Abstract
While mesenchymal stem cells (MSCs) are easily accessible from mesenchymal tissues, such as bone marrow and adipose tissue, they are heterogeneous, and their entire composition is not fully identified. MSCs are not only able to differentiate into osteocytes, chondrocytes, and adipocytes, which belong to the same mesodermal lineage, but they are also able to cross boundaries between mesodermal, ectodermal, and endodermal lineages, and differentiate into neuronal- and hepatocyte-like cells. However, the ratio of such differentiation is not very high, suggesting that only a subpopulation of the MSCs participates in this cross-lineage differentiation phenomenon. We have identified unique cells that we named multilineage-differentiating stress-enduring (Muse) cells that may explain the pluripotent-like properties of MSCs. Muse cells comprise a small percentage of MSCs, are able to generate cells representative of all three germ layers from a single cell, and are nontumorigenic and self-renewable. Importantly, cells other than Muse cells in MSCs do not have these pluripotent-like properties. Muse cells are particularly unique compared with other stem cells in that they efficiently migrate and integrate into damaged tissue when supplied into the bloodstream, and spontaneously differentiate into cells compatible with the homing tissue. Such a repairing action of Muse cells via intravenous injection is recognized in various tissues including the brain, liver, and skin. Therefore, unlike ESCs/iPSCs, Muse cells render induction into the target cell type prior to transplantation unnecessary. They can repair tissues in two simple steps: collection from mesenchymal tissues, such as the bone marrow, and intravenous injection. The impressive regenerative performance of these cells provides a simple, feasible strategy for treating a variety of diseases. This review details the unique characteristics of Muse cells and describes their future application for regenerative medicine.
Mesenchymal Stem Cells
Stem cells are defined as cells that possess the ability to self-renew and differentiate. According to their differentiation ability, stem cells are categorized as totipotent stem cells, represented by fertilized eggs, that are able to generate both embryonic (all cells of the body except for the extraembryonic cells) and extraembryonic cells (trophectodermal; yolk sac, amnion, etc.); pluripotent stem cells, represented by the inner cell mass, that are able to generate all the cells of the body, such as all the cells of the three germ layers (endodermal, ectodermal, and mesodermal), except for the extraembryonic cells; multipotent stem cells that generate some but not all the cells of the body, which could include one or two lineage cell types; and unipotent stem cells, represented by muscle stem cells, that generate only one type of cell (19). Among all of these stem cell types, pluripotent stem cells are highly anticipated to contribute to regenerative medicine due to their ability to generate any cell type in the body, implying their applicability to a wide variety of diseases.
Like neural and hematopoietic stem cells, somatic stem cells can differentiate into cells that comprise the tissue in which they reside (6,38). Although it was initially believed that somatic stem cells could not cross the boundaries between the three germ layers (transdifferentiation), this dogma was challenged by the discovery of the differentiation potential of mesenchymal stem cells (MSCs). MSCs are somatic stem cells harvested from mesenchymal tissues such as the bone marrow (BM), adipose tissue, umbilical cord, and dental pulp (9,15,27,29,37). The first report of the differentiation of BM-MSCs into osteocytes, chondrocytes, and adipocytes in the presence of cytokine cocktails (27) was soon followed by reports that MSCs from nearly all types of mesenchymal tissue are able to differentiate into cells representative of all three germ layers by either gene introduction or cytokine induction. For example, MSCs differentiate in vitro into endothelial cells (23), cardiac muscle cells (21), skeletal muscle cells (3), hepatocytes (24), neuronal cells (4), epithelial cells (32), peripheral glial cells (5), and insulin-producing cells (26). However, the differentiation efficiency of MSCs is generally not high, suggesting that only a small subpopulation of MSCs participate in the differentiation (15).
Another notable feature of MSCs is that very few of the MSCs injected locally or intravenously into various disease models eventually integrate into the damaged tissue and express markers specific to the homing tissue. This phenomenon is particularly observed in the liver and brain, where MSCs must differentiate across lineages from mesoderm into endoderm and ectoderm, respectively, after integration (20,25,33). This feature of MSCs suggests that only a subpopulation of the cells is pluripotent-like.
MSCs are generally known to be positive for the mesenchymal markers cluster of differentiation 105 (CD105)/CD73/CD90 and negative for CD45/CD34/CD14/CD19/human leukocyte antigen-DR (HLA-DR) (11). They are usually obtained from mesenchymal tissues simply by collecting adherent cells, however, and are thus heterogeneous (27). The main problem in the basic research of MSCs is that most analyses have been performed using the heterogeneous bulk MSCs rather than purified populations or single cells, and thus the cells actually responsible for the pluripotent-like phenomenon of MSCs have not been identified.
Discovery of Muse Cells
Multilineage-differentiating stress-enduring (Muse) cells, which were first reported in 2010 by Kuroda et al., correspond to just several percent of the MSCs (16). Muse cells express pluripotency markers, are able to generate cells representative of all three germ layers from a single cell, and their triploblastic differentiation ability at the single-cell level is reproduced over generations, suggesting that they are self-renewable and retain their pluripotency (10,17,22,36). Therefore, these cells fulfill the criteria for pluripotent stem cells. Muse cells distribute sporadically in the connective tissue of nearly every organ and do not seem to associate with a structural niche (36). In the BM, however, they do not locate in the connective tissue, but rather reside in the BM cavity at a ratio of nearly 1:3,000 cells (i.e., comprising ~0.03% of the mononucleated cell fraction) (16).
Importantly, cells other than Muse cells that comprise MSCs do not express pluripotency genes, nor do they cross oligolineage boundaries between the mesoderm, ectoderm, and endoderm (10,22,30,31,36). In other words, MSCs can be clearly segregated into Muse and non-Muse cells based on their pluripotency. Furthermore, the lower rate of MSC differentiation into variety of cell types in vitro and in vivo after integration into damaged organs could be explained by the fact that Muse cells comprise only 1% to several percent of the total MSCs. MSCs have pleiotropic effects as they are heterogeneous. The precise composition of MSCs (e.g., the number of different cell types and their percentage in MSCs) remains unclear, and thus the pleiotropic effects of MSCs are not fully understood. In this sense, the discovery of Muse cells as the cells that are responsible for the phenomena related to the pluripotency of MSCs while the remaining MSCs, namely the non-Muse cells, do not participate in these phenomena is highly significant. The pluripotency of Muse cells compared with non-Muse cells sharply distinguishes these cells among MSCs.
Although in very low frequency, MSCs cultured in α-minimum essential medium (α-MEM) or Dulbecco's minimum essential medium (DMEM) supplied with 10% fetal bovine serum (FBS) spontaneously form cell clusters very similar to embryoid bodies that are formed by embryonic stem cells (ESCs) in suspension culture (16) (Fig. 1A). Such clusters are occasionally pigmented and have hair-like structures, and importantly, the cells beneath the cluster are very similar to ESCs. We attempted to characterize the ESC-like clusters (Fig. 1). Interestingly, cells positive for each of the ectodermal, endodermal, and mesodermal markers were detected in the clusters, suggesting that MSCs contain a small subpopulation of pluripotent-like cells and that those cells are responsible for the cluster formation and triploblastic differentiation within the cluster (16).

Muse cell clusters and their differentiation potential. (A) Human mesenchymal stem cells (MSCs) spontaneously form clusters similar to embryonic stem cell (ESC)-derived embryoid bodies formed in suspension. (B) After isolating multilineage-differentiating stress-enduring (Muse) cells by fluorescence-activated cell sorting based on stage-specific embryonic antigen 3 (SSEA-3), single Muse cells were subjected to single-cell suspension culture. (C) Single Muse cells generated characteristic clusters very similar to the embryoid bodies formed by human ESCs in suspension. (D) When the cell clusters were transferred onto gelatin culture, cells expanded out from the adhered adherent cluster. (E–I) The cells that expanded from the cluster on gelatin culture contained cells positive for (E) cytokeratin 7 (endodermal marker), (F) α-fetoprotein (endodermal), (G) desmin (mesodermal), (H) smooth muscle actin (mesodermal), and (I) neurofilament (ectodermal). Scale bars: 100 μm (A, D), 20 μm (C), 50 μm (E–I). [Pictures adapted and modified with permission from Kuroda et al. (2010), Proceedings of the National Academy of Sciences USA (16) and with permission from Wakao et al. (2011), Proceedings of the National Academy of Sciences USA (36).]
Generally, tissue stem cells are stress tolerant, and while normally dormant they are activated by stimuli such as stress (13). Following their activation, they enter into the cell cycle, differentiate into cells comprising the tissue in which they reside, and contribute to tissue regeneration and repair. We incubated MSCs for over 16 h in trypsin solution containing no nutrients but only digestive enzymes in hydroxyethyl piperazineethanesulfonic acid (HEPES) buffer. If pluripotent-like cells exist among MSCs, those cells may be defined as “stem cells” in the true sense and should thus be very tolerant to stress compared to other MSCs. In fact, this approach substantially increased the ratio of putative pluripotent stem cells while simultaneously eliminating cells other than the putative pluripotent stem cells among MSCs, allowing us to identify the Muse cells (16). The unique characteristics of Muse cells are detailed in the following sections.
SSEA-3 is a Marker of Muse Cells
Muse cells are a pluripotent subpopulation of MSCs, and they have two different personas, namely that of MSCs and pluripotent stem cells. Such duality can be seen in their marker expression: Muse cells express stage-specific embryonic antigen-3 (SSEA-3), a well-known surface marker for human ESCs, and mesenchymal markers such as CD105, CD29, and CD90 (17,22,36) (Fig. 2). Therefore, they can be isolated from mesenchymal tissues and cultured mesenchymal cells as cells double positive for SSEA-3 and one of the mesenchymal markers (e.g., CD105) (Fig. 2). Particularly, when cultured mesenchymal cells, such as BM-MSCs, adipose stem cells, and fibroblasts, comprise the basal population, a single application of SSEA-3 will successfully select Muse cells, as nearly 100% of the MSC population is positive for general mesenchymal markers, and thus there is no need to stain Muse cells with CD105, CD29, or CD90 (17). Muse cells not only express SSEA-3, but they also express the pluripotent markers octamer binding transcription factor 3/4 (Oct3/4), sex-determining region Y box 2 (Sox2), Nanog, and reduced expression 1 (Rex1) (10,16,17,22,36).
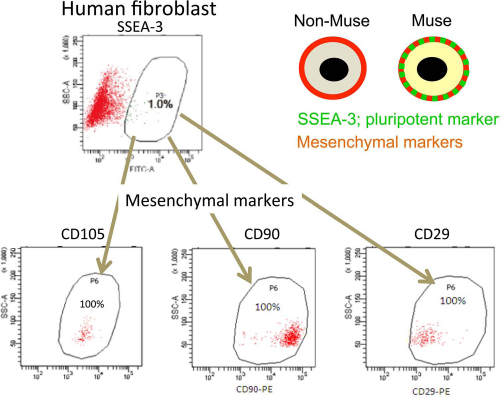
Marker expression in Muse cells. (A) Human fibroblasts contain ~1% of SSEA-3+ Muse cells. These Muse cells are all positive for the mesenchymal markers cluster of differentiation 105 (CD105), CD90, and CD29. Thus, Muse cells are double positive for the pluripotency marker SSEA-3 and mesenchymal markers, but non-Muse cells are positive only for mesenchymal markers. [Pictures adapted and modified with permission from Wakao et al. (2011), Proceedings of the National Academy of Sciences USA (36).]
In our hands, non-Muse cells, cells other than Muse cells among the MSCs, do not express pluripotency markers, including SSEA-3, and are positive only for general mesenchymal markers (36) (Fig. 2). Thus, SSEA-3 can be applied to segregate Muse from non-Muse cells.
Pluripotency of Muse Cells
Pluripotent stem cells can be defined as cells that can generate all kinds of cells in the endodermal, mesodermal, and ectodermal lineages and are self-renewable. As described below, Muse cells have both abilities at a single cell level and thus qualify as pluripotent stem cells.
Spontaneous differentiation of ESCs into triploblastic differentiation is occasionally observed on gelatin-coated culture plates. Similarly, single Muse cell-derived M-clusters expand when cultured on a gelatin-coated adherent system (Fig. 1C, D), and within those expanded cells, cells positive for markers representative of all three germ layers, namely, neurofilament (ectodermal), smooth muscle actin (mesodermal), and α-fetoprotein (endodermal), are recognized (17) (Fig. 1E–I). This differentiation is generated not by cytokine induction, but rather spontaneously, and the positivity for each marker is not very high. Positive ratios for neurofilament and α-fetoprotein are usually only several percent of the total expanded cells, while that for smooth muscle actin, indicative of the mesodermal lineage, the same original background as that of Muse cells, is higher than that of the other two markers, ~10–15% (17). Thus, although Muse cells are prone to differentiate into mesodermal lineage cells, the fact that they can spontaneously differentiate into cells from other lineages helps support the pluripotency of Muse cells.
Not only do Muse cells differentiate spontaneously, they also differentiate at a high rate (~80–95%) into hepatocyte and neural lineage cells as well as into adipocytes and functional melanin pigment-producing cells, namely melanocytes, when the proper cocktail of cytokines is supplied (22,36). Therefore, their differentiation commitment can be regulated by various factors.
Non-Muse cells sharply contrast with Muse cells in terms of their differentiation potential. While non-Muse cells are able to differentiate into osteocytes, cartilage cells, and adipocytes in the presence of certain sets of induction cytokines, their differentiation rate is generally lower than that of Muse cells, and the time required for their differentiation is substantially longer than that required for the differentiation of Muse cells (22). Most importantly, non-Muse cells are unable to cross the lineage boundaries between original mesoderm to ectoderm or endoderm. In fact, even in the presence of cytokine cocktails, non-Muse cells (i.e., cell populations from which Muse cells have been removed) do not differentiate into cells, such as hepatocytes (10,12,22), neuronal cells (10,22,35,36,39), or melanocytes (34), and have only partial intracellular responses to cytokine induction. For example, for the induction of melanocytes, in which it is necessary for Muse and non-Muse cells to cross from the mesodermal to ectodermal lineage, the addition of 10 factors [including wingless-type mouse mammary tumor virus (MMTV) integration site family, member 3A (Wnt3a), endothelin-3, linoleic acid, stem cell factor, and dexamethasone] is sufficient to convert Muse cells into functional melanocytes while only a partial response is evoked in non-Muse cells. Muse cells originally express microphthalmia-associated transcription factor and KIT, and newly express tyrosinase-related protein 1 (TRP-1) and glycoprotein 100 (gp100) at 3 weeks, dopachrome tautomerase at 5 weeks, and finally tyrosinase at the endpoint of 6 weeks, at which point they become pigment-producing functional melanocytes that are positive for the L-3,4-dihydroxyphenylalanine (L-DOPA) reaction. On the other hand, while non-Muse cells react at 3 weeks and newly express TRP-1, the expression of TRP-1 is not sustained and eventually returns to negative within 5 weeks, with the cells reverting back to a fibroblast-like morphology, suggesting only a partial response to melanocyte induction (34). Such a difference between Muse and non-Muse cells is consistent with the fact that Muse cells originally express a subset of pluripotency genes that are not expressed in non-Muse cells.
Since Muse cells are naturally existing somatic stem cells, they generate non-Muse cells during proliferation by asymmetric cell division (16). Even if Muse cells are purified by cell sorting, they will generate non-Muse cells during proliferation in culture, and the proportion of Muse cells will gradually decrease, reaching a plateau to several percent of total cells eventually, which corresponds to the exact proportion of Muse cells in general MSCs as mentioned above. Based on these phenomena, one of the possibilities is that Muse cells are the original pluripotent stage of MSCs, and thus the MSCs that we usually observe are a mixture of original Muse cells and their derivative, non-Muse cells.
Muse cells also exhibit self-renewal of pluripotency. Single human adipose-derived M-clusters generated in suspension culture were collected, and half of the clusters were transferred individually onto gelatin culture to allow the cells to expand out of the M-cluster, and their expression of endodermal, mesodermal, and ectodermal markers, namely, microtubule-associated protein 2 (MAP-2), guanine-adenine-thymine-adenine, binding protein 6 (GATA6), α-fetoprotein, and NK2 homeobox 5 (NKX2.5), was confirmed (10,22). The remainder of the clusters were individually transferred to adherent culture and allowed to proliferate for a certain period, after which they underwent a second round of single-cell suspension in culture to generate second-generation M-clusters (Fig. 3). This experimental cycle was repeated three times, and M-clusters from each step exhibited triploblastic differentiation at the single cell level. Similar results were observed in another experiment when the culture cycles were continued for up to five generations for Muse cells derived from either human BM-MSCs or fibroblasts (16).

Self-renewable ability of Muse cells. Schematic diagram outlines experiments that confirm the self-renewal ability of Muse cells. Microtubule-associated protein 2 (MAP-2; ectodermal), guanine-adenine-thymine-adenine-binding protein 6 (GATA6; endodermal), α-fetoprotein (α-FP; endodermal), and NK2 homeobox 5 (Nkx2.5; mesodermal) gene expression was detected by reverse transcription-polymerase chain reaction from cells expanded from each cluster from first to third generations. Positive controls (Posi con) for MAP-2, α-FP, and Nkx2.5 were human whole embryo and that for GATA6 the human fetus liver. Clusters 1 and 2 in β-actin are from human whole embryo (1) and human fetus liver (2), respectively. Scale bars: 25 μm. RT-PCR, reverse transcription polymerase chain reaction. [Pictures adapted with permission from Ogura et al. (2014), Stem Cells and Development (22).]
Overall, Muse cells exhibit both triploblastic differentiation potential from a single cell and self-renewal, which strongly supports the pluripotency of Muse cells.
Muse Cells Are Nontumorigenic
In our studies, comparing the gene expression among three types of cells, human ESC/induced pluripotent stem cells (iPSCs), and human Muse and non-Muse cells, reveals that the “expression pattern” of pluripotency genes is very similar between Muse and ESC/iPSCs, whereas the “expression level” is moderate in Muse cells compared to ESC/iPSCs. Importantly, non-Muse cells do not express pluripotency genes, which is in sharp contrast to Muse cells (36) (Fig. 4).

Gene expression pattern in human Muse, iPSCs, non-Muse, and non-Muse cells. The expression pattern of pluripotency markers in Muse cells and induced pluripotent stem cells (iPSCs) is similar, but the expression level is higher in iPSCs than in Muse cells. Genes related to cell cycle progression, namely, tumorigenesis, do not largely differ between Muse and non-Muse cells, but are upregulated in iPSCs. [Pictures adapted and modified with permission from Wakao et al. (2011), Proceedings of the National Academy of Sciences USA (36).]
The pattern of gene expression of cell cycle-related factors (namely, tumorigenic factors), however, differs between ESC/iPSCs and Muse cells. Generally, those factors are highly expressed in ESC/iPSCs, while very low expression is observed in Muse cells and the level and pattern are similar to that in non-Muse cells (36) (Fig. 4).
Consistently, telomerase activity, an indicator of tumorigenic activity, is very low in Muse cells compared with iPSCs and Hela cells, and is rather similar to that in non-Muse cells (15). In fact, human BM- and adipose-derived Muse cells transplanted into the testes of immunodeficient mice do not generate any tumors, including teratomas, for up to 6 months (10,16,22). Therefore, Muse cells are considered to have a low risk of tumorigenesis.
Muse Cells Survive and Proliferate Both in Suspension and in an Adherent Environment
The stability of cell survival and proliferation in vitro normally depends on the environment (e.g., whether it is a suspension or adherent system). Hematopoietic cells basically survive in a suspension system and not in an adherent system, while most other cell types, including those of mesenchymal origin, are dependent on an adherent system and will not survive in suspension. Muse cells are unique because they survive and proliferate in both types of environments. Non-Muse cells, on the other hand, are not able to survive in a suspension culture and are only able to stably proliferate in an adherent system. This is a sharp difference between Muse and non-Muse cells.
When Muse cells are transferred to a single-cell suspension culture, they begin proliferating within a couple of days, forming a cluster that is very similar to the embryoid bodies formed from ESCs in suspension (16) (Fig. 1C). Such characteristic clusters formed from a single Muse cell, called M-clusters, not only exhibit an ESC-like morphology, but also contain substantially elevated levels of pluripotent stem cell markers, such as Oct3/4, Sox2, and Nanog, up to 50–100 times higher than those in adherent Muse cells (unpublished data). This finding suggests that a suspension environment potentiates Muse cell pluripotency.
Although the pluripotency of Muse cells is accelerated in suspension, their proliferative activity is limited. When M-clusters reach 70–100 μm in diameter within 10 days to 2 weeks, their growth ceases. However, proliferation resumes upon transfer onto an adherent culture (16). Once the M-clusters adhere to the culture dish, the cells gradually expand out from the cluster (Fig. 1D).
The proliferation speed of Muse cells is ~1.3 days/cell division, both in adherent and suspension systems, nearly the same as that of human fibroblasts. This finding suggests that Muse cells can be grown on a clinically relevant scale (17).
Differentiation Potential of Muse Cells Derived From Different Tissues
Muse cells reside in the connective tissue of nearly every organ except the BM, where they are located in the BM cavity (22) (Fig. 5). MSCs are suggested to locate closely to the subendothelial region of small blood vessels in the connective tissues of nearly every organ (7). While both Muse cells and MSCs share the same home ground, namely, connective tissue, the localization of Muse cells did not seem pertinent to any particular histological structures including vessels; instead they were located sparsely within the matrix (Fig. 5). The relationship of their original histological locations in connective tissue is an interesting point to be clarified in the future.

Tissue distribution of Muse cells. Muse cells labeled by SSEA-3 are observed in the connective tissue of human fat tissue, dermis, and umbilical cord, and in mouse trachea and spleen. Muse cells are contained in the bone marrow cavity (human). Scale bars: 50 μm. [Pictures for the fat and dermis adapted with permission from Wakao et al. (2011), Proceedings of the National Academy of Sciences USA (36).]
To examine whether Muse cells derived from different tissues have a different differentiation potential, we compared the properties of Muse cells derived from the BM, dermal fibroblasts, and adipose tissue (22). Adipose-Muse cells expressed genes related to the differentiation of adipocytes, osteocytes, and skeletal muscle cells, all categorized as mesodermal lineage, at higher levels than did the BM- and dermal-Muse cells. BM-Muse cells, however, contained the highest levels of factors pertinent to the endodermal lineage, such as hepatocytes and pancreatic cells. Both BM- and dermal-Muse cells exhibited higher expression levels of factors related to the ectodermal lineage, such as neuronal-, melanocyte-, and epidermal-related genes, than adipose-Muse cells (22). Therefore, while pluripotency is demonstrated in all BM-, dermal-, and adipose-Muse cells, their differentiation potential is not the same. This suggests that the appropriate tissue source should be selected based on the target cell type or tissue.
Muse Cells Directly Contribute to Tissue Repair
The unique and prominent feature of Muse cells that is not recognized by other types of stem cells, including ESC/iPSCs and somatic stem cells such as neural stem cells and hematopoietic stem cells, is that they have the ability to strongly perceive damage signals released by injured/damaged tissue, home to the damaged tissue when administered intravenously, to readily spontaneously differentiate in vivo into cells compatible with the homing tissue after integration, and can even cross oligolineage boundaries between mesodermal to endodermal or to ectodermal cells. Therefore, Muse cells can work as repairing cells for wide range of tissues and organs.
Intravenously injected human green fluorescent protein (GFP)-labeled Muse cells were demonstrated to home into the liver and muscle of fulminant hepatitis and muscle degeneration models of severe combined immunodeficient (SCID) mice, respectively. These integrated Muse cells spontaneously differentiated into hepatocytes and skeletal muscle cells after integration, replenished new cells, and contributed to eventual tissue repair (16).
In the fulminant hepatitis model, homing and integration of human GFP-Muse cells was intensively recognized around the vessels, suggesting that Muse cells penetrated into the damaged liver via the blood vessels (16) (Fig. 6). Integration of human Muse cells was further confirmed by immunohistochemistry using anti-human Golgi complex and GFP. These integrated GFP-Muse cells expressed human-specific albumin and anti-trypsin, suggesting that they spontaneously differentiated into cells similar to functional human hepatocytes without being induced into hepatocytes before transplantation (16). Muse cells were also shown to integrate as liver progenitor cells in the early phase and then spontaneously differentiate into major liver components, including hepatocytes, cholangiocytes, sinusoidal endothelial cells, and Kupffer cells, in a physical partial hepatectomy model (12). Similarly, in a skeletal muscle degeneration model, intravenously injected human GFP-Muse cells integrated into damaged muscle tissue and spontaneously differentiated into human dystrophin- and paired box 7 (Pax7)-positive cells to replenish new skeletal muscle cells (16).
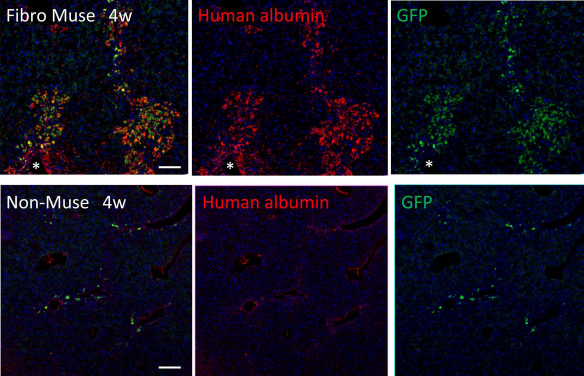
Homing and spontaneous differentiation of Muse cells into hepatocytes in fulminant hepatitis model. Green fluorescent protein (GFP)-positive human Muse or non-Muse cells derived from fibroblasts were infused into the tail vein of a severe combined immunodeficient (SCID) mouse with fulminant hepatitis (at 4 weeks). Human Muse cells abundantly integrated into the damaged liver from nearby blood vessels (*) and expressed human albumin, whereas the majority of non-Muse cells were not retained in the liver and did not express human albumin. Scale bars: 50 μm. [Pictures adapted with permission from Y. Kuroda et al. (2010). Proceedings of the National Academy of Sciences USA (16).]
Locally injected Muse cells were demonstrated to contribute directly to tissue repair. Skin ulcers of a diabetes mellitus model are known to be intractable. Subcutaneous injection of human adipose-Muse cells near the skin defect in nonobese diabetic (Nod) SCID diabetes mellitus mice led to remarkable and rapid skin repair (14). The injected human Muse cells were incorporated into both the dermis and epidermis of the damaged area and differentiated into dermal fibroblasts, blood vessels, and keratinocytes as new functional cells (14). In a stroke model of rat, human Muse cells injected into the region around the ischemia integrated in the host brain and spontaneously differentiated into neuronal nuclei (NeuN)-positive neuronal cells (~65%) (35). Integrated Muse cells were shown to reconstruct pyramidal tract as well as somatosensory circuit, which displayed normalized hindlimb somatosensory evokedpotentials (35).
Importantly, non-Muse cells do not exhibit these activities; they do not remain in host damaged tissues from the early phase to the late stage after injection and thus do not differentiate or replenish lost cells (12,14,16,35) (Fig. 6).
These outstanding properties of Muse cells make regenerative medicine treatments simple and feasible in only two simple steps: 1) collection of Muse cells from tissue or cultured mesenchymal cells by SSEA-3, and 2) injection of Muse cells into the bloodstream. Thus, the induction of cells into purposive cells in a cell processing center prior to transplantation, which is usually required for ESCs and iPSCs, is not necessary in the case of Muse cells (Fig. 7).
Treatment with Muse cells by simple two steps; 1) collection from the bone marrow (BM), fat, or umbilical cord (UC) using SSEA-3 or stress, and 2) injection into the blood stream or local target in the patient.
Muse Cells may Resolve the Problems of MSCs
MSCs are already being used in clinical studies targeting a variety of diseases, including myocardial infarction and cartilage defects (2,28). MSCs can be obtained from easily accessible sources such as the BM and umbilical cord. Muse cells are naturally existing nontumorigenic cells and comprise a small subpopulation of MSCs. Therefore, Muse cells are suggested to have low safety concerns.
In contrast, treatment with MSCs currently faces several problems: 1) low homing rate to the damaged site when supplied into the bloodstream, 2) poor survival for longer period after homing to the damaged tissue where a stressful microenvironment caused by damaged cells is hostile to homed cells, and 3) low rate of differentiation into purposive cells (1,8,18). However, the use of Muse cells is expected to resolve these problems.
When Muse cells are fully utilized, the low homing rate of MSCs by intravenous injection will substantially improve as Muse cells home into the damage site at a higher rate than MSCs due to their ability to perceive damage signals. As reported previously in models of fulminant hepatitis, skeletal muscle degeneration, stroke, and skin regeneration, Muse cells actively migrate to and integrate into the damaged sites with a higher efficiency than non-Muse cells (16). Since non-Muse cells represent ~99% of MSCs, the actions of non-Muse cells can be expected to account for the majority of the representative activities of MSCs. Therefore, Muse cells are expected to produce better results than MSCs.
Muse cells are tolerant to stress and express some factors related to stress tolerance (16). Thus, survival in the hostile environment of damaged tissue will also be improved by using Muse cells. Damaged tissue is usually a very hostile environment in which physiologic homeostasis is disrupted by cell death and infiltration by immunologic cells. Previous reports demonstrated that Muse cells home to damaged tissue and survive after integration whereas non-Muse cells do not (12,14,16,35,39). Therefore, Muse cells are considered suitable stem cells for treating acute or subacute disease phases when the microenvironment is particularly hostile.
Differentiation potential is directly linked to the efficiency of cell-based treatment. In previous MSC transplantation experiments, the ratio of differentiation of integrated MSCs into functional cells in vivo was very low, and their efficiency was mainly determined based on their trophic and immunomodulatory effects. In this respect, MSCs have limited tissue regeneration ability. The low rate of differentiation of MSCs into purposive cells may be resolved by the ability of Muse cells to spontaneously differentiate into purposive cells that are functional according to “the site of theory” after integration. In contrast, non-Muse cells do not remain in the damaged tissue from the early phase and do not replenish lost cells (12,14,35).
As intravenously injected Muse cells specifically integrate into damaged tissue and replenish new cells (16), intravenous injection, which is the simplest and easiest approach, could be applicable for Muse cell treatment. The route of administration may depend on the ability of stem cells to recognize the damaged tissue and to differentiate into the appropriate cells.
Future Perspectives
Induction into purposive cells prior to transplantation in a cell processing center is required for clinical use of ESCs and iPSCs because of their original tumorigenic properties in the undifferentiated state. In addition, cells that failed to differentiate in the final product must be eliminated before transplantation for the same reason. The requirement for multiple steps is a cause for concern regarding quality control of the cell products, as well as time and cost.
Muse cells are unique in their ability to efficiently migrate and integrate into damaged tissue, their nontumorigenicity, and their ability to spontaneously differentiate into cells compatible with the homing tissue, rendering induction into target cells prior to transplantation unnecessary. Muse cells can repair tissues in two simple steps: 1) collection from mesenchymal tissues such as the BM, and 2) intravenous injection. Therefore, Muse cells, with their impressive regenerative performance, may provide a simple feasible strategy for the treatment of a variety of diseases. Furthermore, such a simple and accessible approach may lower the hurdle of regenerative medicine, allowing for application of cell therapy to general medicine (Fig. 7). Summarization of property in ESCs, Muse, and non-Muse cells is shown in Table 1.
Summary of Characteristics in ESCs, Muse, and Non-Muse Cells

ESCs, embryonic stem cells; Muse, multilineage-differentiating stress-enduring; CPC, cell processing center.
From the standpoint of clinical treatment, allogenic cell therapy would be more practical than autologous therapy because autologous therapy would require more time to collect and expand Muse cells for clinical scale and would not be applicable to acute-phase patients. Prompt cell treatment would likely achieve a better outcome, suggesting the high potential of allogenic cell therapy. Additional studies are required to validate the power and longevity of the effect of Muse cells to enhance the feasibility of cell transplantation therapy.
Footnotes
Acknowledgments
This work was supported by a Grant-in-aid from the New Energy and Industrial Technology Development Organization (NEDO), a Grant-in-aid from the Health Labour Sciences Research of the Ministry of Health, Labour, and Welfare, Japan and Research Program on Hepatitis of the Japan Agency for Medical Research and Development (AMED). Mari Dezawa is party to a codevelopment agreement with Clio, Inc. for the use of Muse cells.
