Abstract
Our research group investigates whether human mononuclear cells isolated from umbilical cord blood (HUCBM cells) might be valuable in hepatic regenerative medicine. We recently demonstrated that HUCBM cell transplantation improves histological alterations and function of the liver in rats with acute liver damage induced by D-galactosamine. In the present study, HUCBM cells were transplanted into rats with thioacetamide (TAA)-induced liver cirrhosis, an experimental model that generates an intense fibrosis and mimics the histological and biochemical alterations found in the human disease. HUCBM transplantation had no effect on hepatic histology of cirrhotic animals. In contrast, analysis of plasma albumin and total bilirubin, liver damage markers, revealed a harmful effect of HUCBM cell transplantation in our experimental model of liver cirrhosis. Significantly higher plasma urea concentrations, marker of renal function, were observed in the cirrhotic and control rats intraportally injected with HUCBM cells than in those not receiving this therapy. Histological study revealed tubular and glomerular lesions in kidneys of cirrhotic animals transplanted with HUCBM cells. The glomeruli appeared ischemic, and the tubules showed a severe involvement that included peripheral asymmetric vacuolization and disappearance of the tubular lumen. Taken together, the histological and biochemical data suggest that the cirrhotic rats subjected to HUCBM cell therapy developed a hepatorenal syndrome.
Keywords
Introduction
Liver diseases affect approximately 17% of the world population and entail high social costs (11). Whole liver transplantation, the current therapy for end-stage hepatic disease, is limited by the shortage of organ donors (20). Furthermore, there is no specific treatment for the liver fibrosis that develops in chronic hepatic diseases, regardless of their etiology (22). Accordingly, novel therapies are required to alleviate the suffering of many patients.
Cell transplantation represents one new therapeutic approach. Hepatocytes and hematopoietic stem cells are currently being investigated in regenerative medicine of the liver, and numerous groups have studied the transplantation of hepatocytes in experimental models of liver disease (8,14). Similar experiments have been performed in humans in various countries, with encouraging results (3,5,19).
Hematopoietic stem cells from bone marrow (BMSC) or umbilical cord blood (UCBSC) are also being tested for this purpose. BMSC gave rise to functional hepatocytes in the liver of mice with tyrosinemia (9), and UCBSC produced similar results in mice with liver damage due to overexpression of Fas ligand (13). BMSC were reported to reduce CCl4-induced liver fibrosis in mice (17,18), and were found to improve human liver function in noncontrolled and nonrandomized phase I clinical trials (6,21).
Our research group investigates whether human mononuclear cells isolated from umbilical cord blood (HUCBM cells) might be valuable in hepatic regenerative medicine. We recently demonstrated that HUCBM cell transplantation improves histological alterations and function of the liver in rats with acute liver damage (1). In the present experimental study, we portally injected HUCBM cells into rats with hepatic cirrhosis induced by thioacetamide, an experimental model of chronic liver disease that generates an intense fibrosis and mimics the histological alterations found in the human disease (4,16).
Materials and Methods
Reagents
Dulbecco's modified Eagle's medium (DMEM), fetal bovine serum (FBS), and phosphate-buffered saline (PBS) were from Invitrogen (Paisley, UK). Histopaque was from Sigma (St. Louis, MO). Cyclosporin A was purchased from Fluka (Seelze, Germany), and the anti-asialo GM1 antibody was from Wako (Osaka, Japan).
Isolation of HUCBM Cells From Umbilical Cord Blood
Human umbilical cord blood samples were obtained in heparinized tubes from full-term deliveries, under informed consent, according to approved procedures at the Virgen de las Nieves Hospital (Granada, Spain). Samples were diluted 1:1 in DMEM medium supplemented with 10% FBS. Low-density mononuclear cells were collected after centrifugation at 800 × g for 20 min in Ficoll density gradient (Histopaque, 1.077g/ml) following the manufacturer's instructions. Mononuclear hematopoietic cells were obtained from the interphase and washed twice with sterilized PBS. Pellet was resuspended in lysis buffer (150 mM NH4Cl, 1 mM KHCO3, 0.1 mM Na-EDTA, pH 7.4) and incubated for 5 min at 4°C to deplete erythrocytes. After washing once with PBS, pellet was again resuspended.
Cell viability, determined by the trypan blue dye exclusion method, was 97.40 ± 0.43%. The total average number of viable cells isolated from one umbilical cord was 1 × 108.
Experimental Model of Liver Cirrhosis
All procedures involving rats were approved by the Animal Care Committee of the University of Granada and conformed to European Union Animal Care Regulations. Ninety-five male Wistar rats weighing 100–130 g were divided into two groups: cirrhotic (T) and control (C). The cirrhotic group (n = 49) received 300 mg thioacetamide (TAA)/L drinking water for 4 months, whereas the control group (n = 46) received water for the same time period (4,16).
Transplantation Experiments
The experimental design of the transplantation experiments is depicted in Figure 1. Four months after administration of TAA or water, six rats each from group (C and T) were sacrificed to serve as reference groups (day 0), and to confirm the presence of cirrhosis. The remaining animals were divided into six groups: group C animals (n = 18) drank water and received no treatment; group T animals (n = 18) drank TAA; group TU animals (n = 13) received TAA plus intraportal injection of 10 × 106 HUCBM cells; group TS animals (n = 12) received TAA plus intraportal injection of culture medium without cells; group CU animals (n = 13) received water plus intraportal injection of 10 × 106 HUCBM cells; group CS animals (n = 9) received water plus intraportal injection of culture medium without cells.
Design of the transplantation experiment. Ninety-five male Wistar rats weighing 100–130 g were divided into six groups. Group C received no treatment (control). Group T received TAA. Group CS received immunosuppressive therapy (cyclosporine A and anti-asialo GM1 antibody) and an intraportal injection of culture medium without cells. Group CU underwent immunosuppressive therapy and received an intraportal injection of 10 × 106 HUCBM cells. Group TS received TAA, immunosuppressive therapy, and an intraportal injection of culture medium without cells. Group TU received TAA, immunosuppressive therapy, and an intraportal injection of 10 × 106 HUCBM cells. TAA was administered in the drinking water at a dose of 300 mg/L. TAA treatment was never discontinued, although TAA concentration was lowered to 150 mg/L during the first week immediately after cell transplantation/sham operation (week 17). TAA concentration was again raised to 300 mg/L thereafter. Cyclosporin A was administered intramuscularly and daily starting the day before cell transplantation or sham operation at a dose of 15 mg/kg. Two intraperitoneal injections of 100 μl each of the anti-asialo GM1 antibody were administered the day before and at day 11 after cell transplantation/sham operation. Animals were sacrificed under anesthesia with sodium pentobarbital at 7, 14, and 21 days posttransplantation. The arrows indicate times of sacrifice. Tx: transplantation. The number of animals (n) sacrificed per group at each time is shown.
TAA treatment was never discontinued, although TAA concentration was lowered to 150 mg/L during the first week immediately after cell transplantation/sham operation (week 17). TAA concentration was again raised to 300 mg/L thereafter.
With the exception of groups C and T, all animals (receiving water, TAA, injection of cells or culture medium, i.e., groups CS, CU, TS, and TU) were subjected to immunosuppressive therapy with both cyclosporin A and an anti-asialo GM1 antibody (1). Cyclosporin A was administered intramuscularly and daily starting the day before cell transplantation or sham operation, at a dose of 15 mg/kg. Two intraperitoneal injections of 100 μl each of the anti-asialo GM1 antibody were administered the day before and at day 11 after cell transplantation/ sham operation.
Animals were sacrificed under anesthesia with sodium pentobarbital at 7, 14, and 21 days posttransplantation. The number of animals sacrificed at each time point appears in Figure 1. Blood was collected in tubes containing heparin and centrifuged to separate plasma from cells. The liver was excised and divided into four pieces. The first piece was for histological analysis. The second was for flow cytometry analysis. The third was kept in RNAlater (Ambion, Austin, TX) and used for total RNA isolation. The fourth was for protein extraction and Western blot analysis. Samples of kidneys were also taken for histological analysis.
Western Blot
Protein extraction and Western blot were performed as described elsewhere (12). Cell lysates were prepared in 0.25 mM sucrose, 1 mM EDTA, 10 mM Tris, and 1% protease inhibitor cocktail (Roche, Mannheim, Germany). Samples containing 40 μg of protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (9% acrylamide) and transferred to nitrocellulose. Nonspecific binding was blocked by preincubation of the nitrocellulose in PBS containing 5% skimmed dry milk for 1 h. The nitrocellulose was then incubated overnight at 4°C with a rabbit polyclonal anti-human cytochrome P450 3A4 antibody (Abcam). Bound primary antibody was detected with an HRP-conjugated anti-rabbit antibody (DAKO, Glostrup, Denmark), and blots were developed using an enhanced chemiluminescence detection system (ECL kit, Amersham Pharmacia, Uppsala, Sweden). Equal loading of the gels was demonstrated by probing the membranes with a rabbit polyclonal anti-human β-actin antibody (Sigma-Aldrich, Madrid, Spain).
Determination of Plasma Markers of Hepatic and Renal Function
Activities of aspartate aminotransferase and alanine aminotransferase, and concentrations of albumin, total bilirubin, urea, and creatinine were determined in plasma using commercial kits and a Cobas Integra 400 autoanalyzer (Roche Diagnostics, Basel, Switzerland).
Histology
Livers and kidneys were rinsed in saline solution, minced, fixed for 6 h at room temperature with 4% paraformaldehyde in 0.01 M PBS, and then transferred into PBS. Small liver and kidney pieces always taken from the same lobe or area were embedded in epoxy resin (Electron Microscopy Sciences, Embed 812) and semithin (1 μm) sections were obtained using a Leica Ultra-cut R ultramicrotome. Morphological evaluation was performed in toluidine blue-stained (1% toluidine blue in 1% borax on a hot plate) sections using a Zeiss Axiophot microscope.
Confocal Microscopy
Human cytochrome P450 3A4 was detected using a rabbit polyclonal anti-CYP 3A4 antibody diluted at 1: 1500 (Abcam) and an anti-rabbit IgG secondary antibody conjugated to rhodamine (rabbit IgG CyTM3-linked, Amersham). Cell nuclei were stained using Hoechst 33342 (Sigma). Confocal images were obtained using a Leitz DMRB microscope equipped with a Leica TCSSP5 scanning laser confocal imaging system.
Flow Cytometry
Presence of human CD45+ cells was investigated in the liver. A piece of the liver weighing 200 mg was rinsed with PBS and digested for 2 h at 37°C with a solution containing 6.10 mM glucose, 0.14 M sodium chloride, 5.36 mM potassium chloride, 0.11 mM calcium chloride, 0.61 mM magnesium sulfate, 1.08 mM disodium hydrogen phosphate, 1.10 mM potassium hydrogen phosphate, 400 UI/ml collagenase IV, and 20 mg BSA/ml. The cell suspension was filtered through a 100-μm nylon mesh, and a 1-ml aliquot was incubated for 20 min with 20 μl of a monoclonal mouse anti-human CD45 antibody conjugated with fluorescein (Serotec FL, Oxford, UK).
Fluorescent cells were analyzed in a FACS Vantage flow cytometer (Becton Dickinson Immunocytometry System, San José, CA) equipped with a Coherent Enterprise laser (150 mW at 488 nm, 60 mW at UV), counting 10,000 cells/s.
Real-Time RT-PCR
Total RNA was obtained by using Trizol (Invitrogen, Barcelona, Spain) and quantified by fluorescent method with the Ribogreen RNA Quantitation Kit (Molecular Probes, Barcelona, Spain). Residual genomic DNA was removed by incubating RNA with RQ1 RNase-free DNase (Promega, Madison, WI). First-strand cDNA was synthesized using High-Capacity cDNA Archive Kit (Applied Biosystems, Weiterstadt, Germany). The negative control (no added transcriptase) was performed in parallel, and the reverse transcription reaction product was subsequently analyzed by PCR agarose gel electrophoresis to ensure no genomic DNA contamination in the RNA preparation. cDNA was amplified using TaqMan Universal PCR Master Mix (Applied Biosystems) on an ABI 7000 (Applied Biosystems). TaqMan primers and probes for human CD45 (Hs00236304_m1), CD34 (Hs00156373_m1), and GAPDH (Hs99999905_m1) genes were derived from the commercially available TaqMan® Gene Expression Assays (Applied Biosystems).
Statistical Analysis
All data are presented as mean values with standard error of the means (SEM). Variables were checked for normality and homogeneous variance by using the Shapiro Will and the Levene tests, respectively. All variables followed normality; therefore, parametric tests were used for the comparisons. Effects of treatments x time were assessed by two-way ANOVA, and comparisons of means by a posteriori Bonferroni tests. A value of p < 0.05 was considered significant. SPSS for Windows 15.0 (2005, SPSS Inc. Chicago, IL, USA) was used for the data analyses.
Results
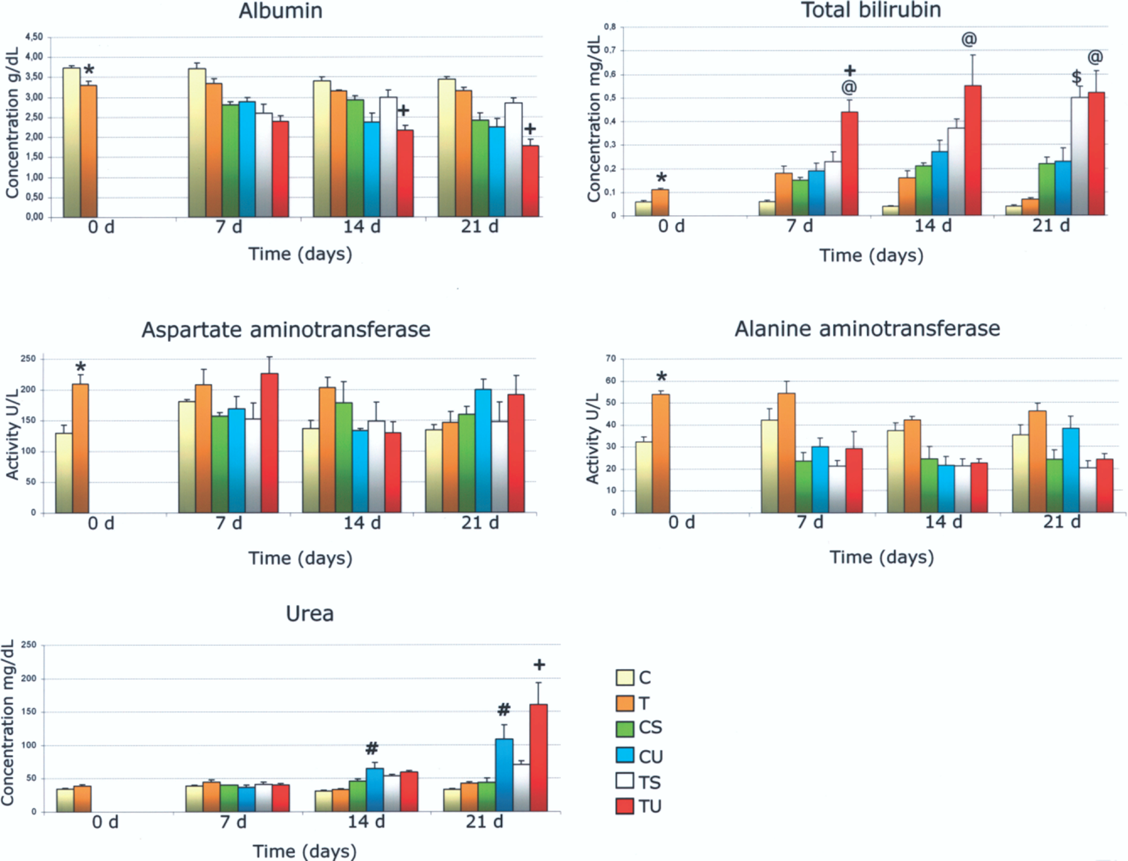
Mean body weight of control rats (group C) increased in a time-dependent fashion, reaching a value of 400.00 ± 33.52 g at week 16. In contrast, body weight of rats that drank TAA (group T) was 208.71 ± 36.08 g. Group T exhibited both hepatomegaly and splenomegaly (not shown). Administration to rats of 300 mg TAA/L drinking water for 4 months induced nodular cirrhosis. Compared with controls, the livers of cirrhotic rats were hardened and stiff, and their surfaces were nodular and yellowish in color (Fig. 2). Toluidine blue staining of semithin liver sections revealed that the animals with experimental cirrhosis had lost their hepatic architecture and displayed severe fibrotic septa, trabecular necrosis, and nodules of hepatocarcinoma (Fig. 3). Figure 4 shows findings for plasma markers of liver damage. Aspartate and alanine aminotransferase activities and total bilirubin concentration were significantly higher in cirrhotic rats versus controls, whereas albumin concentration was lower versus controls (Fig. 4).
Macroscopic appearance of control and cirrhotic rat livers. (Left) Liver of a rat that drank water throughout 4 months (group C). (Right) Liver of a rat that drank TAA throughout 4 months (group T). Two representative livers are shown. Light micrographs showing the liver parenchyma of control and cirrhotic rats. (A) Semithin section of the liver parenchyma of a rat that drank water throughout 4 months (group C). (B) Semithin section of the liver parenchyma of a rat that drank TAA throughout 4 months (group T). Representative images of each group are shown. Magnification: 250x. Liver fibrosis, hepatocyte necrosis, and cancerous nodules are appreciated in group T. Markers of hepatic and renal function. Plasma concentrations of albumin (g/dl), total bilirubin (mg/dl), and urea (mg/dl), as well as activities of aspartate aminotrasferase (U/L) and alanine amino trasferase (U/L). Results are mean ± SEM. *p < 0.05, group C vs. group T. +p < 0.05, group TU vs. group TS. @p < 0.05 group TU vs. group CU. $p < 0.05 group TS vs. group CS. #p < 0.05, group CU vs. group CS.

Cirrhotic and control animals underwent transplantation of 10 × 106 HUCBM cells via portal vein and were sacrificed at 7, 14, and 21 days posttransplantation. Our first goal was to detect human cells in the liver parenchyma of the rats, using flow cytometry, Western blot, and confocal microscopy. Flow cytometry analysis revealed the presence of human marker CD45 (measured as % fluorescence) in the livers of groups CU and TU, although this value was higher in the latter group (Fig. 5).
Detection of human CD45+ cells in rat livers by flow cytometry. The livers of recipient rats were digested with collagenase, incubated with an anti-human CD45 antibody conjugated with fluorescein, and subsequently analyzed in a flow cytometer. Results are expressed in percentages (mean ± SEM). For these experiments, all samples (n) described in Figure 1 were used. $p < 0.05, CU vs. CS. +p < 0.05, grouo TU vs. TS. @p < 0.05 grupo TU vs. CU.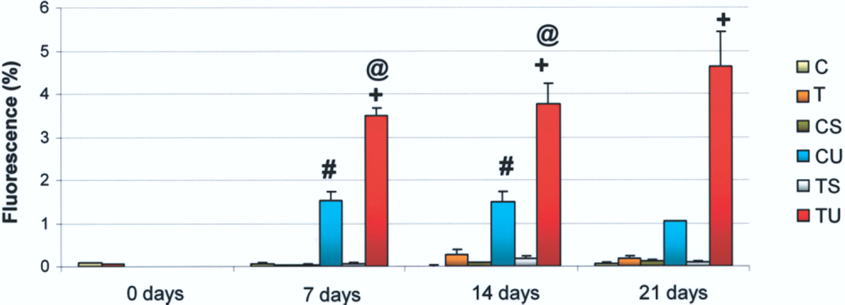
Detection of human cells in the liver parenchyma of the rats was confirmed by Western blot (Fig. 6) and confocal microscopy (Fig. 7), using an anti-human cytochrome P450 3A4 antibody.
Detection of human CYP 3A4 in livers of recipient rats. Representative Western blot showing detection of human CYP 3A4 in the livers of TAA-treated rats subjected to cell transplantation. These experiments were done in triplicate. A lysate from human hepatocytes was used as a positive control. Confocal micrograph showing human cells in the liver of rats with experimental liver cirrhosis at 21 days posttransplantation. A total projection of 25 confocal sections taken every 0.33 μm along the z-axis of two positive cells for human CYP 3A4 localized in the liver parenchyma (shown in red). Hepatocyte nuclei were stained with Hoechst 33342 (shown in blue).

Our next goal was to evaluate the efficacy of cell therapy in the experimental model of chronic liver damage. HUCBM transplantation had no effect on hepatic histology of cirrhotic animals (group TU) at 7, 14, or 21 days, and trabecular necrosis, fibrotic septa, deorganization of liver architecture, and cancerous nodules remained apparent, as in groups T and TS (Fig. 8).
Light micrographs showing the liver parenhyma of control and cirrhotic rats subjected to either HUCBM cell transplantation or sham-operation at 21 days posttransplantation. Semithin sections of the liver parenchyma of control rats subjected to either HUCBM cell transplantation (group CU, top left) or sham operated (group CS, top right), and of rats with TAA-induced liver cirrhosis subjected to HUCBM cell transplantation (group TU, bottom left) or sham operation (group TS, bottom right). Magnification: 500x (top panels) and 250x (bottom panels). Representative images of each group are shown. Whereas histology of groups CS and CU is completely normal, that of groups TS and TU shows the same lesions as in group T.
In contrast, analysis of plasma markers of liver damage revealed that HUCBM cell transplantation exerted a harmful effect in the experimental model of liver cirrhosis, as demonstrated by albumin and total bilirubin concentrations (Fig. 4). Albumin values were decreased in group TU at 14 and 21 days posttransplantation and were significantly lower versus cirrhotic animals that did not receive HUCBM cells (group TS). Further, cirrhotic rats that underwent HUCBM transplantation (TU) exhibited significantly higher values of total bilirubin at 7 days compared with sham-operated cirrhotic rats (TS).
Plasma markers of renal function were also determined (Fig. 4). No significant differences in creatinine were found among groups and times (not shown). Urea concentration was significantly higher in group TU than in group TS (Fig. 4). Interestingly, the same effect was appreciated in control animals, with group CU showing higher urea values versus group CS.
Based on the results obtained for urea, semithin sections of kidneys from all experimental groups were obtained and stained with toluidine blue (Fig. 9). Histological study revealed a mild lesion (vacuolization) of renal tubular epithelial cells in all four groups that received cyclosporin A plus anti-asialo GM1 antibody. However, cirrhotic animals subjected to HUCBM transplantation (group TU) exhibited alterations of tubules and glomeruli: the latter appeared ischemic and the former showed a more severe involvement, which included the aforementioned peripheral asymmetric vacuolization and disappearance of tubular lumen. Human CD45, CD34, and GAPDH mRNAs were not detected in the kidneys of groups TU or CU by real-time RT-PCR (not shown).
Light micrographs showing the renal parenchyma of control and cirrhotic rats subjected to either HUCBM cell transplantation or sham operation at 21 days posttransplantation. Representative images of each group are shown. (Top left panel) Renal semithin section of a rat that drank water throughout 4 months (group C). Magnification: 640x. (Top right panel) Renal semithin section of a rat that drank TAA throughout 4 months (group T). Magnification: 640x. Glomeruli and tubules are completely normal in groups C and T. Treatment with TAA did not induce renal damage. (Middle left panel) Renal semithin section of a control rat subjected to sham operation (group CS). Magnification: 500x. (Middle right panel) Renal semithin section of a TAA-treated rat subjected to sham operation (group TS). Magnification: 500x. Whereas glomeruli are intact, tubules show a slight lesion (peripheral asymmetric vacuolization) in groups CS and TS. (Bottom left panel) Renal semithin section of a control rat subjected to HUCBM cell transplantation (group CU). Magnification: 500x. (Bottom right panel) Renal semithin section of a TAA rat subjected to HUCBM cell transplantation (group TU). Magnification: 640x. Whereas in group CU peripheral asymmetric vacuolization is apparent, group TU shows a more severe tubular damage (disappearance of the lumen) and ischemic glomeruli.
Discussion
Oral administration of 300 mg TAA/L drinking water for 4 months induced nodular cirrhosis in Wistar rats. The livers of cirrhotic rats had steatosis, as suggested by their yellowish color, and they were hardened and stiff. Hepatic histological analysis revealed that the animals with experimental cirrhosis had lost their hepatic architecture, which displayed severe fibrosis, hepatocyte necrosis, and cancerous nodules. Liver damage was confirmed by analysis of plasma markers of hepatic function.
Cirrhotic and control animals underwent intraportal transplantation of HUCBM cells. Rejection of the human cells by the recipient rats was avoided with an immunosuppressive treatment based on cyclosporine A and an antibody that depletes natural killer lymphocytes, as previously validated in an acute model of liver disease in which animals received an injection of 2 × 106 HUCBM cells (1). In the present study we delivered a fivefold higher amount of cells to test whether the number of human cells that reached the liver could be increased. The number of transplanted HUCBM cells (10 × 106) is far below the safety limit described by others for hepatocyte transplantation (15). Presence of human cells in the liver parenchyma of the rats was confirmed by flow cytometry, Western blot, and confocal microscopy. Flow cytometry results indicated that the number of human cells engrafted in the rat livers was higher in the present model of chronic liver disease than in the acute model (1), with a maximum fluorescence of 2.91% in the latter versus 4.62% in the former.
After finding that human cells had engrafted in the rat livers, we then evaluated the efficacy of cell therapy in the experimental model of chronic liver damage. HUCBM transplantation had no effect on hepatic histology of either control or cirrhotic animals, and groups TU, TS, and T showed a similar histology, with the same alterations (necrosis, fibrosis, deorganization of liver architecture, and cancerous nodules). The liver histology of groups C, CS, and CU was completely normal.
However, plasma concentrations of two liver function markers, albumin and total bilirubin, suggested that HUCBM therapy exerted a damaging effect in animals with TAA-induced liver cirrhosis.
Urea was another plasma marker that pointed to a detrimental effect of HUCBM transplantation, because levels were higher in cirrhotic rats that had cell therapy than in cirrhotic sham-operated rats. Strikingly, the same effect was observed in the control animals. Kidneys from all experimental groups were histologically analyzed to confirm this evidence of a deteriorated renal function, showing mild kidney damage in the renal tubules of all four experimental groups that received immunosuppressive therapy. The most striking histological finding was the alteration of glomeruli and the more severe involvement of renal tubules in the cirrhotic animals that underwent HUCBM transplantation (TU).
The histological results of the experimentals groups that received immunosuppressive therapy (CS, TS, and CU) reflect the known nephrotoxic effects of cyclosporin A (10), even though we used the same dose (15 mg/kg/day) and during a similar time period as authors who did not find these effects (14). This immunosuppressant has two main nephrotoxic effects (10): i) an acute and reversible vasoconstriction of glomerular capillaries, which reduces blood flow and filtration rate and may eventually induce acute tubular necrosis; and ii) chronic and irreversible tubulointerstitial fibrosis, which produces chronic liver failure. We found the first (tubular) lesions but not the second (fibrosis) in our experimental model of chronic liver damage; therefore, we consider the nephrotoxicity observed to be incipient. However, the cirrhotic animals that received HUCBM therapy (TU) showed the most severe histological and functional renal alterations, consistent with an acute tubular necrosis leading to renal failure. The hepatic function was also impaired in this group.
Taken together, the histological and biochemical data indicate that cirrhotic rats receiving HUCBM cells developed a hepatorenal syndrome. This syndrome is a complication of patients with advanced liver disease and portal hypertension, and is characterized by renal vasoconstriction, intense reduction of glomerular filtration rate, and water and sodium retention (7). Kidney function is markedly impaired because of a severe reduction of renal blood flow, which is pathogenically related to the impairment in the systemic circulatory function. Hepatorenal syndrome is the cirrhosis complication with the worst prognosis and reduced life expectancy. Because group CU exhibited normal hepatic function and only mild cyclosporin-induced renal lesions, we believe that the increase in urea concentration observed in this group may be explained by differentiation of engrafted HUCBM cells into functional hepatocytes (2). In fact, amplification of human markers (CD45, CD34, and GAPDH) was not detected by real-time RT-PCR in the kidneys of either group TU or CU, ruling out the engraftment of human cells as the cause of the harmful effect observed in this organ.
Overall, our results suggest that HUCBM cell transplantation had a deleterious rather than a beneficial effect on TAA-induced liver cirrhosis.
Footnotes
Acknowledgments
This work was supported by grants PI050520 (co-financed by Fondo de Investigación Sanitaria and Fondo Europeo de Desarrollo Regional, FEDER) and P06-CTS1365 from Consejería de Innovación, Ciencia y Empresa, Junta de Andalucía (to L.F.); SAN196/LE04/07 from Consejería de Sanidad de la Junta de Castilla y León (to S.S.C); and UXXI2006/0002 from Federación de Cajas de Ahorro de Castilla y León (to J.G.G.). M. V. García-Mediavilla was supported by CIBERehd. CIBERehd is funded by the Instituto de Salud Carlos III, Spain.
