Abstract
Despite the high prevalence and devastating outcome, there remain a few options for treatment of ischemic stroke. Currently available treatments are limited by a short time window for treatment and marginal efficacy when used. We have tested a human umbilical cord blood-derived stem cell line that has been shown to result in a significant reduction in stroke infarct volume as well as improved functional recovery following stroke in the rat. In the present study we address the mechanism of action and compared the therapeutic efficacy of high- versus low-passage nonhematopoietic umbilical cord blood stem cells (nh-UCBSCs). Using the middle cerebral arterial occlusion (MCAo) model of stroke in Sprague–Dawley rats, we administered nh-UCBSC by intravenous (IV) injection 2 days following stroke induction. These human cells were injected into rats without any immune suppression, and no adverse reactions were detected. Both behavioral and histological analyses have shown that the administration of these cells reduces the infarct volume by 50% as well as improves the functional outcome of these rats following stroke for both high- and low-passaged nh-UCBSCs. Flow cytometry analysis of immune cells present in the brains of normal rats, rats with ischemic brain injury, and ischemic animals with nh-UCBSC treatment confirmed infiltration of macrophages and T cells consequent to ischemia and reduction to normal levels with nh-UCBSC treatment. Flow cytometry also revealed a restoration of normal levels of microglia in the brain following treatment. These data suggest that nh-UCBSCs may act by inhibiting immune cell migration into the brain from the periphery and possibly by inhibition of immune cell activation within the brain. nh-UCBSCs exhibit great potential for treatment of stroke, including the fact that they are associated with an increased therapeutic time window, no known ill-effects, and that they can be expanded to high numbers for, and stored for, treatment.
Introduction
Stroke is the fourth leading cause of death and the primary cause of long-term disability in the US. Each year, more than 795,000 people have a stroke, with the majority of events being first-time occurrences 1 . Among all types of stroke 87% are ischemic strokes, which are caused by either a transient or permanent reduction of blood flow and oxygen supply to the brain, resulting in death of neural tissue. This reduction is most commonly due to arterial blockage due to clotting or narrowing of the blood vessels that supply the brain.
Despite the high prevalence of stroke there are very few options for treatment. One of the primary therapies available to stroke patients is tissue plasminogen activator (tPA), which is a thrombolytic agent and is only effective within 4.5 h following stroke onset 2 . Due to the narrow therapeutic time window, only 3–5% of stroke patients are able to receive this treatment 1 . Currently, there are no therapies that are able to provide neuroprotection to the affected brain tissue following stroke.
Cell-based therapies are emerging as promising strategies that can be administered within the first few days (compared to hours) following stroke onset. A variety of stem cell types have been tested as potential therapeutic agents for ischemic stroke, each with unique advantages and limitations. Embryonic stem cells (ESCs) are considered the gold standard for pluripotency as they are able to differentiate into any cell type. However, the administration of undifferentiated ESCs leads to teratoma formation, and it is extremely difficult to create a homogenous ESC culture that is completely free of undifferentiated cells 3 . ESCs that have been transplanted into the brain following stroke have been shown to exhibit neuronal markers4,5, though studies have not found these transplanted cells to exhibit functional connections 6 .
Induced pluripotent stem cells (iPSCs) are another potential cell-based therapy for ischemic stroke. These cells are adult fibroblasts that are genetically reprogrammed into an embryonic stem cell-like state7,8. One of the primary benefits of these cells is the elimination of the need for immune suppression, as the fibroblasts can be taken from the patient needing the treatment. Unfortunately, studies have shown that despite this, epigenetic changes to these cells can still result in immune rejection 9 . An additional drawback of using reprogrammed adult fibroblasts is the possibility of teratoma formation following administration 10 .
Neural stem cells (NSCs) can be isolated from the subventricular zone and subgranular zone in both embryos and adults 11 . They are not pluripotent, but are able to differentiate into all types of central nervous system (CNS) cells. In a previous study, administration of NSCs following stroke did show improvement in stroke outcome, though the survival rate of the administered cells was extremely low, at around 1–3% of transplanted cells 12 .
Several types of bone marrow-derived stem cells are being studied as potential stroke therapies. Bone marrow-derived mesenchymal stromal cells (BM-MSCs) have been shown to migrate toward a site of injury in vitro 13 , though these cells show poor survival rates when administered in vivo 14 . Multipotent adult progenitor cells (MAPCs) are also bone marrow derived and have been shown to be able to differentiate into neurons 15 . Administration of these cells has been shown to improve recovery following stroke 16 , though they also show low engraftment rates and have the potential for teratoma formation 17 .
Cells derived from the mononuclear fraction of human umbilical cord blood (UCB-MNCs) are becoming prominently studied as a potential therapeutic agent for stroke because they overcome many of the limitations presented by the other cells types discussed above. Many studies have shown that administration of varying fractions of UCB provides an improvement in stroke outcome when administered within 72 h following a stroke18–20. A limitation of UCB is that the quantity available from each donor may require multiple donors for treating an individual patient. In a previous study we addressed this issue by identifying and characterizing CD34– stem cells within UCB that could be expanded in vitro 21 . This cell fraction also expresses the stem cell markers (sex-determining region Y)-box 2 (Sox-2), Rex-1, and octamer-binding transcription factor 4 (Oct-4) 21 . We refer to these cells as nonhematopoietic umbilical cord blood stem cells (nh-UCBSCs). We demonstrated that systemic administration of nh-UCBSCs 48 h after ischemic brain injury could significantly reduce infarct volume and ameliorate associated neurological deficits. In the current report we describe the mechanisms of action underlying the neuroprotective effects of nh-UCBSCs in treating ischemic brain injury.
Materials and Methods
Animal Subjects
Female Sprague–Dawley (SD) rats weighing 275–300 g were purchased from Charles-River Laboratory (Wilmington, MA, USA). All animals were housed and maintained according to the guidelines of the University of Minnesota Institutional Animal Care and Use Committee (IACUC). Research Animal Resources (RAR) staff provided routine veterinary care in addition to daily clean bedding and fresh food and water.
Surgical Induction of Stroke
Rats were anesthetized with a ketamine/xylazine cocktail (0.85 ml/kg; 75 mg/ml ketamine, Ketaject; Phoenix Pharmaceuticals, Burlingame, CA, USA), 10 mg/ml xylazine (Xyla-ject; Phoenix Pharmaceuticals) intramuscularly. The rat's head was stabilized in a Koph head holder in a supine position after the fur of the neck area was shaved and sterilized. Midline skin in the neck was incised and retracted laterally to expose subdermal structures. The right common carotid artery was exposed after the digastric and sternomastoid muscles were retracted. After carefully separating the right vagus nerve, the right common carotid artery (rCCA) was traced distally to its bifurcation, the superficial branch of which is the external carotid artery (ECA). While tracing distally along it, the endpoint was reached, which branches out into the superior thyroid artery, external maxillary artery, and terminal lingual artery. The right ECA was freed by cauterization and cut at this point, so that the ECA stem was long enough to be able to introduce the thread occluder for the temporary ligation. The right internal carotid artery (rICA) has an extracranial branch before it enters the cranium—the pterygopalatine artery—which was temporarily ligated for a complete ischemic lesion. The rECA was cut at its distal stem so that a silicon-coated surgical thread could be inserted into its lumen. The thread was inserted into the bifurcation, up into rICA, to occlude the middle cerebral artery (MCA). The occluder is kept in place for 1 h before removal. The open arteries are then cauterized, the wound closed, and the animal monitored until fully sternal and recovered.
Laser Doppler
In order to ensure that the MCA occlusion (MCAo) was having the same effect on all animals, we used MoorLab laser Doppler to monitor blood flow of the right hemisphere of the brain (Moor Instruments, Wilmington, DE, USA). Once baseline blood flow was established, the MCA was occluded as described above. Blood flow was maintained at 20–25% of baseline during the occlusion period in order for individual animals to be kept in the study.
Behavioral Analysis
The Neurological Severity Score (NSS) (modified from De Ryck 22 ) was used to evaluate neurological deficits following MCAo. The NSS test consists of eight subtests evaluating motor and sensorimotor function of the animals' limbs. Left and right side limbs were each scored as either 2 (normal function), 1 (impaired function), or 0 (unable to perform). The scores from these subtests were tallied for the right side and left side limbs. The animals' functional score was presented as a ratio of the total score of the left side limbs to the right side limbs.
Measurement of Infarct Size
Paraformaldehyde (PFA; Sigma-Aldrich, St. Louis, MO, USA)-perfused brains were paraffinized (Fisher Scientific, Waltham, MA, USA) and cut into 5-μm sections by microtome (Leica, Wetzlar, Germany) and mounted sequentially on gelatin-coated slides. A 5-μm slice was mounted every 25 μm throughout the length of the brain, slicing coronal sections from the rostral to caudal end, and a total of 30 slices were quantified in each brain. The slides were then stained with hematoxylin (Fisher Scientific) and eosin (Leica) to determine infarct size. Briefly, slides were deparaffinized through three changes of xylene (Sigma-Aldrich) (3 min each), hydrated through graded alcohols for 30 s each, and then run through a gentle flow of tap water for 1 min. Slides were stained with hematoxylin for 3 min, rinsed with water for 1 min, acid water for 15 s, water for 1 min, ammonium water for 15 s, tap water for 1 min, and eosin for 1 min. Slides were then dehydrated through graded alcohols to 100% alcohol and cleared in three changes of xylene (30 s each). Slides were coverslipped with Permount (Fisher Scientific) and allowed to air dry. Images of sequential sections from each brain were collected using a Nikon Eclipse E600 (Nikon, Tokyo, Japan) equipped with SPOT (Diagnostic Instruments Inc., Sterling Heights, MI, USA). ImageJ [National Institutes of Health (NIH), Bethesda, MD, USA] was used to obtain volume measurements.
Preparation and Administration of nh-UCBSCs
Human umbilical cord blood stem cells (UCBSCs) were obtained and grown as described in our previous publications 21 . The cell suspension injectate was made immediately before use with a final concentration of 2 × 106 cells/ml in sterile saline. Those animals that were chosen randomly to be in the treatment group received 1 × 106 cells in 0.5 ml of sterile saline by way of the saphenous vein. Injected animals were briefly anesthetized with isoflurane gas for the duration of the injection (<1 min) and monitored until fully awake and able to hold themselves in a sternal position.
Flow Cytometry
Brain mononuclear cells were isolated from whole rat brains using a density gradient as described by Pino et al. 23 . Briefly, whole brains were homogenized and suspended in a 30% Percoll solution (Sigma-Aldrich) with media. This mixture was gently pipetted over 1 ml of 70% Percoll to create the density gradient. After centrifugation at 500 × g for 30 min, the cell interface was removed. Once the mononuclear fraction was isolated and counted, a 96-well plate was seeded with 5 × 105 cells per well. The seeded cells were then blocked using a mouse anti-rat Fc block (BD Biosciences, San Jose, CA, USA) for 5 min at 4°C. Cell surface antibodies were then added and incubated for 15 min at 4°C. Wells were then spun down and washed three times with FACS buffer [phosphate-buffered saline (PBS) with 2% fetal bovine serum (FBS) (Fisher Scientific)]. Cells were fixed using the cytofix/cytoperm kit (BD Biosciences) according to manufacturer's instructions. Cells were washed again three times. Intracellular antibodies were added and incubated for 20 min at 4°C. Cells were washed three more times and resuspended in FACS buffer with AccuCount beads (Spherotech, Lake Forest, IL, USA) for analysis on a flow cytometer (BD FACSCanto). Absolute cell counts were derived using the counting beads. Cells were stained using CD45 and CD11b (BD Biosciences) to identify microglia and macrophages, CD3, CD4, and CD8a (eBiosciences, San Diego, CA, USA) to identify T cells and their subtypes. Tregs were identified by the expression of FoxP3 in CD3+CD4+ brain immunocytes. CD25 was used as an activation marker to identify activated CD3+ T lymphocytes. Mature B cells were identified using a triple color reagent to select CD3–CD161–CD45RA+ cells (ABD Serotec, Kidlington, UK). Isotype specific antibodies were used for all dyes and panel combinations to control for nonspecific antibody binding. Immunostained cell populations were analyzed using FlowJo software (TreeStar, Ashland, OR, USA).
Statistical Analysis
One-way analysis of variance (ANOVA) was used to determine differences between groups. Values of p < 0.05 were considered significant. Outliers in all data sets were determined using Grubb's test and excluded from analysis. The only outliers detected were within the flow cytometry results, and no more than one data point was excluded per group. The sample size reported does not include any excluded points. The Bonferroni–Holm post hoc test was used to determine significance between groups. All data are plotted as means with SEM. Statistics were calculated using GraphPad PRISM software (GraphPad Software, Inc., La Jolla, CA, USA).
Results
High Passage nh-UCBSC Administration Reduces Infarct Volume and Improves Functional Recovery
In our previous study investigating the efficacy of nh-UCBSCs in ischemic stroke 21 we used cells that were passaged to generate approximately eight population doublings. In the present study we also used nh-UCBSC at 25 population doublings to determine if the expanded cells maintained their therapeutic efficacy. As in the previous study human nh-UCBSCs were administered intravenously 48 h following MCAo surgery (Fig. 1). nh-UCBSCs that were used did not exhibit any karyotype abnormalities (Fig. 2). Animals were sacrificed 7 days following MCAo surgery for histological analysis of infarct size (Fig. 3A and B). Quantification of infarct size showed a significant decrease in infarct volume in both nh-UCBSC groups compared to the saline-treated group (Fig. 3C). Behavioral function was assessed 24 h and 7 days following MCAo surgery with the Neurological Severity Score (NSS). At 24 h following stroke, all treatment groups showed equivalent functional impairment. At 7 days following MCAo surgery, the saline-treated group showed no functional improvement. In contrast, the nh-UCBSC-treated animals showed significant functional improvement in the NSS test (Fig. 3D).

Experimental timeline. Temporary focal ischemia was induced by MCAo surgery on day 0. Behavioral testing using the Neurological Severity Score (NSS) was conducted on day 1. On day 2, animals were given either an IV injection of 1 million nh-UCBSCs or no treatment without immunosuppression. On day 7 following MCAo surgery, behavioral testing was conducted again, and animal subjects were sacrificed for histological assessment.

Karyotyping reveals no genetic transformation of nh-UCBSCs following extended time in culture. Low-passage cells were at passage 4, high-passage cells were at passage 14.

Stroke infarct volume measured by H&E staining. Staining was done 7 days following MCAo. Representative coronal maps of (A) saline-treated and (B) nh-UCBSC-treated MCAo animals. (C) Quantification of infarct volume in saline-treated, low-passage UCBSC-treated, and high-passage UCBSC-treated animals (n = 6 in each group). Maps adapted from Paxinos and Watson 61 . Low-passage and high-passage UCBSC groups have significantly smaller infarct volumes than saline-treated animals (p < 0.05). Average values (±SEM) from pooled data are presented. (D) Treatment with high- and low-passage UCBSC improves functional recovery 7 days following MCAo. Behavior assessed by the Neurological Severity Score (NSS) is represented as a ratio of the scores of the left and right limbs. Both low- and high-passage UCBSC-treated animals showed significant improvement in motor function compared to the saline group, which showed no improvement at 7 days (p < 0.05). Average values (±SEM) (n = 5) are presented.
To determine whether the expansion of the cells diminished the therapeutic efficacy of nh-UCBSCs, cells at eight population doublings and 25 population doublings were compared. There was no significant difference observed between the two groups in terms of reducing infarct size (Fig. 3C) and improving neurological functional recovery (Fig. 3D). All subsequent experiments were performed using only high-passage cells.
nh-UCBSC Treatment Reduces the Number of Macrophages in the Brain Following Stroke
To determine alterations in the number of macrophages in the brain following ischemia and nh-UCBSC treatment, flow cytometry analysis was conducted. Flow cytometric analysis of brain tissue shows an increase in the number of CD11b high/CD45 high macrophages in stroke animals (Fig. 4A–C). This population is reduced in the nh-UCBSC-treated animals, though the macrophage population is still significantly higher than that of the control group (Fig. 4E).

Number of macrophages and microglia in the stroke brain are significantly increased at 7 days following stroke. UCBSC treatment reduces the total number of macrophages and microglia in the brain at 7 days following stroke. Flow cytometry showing macrophage and microglia populations in (A) healthy control brain, (B) saline-treated stroke brain, and (C) nh-UCBSC-treated brain 7 days following stroke. Gates were established based on isotype-stained brain samples. (D) Quantification of the absolute numbers of microglia in each group shows an increase in microglia in stroke brains 7 days following stroke (*p < 0.05 for stroke vs. control) and a decrease in the nh-UCBSC-treated group versus the stroke group, which was not statistically significant. The microglia number after nh-UCBSC treatment was still elevated versus control (*p < 0.05 for stroke + UCBSC vs. control). (E) Quantification of macrophage cell numbers in each treatment group showed a significant increase in macrophages in the stroke brain 7 days following stroke, which does not decrease significantly with UCBSC treatment. Average values (±SEM) (n = 5) are presented.
nh-UCBSC Treatment Reduces the Number of Microglia in the Brain Following Stroke
Flow cytometry was also conducted to determine the number of microglia in the brain after ischemia and treatment with nh-UCBSCs. Flow cytometry analysis of brain tissue revealed an increase in the absolute numbers of CD11b high/CD45 intermediate microglia in the stroke brain (Fig. 4A–C). This population decreased in cell number in the nh-UCBSC-treated group, but these changes were not statistically significant and still remained higher than the control (Fig. 4D).
nh-UCBSC Treatment Normalizes the Number of T Cells in the Brain Following Stroke
Flow cytometric analysis was conducted to quantify T cell infiltration into the brain after ischemia and treatment with nh-UCBSCs (Fig. 5A–C). Analysis of CD3+ T cells showed a significant increase in the total numbers of CD3+ T cells in ischemic animals 7 days following MCAo surgery and a reduction to levels seen in nonischemic brains of the CD3+ T cell population in the nh-UCBSC-treated animals at 7 days (Fig. 5D).

Administration of nh-UCBSC normalizes the number of CD3+ T cells present in the brain 7 days following MCAo. Flow cytometry shows the CD3+ T-cell population in the brain of (A) healthy control rats, (B) stroke animals, and (C) nh-UCBSC-treated stroke animals. Gating was established based on isotype-stained brain samples. (D) Quantification of the absolute number of CD3+ T cells in control, stroke, and UCBSC-treated stroke brains shows a significant increase in the total number of CD3+ T cells in the brain 7 days following MCAo (*p < 0.05 for stroke vs. control, and stroke vs. stroke + UCBSC) and a normalization of the T-cell population in the UCBSC-treated animals. Average values (±SEM) (n = 5) are presented.
Flow cytometry analysis showed a significant increase in CD4+ T cells in the stroke brain 7 days following MCAo (Fig. 6A–C). The number of CD4+ T cells was reduced to nonischemic levels with nhUCBSC treatment (Fig. 6D). In contrast, no changes in the absolute number of CD8a+ cytotoxic T cells were observed among the treatment groups (Fig. 6E).

nh-UCBSC treatment normalizes the number of CD4+ helper T cells 7 days following stroke. Population of CD4 (defined as CD3+CD8a–CD4+) and CD8a (defined as CD3+CD4–CD8+) T cells in (A) healthy control brain, (B) MCAo brain, and (C) nh-UCBSC-treated MCAO brain 7 days following stroke. Quad gates were established based on isotype-stained brain samples. (D) Quantification of CD4+ T-cell populations show a significant increase in cell number in the MCAo group (*p < 0.05 for stroke vs. control, and stroke vs. stroke + UCSBS) and a normalization of the population with nh-UCBSC treatment. (E) Quantification of CD8a+ cytotoxic T cells showed no significant difference in cell number between treatment groups. All gates were determined using isotype-stained control tissue. Average values (±SEM) (n = 5) are presented.
CD4+ helper T cells were costained with the activation marker CD25 to determine their activation status during ischemic stroke and with nh-UCBSC treatment (Fig. 7A–C). Flow cytometry analysis showed that the absolute number of CD4+/CD25+-activated helper T cells significantly increased in the stroke brain at 7 days, and the number of these cells was reduced to nonischemic levels in the nh-UCBSC-treated group (Fig 7D).

Population of activated CD3+CD8–CD4+ CD25+ helper T cells are significantly increased in the brain 7 days following stroke and normalized in nh-UCBSC-treated stroke brains. Flow cytometry of CD4+CD25+ cell populations in (A) healthy control brain, (B) ischemic brain, and (C) nh-UCBSC-treated stroke brains 7 days following MCAo. Quad gates were established based on isotype-stained brain samples. (D) Quantification of CD4+CD25+ cell populations. Cell number is significantly increased in stroke brains 7 days following stroke (p < 0.05) and is normalized with nh-UCBSC treatment (p < 0.05 for stroke vs. stroke + UCBSCs, and p > 0.05 for control vs. stroke + UCBSCs). All gates were defined using isotype-stained control tissue. Average values (±SEM) (n = 5) are presented.
CD8a+ cytotoxic T cells were costained with the activation marker CD25 (Fig. 8A–C). Flow cytometry analysis showed that there was no change in the number of activated cytotoxic T cells in the brain across treatment groups (Fig. 8D).
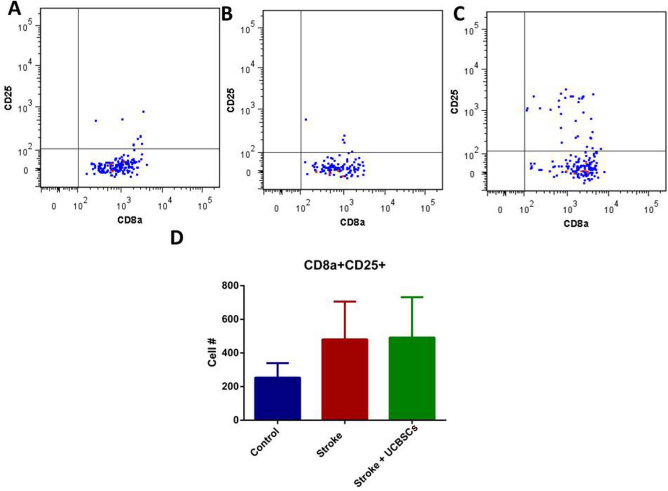
Populations of CD3+CD4–CD8+CD25+-activated cytotoxic T cells do not change significantly 7 days following ischemia. Flow cytometry of CD8a+CD25+ cell populations in (A) healthy control brains, (B) stroke brains, and (C) nh-UCBSC-treated brains 7 days following stroke. Quad gates were established based on identically stained brain samples with isotype-specific control antibodies. (D) Quantification of CD8+CD25+ cell population in the brain 7 days following stroke. Populations showed no significant difference between treatment groups. All gates were determined using isotype-stained control tissue. Average values (±SEM) (n = 5) are presented.
Flow cytometry was used to evaluate the presence of regulatory T cell populations by gating for CD3+, CD4+, and FoxP3+ cells (Fig. 9A–C). Quantification of this population shows an increase in the absolute number of regulatory T cells in the stroke brain 7 days following MCAo, and a reduction in the cell number of this population in the UCBSC-treated group. These changes in the Treg population were not statistically significant (Fig. 9D).

nh-UCBSC treatment normalizes CD3+CD4+FoxP3+ expression 7 days following stroke. Flow cytometry of (A) healthy control brain, (B) stroke brain, and (C) nh-UCBSC-treated brain 7 days following MCAo surgery. Quad gates were established based on isotype-stained brain samples. (D) Quantification of FoxP3+ expression in all treatment groups shows an increase in expression in the ischemic brain, though that change is not statistically significant. Average values (±SEM) (n = 5) are presented.
nh-UCBSC Treatment Normalized the Number of B Cells in the Brain Following Stroke
Flow cytometry analysis of mature B cells showed a decrease in the absolute number of B cells in the ischemic brain 7 days following MCAo, and B cell numbers increased toward the control levels of B cell numbers in the UCBSC-treated group (Fig. 10A–C). The changes in B cell numbers were not statistically significant (Fig. 10D).

Total number of mature B cells (defined as CD3–CD161–CD45RA+) present in the brain decreases 7 days following stroke and normalizes with nh-UCBSC treatment. Flow cytometry of mature B cells in (A) healthy control brain, (B) stroke brain, and (C) nh-UCBSC-treated stroke brain 7 days following MCAo surgery. Gating was established based on isotype-stained brain samples. (D) Quantification of total cell number of mature B cells in the brain in all treatment groups shows a decrease in cell number in the ischemic brain and a normalization with nh-UCBSC treatment, though these changes are not statistically significant. Average values (±SEM) (n = 5) are presented.
Discussion
Our results confirm that administration of nh-UCBSCs 48 h after stroke onset significantly reduces infarct size and improves functional recovery in a rat model of transient focal ischemia. In order to determine the practical potential of transitioning nh-UCBSC therapy into the clinic, we determined that nh-UCBSCs do not lose therapeutic efficacy when expanded in culture for an extended period of time. This would allow clinicians to expand cells from one donor to be able to treat multiple patients—giving nh-UCBSC therapy a distinct advantage over other proposed cord blood cell-based therapies, which require a new donor for each patient, and tissue matching.
The therapeutic benefit of nh-UCBSCs when delivered at 48 h after stroke is consistent with our previous study 21 using low-passage nh-UCBSCs. Several groups have studied different time periods of administration of the mononuclear fraction of umbilical cord blood stem cells following stroke in order to determine the optimal therapeutic time window. Newcomb et al. found functional improvement, and reduced infarct size was optimized when cells were administered 48 h following ischemic stroke 24 . They also found that the time of administration was more important to an improved stroke outcome than cell dose, as the optimal cell dose at any other time point tested did not show significant improvement. Boltze et al. confirmed these results and found that improvements in functional recovery and significant decreases in infarct size were observed if the mononuclear fraction of umbilical cord blood stem cells was administered within 72 h following stroke onset 18 . Together these results demonstrate the ability for cell therapy to greatly expand on the current time window of therapy with tPA at 4.5 h following a stroke event 2 .
Our results also demonstrated that the neuroprotective effects of nh-UCBSC administration were achieved without the utilization of any immunosuppressive agent. This observation is consistent with the work by Pan and colleagues 25 showing that immunosuppressive agents are not required for achieving therapeutic efficacy with human cord blood in cases of a rat model of ischemic stroke. They compared major histocompatibility complex (MHC) levels in animals that received injections of UCBSCs with and without cyclosporin A. They found that there was no significant difference in the expression of MHC I between groups, and there was no difference in cell survival between groups. Vendrame et al. 26 also assessed the efficacy of the mononuclear fraction of cord blood in ischemic brain injury and found neuroprotective effects without the use of immunosuppressive compounds. Thus, immune suppression is not required to prevent acute rejection of nh-UCBSCs in a rat model of ischemic stroke. nh-UCBSCs were also tested as a therapy for myocardial infarction by Chen et al. 27 . This group administered the cell therapy at 48 h following ischemia and did not use any immunosuppressive agents. They found that the administration of nh-UCBSCs showed an increase in overall heart function and preserved fiber orientation in the injured tissue area, similar to that of the sham-operated animals.
As cell-based therapies are gaining traction in the field of novel stroke treatments, multiple cell types have been tested. One of the most commonly investigated cell types is BM-MSCs. These cells have been shown to migrate to the site of injury post-injection, though they often have very poor rates of cell survival. Despite this, multiple studies have observed improved outcomes for stroke following BM-MSC administration14,28. Multipotent adult progenitor cells (MAPCs) have also demonstrated an ability to improve stroke outcome following administration, though no evidence of engraftment into the injured tissue has been found16,17. Several groups have studied UCBSCs, and all have found these cells to improve stroke outcome, but as with previously discussed cell types, little to no evidence of engraftment of these cells into the injured brain tissue has been found19–21.
Despite the repeated success of cell-based therapies in preclinical trials the mechanism of action of these cells is widely debated. Initially, it was thought that by introducing stem cells into the postischemic brain, these new cells would be able to engraft into the damaged area and replace lost tissue, thus recovering function in the damaged areas of the brain. While some studies have shown evidence of these exogenous cells in the postischemic brain after treatment, the evidence that these cells engraft to become functional cells is limited. What is more commonly found is that systemically administered cells show a benefit to stroke outcome, but are generally not found—or only found in small numbers—in the brain after administration 29 . This continued finding and the short period of time in which benefit is achieved have suggested that the mechanism of action is not likely due to engraftment and cell replacement, but due to some other mechanism. More recently, evidence has shown that a possible explanation for the mechanism of action of cell-based therapies is due to these cells exhibiting an influence on the postischemic environment via the immune response and the inflammatory cascade 30 .
Our group has conducted analysis of mRNA in animals with ischemic stroke, which has demonstrated a robust immune response in the ischemic brain associated with the infiltration of macrophages and T cells, as well as the proliferation of microglia 31 . This observation is consistent with our current understanding of the role the immune system plays in the poststroke brain. Ischemic stroke causes a disruption of the blood–brain barrier (BBB), thus allowing peripheral immune cells to infiltrate into the brain and influence the local immune response. These results are congruent with studies in both animals and humans that have found similar increases in these cells in the circulating blood as well as in the brain32,33.
T-cell infiltration into the brain has been shown at 24–48 h after ischemia 34 . This involvement of the perivascular space activates resident macrophages and microglia, which begin to clear dead cells, thus increasing inflammation and stimulating the influx of more leukocytes. The ischemic cell death that occurs in the brain following stroke is another source of increased inflammatory signaling. Studies have shown that T cells, but not B cells, are involved in the evolution of the brain infarction following stroke34,35. These observations combined with studies in which T-cell-deficient mice are protected from stroke damage suggest that infiltration of peripheral T cells plays a crucial role in stroke pathogenesis 36 . These infiltrating T cells are a major source of interferon γ (IFNγ), which causes neurotoxicity and eventual cell death37,38. The results of our study show the aforementioned influx of T cells in the ischemic brain. This influx has been reported to occur at approximately the same time period at which we administer nh-UCBSC treatment, following which we do not see the characteristic increase of T cells present in the ischemic brain. This suggests that a mechanism by which the nh-UCBSC treatment works involves the inhibition of the influx of T cells—at 48 h following stroke—though whether this effect is direct or indirect has yet to be determined. If these cells indeed ameliorate stroke deficit through blunting of immune cell influx into the brain following stroke, it would also prevent the propagation of an inflammatory microenvironment surrounding the ischemic area.
Monocytes from bone marrow, which can differentiate into either macrophages or dendritic cells, have been shown to migrate into infarct areas in the brain following ischemic stroke 39 . These cells are found in the border of the infarct area within 24 h following a stroke, and mature phagocytes are found directly within the infarct core 40 . Studies have shown that the phagocytes found within the infarct core are resident microglia that have transformed into phagocytes41,42. Infiltrating peripheral macrophages are most abundant in the brain at 3–7 days following focal ischemia41,43. These results suggest that resident microglial activation precedes peripheral monocyte and macrophage infiltration and that the peripheral macrophages are likely to contribute to the delayed phase of inflammation and brain injury that occurs at 3–7 days following focal ischemia. The results of our studies corroborate these findings, showing an influx of macrophages in the brain as shown by flow cytometry, as well as an increase in the expression of macrophagerelated mRNA transcripts in our stroke animals at 7 days following a stroke 31 . The timing of our nh-UCBSC treatment at 48 h following a stroke coincides with the time period during which peripheral monocytes are infiltrating into the ischemic brain. As our results show a significant decrease in the number of macrophages in the brain at 7 days following stroke in our nh-UCBSC-treated animals, this suggests that the treatment has some effect on the infiltration of these peripheral cells, thus blunting the added inflammation and injury that typically results from peripheral monocyte/macrophage infiltration.
One of the earliest cellular responses to ischemic stroke is the activation of resident microglia, which occurs within the first hour following stroke onset. There are two subtypes of activated tissue macrophages: the classically activated M1 and the alternatively activated M2 phenotype. Ischemic neurons prime the polarization of microglia toward the M1 subtype, which acts as a proinflammatory mediator and enhances neural injury. In contrast, the M2 phenotype has been shown to provide neural protection via the promotion of an anti-inflammatory microenvironment 44 . Our results show the characteristic increase in activated microglia in the ischemic brain 7 days following stroke. The population of microglial cells decreases in the brains treated with nh-UCBSCs, although this decrease is not significant. This decrease suggests that the nh-UCBSC treatment either directly or indirectly ameliorated the activation of microglia in the postischemic brain.
Our body of data suggests that the changes we observe in immune cell populations following nh-UCBSC treatment poststroke are related to the inhibition of peripheral immune cell recruitment into the ischemic brain. This could occur via the inhibition of monocyte chemoattractant protein-1 (MCP-1) and C-C chemokine receptor type 2 (CCR2), both of which are involved in the migration of peripheral inflammatory cells following stroke. Studies of mice that are deficient in both MCP-1 and CCR2 have shown a significant reduction in infiltrating macrophages following stroke 45 . Another possibility is that nh-UCBSCs are able to blunt the polarization of microglia and macrophages to their proinflammatory M1 phenotype. BM-MSCs have been shown to secrete low levels of immunomodulatory molecules such as CCL2 and interleukin-6 (IL-6), both of which promote M2 macrophages 46 .
Our results also showed a decrease in the number of mature B cells in the brain following stroke and that the number of mature B cells seemed to be restored to control levels following nh-UCBSC treatment. While the role of B cells in the development of stroke is still widely debated, regulatory B cells have been recently reported to have benefits in the ischemic brain 24–48 h following stroke 47 . In addition, it has been shown that the absence of B cells leads to an increased number of activated T cells, microglia, and monocytes in the brain. The IV administration of B cells prior to stroke resulted in a decrease in infarct size as well as a decrease in the number of activated immune cells in the brain 48 . A possible mechanism for the protective effect of B cells is the production of the anti-inflammatory cytokine IL-10 in the stroke brain 49 . These results, combined with our findings, suggest that at least some fraction of B cells play a role in protecting the ischemic brain, possibly by combatting the inflammatory cascade with the release of anti-inflammatory signals.
Another potential molecular mechanism by which nh-UCBSCs may have achieved the immunosuppressive effects observed here is via expression of indoleamine 2,3-dioxygenase (IDO). Several studies have shown that MSCs derived from human tissues express IDO when stimulated by proinflammatory cytokines such as tumor necrosis factor-α (TNF-α), IL-1β, and IFN-γ 50,51. In addition, Li et al. 52 have shown that knocking out IDO results in an increased immune response, suggesting a critical role for IDO in immune cell response in inflammatory environments. These cytokines are abundant in a poststroke environment, suggesting that IDO-mediated immunosuppression may be a likely cause for the reduced numbers of immune cells observed in this study.
The body of our work suggests that the administration of nh-UCBSCs following stroke has a profound effect on the immune response to ischemia, reflected largely in the alteration of immune cell phenotypes present in the brain following stroke. One of the primary mediators of the release and activation of immune cells following brain injury is the spleen. Seifert and colleagues 53 have shown that NK cells and monocytes from the spleen traffic to the brain 48 h following injury and that T cells migrate to the brain by 96 h following injury. Offner et al. 54 reported similar findings, noting an increase in macrophages and CD4+FoxP3+ T cells in the blood stream at 96 h following stroke, as well as a decrease in B cells, suggesting the migration of immune cells from the spleen to the site of injury following stroke. In addition, multiple studies have reported a reduction in spleen size in the acute phase of ischemic stroke, corresponding to the migration of immune cells to the brain, as shown by a decrease in CD8+ T cells in the spleen 26 . When these animals were treated with human UCB-MNCs, the spleen showed a decrease in expression of the proinflammatory cytokine TNF-α, and an increase in the expression of the anti-inflammatory cytokine IL-10, suggesting a switch from a Th1 to Th2 response in the spleen. A human study also showed a decrease in spleen size in the acute phase following stroke, followed in some cases by spleen expansion. It was shown in patients that those who showed splenic expansion following stroke also had a corresponding good clinical recovery 55 .
To further support the importance of the role of the spleen in the immune response to stroke, several studies have shown that animals that received a splenectomy prior to stroke showed a decreased immune response and improved stroke outcome56–59. In addition, studies have shown that treatment with the mononuclear fraction of human umbilical cord blood cells (hUCBCs) following stroke reverses the stroke-related changes in the spleen, such as a decrease in T-cell number in the spleen, a decrease in monocytes in the blood stream, and a decrease in the macrophages and microglia in the brain 60 . These findings suggest that the inhibition of the immune response propagated by the spleen following ischemic injury could be crucial to ameliorating stroke-related brain injury. It is likely that the administration of nh-UCBSCs at 48 h following stroke plays a role in the inhibition of the splenic response, resulting in improved stroke outcomes.
In conclusion, our results have further demonstrated the potential for nh-UCBSCs to provide effective therapy for stroke when administered 48 h following the onset of ischemia. Our analyses of immune cell phenotypes in the post-stroke brain have shown that nh-UCBSC treatment ameliorates the upregulation of immune cell presence in the brain. These results suggest that the influx of immune cells at 48 h following a stroke plays a major role in the development of the infarction and that blunting of these cell populations in the brain following stroke effectively ameliorates stroke-induced deficits.
Footnotes
Acknowledgments
These studies were funded in part by NIH Grants T32 DA007097, T32 AG029796, R41 NS056626, Suzanne M. Schwarz, and Walter and Ginger Bailey. P.R.S. and W.C.L. are founder and consultant, respectively, of Saneron CCEL Therapeutics, Inc. and are inventors on patents related to cord blood. P.R.S. is the Co-editor-in-chief of Cell Transplantation. Neither P.R.S. nor any member of the editorial office or editorial board affiliated with the authors' institutions was involved with the review process and/or decision making on this manuscript. The remaining authors declare no conflicts of interest.
