Abstract
Large-scale production and noninvasive methods for harvesting mesenchymal stem cells (MSCs), particularly in elderly individuals, has prompted researchers to find new patient-specific sources for MSCs in regenerative medicine. This study aims to produce MSCs from human induced pluripotent stem cells (hiPSCs) and to evaluate their therapeutic effects in a CCl4-induced mouse model of fulminant hepatic failure (FHF). hiPSC-MSCs have shown MSC morphology, antigen profile and differentiation capabilities, and improved hepatic function in our model. hiPSC-MSC-transplanted animals provide significant benefit in terms of survival, serum LDH, total bilirubin, and lipid peroxidation. hiPSC-MSC therapy resulted in a one-third reduction of histologic activity index and a threefold increase in the number of proliferating hepatocytes. This was accompanied by a significant decrease in the expression levels of collagen type I, Mmp13, Mmp2, and Mmp9 genes and increase in Timp1 and Timp2 genes in transplanted groups. hiPSC-MSCs secreted hepatocyte growth factor (HGF) in vitro and also expressed HGF in evaluated liver sections. Similar results were observed with human bone marrow (hBM)-derived MSCs. In conclusion, our results have demonstrated that hiPSC-MSCs might be valuable appropriate alternatives for hBM-MSCs in FHF liver repair and support liver function by cell therapy with a large-scale production capacity, patient-specific nature, and no invasive MSC harvesting.
Keywords
Introduction
Fulminant hepatic failure (FHF) is characterized by severe and abrupt loss of liver metabolic and immunological functions. The most common etiologies of FHF are drug abuse (paracetamol, in particular) (17) in patients that commit suicide and viral hepatitis, which affects mostly young adults with a high mortality. Although it is a life-threatening manifestation, no straightforward treatments have been developed, with the exception of liver transplant (LT) (5). A serious shortage of liver donors in combination with other risks of organ transplantation mandates alternative therapeutic approaches.
The transplantation of mesenchymal stem cells (MSCs) to repair injured tissues has raised the hopes for curing liver failure [for review, see Vosough et al. (40)]. To repair the liver, MSCs have been derived from different tissues such as human bone marrow (hBM) (2), umbilical cord blood (33), and adipose tissue (1, 3). Such therapeutic treatments have led to host liver recovery and induced endogenous regeneration programs (1, 26, 39). However, harvesting MSCs from most human sources requires an invasive procedure. The numbers of MSCs that can be obtained from a single donor are limited, and the capacity of these cells for long-term in vitro proliferation is rather poor (12). Additionally, a large number of cells are needed for transplantation (37). The availability of a suitable donor and reduction in the ability of MSCs to self-renew with increased aging are other limitations of this approach (12).
To overcome these shortfalls, the generation of MSCs/ mesenchymal progenitors from pluripotent stem cells such as human embryonic stem cells (hESCs) or human induced pluripotent stem cells (hiPSCs) is necessary. More recently, enriched populations of MSCs have been derived from hESCs by various methods (4, 10, 12, 19, 21, 23, 34, 38, 44, 47). These hESC-MSCs are karyotypically stable and are immunophenotypically and functionally similar to hBM-MSCs (9, 38, 47). Interestingly the transcriptome of hESC-MSCs is similar to hBM-MSCs (47) and have a greater proliferative capacity when compared with human BM-MSCs (12), in addition to homogenous tissue production and lack of in vivo teratoma formation (18, 47). Additionally, hESC-MSCs may be used for human gene therapy, because hESCs have the potential to proliferate indefinitely without senescence. In comparison with adult hBM-MSCs, they also possess more immunosuppressive effects (32) toward natural killer (NK) cells as well as T lymphocytes (6, 46), which are suggestive of lessened allograft rejection. It has been demonstrated that the engraftment capability of hESC-MSCs in a recipient liver is higher than BM-MSCs in an acute liver injury; moreover, the number of engrafted cells are unaffected by the number of passages, which indicates their ability for possible clinical applications (35). These all highlight the significant potential usage of hESC-MSCs in regenerative medicine and may represent a new potential cell source for therapeutic use.
On the other hand, the derivation of hiPSCs that are highly similar to their respective embryo-derived ESCs (29) has opened a new window for regenerative medicine. These cells can be produced from most somatic tissues using different approaches [for review, see Gonzalez et al. (8)]. MSCs/mesenchymal progenitors have also been produced from hiPSCs and are comparable to hBM-MSCs in terms of surface marker expression and differentiation potential [for review, see Jung et al. (13)]. It has been demonstrated that transplanting iPSC-MSCs into the hindlimb of an ischemic mouse model significantly attenuated injury and promoted vascular and muscle regeneration (22). Additionally, iPSC-MSCs display remarkable inhibition of NK cell proliferation and cytolytic function (6). Therefore, highly potent hiPSC-MSCs could be a valuable cell source for clinical applications and tissue engineering investigations. In this regard, hiPSC-MSCs would be a readily accessible source for autologous transplantation and even a histocompatible source for allotransplantation after human leukocyte antigen (HLA) typing.
To our knowledge, no attempts have been made to determine whether the transplantation of hiPSC-MSCs improves liver failure in an animal model. This study aims to evaluate, for the first time, the therapeutic role of hiPSC-MSCs in an in vivo FHF phenotype and compare them with BM-MSCs.
Materials and Methods
Cell Culture and Derivation of Mesenchymal Progenitors
The male hiPSC line Royan hiPSC1 (36), at passages 40–50, was used in these experiments. Cells were expanded and passaged under feeder-free culture conditions on Matrigel-coated dishes (Sigma-Aldrich, St. Louis, MO, USA) in hESC medium that contained 100 ng/ ml basic fibroblast growth factor (bFGF; Sigma-Aldrich) as previously described (25). The medium was changed every other day, until day 7. Before passaging hiPSCs on day 7, spontaneously differentiated cells that appeared in the edges of the colonies were mechanically scraped (24). The obtained cells were cultured in human MSC medium that contained Dulbecco's modified Eagle's medium (DMEM; 12800-116), 15% fetal bovine serum (FBS; 16000-044), 1 mM nonessential amino acids (11140-050), 2 mM l-glutamine (25030-164), and 1% penicillin/ streptomycin (15140122) (all from Invitrogen, Carlsbad, CA, USA) and β-mercaptoethanol (0.1 mM; Sigma-Aldrich, M7522). Cells were passaged by trypsin/EDTA (Invitrogen, 25200-072) treatment.
Antigen Profiling and Functional Differentiation of hiPSC-MSCs
hiPSC-MSCs underwent dissociation in 0.05% trypsin-EDTA. Then, cells were washed three times in phosphate-buffered saline (PBS; Gibco, Gaithersburg, MD, USA, 21600-010), supplemented with 1% heat-inactivated FBS, and 2 mM EDTA (Merck, Darmstadt, Germany; 1084211000) and then fixed in 4% paraformaldehyde (Merck; 1040051000) for 15 min. For permeabilization, Triton X-100 (0.1% v/v; Merck; 11869) was used for 10 min at room temperature. Nonspecific antibody binding was blocked for 15 min at 4°C with combination of 10% heat-inactivated goat serum (made in house) in staining buffer. For each analysis, we used 1–5 × 105 cells per sample. Cells were incubated with primary antibodies [or the appropriate isotype matched controls (Table 1)] 30 min at 4°C. Then, cells were washed three times in PBS and for indirect staining [octamer binding transcription factor 4 (OCT4), NANOG, stage-specific embryonic antigen 4 (SSEA4), and TRA-1-60] cells were incubated with appropriate secondary antibody 30 min at 4°C. Flow cytometric analysis was performed with a BD-FACS Calibur Flow Cytometer (FACS Calibur, BD, Franklin Lakes, NJ, USA). The experiments were replicated at least three times. Acquired data were analyzed by using WinMDI 12.9 software (freeware from Joe Trotter, The Scripps Research Institute, La Jolla, CA, USA).
List of Antibodies Used in This Study for Flow Cytometry

OCT4, octamer binding transcription factor-4; CD45, cluster of differentiation 45; SSEA-4, stage specific embryonic antigen-4; FITC, fluorescein isothiocyanate; PE, phycoerythrin. Santa Cruz (Dallas, TX, USA); e-Bioscience (San Diego, CA, USA); R&D Systems (Minneapolis, MN, USA); BD Pharmingen (San Diego, CA, USA); Abcam (Cambridge, MA, USA); Sigma-Aldrich (St. Louis, MO, USA); Invitrogen (Carlsbad, CA, USA).
We seeded 104 cells per well in six-well plates (TPP, Trasadingen, Switzerland; 92006) to evaluate multipotent differentiation capability. To induce osteogenic differentiation, cells were cultured with MSC medium that also contained 1 μM dexamethasone, 0.5 μM ascorbic acid, and 10 mM β-glycerol phosphate (all from Sigma-Aldrich) for 21 days. For adipogenic induction, cells were cultured in MSC medium supplemented with 50 μg/ml indomethacin (Sigma-Aldrich), 50 μg/ml ascorbic acid, and 100 nM dexamethasone for 15 days. Osteogenesis was assessed by alizarin red (Sigma-Aldrich; A5533) and adipogenesis by oil red (Sigma-Aldrich, O0625) staining. For chondrogenesis differentiation, hiPSC-MSCs were centrifuged (Universal 320R; Hettich, Tuttlingen, Germany) in 0.2 ml of medium at 500×g for 10 min in 15-ml Falcon tubes (TPP; 91015) to form a pellet. The pellets were cultured for 21 days in MSC medium supplemented with 0.01 μM dexamethasone, 397 μg/ml ascorbic acid-2-phosphate (Sigma-Aldrich), 1 mM sodium pyruvate (Invitrogen), 10 ng/ml transforming growth factor (TGF-β1, Sigma-Aldrich), and 1% insulin-transferrin-selenium (Invitrogen).
Cell Culture and Maintenance of BM-MSCs
Human BM-MSCs was prepared from Royan Institute Stem Cell Bank and propagated in the common MSC medium. Bone marrow samples were obtained from two male patients, 49 and 50 years old. Informed consent was obtained from the original patient(s). The study was conducted with the approval of the institutional review board and ethical committee at Royan Institute.
Cell Labeling and Transplantation
hiPSC-MSCs at passage 5 were used for transplantation, because the primary cultures of MSCs without considering the source are mostly heterogeneous, and late passages have lost their capacity for self-renewal (14). Cells were enzymatically detached from plates by using 0.05% trypsin-EDTA, then labeled with a PKH67 Fluorescent Cell Linker Kit (Sigma Aldrich) according to the manufacturer's description and suspended in phosphate-buffered saline (PBS).
In Vivo Experiment Design (FHF Model and Grouping)
To find out the sublethal dose of carbon tetrachloride (CCl4; Merck; 1022231000) for the FHF experimental model, different doses (0.3–1.8 ml/kg) of CCl4 were administered to 8-week-old NMRI male mice (Pasteur Institute, Tehran, Iran) by gavage with the permission of the Royan Institute's ethical committee. CCl4 lethality was monitored for 6 days to establish a time window for cell transplantation. PKH-labeled hiPSC-MSCs (2 × 106) were transplanted intravenously via the tail vein 24 h post-CCl4 gavage. Five groups of 10 mice each were divided into normal, control, sham, hBM-MSC transplant, and hiPSC-MSC transplant groups. The normal group received no CCl4 gavage, and the control group received a single CCl4 gavage (0.6 ml/kg). Transplant groups received 2 × 106 cells just 24 h after a single CCl4 gavage while the sham group received only PBS. Examinations were performed on days 1, 3, 6, and 12 after cell transplantation. All animals in the control, sham, and transplanted groups received intraperitoneal injections of cyclosporine solution (sandimmune; Novartis Pharmaceuticals, Basel, Switzerland) at a dose of 20 mg/kg/24 h beginning 5 days prior to transplantation and continuing after transplantation. At the end of the procedure, animals were sacrificed under deep anesthesia and their blood serum collected. Additionally, liver samples were taken for histopathologic and lipid peroxidation analysis as well as cell engraftment, recipient hepatic cells proliferation, and engrafted cell secretion evaluations.
Serum Albumin (ALB), Alanine Transaminase (ALT), Aspartate Aminotransferase (AST) Lactate Dehydrogenase (LDH), Bilirubin, and Liver Lipid Peroxidation
Serologic analysis was performed using the following kits and according to the manufacturers' instructions. Serum albumin (ALB) levels were measured by the bromocresol green method using a commercial kit (Pars Azmun, Tehran, Iran). Serum LDH, ALT, and AST enzyme activities were assessed by the DGKC (Deutsche Gesellschaft für Klinische Chemie) method using a commercial kit (Pars Azmun). Serum bilirubin levels were also evaluated by the photometric DCA (dichloroani-line) method using a commercial kit (Pars Azmun). Lipid peroxidation was performed by thiobarbituric acid reactive substances (TBARS) assay kit (10009055; Cayman Chemical, Ann Arbor, MI, USA) based on the manufacturer's procedure. Briefly, 25 mg of liver tissue was sonicated to extract liver homogenate and then mixed with 0.2 ml of 8.1% sodium dodecyl sulphate, 1.5 ml of 20% acetic acid, and 1.5 ml of 0.8% TBA (Tris/Borate/EDTA), after which the volume was adjusted to 4.0 ml with distilled water. Liver tissue was boiled at 95°C for 60 min, and then the reaction solution was extracted with 1.0 ml of distilled water and 5.0 ml of n-butanol and pyridine (15:1 v/v). Absorbance was determined at 532 nm.
hiPSC-MSC Engraftment in Liver
Recipient liver tissues were fixed in 4% paraformaldehyde and then soaked in 30% sucrose (Merck; 1076871000) for 24 h at 4°C. Liver tissues were embedded in Tissue-Tek OCT™ compound (Sakura Finetek USA, Inc., Torrance, CA, USA), and 8-μm-thick sections were cut by cryostat (Leica, Wetzlar, Germany; CM1850) at −20°C. The sections were mounted on glass slides (Sail Brand, Jiangsu, China; 7107), and nuclei staining were performed using 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI, Sigma-Aldrich D-8417). Specimens were examined by fluorescent microscope (BX51, Olympus, Center Valley, PA, USA) and randomly picked fields of sections (30 individual fields of three independent samples) were captured from each animal, using a magnification of 20× objective lens. Quantification was estimated by the percentage of PKH+ cells (transplanted cells) in comparison to the total cells, which stained with DAPI in the fields.
Histopathology of the Liver
Liver tissues were processed per standard protocols for paraffin embedding and sectioned into 6-μm sections. The sections were stained with hematoxylin (Sigma-Aldrich; H3136) and eosin (Sigma-Aldrich; E4380) (H&E) and Masson's trichrome (MT) according to standard protocols using fast green (Sigma-Aldrich; F7258).
Hepatocyte necrosis or inflammation was also graded by the histologic activity index (HAI, Knodell score), which is characterized by three individual scores (15). The first component (perioportal and/or bridging necrosis) is scored 0–10. The next two components (intralobular degeneration and portal inflammation) are scored 0–4. The combination of these three markers indicates the amount of inflammation in the liver as follows: 0, no inflammation; 1–4, minimal inflammation; 5–8, mild inflammation; 9–12, moderate inflammation; and 13–18, marked inflammation.
Quantitative RT-PCR Analysis
Total RNA of the liver samples was isolated using the RNeasy Kit (Qiagen, Germantown, MD, USA; 74104). Two micrograms of total RNA was used for reverse transcription reaction with the RevertAid First Strand cDNA Synthesis Kit (Fermentas, Vilnius, Lithuania; K1621) and random hexamer primer (Fermentas), according to the manufacturer's instructions. Quantitative PCR reactions were run in duplicate with the Power SYBR Green Master Mix (Applied Biosystems, Foster City, USA; 4364344) and analyzed with the 7500 Real-Time PCR System (Applied Biosystems, 7500). We collected samples from at least three independent experiments. Expression values of mouse collagen type I, matrix metalloproteinase (Mmps) 2, 9, and 13, and tissue inhibitors of matrix metalloproteinase (Timps) 1 and 2 were normalized to the average expression of the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (Gapdh) by the comparative CT method (2-ΔΔct). The primer sequences are presented in Table 2.
List of Primers Used in This Study for Quantitative Real-Time Reverse Transcriptase-Polymerase Chain Reaction

Mmp13, matrix metalloproteinase 13; Timp1, tissue inhibitor of metalloproteinase 1; Gapdh, glyceraldehyde 3-phosphate dehydrogenase; PPARγ, Peroxisome proliferator-activated receptor γ; LPL, lipoprotein lipase; COL2, collagen type II; ACAN, aggrecan; BGLAP (OCN), bone gamma-carboxyglutamate protein (osteocalcin); ALP, alkaline phosphatase.
Expression values of hiPSC-MSCs for human peroxi-some proliferator-activated receptor γ (PPARγ), PPARα, lipoprotein lipase (LPL), collagen type II (COL2), aggrecan (ACAN), osteocalcin (OCN), and alkaline phosphatase (ALP) normalized to expression of GAPDH. The primer sequences are presented in Table 2.
Proliferation Analysis of Resident Hepatic Cells by Bromodeoxyuridine Incorporation
Bromodeoxyuridine (BrdU; Roche, Mannheim, Germany; 11296736001) dissolved in saline was intraperitoneally injected into mice 12 h after cell transplantation. BrdU was injected at 50 mg/kg body weight twice daily until the mice were killed. Liver tissues were cryosectioned at 4–5 μm. For immunohistochemical staining of BrdU, endogenous peroxidase activity was blocked with H2O2 for 10 min at 37°C and rinsed with PBS. DNA was denatured in 2N HCl for 30 min at 37°C, rinsed with PBS, and then enzymatically pretreated with 0.1% trypsin for 20 min at 37°C. Tissues were blocked for nonspecific binding by 5% goat serum in PBS and then incubated with anti-BrdU antibody (1:1,000; Sigma-Aldrich) for 2 h at 37°C, followed by biotinylated secondary antibody (1:1,000; Sigma-Aldrich) for 40 min at room temperature and with avidin/ horseradish peroxidase (HRP) conjugate for 30 min at room temperature. Positively stained cells were detected using 3,3′diaminobenzidine tetrahydrochloride substrate (Sigma-Aldrich). Hematoxylin was used as the counterstain.
Hepatocyte Growth Factor (HGF) Secretion and Expression by hiPSC-MSCs
For evaluation of HGF secretion by hiPSC-MSCs, cells were allowed to grow to 70–80% confluency [approximately 1 × 105 MSCs per 75-cm2 flask (TPP; 90075)]. Then the cells were washed twice with Ca2+- and Mg2+-free PBS and cultured in 10 ml DMEM supplemented with 0.05% bovine serum albumin (Sigma-Aldrich). After 24 h, conditioned medium was collected and concentrated 25-fold using ultrafiltration units (Millipore, Billerica, MA, USA; UFC800324). HGF were measured by ELISA kit (RayBiotech, Norcross, GA, USA; ELH-HGF-001) based on manufacturer instruction.
To assess expression of HGF by transplanted hiPSC-MSCs, a total of 6-μm sections of paraffin-embedded liver tissues were prepared for immunohistochemical staining. Endogenous peroxidase activity was blocked with H2O2 for 10 min at 37°C and rinsed with PBS. Tissues were blocked for nonspecific binding by 5% goat serum in PBS and then incubated with HGF primary antibody (1:1,000; R&D Systems; Minneapolis, MN, USA; AF-294-NA) and rinsed with PBS. HGF-secreting cells were labeled with HRP/DAB system (R&D Systems; CTS008) based on the manufacturer's procedure. Hematoxylin was used as the counterstain.
Statistical Analysis
Data have been presented as the mean ± SD with at least three biological independent repeats. One-way repeated measures analysis of variance (ANOVA) followed by the Tukey's post hoc test multiple group comparison was used to analyze group differences of the resultant data. The difference between groups was considered as statistically reliable if p ≤ 0.05.
Results
Generation and Characterization of hiPSC-MSCs
Here, MSCs were generated by isolation of outgrowing spontaneous differentiating cells from hiPSC colonies after 7 days. The differentiating cells were mechanically scrapped and cultured in MSC growth medium to obtain MSCs. After four passages, mesenchymal cells displayed spindle-shaped morphology with long extensions, which indicated an epithelial to mesenchymal transition over passages (Fig. 1).

Generation of mesenchymal progenitors from hiPSCs. (A) A human induced pluripotent stem cell (hiPSC) colony. (B) Spontaneously differentiated outgrowths are separated from nondifferentiated parts by dash points and indicated by *. (C) Higher magnification of (B). (D) hiPSC-derived mesenchymal stem cells (hiPSC-MSCs) at passages 1 (P1), 3, and 5. Note the morphologic epithelial–mesenchymal transition from (A) to (D).
Antigen profiling of hiPSC-MSCs indicated a loss in pluripotent markers (OCT4, NANOG, SSEA4, and TRA-1-60) in addition to the absence of hematopoietic surface markers [cluster of differentiation 34 (CD34) and CD45] and endothelial marker (CD31). However, these cells highly expressed MSC markers such as CD105, CD90, CD73, CD44, and CD29 (Fig. 2).

Immunophenotyping of mesenchymal progenitors from hiPSCs. Flow cytometry of the hiPSC-MSCs revealed negligible expression of the pluripotent stem cells markers, Octamer binding transcription factor 4 (OCT4), NANOG, stage-specific embryonic antigen 4 (SSEA4), and TRA-1-60, hematopoietic marker cluster of differentiation 45 (CD45) and CD34, and the endothelial cell marker CD31. The MSC markers CD90, CD105, CD73, CD44, and CD29 were detected. FITC, fluorescein isothiocyanate; PE, phycoerythrin.
hiPSC-MSCs were characterized by their ability to undergo adipogenic, chondrogenic, and osteogenic differentiation by expression of specific genes. The markers of adipocytes, PPARγ, PPARα, LPL, chondrocytes, COL2a, ACAN, and osteocytes, OCN, and ALP were upregulated (Fig. 3A). Adipogenic differentiation was also confirmed by the accumulation of intracellular lipid droplets (Fig. 3B) and validated by oil-red staining (Fig. 3C). Additionally, osteogenic differentiation yielded an extracellular precipitate, identified as calcium deposits by alizarin red staining (Fig. 3D).
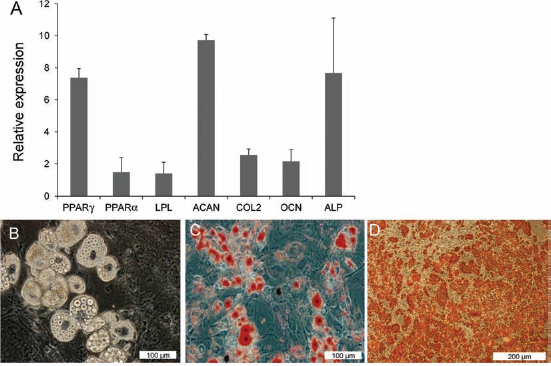
Differentiation potentials of mesenchymal progenitors from hiPSCs. hiPSC-MSCs were differentiated directly into adipocytes, chondrocytes, and osteoblasts. (A) Quantitative real-time RT-PCR for differentiation markers. (B) Adipogenic differentiation of hiPSC-MSCs as seen by phase contrast and (C) oil red-O staining. (D) Osteogenic differentiation of hiPSC-MSCs as indicated by alizarin red staining. PPARg, peroxisome proliferator-activated receptor g; PPARa, peroxisome proliferator-activated receptor a; LPL, lipoprotein lipase; ACAN, aggrecan; COL2, collagen type II; OCN, osteocalcin; ALP, alkaline phosphatase.
hiPSC-MSCs Rescue Lethal Fulminant Hepatic Failure
Mice were given CCl4, which is known to induce oxidative stress, hepatocyte necrosis, extensive vacuolar degeneration, and inflammatory cell infiltration in most zones of the parenchyma (41). H&E staining of liver sections confirmed serious acute liver damage (Fig. 4A).

Evaluation of transplantation of hiPSC-MSCs on the survival of fulminant hepatic failure (FHF) mice model. (A) H&E-stained liver sections in carbon tetrachloride (CCl4) and hiPSC-MSC transplanted groups 3 and 6 days posttransplantation, respectively. (B) The survival curve of CCl4-treated mice to determine sublethal doses for the acute liver failure model. All animals were treated with intraperitoneal injections of cyclosporine (20 mg/kg) starting 3 days prior to CCl4 administration to the end of the survey period. (C) Survival curve of CCl4-administered mice posttransplantation indicating that the cell transplanted groups were rescued, whereas nontransplanted groups died. Human foreskin fibroblasts (hFF) were transplanted as the cellular control group. Human bone marrow-derived mesenchymal stem cells (hBM-MSCs) were also transplanted for comparison.
The CCl4 lethality in NMRI mice was tested by CCl4 administration. We noted that doses of ≤0.3 ml/kg of CCl4 were not sufficiently lethal, while doses of ≥0.9 ml/ kg caused rapid death. The survival curve of CCl4 doses showed that all animals that received doses of 0.6 ml/ kg of CCl4 died after 6 days post-CCl4 administration (Fig. 4B). Therefore, this dose was determined to be the optimal dose for lethal FHF with an appropriate time window (6 days) for the transplantation of hiPSC-MSCs. Therefore, all subsequent experiments were performed at this dose. Transplantation of both hBM-MSCs and hiPSC-MSCs allowed for survival of 12 days posttransplantation compared to the control, sham, and human foreskin fibroblast (HFF) transplanted mice (Fig. 4C).
hiPSC-MSCs Improve the Biochemical Factors and Ameliorate Necrosis
Analyses of ALT, AST, and total bilirubin indicated highly increased levels in both control and sham groups at days 1 and 3 postinjury, while in the cell transplant groups they reduced significantly at day 3 compared to the control and sham groups (p ≤ 0.05) (Fig. 5A). LDH analysis also demonstrated significantly marked elevation of LDH on the first day posttransplantation. LDH levels gradually decreased to normal by day 12 in the cell-transplanted groups compared to the control and sham groups. There was a significant decrease between days 1 and 6 LDH levels and between days 1 and 3 and day 12 in both transplanted groups (p ≤ 0.05) (Fig. 5A). Lipid peroxidation analysis showed a significant dramatic elevated level at days 1 and 3 post-CCl4 administration in all groups compared to the normal group. However, this decreased significantly in the transplanted groups by days 6 and 12 (Fig. 5A). The changes in blood albumin levels in all groups were not significant compared to the normal group (data not shown).

Evaluation of blood serum, lipid peroxidation, and necrosis post-cell transplantation. (A) Blood serum alanine transaminase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), and total bilirubin increased significantly by day 1 in all groups, and decreased significantly over time toward normal levels in the cell transplanted groups. Lipid peroxidation analysis as seen by malondialdehyde (MDA) concentration in the liver samples decreased significantly in the cell-transplanted groups 6 and 12 days posttransplantation. Histologic activity index (HAI) indicated necrosis, which was initially elevated, and showed the decrease in moderate necrosis in the cell transplanted groups toward minimal necrosis. (B) H&E-stained liver sections were used to determine HAI on days 3, 6, and 12 after hiPSC-MSC transplantation. Arrow and arrow head show focal necrosis and hiPSC-MSCs homing, respectively. Data were presented as mean±SD. α, Significant difference with same group in other day(s); b, significant difference between groups in a day; *significant difference from the normal group.
To explore whether cell transplantation could ameliorate CCl4-induced liver necrosiswe compared liver sections in mice that underwent transplantation with either cell type, control and sham groups. The HAI as evaluated by H&E-stained sections (Fig. 5A, B) indicated significantly elevated levels of inflammation on days 1 and 3 posttransplantation in all groups. Inflammation was mild to moderate compared to normal (no inflammation). Moderate necrosis significantly decreased to the levels of minimal necrosis in both transplanted groups by days 6 and 12 posttransplantation (Fig. 5A). We observed transplanted cells in necrotic areas around the central veins. This indicated the affinity of transplanted cells to reconstruct a necrotic area and restore the primary liver architecture, thus regenerating necrotized regions of the liver.
There was no significant increase or decrease in fibrosis score and fibrotic areas in all groups at all days posttransplantation (data not shown).
hiPSC-MSCs Engrafted in Recipient Liver
To evaluate the engraftment and therapeutic properties of hiPSC-MSCs and hBM-MSCs in terms of liver regeneration, PKH-labeled cells were transplanted into mice with FHF. PKH-labeled hiPSC-MSCs were detected in recipient livers in all days posttransplantation (Fig. 6A), which revealed that transplanted cells could engraft in a recipient liver. A comparison of the absolute numbers of PKH+ cells in all groups within the recipient livers showed that after transplantation, 1.3 ± 0.4% (day 1) and 3.6 ± 1.0% (day 12) of transplanted hBM-MSCs and 1.9 ± 0.5% (day 1) and 2.6 ± 0.7% (day 12) of transplanted hiPSC-MSCs were detected (Fig. 6B). At day 6 posttransplantation, the maximum observed engrafted cells were 4.1 ± 0.9% hBM-MSCs and 4.0 ± 0.8% hiPSC-MSCs (Fig. 6B).

Tracing of PKH-labeled hiPSC-MSCs in recipient liver 12 days posttransplantation, Cells were labeled with PKH67 (green) prior to transplantation. Transplanted cells were detected in recipient liver posttransplantation by fluorescent microscopy (A). The ratio of PKH + cells to total cells (B). Nuclei were counterstained by DAPI (blue).
We could not find a double-positive PKH+ cell and immunofluorescence staining using anti-ALB antibody in liver sections. Additionally, there was no neoplasia due to cell transplantation detected in any of the injected animals during our study and also 4 weeks posttransplantation.
To explore whether cell transplantation influences the imbalance between endogenous fibrogenic/fibrolytic factors such as collagen, matrix metalloproteinases, and their tissue inhibitors, we evaluated the related genes by real-time RT-PCR (Fig. 7). Meanwhile expression analysis of collagen type I indicated significantly elevated levels of collagen in all groups on day 1. This significantly decreased from day 3 posttransplantation. We observed the same trend in Mmp13, Mmp9, and Mmp2 expression analyses while Timp1 and Timp2 indicated elevated levels of gene expression in the transplantation groups.
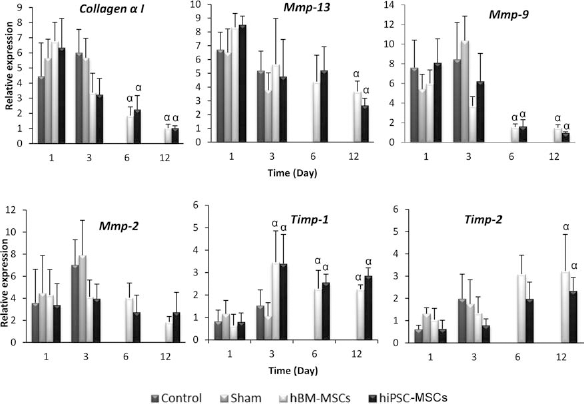
Temporal expression pattern of endogenous genes in liver posttransplantation by real-time RT-PCR. Data indicating that by day 12 posttransplantation, elevated expression of collagen a I, matrix metalloproteinase-13 (Mmp-13), and Mmp-9 decreased in the cell transplanted groups, whereas expression of tissue inhibitor of metalloproteinase 1 (Timp-1) was maintained and Timp-2 increased by day 12 posttransplantation. Data were calculated in relation to the glyceraldehyde 3-phosphate dehydrogenase (Gapdh) gene, which was used as an internal standard. a, Significant difference with same group on other day(s); *significant difference from normal group. Significant difference: p ≤ 0.05.
Transplantation of hiPSC-MSCs Enhanced Repopulation of Resident Hepatic Cells That Secreted HGF
We compared liver sections between the groups to investigate whether transplanted cells were capable of inducing endogenous hepatic cells to regenerate. Immunostaining for BrdU-labeled cells indicated significantly increased proliferating hepatic cells in cell transplanted groups on days 6 and 12 in contrast to the control and sham groups (Fig. 8A). The maximum amount of proliferating cells (3.4 ± 0.2%) were observed on day 12 post-hiPSC-MSC transplantation, which significantly increased compared to the normal and hiPSC-MSC group on days 1 and 3 posttransplantation (p ≤ 0.05) (Fig. 8B).

Evaluation of mitotic cells in recipient livers. (A) Liver sections stained with anti-bromodeoxyuridine (BrdU) indicating proliferation (brown stained nuclei) in resident endogenous hepatic cells for control (day 3), sham (day 3), hBM-MSCs (day 12), and hiPSC-MSCs (day 12) groups posttransplantation. (B) Quantification of mitotic cells in the recipient livers indicating an increased number of proliferating cells on days 6 and 12 in the cell transplanted groups posttransplantation. Thirty fields of three independent samples counted per group at 20× magnification. a, Significant difference with same group in other day(s); *significant difference with normal group. Significant difference: p ≤ 0.05.
To find the possible role of hiPSC-MSCs in ameliorating liver injury by possible hepatotrophic factor secretion, we examined HGF secretion by these cells in vitro and its expression in vivo. HGF was detected similarly in conditioned media of both hiPSC-MSCs and hBM-MSCs (Fig. 9A). Furthermore, we detected the presence of transplanted hiPSC-MSCs within the recipient liver that have the ability to express human HGF (Fig. 9B).
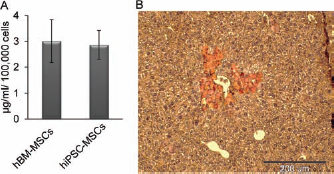
Assessment of secretion and expression of human HGF by hiPSC-MSCs in vitro and in vivo. (A) Detection of hepatocyte growth factor (HGF) secretion in conditioned medium of hiPSC-MSCs and hBM-MSCs using ELISA. (B) Expression of human HGF in transplanted hiPSC-MSCs (brown cells) in liver sections 12 days posttransplantation as stained by immunohistochemistry.
Discussion
The first report in cell-based therapy in mouse model of liver failure was published over a decade ago by bone marrow transplantation (27). This has triggered a progressive and challenging field to locate the most desirable cell source as an alternative for liver transplantation. Several investigations have determined that MSCs are a promising source for liver cell-based transplantation therapies.
Our data indicate that MSCs derived from hiPSCs have several similar characteristics and regenerative properties when compared with hBM-MSCs. They could also rescue the FHF mice model. Based on our knowledge, this is the first report that has investigated the regenerative potential of hiPSC-MSCs in an acute liver failure model. According to our findings, mechanical isolation of spontaneously differentiated cells in hiPSCs colonies and passaging them in MSC medium makes a readily accessible in vitro cell source with the characteristics of all MSCs. hiPSC-MSCs revealed the same morphology, surface markers profile, and osteo-chondro-adipogenic differentiation capability as hBM-MSCs and hESC-derived mesenchymal-like cells. These results support previous studies on hESC-MSCs (4, 10, 12, 19, 21, 23, 34, 38, 44, 47) and hiPSC-MSCs (22).
Since the derivation of hiPSCs, researchers have desired to utilize them in clinical applications; however, viral integration in hiPSCs has made this goal hard to attain. Derivation of MSCs from transgene-free hiPSCs could be an impressive step toward preclinical and clinical patient-specific cell therapies, particularly among elderly individuals who lack stem cell resources (30) and those patients in need of several bone marrow aspiration surgeries.
Currently, clinically applicable hiPSCs do not exist, but are under development. Several key issues aimed at overcoming safety concerns have been achieved for the production of hiPSCs [for reviews, see Philonenko et al. (28) and Plath and Lowry (29)]. These include the use of nonintegrating viruses (such as adenoviruses or episomal plasmid transfection), treatment of cells with cell-penetrating recombinant reprogramming factor proteins, transposon-based systems, plasmid delivery, repeated transfection of modified mRNA encoding reprogramming factors, and miRNAs.
Another essential prerequisite for future widespread application of hiPSC-derived cells is the urgent consideration required disease such as FHF. The banking of hiPSC-MSCs from different HLA individuals could overcome the allograft transplantations (28).
This study also assessed the therapeutic effects of hiPSC-MSCs in a CCl4-induced acute liver failure mice model at days 1, 3, 6, and 12 post-cell transplantation. In comparison with the control and sham groups which died before day 6, cell transplanted groups survived until the end of the study and exhibited reduced levels of ALT, AST, LDH, total bilirubin, and lipid peroxidation as well as better manifestations in HAI. The significant difference in ALT, AST, LDH, bilirubin, and lipid peroxidation [malondialdehyde (MDA)] concentrations seen at days 1 and 3 posttransplantation was due to CCl4 toxicity. This toxicity has been shown to increase lipid peroxidation in the hepatocyte plasma membrane by destroying it, causing the release of cytoplasmic enzymes, ALT, AST, and LDH into the blood (26, 31). On the other hand, CCl4 inhibits reactivity of enzymes related to uptake and conjugation of bilirubin causing elevated levels of this product in the blood (42). Significant decreases in these three markers in the transplanted groups on days 6 and 12 was possibly due to the recovery potential of hiPSC-MSCs and hBM-MSCs by the reestablishment of an altered liver architecture, stopping more cell death, which was detectable. Several PKH+ cells (1–4%) showed survival and integration of engrafted hiPSC-MSCs into the host tissue 12 days following transplantation as well as assistance with recovering liver functions. Cell transplantation significantly decreased collagen type I elevated gene expression level after 6 days posttransplantation. We detected the same results for Mmp9; significant suppression of MmpP13 also occurred 12 days posttransplantation. Surprisingly, there were increased expressions in Timp1 and Timp2 which was strongly supportive of previously described mechanisms involved in FHF recovery. Matrix metalloproteinases play a crucial role in FHF lethality (45). Increased levels of Mmp9 and Mmp2 can cause the progression of FHF from precoma to coma stage and finally death (45). However, FHF models treated with MMP inhibitors such as BB-94 or mice deficient in Mmp2, Mmp3, and Mmp9 showed lower levels of necrosis and apoptosis, and better survival (43). Our results have suggested the possible ameliorating effects of hiPSC-MSCs transplantation in suppression of Mmps as well as increased expression of Timps that could increase the window of time in the FHF model for spontaneous recovery and ultimately survival.
HAI scoring of necrosis in different days revealed significant decrease in the cell transplantation groups on days 6 and 12 posttransplantation, which may be caused by suppression of Mmps, expression of Timps (43), and increased repopulation of endogenous hepatic cells (16). Investigations over hepatic repopulation indicated significantly increased mitotic activity in the transplanted groups on days 6 and 12 posttransplantation compared to both normal and the transplanted groups on days 1 and 3. There was a negative correlation between HAI and repopulation of resident hepatic cells that revealed which necrotic areas within the liver were occupied by transplanted stem cells and newly dividing hepatic cells that could possibly rearrange the hepatic structure and remove necrotic areas.
A question raised is whether treatment of liver injury with hiPSC-MSCs is mediated through in situ differentiation of the donor cells or facilitated via paracrine effects. We could not detect ALB-positive transplanted cells during our study. These findings have suggested a possible hepatotrophic factor from hiPSC-MSCs that could defeat FHF lethality, necrosis and oxidative stress caused by CCl4. There have been several investigations regarding stem cell secretions that possibly support hepatocyte maintenance [see review by Gomez-Aristizabal et al. (7)]. One of the most popular stem cell secreting growth factors that could promote hepatic cell regeneration in liver injuries is HGF that was detectable in our investigation both in vitro and in vivo. It has been indicated that stem cells transfected with lentiviral vectors expressing HGF could significantly promote liver functions and also recover hepatic fibrosis in vivo (11). We have shown that HGF secreting hiPSC-MSCs could repopulate liver resident cells and also attenuate liver inflammation due to antinecrotic effects over stressed hepatocytes (20).
More recently, it was demonstrated that intrahepatic delivery of MSC-conditioned medium (CM) could induce mouse isolated oval cell proliferation in vitro and improve liver function in an acute hepatic failure (AHF) mouse model (48). Proteome analysis of CM derived from MSCs isolated and expanded from second trimester amniotic fluid (AF-MSCs) indicated the presence of anti-inflammatory factors such as interleukin 10 (IL-10), interleukin 1 receptor antagonist (IL-1ra), IL-13, and IL-27, all which may induce liver recovery. Inhibition of IL-10 secretion from the cells did not cause an improvement in the AHF mouse model 24 h after cell transplantation. Furthermore, exogenous injection of rhIL-10 led to liver repair (48). Additionally, it was demonstrated that MSC-CM protected the liver from excessive damage and reduced mortality associated with a rat model of d-galactosamine-induced FHF by an anti-inflammatory effect; thus, it inhibited hepatocellular death and immune cell infiltration and stimulated regeneration both in vitro and in vivo (26, 39). From our findings, it is clear that para-crine effects and secreted molecules from donor cells may be effective for the rapid improvement in the liver rather than the cell differentiation process at the site of injury, as described previously (3). Systematic proteomic analysis combined with fractionation studies of CM derived from hiPSC-MSCs is necessary to identify key therapeutic components. However, in our case it remains to determine whether CM from hiPSC-MSCs improves hepatic failure phenotype. Although hiPSC-MSCs hepatic functional integration indicated their authentic ameliorative impact in AHF mice model, a therapeutic milestone still remains to show their tumorogenic-free nature, which significantly dampens enthusiasm over their clinical usage.
In conclusion, for the first time, this study has shown the therapeutic effect of hiPSC-MSCs in the mouse AHF model. Patient-specific hiPSC-MSCs can be produced as an “off-the-shelf” format (hiPSC-MSCs banked from different HLAs) for the treatment of FHF. Future investigations for safe hiPSC-MSCs generation will bring impressive insights in pluripotent stem cell applications in clinical trials as patient-specific sources for all future regenerative medicine therapies in which MSCs could be beneficial. However, several worldwide researches with term follow-ups of the in vivo safety and efficacy must be performed before their clinical application in regenerative medicine.
Footnotes
Acknowledgments
This study was funded by grants provided from Royan Institute and Iranian Council of Stem Cell Research and Technology. We also thank Mostafa Najjar-Asl, Azam Samadian, and Mozhdeh Sojoudi for their technical assistance. The authors declare no conflicts of interest.
