Abstract
Neurovascular niches serve as the hosts for adult neural stem cells in both the hippocampus and subventricular zone. The rostral migratory stream (RMS) vasculature has been found to be important for neuroblast migration, while its roles in hosting putative neural stem cells have not been investigated. Here we investigated the organization of RMS vasculature and its contribution to the production of new neurons. A single pulse of bromodeoxyuridine (BrdU) administration revealed locally formed new neurons within RMS were located adjacent to blood vessels. In addition, BrdU label-retaining cells that are putative neural stem cells were also found close to the vasculature. Sodium fluorescein perfusion assay demonstrated that the blood-brain barrier (BBB) organization was especially “leaky” in the neurogenic niches. Immunohistochemical visualization of some BBB component molecules indicated a thinner BBB in the RMS region, compared to that in the frontal cortex of adult rats. Finally, the expression of vascular endothelial growth factor was strong and specialized in the RMS region, implying that the region was active in cell proliferation and migration. Here we show that the RMS vasculature associated with surrounding astrocytes provides a highly organized neurovascular niche for adult neural stem cell proliferation, in addition to the function of neuroblast migration support. This result points to a new vasculature supporting neurogenic region in the brain.
Keywords
Introduction
Adult neurogenesis persists in mammalian brain. Previous studies showed that new neurons were consistently generated in the dentate gyrus of hippocampus and in the subventricular zone (SVZ). Neurogenesis in the hippocampus led to the formation of new granule cells, which might participate in learning and formation of new memories, while new neurons generated in the SVZ could migrate along the rostral migratory stream (RMS) to the olfactory bulb (OB), where they would finally differentiate into interneurons and replace dead cells in the OB (24,25,45). Recent studies further demonstrated that the RMS could also give rise to new cells in the OB in adult mice, which is independent from SVZ neurogenesis processes (3). Additionally, some isolated cells from the RMS region were found to have multipotent stem cell properties (14). However, the structural bases of this potential neurogenic area have not been fully investigated.
During embryonic brain development, the intermediate progenitor cells (IPCs) locate closely to nascent blood vessels and divide near the vessels (17,36). It is found that the radial glia-secreted vascular endothelial growth factor (VEGF) guides the angiogenesis and therefore the positioning of IPCs (17). The tight association between angiogenesis and neurogenesis was found in adult songbird brain as well as in the suprachiasmatic nucleus (22). In adult mammalian neurogenesis, the roles of vasculature has been acknowledged in both the hippocampus and SVZ (30,38), as well as the postinjury neurogenesis processes (33). Previous studies found that the RMS vasculature provided the scaffold for neuroblast migration by setting up a brain-derived neurotrophic factor (BDNF) gradient, guiding the migration of new neurons (35,40). The present study aimed to examine the contribution of RMS vasculature to local neurogenesis. The results showed that the RMS vasculature with its “leaky” blood-brain barrier (BBB) is specialized for adult neurogenesis, Putative neural stem cells existed in the RMS region and located adjacent to blood vessels. The secretion of VEGF by surrounding astrocytes might be one master molecule in shaping the area of vasculature niche. The tight interactions of neural stem cells and associated vasculature structures would suggest that RMS is an independent and specialized neurogenic niche during adult neurogenesis.
Materials and Methods
Animals
A total number of 60 adult (2 months old) male or female Sprague–Dawley rats were used for all experiments. All surgical procedures were approved by the Animal Research Ethic Committee for the Use of Live Animals in Teaching and Research at the University of Hong Kong. The animals were provided by the Laboratory Animal Unit of the University of Hong Kong (Hong Kong, China).
Bromodeoxyuridine (5-Bromo-2-Deoxyuridine, BrdU) Label-Retaining Cells Assay
Rats were given a single or multiple (six times in 12 h) doses of BrdU (Sigma, Hong Kong, China), 100 mg/kg in 0.9% saline solution, intraperitoneal injection, to label proliferating cells. The rats were sacrificed either 2 h post-injection (with single injection, n = 20) or 1 month post-injection (with six times injection, n = 20).
Tissue Processing
The rats were sacrificed with an overdose of sodium pentobarbital (160 mg/kg; Sigma) and then subjected to transcardiovascular perfusion through the left ventricle with 0.9% saline solution for 5 min. An incision was made on the right atrium to release the pressure. After perfusion, brains were immediately removed, oriented on a cold metal plate holder with embedding medium OCT (Tissue-Tek, Sakura, Alphen aan den Rijn, Netherlands), and frozen in liquid nitrogen. Then 40-μm frozen sections were cut on a Leica cryostat (Leica, Wetzlar, Germany) at the temperature of −16°C, dried at room temperature, and fixed in a mixture of acetone (Sigma) and ethanol (3:1) for 10 min at room temperature. The sections were washed in 0.01 M phosphate-buffered saline (PBS) (prepared in the laboratory; all reagents purchased from Sigma) prior to immunohistochemistry.
Sodium Fluorescein Perfusion Assay
To determine the leakiness of the blood vessels in the RMS and the OB, one group of animals (n = 10) was transcardiovascularly perfused for 5 min with 1 g/L sodium fluorescein (Sigma) diluted in 0.9% saline solution that was modified to increase signal-to-noise ratio from the previously described procedure (16).
Ink–Gelatin Perfusion Assay
Sacrificed rats were transcardiovascularly perfused with 37° Indian ink (Factfish, Hong Kong, China) with 3% gelatin (20 ml; Sigma) followed by 37° Indian ink with 5% gelatin (30 ml) for visualization of microblood vessels in the brain (n = 10). Successful perfusion was indicated by blackening of the eyes, snout, and ears. Then the main artery was closed with forceps, and the rat was immersed in icy water for 20 min. The brain was then dissected out and postfixed in 4% paraformaldehyde (PFA; Sigma) in 0.1 M PB overnight at 4°C. The tissues were cryoprotected with 10%, 20%, and 30% sucrose (Sigma) solution in 0.1 M PB. Finally, the tissue was cut on a Leica microtome at the thickness of 40 μm, and the frozen sections were mounted on precoated slides (Dako, Hong Kong, China).
Immunohistochemistry
Sections were processed for immunohistochemistry using different reagents for specific visualizations as described below. However, the pretreatment steps were similar and are as follows. Sections were washed in 0.01 M PBS for 5 min before immersion in a blocking solution containing 0.3% triton (Sigma), 1% bovine serum albumin (Sigma), and 10% goat serum (Sigma). Then they were incubated in a diluted primary antibody solution (see Table 1) overnight at 4°, and then washed in PBS before incubation with diluted secondary antibody solution (see Table 2) at room temperature for 2 h. Finally, the sections were washed in PBS and mounted with fluorescein mounting medium (Dako, Carpinteria, CA, USA).
Primary Antibodies Used
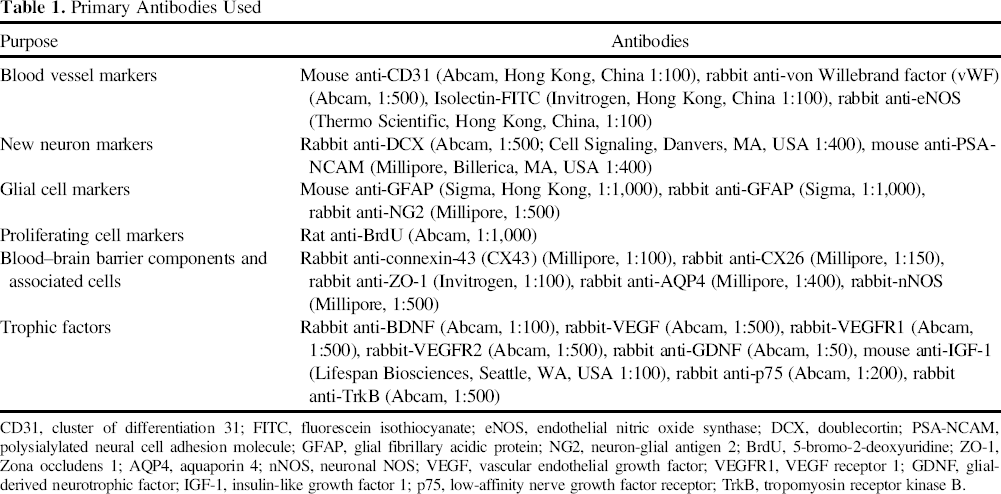
CD31, cluster of differentiation 31; FITC, fluorescein isothiocyanate; eNOS, endothelial nitric oxide synthase; DCX, doublecortin; PSA-NCAM, polysialylated neural cell adhesion molecule; GFAP, glial fibrillary acidic protein; NG2, neuron-glial antigen 2; BrdU, 5-bromo-2-deoxyuridine; ZO-1, Zona occludens 1; AQP4, aquaporin 4; nNOS, neuronal NOS; VEGF, vascular endothelial growth factor; VEGFR1, VEGF receptor 1; GDNF, glial-derived neurotrophic factor; IGF-1, insulin-like growth factor 1; p75, low-affinity nerve growth factor receptor; TrkB, tropomyosin receptor kinase B.
Secondary Antibodies Used

BrdU Immunohistochemistry
For BrdU immunohistochemistry, sections were first incubated in 4 N HCl (Sigma) for 20 min at room temperature and then washed with 0.2 M borate buffer (prepared in the laboratory, all reagents purchased from Sigma) (pH 8.5) for 20 min before another PBS wash for 5 min. Then the sections were processed for immunohistochemistry as described above.
For double/triple immunohistochemistry with BrdU sections, “relatively stable” antigens such as glial fibrillary acidic protein (GFAP), a cocktail of mixed primary antibody solution was applied to tissue directly after the DNA denaturation process. For some “fragile” antigens [antigen reactivity lost after HCl treatment, such as doublecortin (DCX) and polysialylated neural cell adhesion molecule (PSA-NCAM)], sections were first stained with the relative antibody to form a stable “antigen–primary antibody–secondary antibody” complex; then the sections were “fixed” in 4% PFA for 10 min before a PBS wash. Finally, the sections were processed for BrdU immunohistochemistry as previously described above (19).
Imaging
The slices were visualized with a Zeiss Axiophot epifluorescence microscope (Zeiss LSM 510). The confocal microscope was used to collect Z-stacks for 3D volumes for analysis. Both bright field and fluorescent images were collected sequentially.
Results
General Morphology of Blood Vessels in RMS
We examined ink-gelatin-perfused brain sections under light microscopy to investigate the general morphology of blood vessels in RMS (Fig. 1). We found that the RMS vessels contain a network of thinner vessels that are linearly aligned and directed to the OB, around 5 to 10 mm in diameter (Fig. 1C, D). In contrast to this well-arranged pattern, cortical vessels branched in multiple directions with no uniform pattern formed (Fig. 1B). This linear pattern was further confirmed by immunohistochemical staining of some other blood vessel markers, such as cluster of differentiation 31 (CD31), von Willebrand factor, isolectinfluorescein isothiocyanate, and wheat germ agglutinin–rhodamine (data not shown).
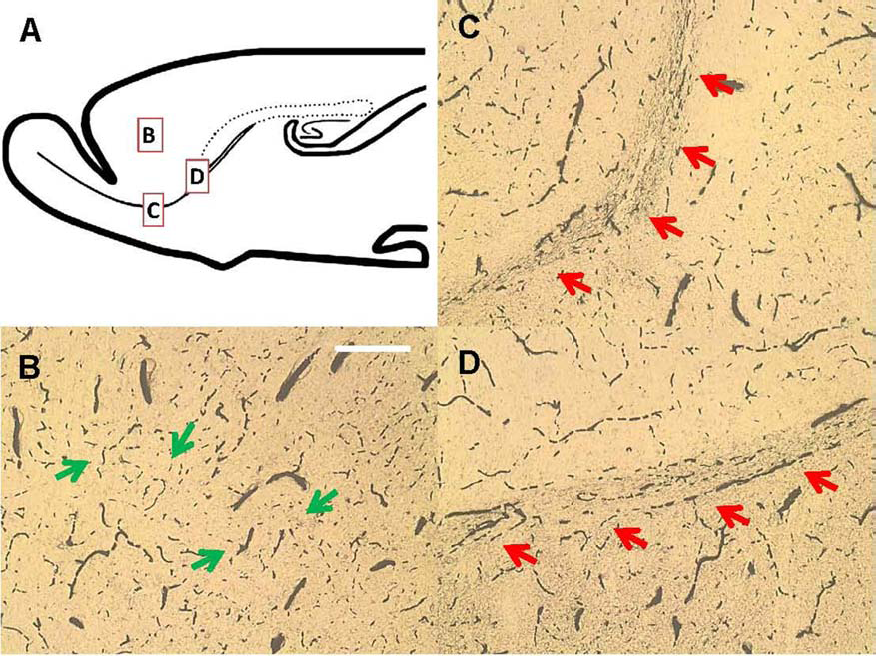
Brain vasculature revealed by ink–gelatin perfusion method. (A) Scheme of a rat brain, showing the cortex (B), and two RMS regions (C, D). RMS vasculature shows a scaffold-like pattern in an organized manner, while cortex vasculature was not organized (scale bar: 200 μm). The arrows indicate the pattern of the blood vessels of interest.
Neural Stem Cells in RMS Lie Adjacent to Blood Vessels
We analyzed the distribution pattern of the immediately proliferating cells within the RMS area by sacrificing animals shortly after a single pulse of BrdU injection. We found that most proliferating cells were adjacent to blood vessels (Fig. 2A–C). In order to find out whether the proliferating cells are migrating neuroblasts generated in the SVZ or local neural stem cells within the RMS, we performed a label-retaining cell (LRC) assay and sacrificed animals 1 month after multiple pulses of BrdU injection (38). The newly generated cells migrated away, and the BrdU-containing cells in situ are considered as putative neural stem cells. Indeed, we found BrdU-labeled cells in the RMS 1 month after BrdU injection (Fig. 2D–F), located nearby blood vessels, with GFAP expression, suggesting that these neural stem cells are actually astrocytes, which is consistent with previous reports in hippocampus and SVZ neurogenic niches (10).

RMS vasculature serves as the cell proliferation niche and migration scaffold. BrdU-labeling of immediate proliferating cells (A–C) and label-retaining cells (LRCs) (D, E). Green: CD31 staining; red: BrdU staining. (A) Within 2 h, immediate proliferating cells populate the vasculature in the RMS region (scale bar: 200 μm). (A) CD31 staining, (B) BrdU staining, and (C) the merged image. (D) One month after pulse labeling, LRCs are observed in the RMS vasculature, adjacent to blood vessels (BVs) (scale bar: 20 μm). (E) Confocal 3D view of (D) and the LRC (arrowhead in D) in relation to the BVs. Panels that are attached (upper and right) show depth of the Z-stack centered at that cell location. The yellow in the cell indicates that it is next to the BV. (F) The summary of distance relationships between the LRCs and BVs. Most LRCs are located very close to the BVs. (G, H) RMS vasculature provides the scaffold of immature neuron migration. Green: isolectin labeling of BVs; red: polysialylated neural cell adhesion molecule (PSA-NCAM) staining. (G) Immature neurons form chains that cross the vasculature scaffold (scale bar: 20 μm). (H) Confocal 3D view of an immature neuron (arrowhead in G) in relation to the BVs. Panels that are attached (upper and right) show depth of the Z-stack centered at that cell location. The new neurons are located in close relationship to BVs.
Migrating Neurons Form Chains Surrounding the Blood Vessel Scaffold
It was reported that newly formed neurons could form chains and migrate along the RMS in a glia-independent manner (41). We examined the spatial relationship between these chains of new neurons and RMS blood vessels. We found that in the RMS region, the chains of new neurons attached to, and migrated over, a blood vessel scaffold (Fig. 2G, H), showing a very close relationship between new neuron migration and blood supply, which is distinct from the fact that in the embryonic RMS the vasculature does not support cell migration (29).
RMS Blood Vessels Are “Leaky”
To understand the unique property of the RMS vasculature from other blood vessels, we tested the permeability of blood vessels in the RMS region by transcardiovascularly perfusing animals with sodium fluorescein solution (16) (Fig. 3). After the perfusion, tissues with leaky blood vessels would gain and retain fluorescence signal when compared to brain tissues with intact BBB. We found that the choroid plexus (Fig. 3C), or neurogenic regions such as the SVZ (Fig. 3B), demonstrated a strong signal in vessel-surrounding areas, indicating the leaky nature of the tissue. While the prefrontal cortex showed no such permeability (Fig. 3A), the RMS region contains mild levels of fluorescence mimicking blood vessel patterns (Fig. 3D), suggestive of enhanced vessel permeability.

RMS vasculature shows enhanced “leakiness” to small molecules, demonstrated by sodium fluorescein perfusion approach. The scheme shows the different brain regions that are examined. (A) Prefrontal cortex, (B) lateral ventricle, (C) third ventricle, (D) RMS (scale bar: 200 μm). RMS region exhibits a high level of fluorescence, similar to the ventricular regions, suggesting increased vasculature permeability compared to the prefrontal cortex.
Organization of BBB in RMS Vasculature
The BBB sets the level of blood vessel permeability. BBB is mainly composed of endothelial tight junctions, endfeet from astrocytes, as well as associated cells such as pericytes (1). We first examined the expression of tight junction marker Zona occludens 1 (ZO-1) on RMS blood vessels with immunohistochemistry (Fig. 4). The results showed that the ZO-1 staining pattern in the RMS was much less dense than that in the cortex (Fig. 4C, D). In the RMS, the reduction in signal density also appeared to form a well-organized pattern that differed from normal BBB.

RMS vasculature shows decreased BBB components, including the astrocyte endfeet and the tight junction over the BVs. Green: AQP4 staining; red: ZO-1 staining. (A and C) RMS; (B and D) frontal cortex. Both AQP4 and ZO-1 staining patterns are different in the RMS compared to the cortex, showing a scaffold-like pattern, and are thinner (scale bar: 50 μm).
In addition, the marker of astrocytic endfeet, Aquaporin 4 (AQP4), also exhibited a similar pattern to the ZO-1 expression in the RMS (Fig. 4A), which is distinct from the cortical regions as well (Fig. 4B). Pericytes visualized with NG2 showed little difference from cortex. However, the microglial cells in the RMS region are mainly perivascular microglial cells that attach tightly to the vasculature with soma or by one to several processes (unpublished data).
Gap junction proteins such as connexin-26 (CX26) and connexin-43 (CX43) have been found to be necessary for newly formed neurons to migrate along radial glial cells during development (11). In the present study with the adult brain, CX43 was found to have a rather specific localization in the RMS region compared to other areas of forebrain and was expressed in vessel-associated astrocytes with patchy localization on RMS vessels (Fig. 5A–E), suggesting that gap junctions could be formed between these astrocytes and blood vessels within the RMS. On the other hand, CX26 was also expressed in blood vessel-associated astrocytes. However, unlike CX43, the expression pattern of CX26 was not organized in either RMS or in the cortex (Fig. 5F–I). It is possible that these gap junctions are necessary for electrical coupling and the exchange of small molecules within the niche microenvironment (26).

Gap junction proteins are expressed in the RMS region. (A–E) Connexin-43 staining, which forms a patchy pattern on BVs. (A–D) Isolectin, connexin-43, PSA-NCAM, and merged image, respectively. (E) Magnified image of (D), showing the localization of connexin-43 in the RMS. (F–I) The connexin-26 protein is also expressed in the RMS region. (F–I) Isolectin, connexin-26, PSA-NCAM, and merged image respectively (scale bar: 20 μm).
VEGF Defines the Boundary of Neuroblasts Migration
VEGF guides the formation of new blood vessels and has been proven to regulate adult neurogenesis when administered into neurogenic regions of the brain (18). Interestingly, we found that VEGF is strongly and specifically expressed in the RMS region when compared to the surrounding brain regions (Fig. 6A). The existence of the molecule was detected around the RMS (Fig. 6B–G). The four-color colocalization image suggested that astrocytes were the source of VEGF within the RMS. We further found that VEGFR1 located primarily on blood vessels, while VEGFR2 mainly located on new neurons (stained by DCX, a new neuron marker) (Fig. 6H–K and L–N, respectively).

VEGF signaling is highly specific for RMS neurogenic region. (A) VEGF is rather selectively expressed in the RMS region, compared to surrounding regions (scale bar: 100 μm); the blue square in (A) indicates the region where (B) is taken. (B–G) The source of VEGF is astrocytes, with strong colocalization shown from confocal images (scale bar: 20 μm); the blue square in (B) indicates the region where (G) is taken. (B–F) VEGF, isolectin, DCX, GFAP, and merged pictures, respectively. (H–N) The VEGF receptors expression pattern in the RMS. VEGFR1 is mainly expressed on BVs (H–K), while VEGFR2 is mainly expressed on new neurons (L–N), suggesting different signaling pathways of VEGF in regulating BV growth and new neuron production/migration (scale bar: 20 μm). (H–K) Isolectin, VEGFR1, GFAP, and merged picture, respectively. (L–N) DCX, VEGFR2, and merged picture, respectively.
Trophic Factors From RMS Vasculature-Associated Astrocytes
We further investigated for the presence of other trophic factors that were known to regulate adult neurogenesis. We found that the RMS region contained numerous trophic factors such as BDNF, insulin-like growth factor 1 (IGF1), and glial-derived neurotrophic factor (GDNF). Colocalization immunohistochemistry identified that these factors localized on vasculature-associated astrocytes (Fig. 7A–D, E–H, and I–K, respectively).

RMS vasculature is associated with the expression of different trophic factors. RMS vasculature is associated with the expression of different trophic factors such as BDNF (A–D), IGF-1 (E–H), and GDNF (I–K). These trophic factors show colocalization with astrocytes. (A–D) Isolectin, BDNF, GFAP, and merged picture, respectively. (E–H) Isolectin, IGF1, GFAP, and merged picture, respectively. (I–K) GDNF, GFAP, and merged picture, respectively (scale bar: 20 μm).
Nitric Oxide Signaling Within RMS Vasculature
We found that endothelial nitric oxide synthase (eNOS) is present at high concentrations in the RMS vasculature (Fig. 8A–C), suggesting that endothelial-derived NO might diffuse locally and contribute to the neurogenesis regulation. In addition, neuronal NOS (nNOS)-positive neurons were found occasionally along the RMS vasculature. However, when compared to nearby tissues, the RMS region exhibited fewer nNOS fiber innervations (Fig. 8D, E).

eNOS and nNOS expression in the RMS vasculature. (A–C) Endothelial nitric oxide synthase (eNOS) is colocalized with the vasculature (scale bar: 20 μm); (A–C) eNOS, isolectin, and merged picture, respectively. (D, E) nNOS cell bodies are not found within the RMS (scale bar: 200 μm), while few axon terminals innervate the RMS region (scale bar: 50 μm).
Discussion
The RMS Vasculature Supports Adult Neurogenesis
The present study showed that RMS vasculature functions as a specialized neurogenic niche. RMS contains a number of proliferating cells along the blood vessels; in addition, BrdU LRCs that are putative stem cells were also found be adjacent to the vasculature. This is consistent with the neurogenic niche in the SVZ, which contains type C transient progenitor cells and type B slow-dividing stem cells (4,32). As is the case for SVZ neurogenesis, the RMS is able to generate both granule and periglomerular cells in the OB; however, the processes are separately regulated when compared with the neurogenesis in the SVZ. For example, odor enrichment led to increased cell proliferation in both the RMS and SVZ, while chemical lesion of the olfactory epithelium only upregulated RMS cell proliferation (3). Moreover, neural progenitor cells transplanted into the RMS region were able to survive and give birth to new neurons within the OB (4). Cell lineage tracing studies also revealed that RMS and different regions within the SVZ could generate distinct subtypes of interneurons in the OB (5). In the present study we showed that the RMS is an independent neurogenic region in rodents, though developmentally it belongs to a part of the ventricle extension.
During postnatal development, the closure of the olfactory ventricle separated the RMS from the SVZ; the specialized vasculature in the RMS might provide an alternative structure with enhanced molecule exchanges to perivascular CSF. Therefore, the neural stem cells are in the best place to sense the changes in the external environment and react accordingly. As a “promigratory and development center,” the RMS vasculature is unidirectional and has many intervasculature spaces for cells to reside or migrate. Recent reports also led to a similar conclusion for mice RMS (35,40,44). We believe that such features may serve as a common mechanism underlying long-distance neuronal migration in higher vertebrates, including humans.
Putative Neural Stem Cells in the RMS
In animals with single BrdU injection and sacrificed 2 h later, the labeled cells are locally proliferating cells rather than cells generated in the SVZ undergoing migration, suggesting that RMS could give birth to new cells. Additionally, 1 month after pulse labeling, the newly generated cells would migrate away, and the resident LRCs in the RMS are thought to be the “grandmother-like” stem cells or putative neural stem cells.
BrdU LRCs that are putative neural stem cells in the neurogenic niche were found to be astrocytes, which were also the case for both the hippocampus and SVZ neurogenic zones (10,25). Interestingly, these slowly dividing cells could keep their complex morphology and elongated processes (Fig. 2), suggesting that these cells would be able to respond to different stimuli, such as the circulating cytokines from the blood vessels. Recent evidences showed that astrocytes could keep the morphology and functional gap junctions during developmental proliferation (13); whether this is also the case for neurogenic LRCs still require further investigation.
Permeability of BBB Is Critical for the RMS Vascular Niche
One important feature of the RMS blood vessels is that they were more “leaky” than that of nonneurogenic regions such as the cortex. Similar vasculature has been observed in injured brain tissues, which is often associated with migrating neuroblasts attracted to the wound area (23). The underlying mechanisms could be comparable, including the enhanced BBB permeability to small molecules, oxygen, and trophic factors from both endothelial cell and vasculature-associated astrocytes. The perivascular microglial cells could be another important source of cytokine release upon brain insults as they are in the first place to sense the information transported within the vasculature. Previous studies have showed that injury could induce changes in BBB permeability, including altered AQP4 expression in the injured regions (28). In our olfactory tract transection model, we transected OB, which inhibited the new neuron migration to the bulb, and the new neurons thus would accumulate within RMS; we also noticed that the expanded AQP4 scaffold structure was colocalized to the increased area of new neuron migration chains (unpublished data).
VEGF regulates the growth and permeability of local blood vessels as well as being neurogenic (34). Therefore, the continuous expression of VEGF could also contribute to the “leaky” characteristic of the RMS vasculature.
RMS Vasculature as the Trophic Niche
VEGF was shown to directly stimulate cell proliferation and increase the survival of neural stem cells (18). It was also found to be important in the formation of injury-induced neurovascular niche as well as perivascular niche for brain tumor stem cells (21,37,39). Infusion of VEGF to normal brain regions induced ectopic neurogenesis and attracted RMS neuroblasts to migrate out from the original route (42). Therefore, the specific control of VEGF expression within the RMS region is important and critical for neurogenesis and cell migration. A recent study proved that VEGF secreted by RMS astrocytes controlled the formation and growth of blood vessels during development of the RMS vasculature (6), highlighting the contributions of surrounding glial cells to neurogenesis. Even though the RMS new neurons migrate in a glial-independent manner (41), the glial cells could play a role as regulators of or permissive cues for neuronal migration.
We also found that in RMS vasculature-associated astrocytes provided abundant sources of trophic factors, including BDNF, IGF1, and GDNF. All of these trophic factors have been proven to regulate neurogenesis, both in embryos and adulthood (15,43). The migrating new neurons also expressed some receptors of these trophic factors, including low-affinity nerve growth factor receptor p75 and trkB (unpublished data), which is consistent with previous findings (12,35,43). Further studies to identify the mRNA localization of these trophic factors would be interesting. In early development the overexpression of p75 is a clear indicator of an early phenotype of a developing cell. As the cells mature, there is a decrease in the p75 number and an increase in other neurotrophic receptors. The regulation of the cells has been shown to be controlled by the expression and the balance of p75 versus trkB.
Nitric Oxide Signaling and Neurogenesis in RMS Region
In the present study we found that eNOS rather than nNOS was located on the RMS vasculature, suggesting the potential involvement of the vasculature in regulating adult neurogenesis via NO signaling. One theory of why endothelial cells could support stem cell proliferation in vivo is that it could release NO, which was regulated by blood flow during times of increased shear stress, as has been shown in the development period (2). The “leaky” vasculature of RMS permits the diffusion of NO into nearby tissues easily and the short half-life of NO implied that stem cells have to be located adjacent to the vasculature. Possibly, the long and linearly arranged blood vessels permitted better perfusion, which increased the local shear stress on endothelial cells that would produce more NO. NO is a shared regulator for many types of stem cells and progenitor cells, including mesenchymal stem cells, embryonic stem cells, endothelial progenitor cells, muscle satellite cells, and neural stem cells (9). It has been found that NO participated in many processes of adult neurogenesis in both the hippocampus and SVZ region (7), but with opposite functions. For example, following brain ischemia, eNOS upregulated neurogenesis of SVZ (8); additionally, after injury, the inducible NOS (iNOS) activated neurogenesis in the dentate gyrus (46). While nNOS seemed to be an inhibitor for adult neurogenesis as the administration of one nNOS blocker, Nω-nitro-L-arginine methyl ester (L-NAME), showed enhanced neural stem cell proliferation in the SVZ and RMS region (27). Therefore, NO might function as the arbiter of neurogenesis, depending on the source of releasing cells. Last but not the least, the question of how the two isoforms of NOS regulate the survival/death of migrating neuroblasts is important as NO is also a regulator of cell apoptosis (20).
NO was found to contribute to the angiogenic properties of VEGF as well (31). As a short-life molecule, NO presented the possibility for accurate and timely precise modulation when combined with VEGF signaling. Modulating NOS activity and VEGF signaling simultaneously therefore provides an intriguing possibility to stimulate endogenous neurogenesis after brain injury. In addition, how the NO signaling would interact with its other partners (e.g., p75) in adult neurogenesis remains to be investigated. For instance, p75-expressing neural progenitor cells represented the BDNF-responsive neurogenic cells (43), and the NOS activity could therefore be implicated in survival of these progenitor cells.
Summary
The present study showed that RMS vasculature could function as an independent neurogenic niche and facilitate the migration of neuroblasts. The BBB organization is specific with enhanced permeability so that the neural stem cells could sense changes in the circulation. In addition, the “leaky” vasculature might support neurogenesis by releasing NO and other trophic factors, partly by associated astrocytes. The previous theory of “glial-independent migration” in the RMS region based on morphology observation therefore might require modification, as the glial cells might also be involved in another indirect manner regulating adult neurogenesis, such as contribution to the vasculature organization. The fact that the RMS neurovascular niche shared many characteristics with stem cell niches in brain tumors and injury further implied that vasculature is one common organization among diverse stem cell niches. This knowledge might provide novel engineering targets in developing treatments to brain injury or neurodegenerative disorders, such as the specialized VEGF signaling. This may also explain why the OB is one of the few places in the CNS that allows for regeneration in the adult.
Footnotes
Acknowledgments
The study was supported by the Department of Anatomy, University of Hong Kong. T.Y., Y.L., D.T., K.S., and R.E.-B. designed the experiment; T.Y. and Y.L. performed the experiment; T.Y., Y.L., D.T., K.S., and R.E.-B. analyzed the data and wrote the paper together. The authors declare no conflicts of interest.
