Abstract
Umbilical cord blood-derived mononuclear cell (UCB-MNC) transplants improve recovery in animal spinal cord injury (SCI) models. We transplanted UCB-MNCs into 28 patients with chronic complete SCI in Hong Kong (HK) and Kunming (KM). Stemcyte Inc. donated UCB-MNCs isolated from human leukocyte antigen (HLA ≥4:6)-matched UCB units. In HK, four patients received four 4-μl injections (1.6 million cells) into dorsal entry zones above and below the injury site, and another four received 8-μl injections (3.2 million cells). The eight patients were an average of 13 years after C5-T10 SCI. Magnetic resonance diffusion tensor imaging of five patients showed white matter gaps at the injury site before treatment. Two patients had fiber bundles growing across the injury site by 12 months, and the rest had narrower white matter gaps. Motor, walking index of SCI (WISCI), and spinal cord independence measure (SCIM) scores did not change. In KM, five groups of four patients received four 4-μl (1.6 million cells), 8-μl (3.2 million cells), 16-μl injections (6.4 million cells), 6.4 million cells plus 30 mg/kg methylprednisolone (MP), or 6.4 million cells plus MP and a 6-week course of oral lithium carbonate (750 mg/day). KM patients averaged 7 years after C3-T11 SCI and received 3–6 months of intensive locomotor training. Before surgery, only two patients walked 10 m with assistance and did not need assistance for bladder or bowel management before surgery. The rest could not walk or do their bladder and bowel management without assistance. At about a year (41–87 weeks), WISCI and SCIM scores improved: 15/20 patients walked 10 m (p = 0.001) and 12/20 did not need assistance for bladder management (p = 0.001) or bowel management (p = 0.002). Five patients converted from complete to incomplete (two sensory, three motor; p = 0.038) SCI. We conclude that UCB-MNC transplants and locomotor training improved WISCI and SCIM scores. We propose further clinical trials.
Keywords
Introduction
Umbilical cord blood-derived mononuclear cell (UCB-MNC) transplants improve walking recovery in rat1–18 and canine19–24 spinal cord injury (SCI) models. Investigators gave the cells by intravenous infusion1,25,26, intrathecal injection27,28, or direct transplantation into the spinal cord3,4,6,15,21. A few investigators used allogeneic canine19,23,29 or fetal rat UCB cells 13 . Some directly infused human UCB cells intravenously1,8,15. Others intraspinally transplanted human UCB-MNCs enriched for CD34+ cells3,5–7,10,16,21,25,30–35 or CD45+ cells 28 , isolated from human4,11,12,19,22,24,27,36–46 or canine UCB19,23,29,47, human UCB cells selected for neural characteristics48–53, somatic stem cells 18 , human UCB-MNCs transfected to express growth factors54,55, or human UCB-MNCs combined with olfactory ensheathing glia 56 or lithium chloride 12 . In addition, many groups have studied human umbilical cord tissue-derived cells14,19,24,29,36–39,45,46,57–79.
UCB-MNCs may improve recovery through multiple mechanisms, including secretion of anti-inflammatory cytokines9,30–32, release of growth factors8,10,11,46, upregulation of matrix metalloproteinases 31 , downregulation of tissue plasminogen activator 32 , prevention of apoptosis 30 , facilitation of myelination7,22,49, reduced gliosis24,47, and increased angiogenesis 35 . Although several groups have claimed that UCB cells differentiate into neural precursors80,81 or neural stem cells (NSCs)82–85, more evidence of neuronal or astroglial production by UCB-derived cells transplanted into animal spinal cords is needed.
Lithium stimulates stem cell proliferation 86 , neurogenesis 87 , and regeneration of long spinal tracts88–90. Systemic lithium treatment increases neurotrophin expression in contused rat spinal cords after transplantation of neonatal rat MNCs, including nerve growth factor (NGF), neutrophin-3 (NT-3), and glial cell-derived neurotrophic factor (GDNF) known to stimulate spinal axonal growth 91 . Deng et al. 12 reported that lithium combined with human UCB-MNCs improves locomotor recovery in rats after SCI. We therefore proposed doing clinical trials to assess safety and efficacy of lithium, UCB-MNCs, and UCB-MNCs plus lithium therapy for SCI.
Several groups have transplanted UCB-MNCs and UCB-derived mesenchymal cells into patients with SCI. In 2005, Kang et al. 92 reported hip and thigh movement recovery after transplanting human leukocyte antigen (HLA)-matched UCB “multipotent stem cells” into the spinal cord of a 37-year-old woman with chronic SCI. In 2010, Ichim et al. 93 transplanted UCB-MNCs into the spinal cord of a patient with chronic SCI. In 2011, Cordes et al. 94 transplanted human CD34+ UCB cells into the spinal cord of a patient with amyotrophic lateral sclerosis. In 2013, Yao et al. 95 reported improved autonomic function and somatosensory evoked potentials 12 months after intrathecal and intravenous injection of UCB-MNCs into 25 patients with chronic SCI (≥6 months). Several groups transplanted umbilical cord-derived mesenchymal cells intrathecally into patients with SCI96–98.
We did phase I and II clinical trials in Hong Kong (HK) and Kunming (KM) to assess the safety and efficacy of transplanting escalating doses of HLA-matched UCB-MNCs into the spinal cords of people with chronic (1–19 years after) complete SCI. In the phase I trial in HK, eight patients received a total of 1.6 or 3.2 million UCB-MNCs transplanted into the spinal cord. The patients (on average 13 years after injury) did not receive any walking training, and we did magnetic resonance diffusion tensor imaging (MR-DTI) to visualize long spinal tracts. In the phase II trial in KM, 20 patients with chronic (average of 7 years after injury) complete C5–T11 SCI were sequentially assigned to five treatment groups in which they received 1.6 (group A), 3.2 (group B), or 6.4 (group C) million UCB-MNCs, 6.4 million UCB-MNCs with a 30-mg/kg bolus dose of MP (group D), or 6.4 million UCB-MNCs with MP and a 6-week course of oral lithium carbonate (group E). The patients then started 3–6 months of intensive locomotor training and were assessed at 6 weeks, 3 months, 6 months, and about 1 year after surgery for changes in the American Spinal Injury Association and International Spinal Cord Society (ASIA/ISCOS) impairment scale (AIS) classification97–100, motor and sensory scores, the walking index of spinal cord injury (WISCI)99–104, the spinal cord independence measures (SCIM III)105–107, modified Ashworth scale (MAS) for spasticity108–118, visual analog scale (VAS) for pain, and severe adverse events (SAEs).
Materials and Methods
Inclusion and Exclusion Criteria
The trials included male and female adults (18–60 years old) with chronic (≥1 year), neurologically stable (≥6 months), neurological levels from the 5th cervical (C5) to the 11th thoracic (T11), and complete SCI according to the ASIA/ISCOS impairment scale (AIS A). We excluded people who were in another trial within 4 weeks, had surgical or medical risks, or were pregnant or lactating. Neurological level is the lowest contiguous segmental level where motor score is >3 and sensory score is normal.
Treatments
Patients were assigned sequentially to five treatment groups (n = 4/group). Group A received four 4-μl injections of UCB-MNCs (100,000 cells/μl). Group B received four 8-μl injections. Group C received four 16-μl injections. Group D received 16-μl injections plus a 30-mg/kg intravenous bolus of methylprednisolone sodium succinate (MP; Solumedrol; Pfizer, New York, NY, USA). Group E received four 16-μl injections plus MP and a 6-week course of 750 mg/day oral lithium carbonate (250-mg tablets; Shanghai Xinyi Huanghe Pharmaceutical Co. Ltd., Shanghai, P.R. China); one patient in group E was mistakenly given placebo tablets (Shenzhen Zhijun Pharmaceutical Co. Ltd., Shenzhen, P.R. China). In HK, eight patients were assigned to group A or B (n = 4/group). In KM, 20 patients were assigned to five groups (n = 4/group), and all had locomotor training 6 h/day, 6 days/week for 3–6 months.
Primary Outcome Measure
The primary outcome measure was ASIA/ISCOS motor and sensory scores119,120. Secondary outcomes include AIS, WISCI 121 , SCIM 122 , MAS for spasticity 108 , and VAS for pain 123 . We identified and categorized adverse events (AEs) by severity, relevance, significance, and outcomes.
Adverse Events, Neurological Assessment, and SCIM Assessment
The surgical teams reported and judged the severity and relevance of AEs. At 6 weeks, 6 months, and 1 year after surgery in HK, a physical therapist and an occupational therapist assessed the patients. In KM, a team of one doctor and one nurse evaluated the patients.
Unit Selection
Stemcyte Inc. (Covina, CA, USA) donated frozen plasma-depleted UCB units, and Vista Biologics (Carlsbad, CA, USA) processed the units for MNCs. We selected units that matched at least four out of six (≥4:6) HLAs, that is, initially by low-resolution HLA-A, -B, and -DR typing and later confirmed with medium-resolution (HLA-A and -B) and high-resolution (HLA-DR) typing after transplantation. All units fulfilled the National Marrow Donor Program (NMDP) standards for UCB transplantation 124 . Units that may have had hepatitis B exposure (e.g., positive maternal antibody) were excluded.
Cell Preparation
Vista Biologics prepared the cells for transplantation. Each frozen cord blood unit was thawed at 4°C, washed to reduce dimethyl sulfoxide (DMSO) concentration from 10% to <1%, treated with human DNAase (Pulmozyme; Genentech, San Francisco, CA, USA), and centrifuged at 465 × g in a Ficoll Paque at 1.077 specific gravity gradient (GE Healthcare Life Sciences, Pittsburgh, PA, USA) to isolate the UCB-MNCs. The cells were suspended (1 million cells/ml) in animal product-free CO2-independent media (Invitrogen, Carlsbad, CA, USA) and shipped at 12°C–28°C. We shipped 38 test units from Carlsbad to HK and KM. About 10% of UCB-MNCs were lost per day of shipment. Shipping at room temperature (RT) enriched the UCB-MNCs for monocytes (35%–45%), CD34+, or CD133+ cells (3%–4%), nucleated red cells (1%–2%), and mesenchymal cells (CD105 ~1%).
Transplantation
Upon arrival at the hospital, the cells were washed in saline. An aliquot was removed to count cells that excluded trypan blue dye (Thermo Fisher Scientific, Grand Island, NY, USA). The cells were suspended in normal saline with 1% human albumin (CSL, Melbourne, Australia) so that each microliter had ~100,000 trypan blue excluding cells. The cells were loaded into a 27-gauge (27G × ¾″ or 0.4 × 19 mm; Sterile Scalp Vein Set; EXEL International Co., Los Angeles, CA, USA) needle attached to a 100-μl Hamilton syringe (Hamilton Company, Reno, NV, USA). After laminectomy, durotomy, and removal of adhesions between the spinal cord and surrounding tissues, the surgeons manually inserted the needle 3 mm deep at a 45° angle (bevel up) into the left and right dorsal root entry zones (DREZ) above and below the injury site, and slowly (1 μl/min) injected 4, 8, or 16 μl of cell suspension into each of the four sites. The dura was sutured to prevent cerebrospinal fluid leakage.
Magnetic Resonance Diffusion Tensor Imaging
In HK, we used a 3-Tesla magnetic resonance imaging (MRI) scanner (Achieva X-series; Phillips Medical Systems International, Best, The Netherlands) to obtain MR-DTI of the spinal cord. Conventional MRIs were first obtained (three-dimensional T2W and T2W) to locate the injury site. Fiber tracts were selected for tractography by manually identifying regions of interest (ROI) above the injury site for descending fibers and below the injury site for ascending fibers. Using software purchased from the manufacturer (Phillips Medical Systems International), we quantified the fractional anisotropy of the selected ROI, yielding ratios that indicated the degrees to which diffusion of water is anisotropic, calculated with eigenvalues (λ1, λ2, λ3…) of the diffusion tensor.
Initial scans were obtained from six volunteers without SCI (six of the investigators volunteered). The FA values of normal spinal cords were reproducible, ranging from 0.65 to 0.55 from C1 through T12, declining in the more distal segments. Standard errors of average FA values were usually ±0.05, suggesting that the measurements are reproducible. In addition, we did MR-DTI scans of seven people with complete SCI who did not receive surgery or cell transplantation. All seven showed a gap of DTI signal at the injury site. One person was recruited to be part of the trial, but we could not find a suitable ≥4:6 HLA match for him. This person agreed to have follow-up MR-DTI, and the gap was unchanged 2 years later.
Clear and distortion-free MR-DTI images were obtainable from five of the eight patients in the HK trial. In the remaining three patients, distortion from fixation instruments did not allow the tractography. Once a set of MR-DTI parameters was found to be satisfactory in a patient, further scans on the patient were done with the same parameters, so that the images could be compared over time. Three patients that showed fiber bundles growing across the injury site were rescanned at 1.5 years after surgery, and one patient of these three was rescanned at 2 years. Two patients had long fiber bundles crossing the injury site and growing progressively and long distances into the proximal and distal spinal cord over 12 months or longer.
Locomotor Training
In HK, the clinical trial patients did not receive locomotor training. In KM, at 14 days after surgery, the patients started locomotor training according to the Kunming locomotor scale (KLS) described previously 125 . The patients initially stood with help (KLS II), then without help (KLS III), walked in a rolling walker with minimal (KLS IV) or no assistance (KLS V), or walked with a 4-point walker (KLS VI) without assistance. Patients walked as much as 6 h a day and 6 days a week for 3–6 months. They typically walked 3 h in the morning and 3 h in the afternoon. The patients left the hospital at 3 or 6 months, the latter if they were still improving at 3 months. One-year follow-up times ranged from 41 to 87 weeks (w41–87).
Statistical Analyses
We used SPSS Statistics version 22 (IBM, Armonk, NY, USA) to do repeated-measures analysis of variance (ANOVA) to assess ASIA, SCIM, and VAS score changes over time (Time) and treatment groups (Group); the Kruskal–Wallis test to compare AIS, MAS, WISCI, and KLS among treatment groups; Wilcoxon signed-rank test to compare AIS, MAS, WISCI, and KLS before and w41–87 after treatment; and Spearman correlation to assess the relationship between KLS and WISCI. Missing data were assumed to equal the last observation, that is, last observation carried forward (LOCF). To assess WISCI score changes (DWISCI) among treatment groups, we used ANOVA and Scheffé's post hoc test (Statview 5.01 for Windows; SAS Institute, Cary, NC, USA). We used the chi-square contingency test (Prism 6; GraphPad Software, La Jolla, CA, USA) to assess HLA matching among treatment groups. All ± values indicate SEs of the mean, values of p < 0.05 were considered significant, and analyses were based on intention to treat. The figures show only scored categories.
Approvals and Registrations
Institutional review boards (IRBs) of the Chinese University of Hong Kong, University of Hong Kong, and Kunming People's Liberation Army Hospital of Chengdu Military Command approved the trial. The Hong Kong Department of Health approved the trial. The Western IRB (Seattle, WA, USA) gave an “approvable” rating for both trials. The Military Medical Ministry and Yunnan Department of Science and Technology approved and awarded grants for the KM trial. The trials were registered on https://www.clinicaltrials.gov as NCT01046786 and NCT01354483.
Results
Patients
Eight patients (1 female, 7 males) participated in HK, and 20 patients (4 females, 16 males) participated in KM. At enrollment, the patients in HK averaged 42.6 ± 2.7 (range 29–53) years of age, and those in KM averaged 36.9 ± 2.4 (range 18–53) years. The years after injury averaged 12.8 ± 2.6 (2–20) and 7.2 ± 1.2 (2–20) years. All 8 patients in HK had “complete” injury as indicated by AIS A classification, and 19/20 patients in KM were AIS A with C5–T11 neurological levels, and 1 was sensory incomplete (AIS C) with C3 neurological level. Figures 1 and 2 summarize the HK and KM data. All had significant spinal fractures, and most had metallic implants to stabilize the spinal fractures. In HK, all patients had a WISCI score of 0. In KM, two patients had a WISCI score of 2 before treatment.

Neurological levels and scores in Hong Kong (HK). Each column represents a patient in the trial (R=right, L=left side of the body): dark green indicates segments with normal sensation and motor function, dark green with white letters indicates the neurological level before treatment, red with white letters indicates changed neurological level 1 year after treatment (48w), light green indicates zone of partial preservation (ZPP) before treatment, and pink indicates ZPP after treatment. Individual patient data are listed, including Age (in years) and Years (after injury), Sex (M = male, F = female), the number of human leukocyte antigen (HLA) matches out of 6 at medium–high resolution (medium for two HLA-A and two HLA-B and high for two HLA-Dr). AIS refers to the ASIA/ISCOS impairment scale, where A is complete, B is sensory incomplete, and C is motor incomplete so that the patients have <50% of normal motor scores below the neurological level. None of the patients received walking training or recovered walking; hence, no Kunming locomotor scores (KLS) or walking index of spinal cord injury (WISCI) scores are listed. Motor score is the sum of muscle grades (0–5) for 10 muscles on each side of the body, totaling 100 points. Touch and Pin, respectively, refer to light touch and pinprick scores (0 = no, 1 = abnormal, and 2 = normal) for 28 dermatomes on each side of the body. MAS is the modified Ashworth scale (0–4) for spasticity. VAS is visual analog scale (0–100) for pain. SCIM is spinal cord independence measure (0–100). The score changes are color coded: red indicates improvement, pink is slightly better, and green is worse. SAE (yellow) refers to severe adverse events, and the codes for specific events are listed on the right.

Neurological levels and scores in Kunming (KM). Each column represents a patient with four patients per treatment group (groups A, B, C, D, and E). Two patients converted from complete to sensory incomplete, and three patients were converted to motor incomplete (AIS C). Five patients had severe adverse events (SAE): WH, wound healing; TF, tibial fracture; KS, knee swelling. †Anal sensation; ‡anal sensation and voluntary anal contraction. See legend in Figure 1 for explanation.
Treatments
Cord blood units were selected on the basis of low-resolution HLA matching (HLA ≥4:6) for HLA-A, -B, and -DR and then checked for medium- (HLA-A and -B) and high-resolution (-DRB1) matching after transplantation. In HK, two patients matched 6:6 and six matched 5:6 at low resolution, but subsequent medium- and high-resolution matching showed one participant matching at 6:6, one at 5:6, and six at 4:6. In KM, 6 patients matched 6:6, 10 patients 5:6, and 4 patients 4:6 at low resolution, and subsequent medium- and high-resolution matching showed 9 patients matching at 5:6, 7 at 4:6, 3 at 3:6, and 1 at 1:6. Low-resolution HLA matches differed among KM treatment groups [χ 2(8) = 15.67, p = 0.0474] but medium- and high-resolution matches did not [χ 2(12) = 13.17, p = 0.3565]. All patients received planned doses of cells. One group E participant (#18) received placebo instead of lithium. Three KM patients did not complete locomotor training, two due to possible tibial fractures (#2, #8) and one due to knee swelling (#20).
Diffusion Tensor Imaging
MR-DTI showed gaps at the injury site (Fig 3). At 6–18 months, two patients had progressive fiber growth crossing the gap. Figure 4 shows pretreatment, 6-month, and 1-year MR-DTI of a patient with a 4th thoracic (T4) neurological SCI level. Figure 5 shows MR-DTIs of an uninjured spinal cord, spinal cords with narrow and wide gaps, fibers crossing the gap, and two spinal cords with narrower gaps at 12 months.

Magnetic resonance diffusion tensor image (MR-DTI) of the spinal cord before treatment. White matter tracts were selected from regions of interest (ROIs) above and below the injury site, and the tract-tracing software was then used to identify adjacent pixels with similar diffusion tensors. Descending tracts are colored purple and green, while ascending tracts are colored blue. A clear gap was present in the spinal cord at C6 vertebral level.

Magnetic resonance diffusion tensor images (MR-DTI) of the spinal cord of a participant before operation (Pre-op), at 6 months (6m), and at 12 months (12m) after treatment. Before operation, MR-DTI showed very atrophic descending fibers (blue), more ascending fibers (green), and a clear gap at the T4 injury site. At 6 months, the gap was still present. At 12 months, both ascending and descending fibers were crossing the gap (upper right). On the lower right image, ascending fibers (dark blue) were removed so that the descending fibers (light blue) could be seen extending into the lumbosacral spinal cord.
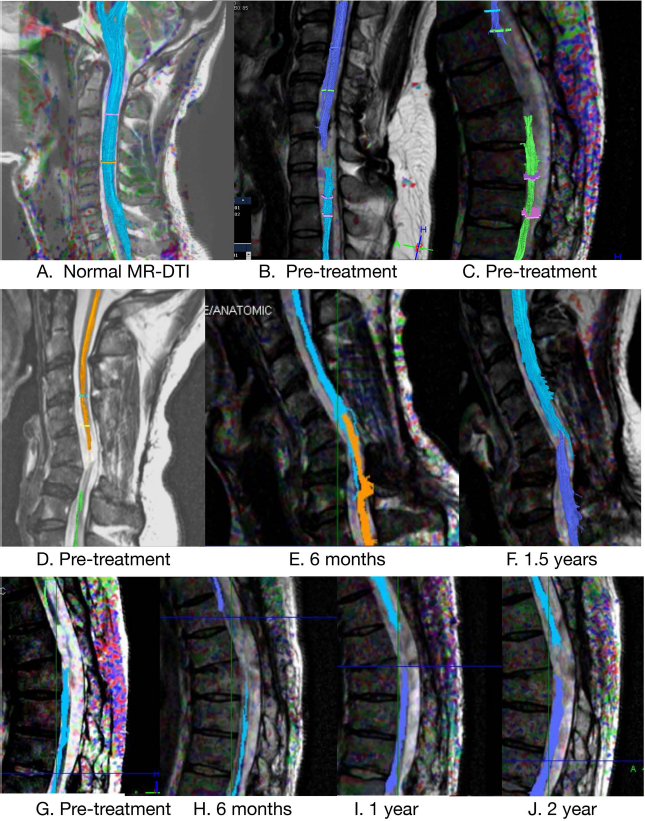
Magnetic resonance diffusion tensor images (MR-DTI) of a normal cervical spinal cord (A), an image of an injured spinal cord with a narrow gap before treatment (B), and an image of an injured spinal cord with a wide gap before treatment (C). (D–F) MR-DTI from a patient before, at 6 months after, and at 1.5 years after treatment. Note the fibers crossing the gap. (G–J) MR-DTI from a participant before, at 6 months, at 1 year, and at 2 years after treatment. Note the narrowing of the white matter gap.
Adverse Events
Nine AEs occurred in three patients in HK. One participant developed neuropathic pain (probably related), hyperthyroidism (probably unrelated), and hypertension (probably unrelated). A second participant developed a thin subdural hematoma and pneumocephalus due to cerebrospinal fluid (CSF) loss during surgery. He also developed a decubitus. Both resolved spontaneously. A third participant developed subarachnoid hemorrhage (definitely related), neuropathic pain (probably related), and colon cancer (not related); the first two resolved spontaneously. In KM, 68 AEs occurred in 19 patients: 43 were unrelated, 17 definitely, 1 probably, and 7 possibly related to treatment. The most common event was postoperative wound swelling and pain in nine patients. All AEs resolved with routine therapies. No patient had motor or sensory loss. In the 28 patients in the two trials, 5 had SAEs. One patient (KM group A, #1) (Fig 2) had slow wound healing and low serum protein; both resolved on a high-protein diet. Another (KM group A, #2) (Fig 2) developed a CSF leak and wound dehiscence that required reoperation. He was later found to have an old tibial fracture and stopped locomotor training. A third patient (HK group A, H02-2) (Fig 1) had blood pressure increases requiring hospitalization. A fourth patient (KM group C, #12) had left leg swelling and thrombosis of vena iliaca externa treated by vena cava filter (unrelated). A fifth participant (HK group B, H01-3) had colon adenocarcinoma discovered at 21 months after treatment (probably unrelated).
Spasticity and Pain
In HK, the eight patients had no or mild spasticity before and after treatment. Three had severe neuropathic pain (VAS >50) before treatment that decreased (88 to 76, 51 to 28, and 67 to 0) after treatment, and two developed neuropathic pain after treatment (0 to 62 and 12 to 24). In KM, five patients had mild spasticity (MAS = 1) and two had moderate spasticity (MAS = 2) before treatment; at w41–87, seven patients had 1-point increases of MAS scores, while two patients had 1-point decreases that were not significant [χ 2(4) = 2.977, p = 0.562]. Before treatment, five patients had VAS scores of 12–50 out of 100. Between w0 and w41–87, the VAS score increased in three patients (range +15 to +69) but decreased in four patients (range −5 to −43), but these increases were not statistically significant [Time: F(1) = 0.015, p = 0.905; Group: F(4) = 0.0470, p = 0.757; Group × Time: F(4) = 1.232, p = 0.339]. Two patients in group E had high VA S scores; lithium reduced VAS scores in both.
ASI A Grade, Levels, and Scores
In HK, one of eight patients (7%) converted from AIS A to B, four gained 2–5 points in touch scores, and two had 2–3 point motor score increases. In KM, two patients switched from AIS A to B (10%) and three from AIS A to C (15%). Neurological levels descended one segment in six patients (30%). Ten patients (50%) gained 1–10 touch points, and nine (45%) gained 1–8 pinprick points between w0 and w41–87. Mean sensory scores increased over time, that is, touch scores increased 1.7 points [Time: F(1) = 9.869, p = 0.007; Group: F(4) = 0.346, p = 0.299; Group × Time: F(4) = 0.535, p = 0.712], and pinprick scores increased 2.6 points [Time: F(1) = 8.984, p = 0.009; Group: F(4) = 8.984, p = 0.284; Group × Time: F(4) = 0.455, p = 0.768]. Motor scores did not change significantly [Time: F(1) = 1.800, p = 0.200; Group: F(4) = 0.145, p = 0.962; Group × Time: F(4) = 0.800, p = 0.544], and one participant gained 2 and another gained 4 points.
Locomotor Training
Patients in HK did not receive locomotor training. In KM, 17 of 20 patients received intensive locomotor training. Before treatment, 4 patients could not stand (KLS I), 15 needed help to stand (KLS II), and 1 walked in a rolling walker with minimal assistance (KLS IV), that is, an assistant pulled on ropes to stabilize knees during walking. By w14–24, 17 of 20 patients (85%) were training at KLS IV (Fig 6). Nine patients went home at 14 weeks because they reached a plateau (n = 6) or stopped training (n = 3) due to tibial fractures in two cases or swollen knee in one case. After going home, four patients did not continue walking and regressed. At w41–87, only 13 (65%) walked at KLS IV, and 2 (10%) walked unassisted with 4-point walkers (KLS VI). KLS at w41–87 differed significantly from w0 (Z = 3.532, p < 0.0005).

KLS and WISCI. KLS represents locomotor training stages: I indicates inability to stand; II, standing with assistance; III, standing without assistance; IV, walking in a rolling walker with minimal assistance; V, walking in rolling walker without assistance; VI, walking in 4-point walker without assistance; VII, walking with crutches; VIII, walking with a cane; IX, unstably walking without devices; and X, walking stably with a device. No patient had a KLS score of V, VII, VIII, IX, or X. WISCI reflects ability to ambulate 10 m (10m) with devices, braces, and assistants: 0 indicates inability to stand or to participate in assisted walking; 1 is ambulating <10 m; 2 is ambulating 10 m in parallel bars with braces and two assistants; 3 is ambulating 10 m in parallel bars with braces and one assistant; 4 is ambulating 10 m in parallel bars with no braces and one assistant; 5 is ambulating 10 m in parallel bars without braces or assistant; 6 is ambulating 10 m with a walker with braces and one assistant; 7 is ambulating 10 m with two crutches, braces, and one assistant; 8 is ambulating 10 m with walker, no braces, and one assistant; 9 is ambulating in a walker with braces and no assistant; 10 is ambulating with one cane or crutch, no braces, and one assistant; 11 is ambulating with two crutches, no braces, and one assistant; 12 is ambulating with two crutches, braces, and no assistant; 13 is ambulating in a walker without braces or assistants. No patients received WISCI scores of 3–5, 8, 10–12, and 14–20. Missing w24 data were assumed to equal w14 data, and w48 refers to w41–87.
Walking Recovery
In HK, no patients walked before or after treatment. In KM before treatment, 16 patients (80%) could not walk (WISCI 0), 1 walked <10 m (WISCI 1), 1 walked 10 m (WISCI 2) in parallel bars with braces and two assistants, and 2 walked 10 m in a walker with braces and no assistants (WISCI 9). At w41–87, five patients had WISCI 0, two were WISCI 2, six (30%) walked 10 m with one assistant (WISCI 6), and seven (35%) walked without assistance (WISCI ≥7). WISCI scores at w41–87 differed from W0 (Z = 3.315, p = 0.001). WISCI and KLS correlated highly (r = 0.925, p < 0.0005).
SCIM Scores
The SCIM has 19 subscores separated into three subtotal scores covering self-care, respiration and sphincter management, and mobility. In HK, SCIM scores did not change significantly. In KM, most patients had increases in their SCIM scores. Repeated-measures ANOVA confirmed that mean SCIM scores increased over time (F = 51.194, p < 0.0005) by 19.6 ± 2.67 points between w0 and w41–87. Fourteen of 19 SCIM subscores, that is, except feeding, grooming, respiration, outdoor mobility (>100 m), and stair management, improved significantly between w0 and w41–87 (Figs. 7 and 8). The self-care subtotal score accounted for 3.6 points, while the respiration and sphincter management subtotal accounted for 9.7 points, and the mobility subtotal accounted for 6.3 points. The bladder management, bowel management, and use of toilet subtotal score accounted for almost half of the SCIM score improvement. At w0, only 2 (10%) patients required assistance for bladder and bowel management, but 12 (60%) became independent by w41–87. Likewise, 65%–70% of patients could transfer from bed to wheelchair and from wheelchair to toilet and tub at w41–87 without assistance, compared to 5%–30% at w0. At w41–87, 35% of patients could walk without assistance or supervision indoors and for moderate distances up to 100 m. For distances >100 m, only two patients walked, and the rest used wheelchairs. Table 1 shows means and standard errors of total SCIM and subscores, as well as t values, Wilcoxon Z values, and p values.

Spinal cord independence measure (SCIM) mobility scores. Indoor (indoor mobility on even surfaces), moderate distances (10–100 m), and outdoors (>100 m): 0 = total assistance; 1 = electric wheelchair or manual-assisted wheelchair; 2 = moves independently with manual wheelchair; 3 = walks with supervision; 4 = walks with walking frame or crutches by swinging; 5 = walks with crutches or two canes with reciprocal gait; 6 = walks with one cane; 7 = needs leg orthosis only; and 8 = walks without walking aids. Stair management (go up or down stairs): 0 = total assistance; 1 = ascends and descends ≥3 steps with support or partial assistance; 2 = ascends and descends at least three steps with rail, crutch, or cane; and 3 = ascends and descends at least three steps without support or supervision. Bed mobility (turn in bed and actions to prevent pressure sores): 0 = total assistance to turn and to sit up in bed, to push up in wheelchair, with or without adaptive devices; 1 = performs one of the above actions without assistance; 2 = performed two to three of above without assistance; and 3 = independent. Ground wheelchair (get into wheelchair from the ground): 0 = total assistance and 1 = transfers independently with or without adaptive devices. SCIM scores that were not assigned to any patient were omitted from the figure.

Spinal cord independence measure toilet and transfer functions. Bladder management: 0 = indwelling catheter; 3 = residual urinary volume (RUV) >100 ml with assisted catheterization; 6 = RUV <100 ml with intermittent self-catheterization (ISC) and external drainage (ED) with assistance; 9 = ISC and ED without assistance; 11 = ISC without ED; 13 = RUV <100 ml, only ED and no assistance; and 15 = RUV <100 ml, no ED. Bowel management: 0 = irregular or very low frequency movements <1/3 days; 5 = regular, requires assistance for suppositories, rare accidents <2/month; 8 = regular without assistance, rare accidents; and 10 = regular, no assistance or accidents. Use of toilet: 0 = total assistance; 1 = partial assistance, does not clean self; 2 = partial assistance, cleans self; 4 = independent but requires adaptive device; and 5 = independent without adaptive devices. Wheelchair–toilet (transfers to and from toilet, locking wheelchair, lifting footrests, and removing and adjusting armrests): 0 = total assistance; 1 = partial assistance or supervision; and 2 = independent. Bed–wheelchair (transfers from bed to wheelchair): 0 = total assistance to lock wheelchair, lift footrests, remove and adjust arm rests, transferring, lifting feet; 1 = partial assistance or supervision and/or adaptive devices; and 2 = independent or does not require wheelchair. Wheelchair–car (approach a car, lock wheelchair, remove arm and footrests, transfers to and from car, and bring wheelchair into and out of car): 0 = total assistance; 1 = partial assistance or supervision/adaptive devices; and 2 = independent.
SCIM Scores and Subscores

R & S refers to respiration and sphincters, R & T refers to room and toilet, I & O refers to indoors and outdoors. Week 0 is baseline score, while Week 48 represents the mean scores ± SEM of weeks 41–87 with 95% confidence interval (CI 95%). Δ is the difference (mean ± SEM) between Week 0 and Week 48 scores. We used paired samples t-test and Wilcoxon signed ranks test to compare findings at Week 0 and Week 48.
Treatment Effects
Comparison of walking scores among the treatment groups revealed that all eight patients in groups B and C showed improvement of WISCI scores by 6 points or greater. Only two patients showed improved walking in groups A and D, and only one participant in group E. Although this was not statistically significant, the data do not support beneficial effects of MP or lithium. Almost every participant except for one in group B showed improved SCIM scores. SAEs did not differ among the groups, occurring in four of the five treatment groups, with two in group A, one in group B, one in group C, and one in group D. Figure 9 shows the ANOVA of change of WISCI scores in the five treatment groups. Mean change of WISCI scores increased from 3.5 to 6.1 to 9.1 in groups A, B, and C, fell to 2.9 in group D, and fell to 0.4 in group E.

Analysis of variance (ANOVA) of change of WISCI between week 0 and week 48. The ANOVA table indicated an F value of 6.765 (p = 0.0026) among treatment groups (Tx). The lower left graph shows means and standard errors of mean. Group A received the lowest dose of 1.6 million cells, B received a higher dose of 3.2 million, groups C–E received the highest dose of 6.4 million, group D received the highest cell dose plus 30 mg/kg methylprednisolone (MP), and group E received the highest cell dose plus MP and a 6-week course of oral lithium carbonate.
Discussion
The HK trial showed that 4–8 μl of UCB-MNCs can be safely injected into the spinal cord above and below the injury site. MR-DTI suggest that white matter gaps decreased at the injury site, and two patients showed bundles of fibers growing across the injury site into surrounding spinal cord at 6–18 months. However, the patients did not have recovery of motor function. The KM trial showed that 4, 8, and 16 μl of UCB-MNCs can be safely injected into the spinal cord. Over half of the patients recovered walking with minimal or no assistance by 6–12 months after UCB-MNC transplants and locomotor training, as well as increased independence in activities of daily living, including self-care, bowel and bladder management, and mobility. This is an unprecedented recovery for complete chronic SCI.
Walking recovery is rare in patients with chronic complete SCI102,126,127. The finding that 15/20 patients (75%) with chronic complete SCI could walk 10 m and 7 (35%) walked 10 m without manual assistance a year after treatment is unprecedented. SCIM indoor mobility subscores confirmed that seven patients (35%) walked indoors without assistance at 1 year after treatment. Likewise, mobility subscores for moderate distances showed the same seven patients (35%) walking 10–100 m without assistance, two (10%) walking with supervision, and the rest using wheelchairs. For distances of >100 m, only two patients (10%) walked, while the rest used wheelchairs. Thus, 35% of patients used walking for indoors and for moderate distances <100 m, but most patients preferred wheelchairs for longer distances >100 m.
Late conversions from AIS A to B or C are also rare. As Kirshblum et al. 128 pointed out, among 987 patients who were neurologically complete (AIS A) at 1 year, only 3.5% improved to AIS B and 1.05% improved to AIS C and D by 5 years. In HK, one of eight patients converted from AIS A to B (12.5%). In KM, two patients converted from AIS A to B (10%) and three from AIS A to C (15%), a conversion rate of 25%. These changes were statistically significant (Z = 2.070, p = 0.038) but did not vary among treatment groups.
SCIM scores indicated significant improvements in independence for bladder management. Before treatment, 18 patients (90%) required assistance for bladder management, and 15 patients (75%) used catheters. At w14–24, 16 patients (80%) were still catheterizing. At 1 year after treatment, 12 (60%) patients did not need assistance for bladder care, and 11 patients (55%) no longer used catheters. Three patients (15%) did not use catheters or drainage devices.
Bowel management also improved. Before treatment, six patients (30%) had irregular or low-frequency bowel movements (<1/3 days), and 90% required assistance. By discharge from hospital at 14–24 weeks, all patients became regular, but 75% still needed assistance and had occasional accidents. By w41–87, however, 60% did not require assistance and rarely had accidents. Four patients (20%) had no accidents. We plan to bring the patients back for further evaluation.
MP and lithium may have reduced walking recovery. Animal studies suggest that MP improves survival of transplanted cells 129 , and lithium should improve walking recovery 90 after acute SCI. Only two of eight patients (25%) in groups D (6.4 million cells plus MP) and E (6.4 million cells plus MP and lithium) recovered walking to 6 points on WISCI, compared to eight of eight patients (100%) in groups B (3.2 million cells) and C (6.4 million cells). Lithium reduced neuropathic pain in two patients in group E, consistent with our earlier report 130 .
Change in WISCI scores (ΔWISCI) increased with cell dose. As Figure 9 shows, ANOVA of ΔWISCI in the five treatment groups showed progressive increases in ΔWISCI in groups A, B, and C, but ΔWISCI decreased in groups D and E. Post hoc tests (Scheffé's) suggest that groups C and E differed significantly at p < 0.0051. However, one patient (#18) in group E inadvertently received placebo rather than lithium tablets, one patient (#19) was already walking at WISCI 9 when the trial began, and one patient (#20) stopped walking training due to knee swelling. Thus, only one patient in group E represented a valid comparison with group C. Further clinical trials are needed to determine whether lithium is effective.
Two other findings are noteworthy in the KM trial. First, two patients had to discontinue locomotor training due to old bone fractures. We should screen patients in future trials for old bone fractures. Second, many patients did not show improved motor scores despite recovery of ability to walk and other programmed spinal cord functions, that is, micturition and defecation. We hypothesize that UCB-MNC transplants stimulated growth of axons that activate lumbosacral central pattern generators for walking, micturition, and defecation but only in patients who received intensive locomotor training 131 . This would explain why most patients could walk but could not voluntarily contract individual muscles or feel specific sensory signals in their legs at 1 year after treatment. It is possible that some patients will recover more voluntary motor and sensory function later.
Our trials left several critical questions unanswered. First, can intensive locomotor training alone improve locomotor function in people with chronic complete SCI? Several years ago, most doctors would have replied that locomotor training alone cannot restore locomotion to people with chronic complete SCI. Second, does untethering surgery improve the effects of intensive locomotor training? In our trials, all patients that received transplants also received untethering surgery. Many neurosurgeons have reported beneficial effects of untethering surgery in patients with spina bifida or syringomyelic cysts116–131. Third, does lithium improve locomotor recovery when combined with UCB-MNCs and intensive locomotor training? We have previously observed that a 6-week course of lithium does not improve motor or sensory function in patients with chronic complete SCI, but these patients did not receive any locomotor training 114 . If lithium does not improve function when combined with UCB-MNCs and locomotor training, we should exclude it from future phase III trials.
We have proposed further phase II trials to answer these questions. The first trial will ascertain whether locomotor training alone or untethering surgery plus locomotor training restore walking in people with chronic complete SCI. This trial (NCT02663310, http://clinicaltrials.gov) is underway in KM, comparing walking outcomes of 30 people with chronic complete SCI, randomized to untethering surgery or no surgery, followed by 6 months of intensive locomotor training. We have applied for two phase II trials, one in India and the other in the US, to ascertain whether lithium improves locomotor recovery of patients with chronic SCI, randomized to UCB-MNC transplants or UCB-MNCs plus a 6-week course of lithium, followed by 6 months of intensive locomotor training.
These trials will provide the following important information needed for the design of pivotal phase III trials of UCB-MNC treatment. First, if the trials show no significant benefits of adding lithium to UCB-MNC transplants, lithium should be omitted from the phase III trials. Second, if locomotor training alone or untethering surgery plus locomotor training improves walking recovery in patients with chronic complete SCI, it would provide justification for a surgery control group involving untethering only. Finally, intensive locomotor training (6 h a day, 6 days a week for 6 months) has not been practiced outside of KM. It is important to establish that such training is feasible elsewhere since our trials to date suggest that intensive locomotor training is essential for recovery of walking.
In summary, our data indicate that UCB-MNCs can be safely transplanted into the spinal cords of people with chronic SCI, intensive locomotor training is essential for motor recovery, and UCB-MNC transplants combined with intensive locomotor recovery can lead to significant locomotor, bowel, and bladder recovery in people with chronic complete SCI. However, the patients did not recover much voluntary motor function. Some patients recovered sensory dermatomes close to the injury site, and as many as a quarter of the patients recovered anal sensation and voluntary sphincter contraction, converting from AIS A to B and C. Further clinical trials are necessary to determine whether these improvements are due to UCB-MNCs, untethering surgery, or intensive locomotor training.
Footnotes
Acknowledgments
We thank the patients and their families for their hard work and the nurses who cared for them. The Hong Kong Spinal Cord Injury Fund, Stemcyte Inc. (Covina, CA, USA), Yunnan Department of Science and Technology, General Hospital of Chengdu Military Command, Science & Technology Planning and Key Technology Innovation Projects of Guangdong (20148050504006), and the Tongren Hospital in Kunming helped fund the study. We thank Stemcyte Inc. for their generous donation of cells for the trials and support of the trials. We are very grateful to Vista Biologicals for their careful work preparing the cells for transplantation. Conception and study design: Wise Young, John Chen, Wendy Cheng, Waisang Poon, Gilberto K.-K. Leung, Hui Zhu, and Kwok-Fai So. Data acquisition: Hui Zhu, Waisang Poon, Gilberto K.-K. Leung, Yatwa Wong, Yansheng Liu, Stephanie C. P. Ng, Kam Sze Tsang, David T. F. Sun, David K. Yeung, Yaping Feng, Fang Niu, Penglu Tan, Caihong Shen, Zhexi Xu, Shaofeng Tang, Hongkun Gao, Yun Cha, Jan Lai, Dongming Sun, and Wendy Cheng. Analysis and data interpretation: Wise Young, John Chen, and Wendy Cheng. Drafting of manuscript: Wise Young. Revising of manuscript: Wise Young, John Chen, Wendy Cheng, Hui Zhu, Waisang Poon, and Gilberto K.-K. Leung. All the authors participated in approval of the drafts and revisions. The authors declare no conflicts of interest.
