marrow aspiration, the patients were taken to the procedure room, and 10 ml of 0.9% saline was injected into the femoral artery under sterile precautions. At 12 weeks, the patients received 0.5 ml distilled water as a placebo injection subcutaneously for 5 days followed by an IV 0.9% 5 ml saline injection on the sixth day. Follow-up was similar to that of the patients in Group I. Lifestyle and diet advices were reinforced during each visit to both of the groups.
Follow-up
Both the cases and the controls were followed up initially at two weekly intervals for 3 months, then on monthly visits for the next 3 months and then at three monthly intervals. All the patients were advised to perform self-monitoring of blood glucose (SMBG) including a five-point glucose profile once a week. Fasting plasma glucose (FPG) levels were targeted between 70 and 130 mg/dl, postmeal <180 mg/dl, and HbA1c of <7%. Insulin doses were tapered whenever the patient reported hypoglycemic episodes or when the FPG was <70 mg/dl and a postmeal glucose of <120 mg/dl. Patients were asked to contact a single investigator (PK) by telephone or on an outpatient basis for insulin dose adjustment. Anthropometry was recorded during each visit, and HbA1c was repeated every 3 months. Glucagon stimulated C-peptide and fasting plasma insulin (FPI) were repeated at 6 and 12 months. All of the patients in both groups were followed for a period of 12 months without any dropout.
Outcomes
The primary end point was a reduction in insulin requirement by ≥50%, while maintaining HbA1c <7%, and the secondary end points were a change in weight, HbA1c, and stimulated C-peptide levels compared to the baseline.
Statistics
The statistical program for the Social Sciences (Release 16.01, PC Windows; SPSS, Inc., Chicago, IL, USA) was used for the data analysis. Data were expressed as median and interquartile range, unless otherwise specified. Baseline and posttreatment data within the groups were compared using Friedman's test with post hoc Wilcoxon's signed rank test (p value corrected using Bonferroni procedure), and the data between two groups were analyzed using the Kruskal Wallis test. Linear regression analysis was used to identify an association between two variables. A value of p < 0.05 was considered as significant.
Results
Group I
The median age of the patients in Group I (n = 11, 9 male) was 51 (46.5-56.0) years with duration of diabetes 12.0 (10.0-15.5) years (Table 1). Five patients had neuropathy, three had nonproliferative diabetic retinopathy (NPDR), and six had microalbuminuria. Eight patients had hypertension, and one had coronary artery disease. The median duration of insulin treatment and insulin dose was 4.0 (1.8-5.0) years and 42.0 (31.0-64.0) IU/day, respectively. Eight patients were on twice daily premixed human insulin, while three patients were on multiple SC injection [regular + neutral protamine Hagedorn (NPH)]. The baseline HbA1c of the group was 6.9% (6.4-7.1%).
Baseline Clinical and Laboratory Parameters of Cases and Controls

BMI, body mass index; WC, waist circumference; FPG, fasting plasma glucose; HbAlc, glycated hemoglobin; FPI, fasting plasma insulin; HOMA-IR, homeostatic model assessment-insulin resistance; HOMA-β, homeostatic model assessment β-cell function. All values are expressed as median and interquartile range; NS, not significant.
The median volume of bone marrow harvested for SCT was 210 (194-219) ml, which yielded a median of 2.9 (2.2-3.8) × 108 mononuclear cells (Table 2). One patient had splenic artery spasm, two had superior pancreaticoduodenal artery spasm, and one celiac plexus spasm during the cannulation, and the stem cell concentrate was administered in the gastroduodenal artery in these four patients for the above reasons. One patient had an absent superior pancreaticoduodenal artery, and so the stem cells were injected into the inferior pancreaticoduodenal artery via the superior mesenteric artery. One patient had complex celiac plexus anatomy and required a catheter change. In all these patients, postprocedure serum amylase was monitored, and it remained in the reference range, and none of them developed pancreatitis. None of the patients developed hematoma or femoral artery thrombosis. No other adverse event was reported during the study period.
Details of Stem Cell Therapy Procedure

MNC, mononuclear cell; CD34, cluster of differentiation 34; G-CSF, granulocyte colony stimulating factor; TLC, total lymphocyte count.
After SCT, 10 out of 11 patients could maintain mean HbA1c <7% during the study period accompanied with a decrease in the total daily insulin requirement by 66.7% at the end of 12 months. There was a 47.6% reduction in insulin dose in the first 3 months of SCT followed by a 19.1% dose decrement in the next 9 months following peripheral mobilization of stem cells. This decrease in insulin doses was statistically significant at 3 months (p = 0.002), at 6 months (p = 0.008), and at 12 months (p = 0.011) compared to baseline (Fig. 2). After SCT, nine out of 11 (82%) patients in Group I achieved ≥50% reduction in insulin requirement (responders) over a mean duration of 108 days (range: 14-280 days). Five patients had recurrent hypoglycemia and required <0.1 IU per kg per day of insulin and were then switched to sulfonylurea—Gliclazide (T. Diamicron XR 30 mg per day; Serdia Pharmaceuticals, Mumbai, India) so that their blood glucose levels could be maintained within the target range.

Change in weight and insulin dose profile in the cases (Group I) and the controls (Group II). *Significant difference from baseline. #Significant decrease in Group I compared to Group II. Group I is shown with upper quartile and Group II with lower quartile for weight, and Group II is shown with upper quartile and Group I with lower quartile for insulin dose for clarity.
There was an insignificant increase in HbA1c from 6.9% (6.4-7.2%) to 7.1% (6.6-7.5%) at 12 months (Fig. 3). The stimulated C-peptide significantly increased from 2.0 (0.6-2.2) to 3.3 (2.3-3.7) ng/ml at 12 months (p = 0.036) (Fig. 4). There was an insignificant weight gain at 6 months, and the patients had returned to their baseline weight by 12 months. The number of hypoglycemic episodes per patient per month increased after the SCT and subsequently decreased after tapering of insulin doses (Table 3).
Comparison of Clinical and Laboratory Parameters in Cases (Group I) on Follow-up

All values are expressed as median and interquartile range.
Significant difference from baseline. NS, not significant.

Change in HbA1c in the cases (Group I) and the controls (Group II). HbA1c, glycated hemoglobin. Group I is shown with upper quartile and Group II with lower quartile for clarity.

Alterations in stimulated C-peptide in the cases (Group I) and the controls (Group II). *Significant difference from baseline. Group I is shown with upper quartile and Group II with lower quartile for clarity.
The change in insulin doses was positively correlated with stimulated C-peptide (r = 0.8, p = 0.001) at 12 months. However, there was no correlation with change in weight, waist circumference, and MNC count. There was no significant change in HOMA-IR and HOMA-β at the end of the study.
Group II
Median age of the control group (n = 10, 7 male) was 54.0 (52.5-55.8) years, and the duration of diabetes was 20.0 (16.25-20.8) years. Median duration of insulin therapy was 4.5 (1.0-6.8) years with a median insulin dose at study inclusion of 40.5 (31.8-44.3) IU/day (Table 1). Half of the patients were receiving multiple SC insulin injection (Regular + NPH), while remaining on twice-a-day premixed human insulin. Seven subjects had neuropathy, three each had microalbuminuria, macroproteinuria, and NPDR, and two had proliferative diabetic retinopathy. Seven patients had hypertension, and one had coronary artery disease. Median HbA1c at baseline was 6.9% (6.2-7.0%).
After the sham procedure, insulin requirement decreased by 32.1%. There was a reduction of 8.6% at the end of 3 months, which further decreased by 23.5% at 6 months, and this remained relatively stable until the end of the study. The decrease in insulin dose was statistically significant at 6 and 12 months (p = 0.014 and p = 0.008, respectively) compared to the baseline (Table 4, Fig. 2). There was a minor nonsignificant weight gain at 6 and 12 months compared to the baseline weight (Fig. 2). The changes in BMI, body fat %, and waist circumference were not statistically significant at 12 months compared to the baseline. None of the patients could achieve ≥50% reduction in insulin requirement (“nonresponders”), and only six out of 10 patients could achieve HbA1c <7% over 12 months. The HbA1c increased in the controls from 6.9% (6.2-7.0%) to 7.0% (6.9-7.5%) at 12 months, and this increase was not statistically significant (Fig. 3). The changes in HOMA-IR and HOMA-β and stimulated C-peptide at 12 months were not significant compared to the baseline (Table 4, Fig. 4).
Comparison of Clinical and Laboratory Parameters in Controls (Group II) on Follow-up
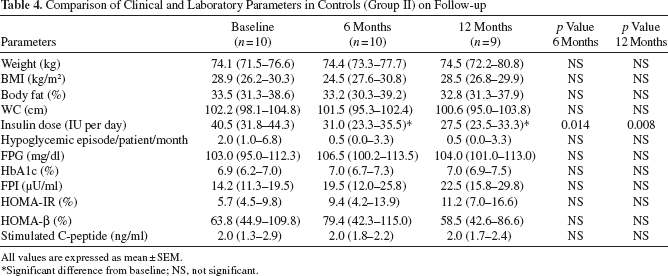
All values are expressed as mean ± SEM.
Significant difference from baseline; NS, not significant.
Comparison Between Cases and Controls
Both of the groups were comparable at baseline with respect to age, sex, duration of diabetes, insulin therapy, insulin dose, weight, HbA1c, C-peptide, and fasting plasma insulin (Table 1). On comparing the two groups at 3, 6, and 12 months, the changes in clinical parameters were not statistically significant. The decrease in insulin dose (Dinsulin dose) was significantly more in the cases compared to the controls at 3 months (p = 0.032; data not shown), at 6 months (p = 0.001), as well as at 12 months (p = 0.004). The change in HbA1c (DHbA1c %) was not significantly different between the two groups. Ten out of 11 patients in Group I and six out of 10 patients in Group II achieved HbA1c <7% at 12 months (p = 0.167). There was no statistically significant change in FPI in either cases or controls. The HOMA-IR was significantly lower in Group I compared to Group II at 6 months (p = 0.044) but not at 12 months (p = 0.113), while HOMA-β did not change significantly during the study. Stimulated C-peptide significantly improved in the cases compared to the controls at 6 months (p = 0.041) and at 12 months (p = 0.032) (Table 5). The decrease in insulin dose (Dinsulin dose) positively correlated with the stimulated C-peptide at 6 and 12 months (r = 0.8, p = 0.001).
Comparison of Parameters Between Cases and Controls
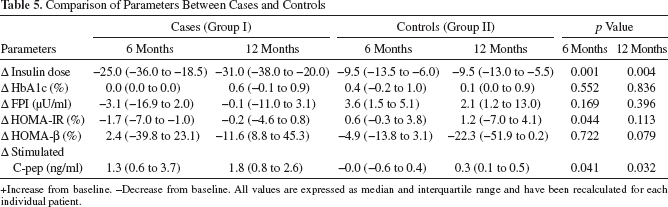
+Increase from baseline. -Decrease from baseline. All values are expressed as median and interquartile range and have been recalculated for each individual patient.
Discussion
This prospective, randomized, single-blinded placebo-controlled study demonstrates that the autologous bone marrow-derived stem cell transplantation results in a reduction in exogenous insulin requirement while maintaining target HbA1c and is accompanied by an improvement in the stimulated C-peptide response in patients with T2DM having three oral antidiabetic drug failure and requiring insulin for glycemic control.
The available medical therapies for the management of T2DM either enhance insulin secretion from the residual β-cells or target insulin resistance. Both sulfonylureas and metformin have not been shown to have a durable effect on glycemic control with the advancing duration of disease. Though thiazolidinediones have been demonstrated to have a protective effect on β-cell function as shown in the Diabetes Reduction Assessment with Ramipril and Rosiglitazone Medication (DREAM) study (18) and in A Diabetes Outcome Progression Trial (ADOPT) (32), currently the thiazolidinediones are in disrepute due to their adverse effects (22,23,29). The newer drugs in the armamentarium of T2DM targeting the enteroinsular axis, namely, dipeptidyl peptidase-4 (DPP-IV) inhibitors and GLP-1 receptor agonistic analogs, have been demonstrated to have a proliferative effect on b-cell mass in mice and have glycemic durability in humans, which is a surrogate marker of improvement in β-cell function and/or mass. However, their long-term safety profile is yet to be established.
There is growing interest in the scientific community for cellular therapies utilizing bone marrow-derived cells for the treatment of diabetes and its complications (15,25). It has been shown that bone marrow-derived stem cell transplantation reverses diabetes in streptozotocin- induced diabetic mice (14,31). A pioneer work by Banerjee et al. showed that multiple infusions of bone marrow- derived stem cells restored euglycemia as early as 6 days and demonstrated islet neogenesis on histology (4).
Very few studies have examined the utility of bone marrow-derived stem cell transplantation for the treatment of T2DM in humans. A previous study by Estrada et al. showed that the combined use of autologous bone marrow-derived stem cell infusion into the dorsal pancreatic artery and hyperbaric oxygen therapy (HBO) resulted in a significant reduction in fasting plasma glucose and insulin doses and an increase in C-peptide levels at the end of 1 year (13). The authors hypothesized that the infused stem cells led to the differentiation of the local progenitor cells and decreased inflammation in the pancreas, which was augmented by HBO therapy. However in this study, there was no placebo arm, and the recruited patients had varying degrees of glycemic control and were receiving different antidiabetic medications. Moreover, the follow-up data were available for only one third of the patients at the end of 1 year. Another study by Wang et al. used combined ABMSCT and HBO in 31 patients of T2DM and showed a decrease in HbA1c of >1.5% as quickly as 1 month after therapy, and it was maintained over the study period of 1 year (33). The C-peptide increased significantly at 3 months but declined to baseline at 1 year. The authors concluded that the combined ABMSCT and HBO therapy led to an improvement in glucose control, a decrease in requirement of insulin and oral hypoglycemic agents, and a transient improvement in β-cell function (33). In a recent study, Hu et al. demonstrated the long-term efficacy and safety of autologous bone marrow mononuclear cells (ABMNCs) in comparison to intensive insulin therapy in patients with T2DM (20). One hundred and eighteen patients were followed-up for a period of 33 months, and they showed that the ABMNC group could achieve significantly lower HbA1c with a reduction in oral hypoglycemic drugs and insulin doses compared to the control group on intensive insulin therapy. However, the study was not appropriately designed as patients were open to choose their mode of treatment, and it was not oriented with an intention-to-target HbA1c <7%. Moreover, the control group was not appropriately matched for the operative procedure (20). In our previous study, we showed >50% reduction in the insulin requirement from the baseline in three fourths of the patients with a reduction in HbA1c from 8.4 ± 0.6% to 7.3 ± 0.8% and a significant increase in stimulated C-peptide after ABMSCT (7). This reduction in insulin requirement and the change in HbA1c persisted for at least 15 months in almost two thirds of the patients (unpublished observation). However, in this study, there was no placebo group and the patients had uncontrolled diabetes with a mean HbA1c of 8.4% at baseline.
The present study showed a significant reduction in insulin requirement while maintaining HbA1c <7%, with an improvement in the stimulated C-peptide. The study was intentionally designed to include subjects with HbA1c <7.5% in order to mitigate any improvement in β-cell function secondary to the improvement in glucotoxicity, as the median HbA1c at baseline in the present study was 6.9%. Further, we could sustain a HbA1c <7% despite the reduction in the insulin doses by >50%, suggesting an improvement in β-cell function as indicated by the significant improvement in C-peptide at 6 and 12 months. There was also a significant reduction in the insulin doses in the control group; however, none of the patients could achieve ≥50% reduction in insulin doses, and this was not associated with a change in weight or improvement in stimulated C-peptide. This reduction in insulin doses in the control group can be attributed to the repeated emphasis on lifestyle modification during their frequent visits to the hospital. HOMA-β did not show any significant change during the study, probably due to the low number of patients. HOMA-IR decreased in the cases significantly at 6 months compared to the controls, and this can be attributed to a better metabolic milieu and/or a direct effect of stem cell therapy on insulin therapy. In this study, we also used a second injection of MNCs after peripheral mobilization with G-CSF, and that resulted in further reduction of insulin doses suggesting that multiple injections of stem cells may be useful for a durable effect.
Stem cell therapy improves β-cell function and/or mass as shown by the improvement in HOMA-β and C-peptide in various studies (13,33). Various animal studies have been carried out to elucidate the mechanism of action of the stem cell transplantation. Banerjee et al. showed neo-islet development after the multiple injections of the bone marrow-derived hematopoietic stem cells in STZ-induced diabetic Wistar rat (4). Another elegant study by Dong et al. showed a significant reduction in blood glucose in STZ-induced diabetic rat after allogenic MSC transplantation. They also showed transdifferentiation of mesenchymal stem cells (MSCs) into insulin-producing cells (12). However, the effect of MSCs on glycemic control was shown to be transient by Azab et al. (2). Although the mechanism of action of stem cell therapy remains elusive, the possible proposed mechanisms for improvement in β-cell mass/function include i) secretion of various growth factors, which may promote angiogenesis and stimulate resident stem cells in the pancreatic duct to differentiate into islets, ii) transdifferentiation into b-cells, and iii) pancreatic and duodenal homeobox 1 (PDX-1) upregulation, thereby enhancing islet differentiation (5,9,11). Recently, Si et al. demonstrated that MSC infusion in the STZ-induced type 2 diabetic rat not only promoted β-cell function, but also ameliorated insulin resistance by enhancing the glucose transporter type 4 (GLUT-4) expression and increasing insulin receptor substrate-1 (IRS-1) and protein kinase B expression in insulin target tissues (28).
Though MSCs are present at a low concentration (<1%) in the bone marrow, their role in the improvement of β-cell function after hematopoietic stem cell transplantation cannot be completely excluded (1,21,26). Jiang et al. have studied the efficacy of placenta-derived MSCs (PD-MSCs) in patients with long-standing T2DM and showed a significant decrease in insulin requirement and increase in C-peptide by 40%, achieving ≥50% reduction in insulin requirement (21).
The potential of stem cell therapy in the management of T2DM is yet to be explored. The data available so far from animal and human studies are encouraging; however, it has enormous limitations. The animal models used for such experiments do not truly represent T2DM. Moreover, the timing of MSC administration after diabetes induction in the rat may also confound interpretation of the results as shown by Si et al. (28). The type of stem cells to be employed, whether hematopoietic, cultured bone marrow-derived MSCs, or placenta-derived MSCs, needs to be defined, as different types of stem cells may have a variable outcome. The preferred route of administration of stem cells, whether targeted or peripheral, also needs to be delineated. In the targeted approach, determining whether administration via the dorsal pancreatic artery, superior pancreaticoduodenal artery, or splenic artery is to be preferred is important, as theoretically this may have a better outcome than simple peripheral administration of stem cells. The number of stem cells required for an optimal response also needs to be determined. The factors that influence the “homing in” and the mechanisms involved in either improvement of β-cell function and/or insulin sensitivity need to be studied further. The glycemic durability demonstrated after stem cell therapy is short lasting; therefore, larger doses or multiple injections at defined intervals may have to be administered to have a long-lasting effect. Last but not least, these patients should remain under close surveillance for the development of any neoplasia, as stem cells being multipotent/pluripotent have a potential for malignant transformation. Therefore, more studies are needed to establish the role of stem cell therapy in the management of T2DM, as well as regarding its efficacy in patients with T2DM of a variable disease duration, its long-term safety, durability, molecular mechanisms, and cost effectiveness.
The strengths of our study include homogeneous population of patients with T2DM, good glycemic control at inclusion, and the presence of the control group. The limitations of the study are a small number of patients, short duration of follow-up, and a lack of metabolic clamp studies to assess the β-cell function and insulin sensitivity.
In conclusion, autologous bone marrow-derived stem cell therapy in patients with T2DM results in a significant decrease in the insulin dose requirement along with an improvement in stimulated C-peptide levels. The efficacy and safety of stem cell therapy needs to be established in more numbers of patients with a longer duration of follow-up.
Footnotes
Acknowledgments
This study was financially supported by Defence Research and Development Organization (DRDO), New Delhi, India. The authors declare no conflict of interest.
