Abstract
Blood–brain barrier (BBB) permeabilizers, such as mannitol, can facilitate peripherally delivered stem cells to exert therapeutic benefits on the stroke brain. Although this BBB permeation-aided stem cell therapy has been demonstrated in the acute stage of stroke, such BBB permeation in the chronic stage of the disease remains to be examined. Adult Sprague–Dawley rats initially received sham surgery or experimental stroke via the 1-h middle cerebral artery occlusion (MCAo) model. At 1 month after the MCAo surgery, stroke animals were randomly assigned to receive human umbilical cord stem cells only (2 million viable cells), mannitol only (1.1 mol/L mannitol at 4°C), combined human umbilical cord stem cells (200,000 viable cells) and mannitol (1.1 mol/L mannitol at 4°C), and vehicle (phosphate-buffered saline) only. Stroke animals that received human umbilical cord blood cells alone or combined human umbilical cord stem cells and mannitol exhibited significantly improved motor performance and significantly better brain cell survival in the peri-infarct area compared to stroke animals that received vehicle or mannitol alone, with mannitol treatment reducing the stem cell dose necessary to afford functional outcomes. Enhanced neurogenesis in the subventricular zone accompanied the combined treatment of human umbilical cord stem cells and mannitol. We showed that BBB permeation facilitates the therapeutic effects of a low dose of peripherally transplanted stem cells to effectively cause functional improvement and increase neurogenesis in chronic stroke.
Introduction
Stroke remains a significant unmet need, with the current anti-blood clot treatment of tissue plasminogen activator (tPA) relegated to a window of only 4.5 h after ischemia onset, thereby limiting benefits to 3–8.5% of the stroke population1,2. Stem cell therapy has emerged as an experimental treatment for stroke, which has been shown to be effective in the laboratory when initiated acutely (within hours to a few days)3–5 and even when intervention is performed in the chronic stage of the disease6–8. Cell replacement, neurotrophic factor secretion, and biobridge formation have been proposed as mechanisms of action underlying the functional benefits of stem cell therapy8–13. The intravenous route of cell delivery has been the preferred approach for acute stroke characterized by an abundance of chemokines/cytokines conducive to stem cell homing14–16, whereas intracerebral transplantation has been generally indicated for chronic stroke when migratory signals have largely subsided in the brain6–8. Therapeutic cell doses are much higher (at least 10-fold) with the peripheral route of cell administration as opposed to direct brain transplantation, owing in part to the presence of the blood–brain barrier (BBB)12,17–20. To this end, BBB permeabilizers, such as mannitol, have been demonstrated to facilitate the functional benefits of a low dose of peripherally administered stem cells in stroke, both adult and neonatal, and in adult intracerebral hemorrhage and traumatic brain injury (TBI) models12,17–20. However, this BBB permeation-mediated stem cell therapy has, to date, only been investigated in the acute stage of the disease, thereby warranting investigations to assess whether BBB manipulation in the chronic stage of the disease similarly facilitates stem cell therapeutics in the stroke brain.
Materials and Methods
Animals
We examined the histological and neurobehavioral effects of chronic intravenous delivery of human umbilical cord blood cells (hUCBCs) (Saneron CCEL Therapeutics, Inc. Tampa, FL, USA) into rats after transient middle cerebral artery occlusion (MCAo). Adult Sprague–Dawley (SD) male rats (Harlan, Indianapolis, IN, USA) weighing 250–300 g served as subjects in this study. The National Institutes of Health (NIH) and Institutional Animal Care and Use Committee (IACUC) guidelines for use of animals in research were followed under an approved USF IACUC protocol. Animals were subjected to right MCAo for 1 h, then randomly assigned to one of the following treatments at 1 month after the MCAo surgery: hUCBCs only (2 million viable cells), mannitol (Sigma-Aldrich, St. Louis, MO, USA) only (1.1 mol/L mannitol at 4°C), combined hUCBCs (200,000 viable cells) and mannitol (1.1 mol/L mannitol at 4°C), or vehicle [phosphate-buffered saline (PBS)] (Sigma-Aldrich) only (n = 10 per treatment condition). Behavioral tests were conducted at 3 days after transplantation, and thereafter animals were euthanatized for histological analysis of hUCBC survival.
Stroke Surgery
Stroke surgery was performed using the middle cerebral artery occlusion (MCAo) technique as described in our previous studies4,17. Animals were anesthetized with a mixture of 1% to 2% isoflurane (Henry Schein Animal Health, Dublin, OH, USA) in nitric oxide/oxygen (69%:30%) (Airgas USA LLC, Tampa, FL, USA) via a face mask, and body temperature was maintained at 37 ± 0.3°C during the surgical procedures. A midline skin incision was made in the neck with subsequent exploration of the left common carotid artery, the external carotid artery, and internal carotid artery. Thereafter, a 4-0 monofilament nylon suture (27.0–28.0 mm; Covidien, Dublin, Ireland) was advanced from the common carotid artery bifurcation until it blocked the origin of the MCA. Animals were allowed to recover from anesthesia during MCAo. Hours after MCAo, animals were reanesthetized and reperfused by withdrawal of the nylon thread. We previously standardized the MCAo model, with stroke animals showing ≥80% reduction in regional cerebral blood flow during the occlusion period as determined by laser Doppler (Perimed, Ardmore, PA, USA). We also found no significant differences in physiological parameters, including PaO2, PaCO2, and plasma pH measurements, in our stroke animals, indicating homogenous stroke insults. Rats that reached the 80% cerebral blood flow reduction during occlusion were used for future studies. Sham animals were also included in this study, which involved anesthetizing rats and making an incision in the neck area, then exposing and isolating the common carotid and internal carotid arteries, ligating the external carotid artery, but without inserting the filament. Thereafter, incisions were closed, and animals were allowed to recover from anesthesia. All animals were euthanatized at 3 days posttransplantation for subsequent imaging analysis.
HUCBC Transplantation
At 1 month after the MCAo surgery, anesthetized (mixture of 1–2% isoflurane in 69%:30% nitric oxide/oxygen ratio) animals received an intravenous (jugular vein) injection of hUCBCs (200,000 cells from Saneron CCEL Therapeutics, Inc. in 10 μl PBS) or vehicle (PBS, same volume) over 10 min. Immediately thereafter, using the same intravenous line, animals received either 1.1 mol/L mannitol (maintained at 4°C) or vehicle (PBS, also maintained at 4°C) over 5 min.
Behavioral Testing
Motor deficits were characterized at day 3 after transplantation using the motor asymmetry elevated body swing test (EBST). We have used the EBST previously and noted that MCAo stroke animals display >75% biased swing activity after stroke surgery, which remains stable for at least 2 months after the insult 17 . EBST is a measure of asymmetrical motor behavior that does not require animal training or drug injection4,17. The rats were held, in the vertical axis, approximately 1 inch from the base of their tails and then elevated to an inch above the surface on which they had been resting. The frequency and direction of the swing behavior were recorded for over 20 tail elevations. A swing was counted when the head of the rat moved more than 10° from the vertical axis to either side. Rats with unilateral ischemic stroke exhibited significant biased swing activity to the right (ipsilateral side), while those with no unilateral ischemic stroke displayed equal number of swings to the right and left. The total number of swings made to the biased side was divided by 20 to get percentages of the swings. For the criterion of successful MCAo model completion, biased swing behavior was set at 75% or higher.
Human Nuclei (HuNu) Staining for Graft Survival Analysis
Every one of six 40-μm-thick coronal tissue sections of the brain, spanning the area of injury, were randomly selected for quantitative analysis. Free-floating sections were washed three times for 5 min in PBS. For HuNu staining, samples were blocked for 60 min at room temperature with 5% normal goat serum (Invitrogen, Carlsbad, CA, USA) in PBS containing 0.1% Tween 20 (PBST; Sigma-Aldrich). Sections were then incubated overnight at 4°C with mouse monoclonal anti-HuNu (1:50, MAB1281; EMD Millipore, Billerica, MA, USA) with 5% normal goat serum. The sections were washed five times for 10 min in PBST and then soaked in 5% normal goat serum in PBST containing corresponding secondary antibodies, goat anti-mouse IgG-Alexa 488 (1:500; Invitrogen) for 90 min. Finally, sections were washed 5× for 10 min in PBST and three times for 5 min in PBS, then processed for Hoechst 33258 (bisBenzimideH 33258 trihydrochloride; Sigma-Aldrich) for 30 min, washed in PBS, and coverslipped with Fluoromount (Sigma-Aldrich). Brain and spleen sections were examined using a confocal microscope (DP70; Olympus, Center Valley, PA, USA). Control studies included exclusion of primary antibody substituted with 5% normal goat serum in PBS. No immunoreactivity was observed in these controls.
MAP2 and Ki-67 Immunofluorescence for Peri-infarct and Neurogenesis Analysis
Every sixth 40-μm-thick coronal tissue section beginning at AP −1.70 and ending at +0.20 relative to bregma was isolated for morphological analysis of the striatum and SVZ. Free-floating sections were washed three times for 5 min in PBS. For Ki-67 and MAP2 staining, samples were blocked for 60 min at room temperature with 5% normal goat serum (Invitrogen) in PBST. Sections were then incubated overnight at 4°C with rabbit polyclonal anti-Ki-67 (1:100; Abcam, Cambridge, MA, USA) and mouse monoclonal anti-MAP2 (1:500; Abcam) with 5% normal goat serum. The sections were washed five times for 10 min in PBST and then soaked in 5% normal goat serum in PBST containing corresponding secondary antibodies, goat anti-rabbit IgG-Alexa 488 (Abcam) and goat anti-mouse IgG-Alexa 594 (1:500; Invitrogen), for 90 min. Finally, sections were washed five times for 10 min in PBST and three times for 5 min in PBS, then processed for Hoechst 33258 for 30 min, washed in PBS, and coverslipped with Fluoromount (Sigma-Aldrich). Control studies included exclusion of primary antibody substituted with 5% normal goat serum in PBS. No immunoreactivity was observed in these controls. For peri-infarct analysis, we demonstrated here that the primary damage produced by the MCAo model was to subcortical areas, including the striatum, as shown previously 17 . Six randomly selected coronal sections between the anterior edge and posterior edge of the infarcted striatum area were collected and processed for triple immunofluorescence labeling (Hoechst, MAP2, and HuNu). The peri-infarct cell survival rate was expressed as a percentage of the ipsilateral hemisphere compared with the contralateral hemisphere. For neurogenesis analysis, unbiased stereology was performed on brain sections immunostained with Ki-67. Sets of one of six sections were taken from the brain region corresponding to the SVZ. Of note, section thicknesses were confirmed as being between 20.0 and 1.0 μm after dehydration, and this did not statistically differ between groups. Ki-67-positive cells were examined using the Cavalieri estimator probe (MBF Bioscience, Williston, VT, USA) of the unbiased stereological cell technique revealing the volume of Ki-67-positive cells in the SVZ in both hemispheres (ipsilateral and contralateral). All samples were optimized to count at least 300 cells per animal with error coefficients <0.07. The Cavalieri estimator probe was executed using a point grid spaced equally both across and down directions. A grid space of 100 μm was used to cover the SVZ region representing our region of interest.
Statistical Analysis
Behavioral scores and infarct volumes were initially analyzed using ANOVA, followed by post hoc Bonferroni t-tests for pairwise comparisons between treatment groups. The level of significance was set at a value of p < 0.05.
Results
Here we show evidence that BBB permeation in the chronic stage of stroke facilitated the therapeutic benefits of peripherally delivered stem cells. Results revealed that vehicle- and mannitol-treated stroke rats displayed significant deficits in behavioral (motor abnormalities) (Fig. 1) and histological (brain damage) parameters compared to sham surgery animals (control) (p < 0.05) (Fig. 2). In contrast, while stroke animals that received hUCBCs alone or combined hUCBCs and mannitol also displayed significant behavioral and histological deficits compared to control animals, these transplanted stroke animals exhibited significantly improved motor performance and significantly better host brain cell survival in the peri-infarct area compared to stroke animals that received the vehicle or mannitol alone (p < 0.05) (Fig. 1). Moreover, we show that BBB permeation in chronic stroke lowers the effective stem cell dose necessary to improve functional outcomes. Of note, the significant improvement in motor performance and significant increase in endogenous cell survival in the peri-infarct area were comparable between stroke animals that received hUCBCs alone and those that received combined hUCBCs and mannitol (p > 0.05) (Fig. 2). Finally, we show that the combined hUCBCs and mannitol significantly increased the number of proliferating cells in the SVZ, as revealed by upregulation of Ki-67-positive dividing cells compared to that observed in the groups transplanted with the hUCBCs alone or the other treatment conditions (p < 0.05) (Fig. 3). Although there was an increased migration of transplanted hUCBCs from the periphery to the stroke brain in the combination treatment as opposed to the stand-alone stem cell treatment (p < 0.05), the overall graft survival was modest (less than 1%).
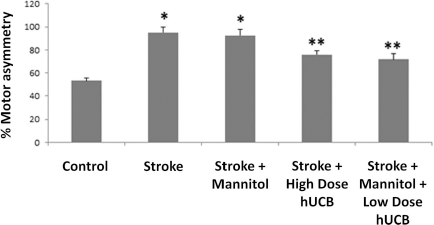
Combination of hUCBCs and mannitol ameliorates chronic stroke-induced behavioral deficits. At 3 days after transplantation (i.e., 1 month and 3 days after stroke) vehicle- and mannitol-treated stroke rats displayed significant deficits in behavior (motor abnormalities). In contrast, stroke animals that received hUCBCs alone or combined hUCBCs and mannitol exhibited significantly improved motor performance compared to stroke animals that received vehicle or mannitol alone. *p < 0.05 significantly impaired versus control; **p < 0.05 significantly impaired versus control, but also significantly behaviorally recovered compared to stroke (vehicle only) and stroke + mannitol.

Combination of hUCBCs and mannitol increases peri-infarct cell survival in chronic stroke. At 3 days after transplantation (i.e., 1 month and 3 days after stroke) vehicle- and mannitol-treated stroke rats displayed significant reductions in peri-infarct cell survival compared to sham surgery animals (control). In contrast, stroke animals that received hUCBCs alone or combined hUCBCs and mannitol showed significantly better brain cell survival in the peri-infarct area compared to stroke animals that received vehicle or mannitol alone. (A–D) Representative samples of stroke + mannitol-treated subventricular zone (SVZ) sections. (E–H) Representative samples of stroke + low-dose hUCBCs. (A, E) Hoechst; (B, F) MAP2; (C, G) HuNu; (D, H) merged. *p < 0.05 significantly reduced cell survival versus control; **p < 0.05 significantly reduced cell survival versus control, but also significantly increased cell survival compared to stroke (vehicle only) and stroke + mannitol. Scale bar: 5 μm.

Combination of hUCBCs and mannitol enhances neurogenesis in chronic stroke. Although high-dose hUCBCs increased neurogenesis as revealed by increased number of Ki-67 newborn cells in the stroke brain compared to control, stroke alone, and stroke + mannitol (*p < 0.05), BBB permeation in chronic stroke further enhanced neurogenesis as seen in those animals treated with combined hUCBCs and mannitol compared to all other treatment groups (**p < 0.05). (A–H) Representative samples of stroke + mannitol-treated SVZ. (I–P) Representative samples of stroke + low-dose hUCBCs. (A, E, I, M) Hoechst; (B, F, J, N) Ki-67; (C, G, K, O) HuNu; (D, H, L, P) merged. Scale bar: 5 μm.
Discussion
We, and others, have previously shown the potentiating effects of mannitol on facilitating the therapeutic effects of stem cells in animal models of acute injury, such as stroke and intracerebral hemorrhage12,17–20. Here we demonstrated that mannitol was equally effective in enhancing the benefits of stem cells for chronic stroke. Multifaceted benefits accompany this BBB permeation. Cell dose requirements to effectively abrogate stroke were reduced by 10-fold (from 2 million cells without mannitol to 200,000 cells with mannitol). From cell manufacturing logistics, such a low therapeutic cell dose is likely to reduce cell production costs and may allow improved use of autologous cells that until now have inherent technical challenges from initial isolation to eventual amplification when harvested from diseased and aged donors21–24. Additionally, a low effective cell dose increases the safety profile of cell therapy because large doses of transplanted stem cells have been implicated in microembolism22,25. Furthermore, in view of the recognized progressive nature of stroke 26 , repeated stem cell treatments may be necessary to arrest the secondary cell death. That the BBB can be safely and efficiently breached with mannitol offers a novel regimen for stroke patients to receive multiple stem cell treatments via peripheral administration, which may be considered as a minimally invasive procedure compared to direct brain transplants currently indicated for patients in the chronic stage of the disease. Mannitol has been routinely used in the clinic to reduce edema in stroke, but with inconsistent results27–29. Combining mannitol with stem cell transplants has been shown to increase brain neurotrophic factors12,17, as well as synaptogenesis, proliferation of immature neurons, and neuronal migration 20 . The observed enhancement of neurogenesis in the subventricular zone of the present chronic stroke animals further supports the notion of bystander effects as important mechanisms of cell therapy12,30,31, which appear to mediate functional benefits of the combined mannitol and hUCBCs, despite the modest number of peripherally transplanted stem cells recruited to the site of injury as seen here and in a previous report 20 . Altogether, the present findings provide evidence that BBB permeation facilitates the functional benefits of a low dose of peripherally transplanted stem cells in chronic stroke, as revealed by improved behavioral performance and reduced neurostructural damage in mannitol-treated and stem cell-transplanted animals, possibly via upregulation of endogenous neurogenesis. That therapeutic benefits were achieved with a reduced therapeutic cell dose advances a much safer cell therapy and caters to the envisioned repeated peripheral administrations of stem cells from the acute to the chronic phase of stroke.
Footnotes
Acknowledgments
C.V.B. is funded by NIH R01 NS071956, NIH R01 NS090962, NIH R21 NS089851, NIH R21 NS094087, DOD W81XWH-11-1-0634, and VA Merit Review I01 BX001407. C.V.B. and P.R.S. hold patents and patent application related to the use of mannitol and stem cell therapy. P.R.S. is a co-founder and C.V.B. is a consultant of Saneron CCEL Therapeutics, Inc. P.R.S. is a Co-Editor-in-Chief of Cell Transplantation. Neither P.R.S. nor any member of the editorial office or editorial board affiliated with the author's institution was involved with the review process and/or decision making on this manuscript.
