Abstract
Ischemic stroke results in violent impairment of tissue homeostasis leading to severe perturbation within the neurovascular unit (NVU) during the recovery period. The aim of this study was to assess the potential of mesenchymal stem cells (MSCs) originating from Wharton's jelly (WJ) to differentiate into functionally competent cells of endothelial lineage (WJ-EPCs). The protective effect(s) of either primary WJ-MSCs or induced WJ-EPCs was investigated and compared after oxygen–glucose deprivation (OGD) of hippocampal organotypic slices (OHC) in the indirect coculture model. WJ-MSCs, primed in EGM-2 (Lonza commercial medium) under 5% O2, acquired cobblestone endothelial-like morphology, formed capillary-like structures and actively took up DiI-Ac-LDL. Both cell types (WJ-MSCs and WJ-EPCs) were positive for CD73, CD90, CD105, VEGFR-2, and VEGF, but only endothelial-like culture expressed vWF and PECAM-1 markers at significant levels. In the presence of either WJ-MSCs or WJ-EPCs in the compartment below OGD-injured slices, cell death and vascular atrophy in the hypoxia-sensitive CA1 region were substantially decreased. This suggests that a paracrine mechanism may mediate WJ-MSC- and WJ-EPC-dependent protection. Thus, finally, we estimated secretion of the neuro/angio/immunomodulatory molecules IL-6, TGF-β1, and VEGF by these cell cultures. We have found that release of TGF-β1 and IL-6 was TLR ligand [LPS and Poly(I:C)] concentration dependent and stronger in WJ-EPC than WJ-MSC cultures. Simultaneously, the uneven pattern of TLR receptors and modulatory cytokine gene expression was confirmed also on qRT-PCR level, but no significant differences were noticed between WJ-EPC and primary WJ-MSC cultures.
Keywords
Introduction
Growing data on complex interactions between resident brain cells, neurons, glia, and endothelial cells, led to the concept of the neurovascular unit (NVU) as an integrated functional entity, the dysfunction of which may be responsible for a variety of brain diseases and influence the effectiveness of their restorative treatment (16). The focus on disturbed functional interactions and need of complex and simultaneous targeting of all the cells involved in NVU may lead to overcoming permanent failure of the majority of recently proposed, single-target medical strategies (14,21). Broadening reports on beneficial effects of mesenchymal stem cells (MSCs) for prevention or even reversion of numerous pathologies (8,28) by use of multifactorial regenerative mechanisms may indicate them as preferred candidates for “holistic” tissue therapy. Selective vulnerability of the hippocampal CA1 neurons to hypoxia/ischemia is a well-known, but still not fully understood, phenomenon. Years ago, it was speculated that specific characteristics of the local vasculature might be responsible for selective CA1 region sensitivity to ischemia (20). However, this idea was later challenged with the observation that delayed death of CA1 neurons could be observed not only in vivo but also in the hippocampal slice culture under an oxygen–glucose deprivation (OGD) model in vitro where the blood flow is totally absent (7,13). Nowadays, we have begun to understand that the integrity of cerebral tissue relays on the existence and stability of the combined interactions of endothelial cells with surrounding nerve cells, glia, and pericytes that support each other in the NVU. A number of in vitro studies have shown that coculturing these cells together may induce typical brain barrier proteins and stimulate secretion of factors essential for maintenance of vascular structures (1,3,4,4,34). Recently, it was reported that CA1 neuronal death in hippocampal slices is followed by selective disintegration of the vascular system in the same region (5,13).
In the present work, to expound upon this original observation, we tested if treatment of the rat OGD-injured hippocampal slices by either Wharton's jelly mesenchymal stem cells (WJ-MSCs) or their epithelial progenitor cell-like (WJ-EPCs) derivatives obtained by culturing WJ-MSCs in specific inductive media can protect neurons and vasculature in the hippocampal CA1 region. Embryonic vasculogenesis was primarily thought to be solely responsible for formation and repair of all vessels during the entire life span by sprouting of the preexisting endothelial cells. However, later discoveries that bone marrow-derived EPCs are released into blood circulation (3) caused attention to shift to the de novo angiogenesis observed under repair processes. The active migration of circulating EPCs toward regions of injury provide new cells able to integrate and then repair damaged vasculature or supply parenchyma with growth and trophic factors that, in turn, stimulate tissue regeneration (2). These new findings have opened the door to therapeutic delivery of cultured EPCs, which may offer a cure to the victims of various ischemia-linked pathologies (15,22,27,4). A possible interaction between vascular atrophy, CA1 neuronal degeneration, and possible protection by WJ-MSCs or induced EPCs due to secretion of trophic [vascular endothelial growth factor (VEGF)] and immunomodulatory [interleukin-6 (IL-6) or transforming growth factor-β (TGF-β1)] factors has been studied in vitro in the OGD model of hippocampal injury. According to the literature, MSCs under inflammatory conditions can modulate or even change their resting immunosuppressive and tolerance-promoting function (30), and human MSCs mainly express toll-like receptor 3 (TLR3) and TLR4. These receptors are believed to be hallmarks of innate immunity, and in an inflammatory environment, they may modulate immunogenic potential of MSCs (17,18). Thus, here we estimated the ability of WJ-MSCs and WJ-EPC-like cells to respond to specific stimulation of TLR3/4 by their agonists, polyinosinic/polycytidylic acid [poly(I:C)] and lipopolysaccharide (LPS), by changing related cytokine (IL-6, TGF-β1, VEGF) secretion patterns. During this study, we were trying to elucidate the following: 1) if human WJ-MSCs induced in vitro to endothelial-like phenotype can be qualified as the functional EPCs according to the commonly accepted criteria; 2) if these induced WJ-EPCs differ from the naive WJ-MSCs in their ability to protect the CA1 hippocampal region after OGD injury; 3) to correlate MSC-linked protection with changes in expression and/or secretion of immuno- and vasoactive cytokines (IL-6, TGF-β1, and VEGF) as well as with expression of innate immunity and endothelial plasticity of hallmark genes.
Materials and Methods
WJ-MSC Isolation and Endothelial Induction
All study protocols were approved by the Ethics Committee of Medical University of Warsaw. Informed consent was obtained from females with full-term pregnancy. Samples of human umbilical cords were collected and processed for MSC isolation using mechanical fragmentation techniques. Umbilical cords (15 cm long) were briefly immersed in sterile phosphate-buffered saline (PBS; Gibco, Warsaw, Poland) supplemented with penicillin streptomycin (1:100; Gibco) and then immediately transferred to the laboratory. The umbilical cords were cut with a sharp, sterile blade into 2- to 3-mm pieces. Using a biopsy punch (Miltex, GmbH, Ratingen, Germany), small cylindrical fragments (2 mm diameter) were removed from the mucous connective tissue (Wharton's jelly matrix), avoiding blood vessels and amniotic epithelium, and transferred to culture dishes. Wharton's jelly fragments were incubated in human MSC growth medium (MSCGM BulletKit; Lonza, Basel, Switzerland) at 37°C in a humidified incubator under a 21% O2 and 5% CO2 atmosphere. After 7–10 days of culture, the first colonies of WJ-MSCs were observed. Then the cells were pursued until subconfluence; nonadherent cells were removed, and stromal cells were detached (0.05% trypsin-EDTA; Gibco), then transferred into 25-cm2 flasks at an initial density of 5 ° 103/cm2 and cultured up to 70–80% confluence before collection for subsequent passages (8). WJ-MSCs cultured for 7 days in EGM-2 medium (Lonza) containing fibroblast growth factor 2 (FGF2), VEGF, heparin, insulin-like growth factor-1 (IGF-1), epidermal growth factor (EGF), hydrocortisone, and ascorbic acid were differentiated toward endothelial precursors.
Organotypic Hippocampal Culture (OHC)
Organotypic slice culture was obtained according to the Stoppini method but was modified and described by our lab (26). Male and female 1-week-old Wistar rat pups were provided by Mossakowski Medical Research Centre Animal Breeding House. At the time of sacrifice, pups were cooled on ice and decapitated. Brains were extracted, and rat hippocampi were isolated, sectioned into 400-μm slices using a McIlwain tissue chopper (Ted Pella, Poznan, Poland), and transferred to Millicell CM (Millipore, Warsaw, Poland) membranes placed in six-well plates (Nunc; Thermo Fisher Scientific, Warsaw, Poland). The culture medium was initially composed of DMEM (50%; Gibco), horse serum (25%; Sigma-Aldrich, Poznan, Poland), HBSS (25%; Gibco), as well as HEPES (Gibco), 5 mg/ml glucose (Sigma-Aldrich), 2 mmol/L L-glutamine (Gibco), and antibacterial–antimycotic solution (1% amphotericin B and 0.4% penicillin–streptomycin; Gibco). In the following 4 days, the serum content in the culture medium was gradually lowered, and from the fifth day in vitro (DIV), the hippocampal slices were cultured in serum-free medium containing neurobasal medium (75%; Gibco), nutritional N2 (1:10; Gibco), B27 (1:100; Gibco), and HBSS, HEPES, glucose, L-glutamine, and antibacterial–antimycotic solution (as mentioned before). Then slices were used for the OGD procedure and coculture experiments.
Oxygen–Glucose Deprivation in OHC Slices in a Transwell Coculture Model System
For the OGD procedure, the membranes with hippocampal slices were transferred to an anaerobic chamber immersed inserted in Krebs–Ringer solution (Sigma-Aldrich) supplemented with 10 mM mannitol (as the glucose substitution; Sigma-Aldrich) and saturated with 95% N2 and 5% CO2. The cultures were kept in an oxygen-free atmosphere (95% N2/5% CO2) for 40 min in order to mimic an ischemic injury. Simultaneously, the control slices were maintained in glucose-containing Krebs–Ringer solution in normoxic conditions. OGD-triggered cell death or vessel damage was analyzed 24 h after injury. To assess cell death in OHC, the fluorescent marker propidium iodide (PI; Sigma-Aldrich) was used. Twenty-four hours after injury, the culture medium was supplemented with PI for 1 h, and PI-labeled cells were counted using a confocal microscope (LSM 510; Carl Zeiss, Warsaw, Poland) equipped with ZEN 2008 software (Carl Zeiss). To evaluate MSC-induced protection, hippocampal slices were cocultured for 24 h with WJ-MSCs or WJ-EPCs (at 80% confluency) in serum-free medium containing DMEM (50%), HBSS (25%), HEPES, 5 mg/ml glucose, 2 mmol/L L-glutamine, and antibacterial–antimycotic solution (1% amphotericin B and 0.4% penicillin– streptomycin). The slices were then fixed with 4% paraformaldehyde (PFA; Carl Roth, Warsaw, Poland) for 30 min at room temperature (RT) and rinsed with PBS. Hippocampal slices were carefully transferred from the culture inserts for antibody labeling of free-floating sections including a 24-well dish with 10% goat serum (GS; Gibco) and 0.2% Triton X-100 (Sigma-Aldrich) in PBS. Slices were incubated in the blocking buffer for 1 h at RT. Fixed slices were labeled overnight at 4°C with primary antibody against rat endothelial cells RECA-1 (Abcam, Straszyn, Poland). Hippocampal slices were rinsed three times with PBS and incubated for 1 h at RT with Alexa 546 fluorochrome (Invitrogen, Thermo Fisher Scientific, Warsaw, Poland) in PBS. Hippocampal cell nuclei were stained with Hoechst 33258 (Sigma-Aldrich). Stained slices were viewed by the confocal laser scanning microscope (LSM 780; Carl Zeiss) and analyzed by ZEN 2012 software (Carl Zeiss). The vessel density in CA1 and CA2/3 hippocampal regions was quantified in minimum five slices, based on fluorescence intensity, and was expressed as percentage of the area of blood vessels per selected region.
Immunocytochemistry
For immunofluorescence staining, the cell culture was fixed in 4% PFA and 0.2% Triton X-100 in PBS for 15 min at RT. Nonspecific reactions were blocked with 10% GS in PBS for 60 min at RT. To examine the expression of endothelial or mesenchymal markers primary antibodies (Table 1) against vascular endothelial growth factor receptor-2 (VEGFR-2, 1:300; Santa Cruz Biotechnology, Lodz, Poland), platelet/endothelial cell adhesion molecule 1 (PECAM-1, 1:100; Santa Cruz Biotechnology), von Willebrand factor (vWF, 1:400; Abcam), vascular cell adhesion molecule 1 (VCAM-1, 1:100; Santa Cruz Biotechnology), and rat endothelial cell antigen-1 (RECA-1, 1:25; Abcam) were added, with primary antibody replaced by PBS in the negative control group. After overnight incubation, a secondary antibody (Alexa Fluor 488/546, 1:500; Invitrogen, Thermo Fisher Scientific) was added for 1 h at RT. Cell nuclei were counterstained with Hoechst 33258 (1:150). Finally, labeled cells were analyzed under a confocal laser scanning (LSM 510) microscope.
Primary Antibodies Used in Immunocytochemistry

FACS Analysis of Cell Surface Markers
WJ-MSCs and WJ-EPCs were characterized using anti-human fluorochrome-conjugated antibodies against CD90, CD73, CD105, CD34, CD11b, CD19, CD45, and HLA-DR (Human MSC Analysis Kit; BD Biosciences, Warsaw, Poland) and analyzed by flow cytometry (FACSCalibur II) and Diva software (BD Biosciences). Corresponding isotype antibody controls (mIgG1, κ FITC; mIgG1, κ PerCP-Cy5.5; mIgG1, κ APC; mIgG1, κ PE; mIgG2a, κ PE) and unstained samples were analyzed in parallel as standards. Cells were digested with Accutase Cell Detachment Solution (BD Biosciences), washed in PBS, and a single cell suspension in cold BD Pharmingen Stain Buffer (BD Biosciences) with 1 ° 106 cells/ml was made. One hundred microliters of the cell suspension was added to each Eppendorf tube. One tube was kept as negative control, and each of the following antibodies were added according to the manufacturer's protocol and incubated for 30 min in the dark at room temperature. The suspended cells were washed twice in stain buffer and then analyzed with a FACSCalibur II fluorescence-activated cell sorter (BD Biosciences). For each sample, 10,000 events were acquired and analyzed with FACSDiva software. The results were expressed as the percentage of cells that were simultaneously positive for mesenchymal markers and negative for hematopoietic markers.
Cell Proliferation Analysis
The cell proliferation rate was estimated by analyzing the metabolic activity of living cells with WST-1 {(4-[3-(4-lodophentyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate} reagent (Roche, Warsaw, Poland). The cells were seeded on 96-well tissue culture plates at an initial density of 1.5 ° 103 cells/cm2 for WJ-MSCs and 2.5 ° 102 cells/cm2 for WJ-EPCs and measured each day for 1 week. For detection, 10 μl of WST-1 solution was added to each sample and incubated, avoiding light, in an incubator at 37°C for 2 h. The optical absorbance of each well was measured using an Omega Plate Reader (BMG LABTECH, Jozefow, Poland) at 420 nm and then recorded. The curve of cell growth was plotted with time as the x-axis and cell number as the y-axis. All experiments were repeated three times in duplicate.
Cumulative population doublings (CPDs), which refer to the total number of population doublings (PDN) of the cells since their primary isolation in vitro, and population doubling time (PDT), which stands for the time interval required for population doubling, were calculated as described previously (8). Briefly, when cell density reached subconfluence, cells were reseeded at an initial density of 2,000 cells/cm2. PDT was calculated based on the total cell number at each passage using the formula (t - t0) ° log 2/(log N - log N0), PDs = log(N0/Ni)/log 2; CPDs = PDs1 + PDs2 + PDs3 + . . . + PDsn; where t - t0 is the culture time (days), N is the number of harvested cells, and N0 is the initial number of cells.
Matrigel Tube Formation Assay
Analysis of tube formation was performed using extracellular matrix Matrigel (Corning, Warsaw, Poland) according to the manufacturer's instructions. WJ-MSCs cultured for 7 days in EGM-2 medium were harvested with trypsin (Gibco) and cultured at an initial density of 50,000 cells/cm2 for 24 h in 24-well plates precoated with 150 μl of Matrigel. Formation of capillary-like structures was analyzed using a phase-contrast microscope (Axio Vert 40C; Carl Zeiss).
Acetylated Low-Density Lipoprotein (Ac-LDL) Uptake Assay
WJ-MSCs cultured for 7 days in EGM-2 medium were incubated with 10 μg/ml of acetylated low-density lipoprotein labeled with 1,1'-dioctadecyl-3,3,3’,3'-tetramethylindo-carbocyanine perchlorate (DiI-Ac-LDL; Molecular Probes, Thermo Fisher Scientific, Warsaw, Poland) at 37°C for 4 h. Then the cells were fixed with 4% PFA, stained with Hoechst 33258, and observed under a fluorescent microscope (Axio Vert.A1; Carl Zeiss).
MSC Secretome After Toll-Like Receptor Stimulation
The WJ-MSC and WJ-EPC cultures were grown in separate wells in corresponding MSCGM or EGM-2 media as described above. For the experiment, these media were changed to neurobasal medium, and cells were stimulated by either the TLR3 agonist poly(I:C) (1 μg/ml; 10 μg/ml) (Tocris, Abingdon, UK) or the TLR4 agonist LPS (10 ng/ml; 50 ng/ml) (InvivoGen, Warsaw, Poland) as recommended by Waterman et al. (31). All cultures were then incubated for 24 h, and media were collected for further analysis.
The concentrations of IL-6, TGF-β1, and VEGF in the media were measured with BD Cytometric Bead Array Flex Sets (BD Biosciences) according to the manufacturer's protocol. Analysis was performed with FACSCalibur cytometer II and FCAP Array software (BD Biosciences).
RT-PCR and Real-Time Quantitative qRT-PCR Analysis
Total mRNA was isolated from 5 ° 105 WJ-MSCs and WJ-EPCs using TRIzol reagent (Invitrogen, Thermo Fisher Scientific). Genomic DNA contamination was eliminated by DNase digestion (DNA-freeTM DNA Removal Kit; Ambion, Thermo Fisher Scientific, Warsaw, Poland) according to the manufacturer's protocol. The RNA purity was then evaluated by reading absorbance in a NanoDrop ND-1000 spectrophotometer and a ratio from 260:280 and 260:230 was determined, for noncontaminated samples, an assumed 260:230 ratio greater than 1.8. RNA (1 μg) was used for cDNA preparations using the High Capacity RNA-to-cDNA Kit (Applied Biosystems, Thermo Fisher Scientific, Warsaw, Poland). RT-PCR was performed according to the Taq PCR Core Kit (Qiagen, Warsaw, Poland) using primers listed in Table 2. PCR products were separated by electrophoresis on 1.5% agarose in 0.5% Tris/Borate/EDTA (TBE; Invitrogen, Thermo Fisher Scientific) buffer and visualized by UltraSafe blue (Syngene, Cambridge, UK) staining in ultraviolet light. PCR product images were acquired with a Gel Expert 4.0.
Primers for Quantitative Reverse Transcription-Polymerase Chair Reaction (qRT-PCR and RT-PCR)

For quantitative RT-PCR, the 7500 Real Time PCR System (Applied Biosystems, Thermo Fisher Scientific) was used. The final reaction contained template cDNA, SYBR Green PCR Master Mix (Thermo Fisher Scientific), and gene-specific primers (Table 2). ACTB (β-actin) was used as an internal control. The cycle threshold values of reference and other specific genes were acquired after PCR. The normalized fold expression was obtained using the 2-ΔΔCt method. The results were expressed as the normalized fold expression for each gene.
Statistical Analysis
Statistical analysis of the raw data was conducted using the GraphPad Prism version 5 software (La Jolla, CA, USA). The mean ± SEM was calculated for all samples, and significance was determined using Student's t-test or ANOVA followed by Bonferroni post hoc test. The values were considered as significant with a value of p < 0.05. All of the animal experiments carried out complied with EU guidelines for the use of animals in research and were approved by the Fourth Warsaw Local Ethics Committee. Every possible effort was made to minimize animal suffering and the number of animals used.
Results
The Effect of Angiogenic Induction on Cell Morphology and Proliferative Potential
Umbilical cord punctured minislices in culture contain a heterogeneous population of mesenchymal cells, where the majority of them, due to their high migratory ability, grow as a fibroblast-like, spindle-shaped cell population (WJ-MSCs). WJ-MSCs reached 90% confluence at 7–10 DIV in the appropriate media (Lonza). After 2 weeks in culture, WJ-MSCs were induced to differentiate into endothelial cells in EGM-2 medium as described in Materials and Methods. EPC-like cells were then characterized phenotypically and compared with the initial, noninduced WJ-MSC population obtained according to methods described in our previous work (8). Over the course of a few days, primed cells changed their morphology to a cobblestone-like phenotype typical for endothelial progenitors and became three times smaller compared to the initial WJ-MSC population (Fig. 1A). Surprisingly, this typical EPC-like morphology, described also by other authors (6,23), did not influence the expression of the MSC-specific surface markers as measured by a commercial FACS-based test (Fig. 1C). A sustained absence of expression of CD34 hematopoietic lineage markers, classically reported in EPCs, was noticed in our EPC-like cultures.

Phenotypic characteristics of mesenchymal stem cells originating from Wharton's jelly (WJ-MSCs) and differentiated into cells of endothelial lineage (WJ-EPCs). (A) Phase contrast imaging of noninduced WJ-MSCs (left) and primed WJ-EPCs (right). (B) PDT and cumulative population doublings of WJ-MSCs (turquoise) and WJ-EPCs (blue). The results are presented as mean values of seven experiments ± SEM, ***p < 0.001. (C) Noninduced and induced WJ-MSCs expressed CD90, CD73, CD105, and showed no expression of CD11b, CD19, CD45, HLA-DR, and CD34 markers, measured as fluorescence intensity (MFI). The positively stained cells are expressed as a percentage in the middle of the frames. The blue histograms depict tested specimens, and gray histograms represent isotype controls. Analyses were carried out with FACScalibur II flow cytometer (BD Biosciences). (D, E) Both cell types were positive for VEGFR-2, VEGF-A, VCAM-1, but only WJ-EPCs expressed specific endothelial cell markers vWF and PECAM-1 in vitro. (F) WJ-EPCs seeded on Matrigel in EGM-2 created network formation. Pictures were taken at different time points: 2 h (left image) and 7 h (right image). (G) Endothelial-like differentiated cells showed uptake of DiI-Ac-LDL with varying intensity. Scale bars: 50 μm.
Differentiation toward an endothelial-like phenotype had a noticeable effect on the growth rate and proliferation potential of WJ-MSCs, as indicated by the higher number of cumulative population doublings observed after defined cell growth period. For noninduced WJ-MSCs, it was 4.8 doubling time after 4 days and 8.5 doubling time after 7 days, whereas for WJ-EPC, it was 15.8 and 37.7 doubling time after 4 and 7 days, respectively. Also, an average PDT in the second passage was four times shorter for WJ-EPCs (0.79 ± 0.2 days) than for WJ-MSCs (4 ± 1.7 days) (Fig. 1B).
Comparison of Mesenchymal and Endothelial Marker Expression in WJ-MSCs Versus WJ-EPC-Like Cultures
Flow cytometry (FACS) and molecular PCR analyses confirmed that both cell populations (WJ-MSCs and WJ-EPCs) were positive for expression of antigens typical for MSCs (CD73, CD90, CD105) but negative for hematopoietic lineage markers like CD34, CD45, CD11b, CD19, and HLA-DR, in agreement with International Society for Cellular Therapy (ISCT) established minimal criteria for MSC culture characteristics. Both populations also expressed VEGF-A and its receptor VEGFR-2 as well as VCAM-1 (Fig. 1C, D). However, after 7 days of culturing in EGM-2 medium, the WJ-EPC-like cells expressed genes and proteins typical for the endothelial progenitor population such as PECAM-1 and vWF (Fig. 1D, E).
Functional Endothelial Differentiation
Functional endothelial differentiation of EPC-like cell culture was confirmed by the LDL-uptake test and Matrigel angiogenesis assay. The WJ-MSCs and WJ-EPC-like cells were incubated for 4 h in a medium containing 10 μg/ml of DiI-Ac-LDL. Intracellular appearance of the red complexes was then identified only in EPC-induced cells, whereas WJ-MSCs were still unable to take up this red label (Fig. 1G). Next, the ability of cells to form capillaries in semisolid medium was tested in the Matrigel angiogenesis assay. After 7 days of endothelial differentiation, the cells were trypsinized and seeded on the ECMatrix gel in EGM-2 medium. Noninduced WJ-MSCs growing in MSCGM medium served as a negative control. The first signs of the network formation appeared 2 h after injection of WJ-EPC-like cells into the ECMatrix. Formation of the mature vascular network structure with thick and branched tubules were observed at later time points (Fig. 1F). Control primary WJ-MSC cultures did not show capacity to form such a capillary-like network under similar conditions.
Vascular Density in OHC Slices and Protection by WJ-MSC and WJ-EPC Indirect Coculture
Despite the absence of blood flow in organotypic hippocampal slices, the microvessel structures were well maintained and expressed RECA-1. In the intact hippocampal slices, vessel density was similar in the CA1 as well as in the CA2/3 regions. OGD resulted in significant, selective loss of vessel structure exclusively in the CA1 region. Vascular density in the CA2/3 remained stable in both control and injured tissue. The average vessel density in the injured CA1 was 1.9-fold lower compared with the reference intact regions (Fig. 2A). Intact rat organotypic hippocampal slices cocultured with xenogeneic human WJ-derived cells (MSC- and EPC-like) did not exhibit any changes in vessel architecture within CA1 as well as CA2/3 regions (Fig. 2B). On the other hand, coculture of OGD-injured OHCs either with hWJ-MSCs or hWJ-EPCs contributed significantly to maintaining slice vascularity. The vascular density in injured CA1 remained 1.5- and 1.4-fold higher in slices cocultured with WJ-EPCs or WJ-MSCs, respectively, than in OGD slices alone (Fig. 2C).

Blood vessel density in CA1 and CA2/3 hippocampal regions after OGD injury. Forty-eight hours after OGD injury, vessel disruption in the CA1 region was observed, with no reaction in the reference CA2/3 region. (A) Vessel density was determined by counting average area (%) of cells immunolabeled with antibody RECA-1 blood vessels in CA1 and CA2/3 regions of OHC (A). (B, C) Coculture of OGD-injured OHC either with WJ-MSCs or WJ-EPCs resulted in a significant supportive/protective effect on the slice vascularity. The results are presented as mean values from five independent experiments ± SEM. **p < 0.01, ***p < 0.001.
Neuroprotective Effect of WJ-MSCs and WJ-EPCs in Injured OHC Indirect Coculture
Organotypic hippocampal slices were exposed to OGD, which caused selective tissue damage in the CA1 region. Twenty-four hours after the insult, the majority of the cells in the CA1 layer were strongly positive for PI staining, indicating dying cells (Fig. 3). WJ-MSCs or WJ-EPCs cocultured with hippocampal slices 24 h after metabolic injury showed a significantly reduced number of dead cells. The best protective effect was observed in the WJ-MSC coculture in which there was a 76.7% decrease in cell death compared with unprotected culture. The influence of WJ-EPCs was slightly weaker and covered only 70% of dying CA1 cells observed in unprotected injured slices (Fig. 3).

Evaluation of neuroprotective properties of WJ-MSCs/EPCs. PI incorporation into the CA region of the hippocampal slices (top). Coculture with WJ-MSCs/EPCs resulted in a significant decrease in the amount of dead cells. The best protective effect was observed in the coculture with noninduced WJ-MSCs. All data were expressed as mean ± SD; n = 19. **p < 0.01.
Comparison of WJ-MSC and WJ-EPC Secretome Modulated by TLR3 and TLR4 Stimulation
In order to look for putative adjuvant agents responsible for the protective effects of cocultured cells in the OGD hippocampal injury model, we focused our attention on a few key secreted factors. We chose the potent anti-inflammatory cytokine TGF-β1, mixed function modulatory IL-6, and neuro/angioprotective growth factor VEGF. Both WJ-derived cells cultured with either MSC or EPC phenotypic characteristics exhibited substantial, but quite different, secretion patterns of TGF-β1 and IL-6 cytokines (Fig. 4). This secretion was significantly more pronounced in WJ-EPC than in WJ-MSC cultures. In contrast, VEGF was secreted at a comparable level in both WJ-MSC and WJ- EPC cultures.
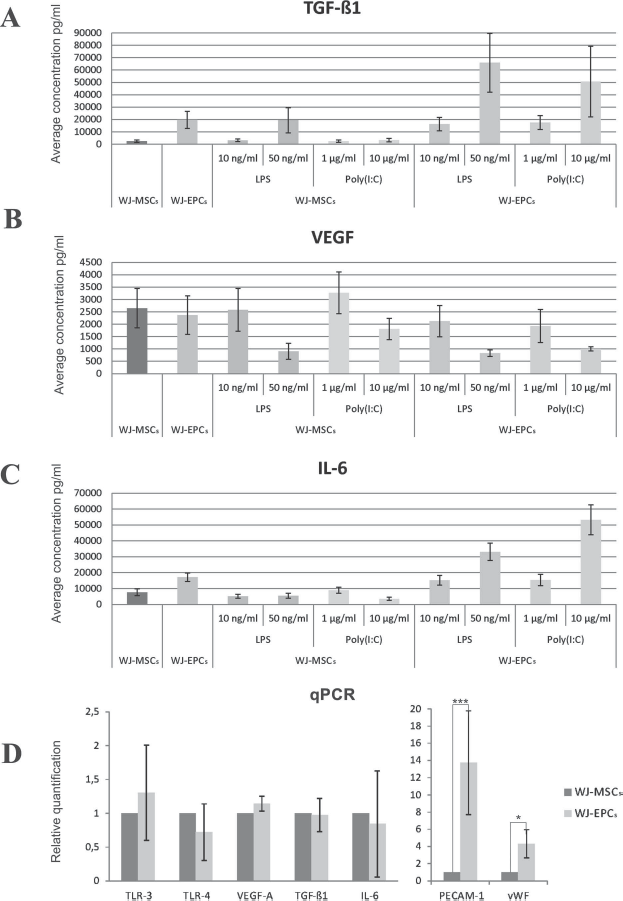
Comparison of levels of cytokines TGF-β1, VEGF, and IL-6 secreted by WJ-MSC and WJ-EPC cultures modulated by LPS and poly(I:C) treatments together with levels of gene expression (qPCR) of the related compounds. (A, C) Cytokine TGF-β1 and IL-6 concentrations measured in culture supernatant were highher for WJ-EPC than for WJ-MSC cultures and additionally stimulated by TLR ligands in their higher concentrations. (B) VEGF secretion pattern did not differ in WJ-MSC and WJ-EPC cultures and was significantly inhibited by increased concentrations of TLR ligands. Each bar represents the mean ± SD of data from two independent experiments repeated three times. (D) mRNA expression was significantly increased in WJ-EPCs compared with WJ-MSCs only for EPC marker genes encoding vWF and PECAM-1. The relative gene expression of WJ-EPCs normalized to the reference gene ACTB was compared to WJ-MSCs as calibrator group. The results are presented as mean values of four experiments from two separate isolations ± SD. *p < 0.05, ***p < 0.001.
Since our experiments were focused on the effect of WJ-derived cell coculture on postinjured OHC slices where the tissue inflammatory response may be exaggerated, we estimated the effects of TLR3/4 stimulation on the secretion pattern of chosen cytokines. In WJ-EPC cultures, we found increased release of TGF-β1 and IL-6 observed only in a higher concentration of TLR3/4 ligands (Fig. 4A and C). In both investigated cell types, the decreased secretion of VEGF at higher level of TLR ligands (Fig. 4B) was observed. The low concentration of either LPS or poly(I:C) (10 ng/ml and 1 μg/ml, respectively) never stimulated any cytokine release.
In parallel experiments, we quantified and compared the levels of gene expression in EPC-like culture versus that of primary WJ-MSCs (Fig. 4D). The relatively low level of transcriptional activity was found for TLR3 and TLR4 receptors as well as for TGF-β1, VEGF, and IL-6 cytokine expression in both types of investigated cultures. In contrast, only induced WJ-EPCs displayed a strong expression of specific endothelial marker genes, PECAM-1 and vWF. This last finding corresponds well with the other data reported here (Fig. 1) and supports genuine endothelial character of the induced WJ-EPCs.
Discussion
One of the most valuable properties of WJ-MSCs in terms of cell therapy is high plasticity and their ability to be induced into diverse phenotypes, meaning they are not restricted only to mesodermal differentiation, but also to other germ layer-specific lineages (11,12,29). Thus, the ability of WJ-MSCs to adopt an endothelial phenotype in an appropriately stimulating environment is not surprising, but it is still controversial and difficult. Classical endothelial cells and their precursors (EPCs) are mainly described as being derived from the bone marrow population of hematopoietic progenitors circulating in blood (2,3,33). The expression of CD34, CD133, and kinase insert domain receptor (KDR), but not the other more mature, hematopoietic lineage connected markers (CD11b, CD45), usually define classical EPC phenotype (34).
However, several studies have described another angiogenic cell subpopulation, named early EPCs, which do not express classical endothelial markers such as CD34 and CD133. These cells are adherent and, in addition to the typical endothelial proteins, also express certain MSC and monocytes markers (10). These properties and functions have been found not only in WJ-derived EPCs (32) but also in umbilical cord blood, placenta (19, 25), or other fetal and perinatal tissues in vivo and in vitro. Thus, we assume that EPCs differentiated from a primary WJ-MSC culture could be considered this subtype of early EPCs due to all of their tested functional and molecular attributes proven here (Fig. 1). This functional angiogenic potential of WJ-EPCs was positively verified in vitro by the widely used LDL-DiI uptake test as well as their high ability to form a vascular network in Matrigel culture.
Similarity of the protection exerted by both native hWJ-MSCs and their induced endothelial-like derivatives observed in our model of OGD-injured hippocampus may indicate common and interrelated pathophysiological mechanisms of action. The specific spatiotemporal characteristics of the injury and their protection focused mainly on the CA1 region and were quantified here by the number of PI-stained (dead) cells or by a decrease in blood vessel density (Figs. 2 and 3) was similar to that observed in a previous study (7). Moreover, indirect Transwell coculture of rat OHC slices with either WJ-derived MSCs or EPCs has no influence on any vital parameters monitoring slice integrity in control. In contrast, OGD stress resulting in cell death and vascular atrophy in the CA1 region (Figs. 2 and 3) was significantly and evenly attenuated by the presence of both types of WJ-derived cells in the lower Transwell compartment. Interestingly, the protective effect of WJ-MSCs was slightly more pronounced in terms of neuronal death inhibition, whereas coculture with WJ-EPCs yielded better CA1 hippocampal vasculature.
In order to ascertain the paracrine effectors secreted by WJ-MSCs and WJ-EPCs with regard to protection against OGD hippocampal injury, we showed that both types of cultured cells release the potent anti-inflammatory cytokine TGF-β1, the mixed function modulatory IL-6, and the neuro/angioprotective growth factor VEGF (Fig. 4). Moreover, the secretion pattern of both types of WJ-derived cultures remain resistant to stimulation by a low concentration of TLR3/4 ligands [LPS and poly(I:C)] recommended previously as optimal for activation of MSC cultures (31) (Fig. 4). Significantly, at higher concentrations of the ligands, only the WJ-EPC culture could respond slightly by activation of TGF-β1 and IL-6 release, whereas VEGF secretion remained insensitive to stimulation. The relatively low affinity of TLR3/4 to LPS and poly(I:C) ligand stimulation in WJ-MSCs can be linked with exceptionally low expression of these receptors reported by others in WJ-MSCs (18,24) and confirmed here for the induced WJ-EPC population. Low expression of TLR genes may be characteristic for MSCs originating from the early developmental stages like WJ-MSCs and their induced endothelial derivatives. Such exceptionally low expression and activity of TLR receptors may suggest maintenance in these cells’ default immunosuppressive function hold even under an inflammatory environment.
Altogether, our data demonstrated that WJ-MSCs could be directed (induced) toward an EPC-like phenotype under permissive environmental influence in vitro and probably also in vivo (32). Both types of cells, WJ-MSCs and induced WJ-EPCs, can release soluble factors that function to protect the hippocampus against OGD impairment. In addition, their innate immunosuppressive potential, evidenced by the basal TGF-β1, IL-6, and VEGF secretion, was only minimally and positively affected by proinflammatory LPS and poly(I:C) stimulation.
Footnotes
Acknowledgment
The work was supported by National Science Center grant No. 2011/01/B/NZ3/05401 and National Centre for Research and Development grant No. Strategmed 1/234261/2/NCBR/2014. The authors declare no conflicts interest.
