Abstract
Our objective was to evaluate the safety and clinical efficacy of autologous M2 macrophage transplantation in nonacute stroke patients. We also evaluated whether the intrathecal administration of macrophages influences the production of cytokines by peripheral blood cells and whether the levels of cytokines correlate with stroke severity and responsiveness to cell therapy. In this study, 13 patients (12 males and 1 female with a median age of 63 years) diagnosed with ischemic (n = 10) or hemorrhagic (n = 3) stroke were subjected to cell transplantation therapy (study group). On average, 21.9 × 106 autologous M2 macrophages were injected intrathecally. Thirteen matched case-control stroke patients who did not receive cell therapy comprised the control group. We did not observe any serious adverse events (i.e., intrahospital mortality, neurological worsening, and seizures) related to the cell injection. One patient in the study group and two patients in the control group died during the 6-month follow-up period due to recurrent stroke. In the study group, the NIHSS score decreased from 11 to 6 (p = 0.007) in 6 months after the therapy, whereas the patients in the control group showed a less pronounced neurological improvement (the NIHSS score decreased from 11 to 8, p = 0.07). The obvious positive response (the improvement of the NIHSS score ≥3) in the study group was observed in 75% versus 18% in the control group (pFET = 0.03). M2 cell introduction did not significantly affect the production of various cytokines. Nevertheless, pretreated levels of IL-8, IL-10, and IL-4 correlated with stroke severity. Moreover, responder patients had lower spontaneous production of IL-10, FGF-β, PDGF, VEGF, and higher stimulation indexes of IL-1β, TNF-α, IFN-γ, and IL-6 than nonresponders. These findings suggest that the intrathecal administration of autologous M2 cells in stroke patients is safe and leads to a better neurological recovery, which could be mediated through the immunomodulatory activity of M2 macrophages.
Introduction
Stroke continues to be the leading cause of death and long-term disability in adults worldwide 1 . Currently, the only FDA-approved therapy for acute ischemic stroke is thrombolysis, but this therapy is limited by a narrow window of time and only targets a single mechanism, which is clearly insufficient for managing such a multifaceted pathological condition 2 . Hence, recent studies have shifted from investigations of acute neuroprotection toward neurorestorative approaches, with special focus on cell-based therapies aimed at improving the neurological outcomes in stroke patients. Actually, numerous preclinical studies using cell therapy for acute and chronic models of ischemic stroke were demonstrated to promote neurological recovery3–5, most likely via indirect bystander effects6–8.
Recent clinical trials established the safety of various types of stem cell products and demonstrated that stem cell therapy was associated with significant improvements in behavioral and functional activity in stroke patients, although clinical benefits of stem cells need further analysis9–11.
It has recently become clear that along with stem cells, monocytes/macrophages could also be implicated in the repair of brain tissue and cerebral vasculature12–14. Macrophages are known to exist as polarized populations, including M1 and M2 subsets. While M1 macrophages are powerful inflammatory cells that produce proinflammatory cytokines and phagocytose pathogens, M2 macrophages modulate inflammatory responses and contribute to angiogenesis and tissue repair15,16. In the central nervous system (CNS), local microglia and newly recruited macrophages can polarize toward the M2 phenotype, thereby promoting functional improvement. Originally, the capacity of macrophages to stimulate neural repair was demonstrated in spinal cord injury (SCI) models17–19. Later experimental studies in models of stroke demonstrated that M1-polarized macrophages induced neuronal death whereas M2 cells protected neurons from apoptosis 20 , and administration of monocytes from human umbilical cord blood-derived mononuclear cells (hUCB-MNCs) reduced infarct size and restored motor function 14 . Similarly, the recovery of astrocytes, oligodendrocytes, endothelial cells, and myelin in the lipopolysaccharide (LPS)-injured brain was shown to occur following the infiltration of monocytes that displayed a repair-related phenotype 21 .
Moreover, several lines of evidence indicated that the clinical effects of stem cells may largely be mediated by monocytes/macrophages. First, mesenchymal stem/stromal cells (MSCs) were reported to induce macrophages to adopt an anti-inflammatory phenotype 22 . When injected intravenously, MSCs stimulated lung macrophages and so led to a systemic increase in anti-inflammatory cytokines. This, in turn, altered the intracerebral microenvironment and augmented the level of M2 (anti-inflammatory) macrophages 23 . Second, monocyte/macrophages may be responsible for the functional improvements seen in stroke models following transplantation of bone marrow or umbilical cord blood mononuclear cells (BM-MNCs and UCB-MNCs, respectively). Specifically, monocyte-depleted MNCs display a reduced capacity to improve the neurological outcome compared to the whole MNC fraction in a rat stroke model 14 . Another possibility is that many transplanted stem cells undergo apoptosis, and their engulfment induces anti-inflammatory responses in both resident and recruited macrophages, thereby boosting the production of macrophage-derived growth factors supporting neural repair, including vascular endothelial growth factor (VEGF), insulin-like growth factor 1 (IGF-1), platelet-derived growth factor (PDGF), and erythropoietin (EPO) 24 . Altogether, these data indicate that in terms of immunoregulation and stimulation of neural repair, monocytes/macrophages could represent good candidates for cell-based therapies13,25.
While the therapeutic potential of stem cells in stroke has been demonstrated in multiple clinical studies (reviewed in Jeong et al. 10 ), to our knowledge, there are no clinical reports on the use of macrophages in the treatment of chronic stroke. Earlier, we showed that culturing macrophages in low growth factor conditions results in the induction of M2-like macrophages. These cells differ from M1 cells in that they produce lower levels of many proinflammatory cytokines and chemokines including tumor necrosis factor-α (TNF-α), interleukin 6 (IL-6), interferon-γ (IFN-γ), IL-18, and IL-8, and they are unable to stimulate T-cell proliferation in a mixed lymphocyte reaction and to induce a Th1/Th17 response; however, they secrete high levels of IGF-1 as well as VEGF 25 . Intrathecal delivery of these cells was shown to be safe and improved motor functions and mental faculties in children with cerebral palsy 26 .
The present study aims at assessing the safety and feasibility of using autologous M2 macrophages in a prospective cohort of stroke patients. In the current study, we also focus on the evaluation of cytokine production in poststroke patients to gain insight as to whether intrathecal administration of macrophages influences the profiles of cytokines secreted by peripheral blood cells (PBCs) and whether the levels of cytokines correlate with stroke severity and responsiveness to the cell therapy.
Materials and Methods
Clinical Study Design
This was a prospective phase I/II nonrandomized open-label clinical study of safety and efficacy of autologous M2 macrophages in nonacute stroke patients. The protocol was approved by the Research Ethics Committee of the Institute of Fundamental and Clinical Immunology. Written informed consent was obtained from the recruited patients. This study was registered at ClinicalTrials.gov (NCT01845350). Patients were selected on the basis of the World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Research and patient care costs were covered by the Institute of Fundamental and Clinical Immunology.
Patients and Selection Criteria
Individuals of both sexes were eligible for the study according to the following inclusion and exclusion criteria. The inclusion criteria were as follows: (1) ischemic or hemorrhagic first-ever stroke in the middle cerebral artery (MCA) territory as evidenced by computed tomography (CT) or magnetic resonance imaging (MRI); (2) age more than 18 years; (3) nonacute period after stroke (more than 1 month and less than 12 months); (4) baseline National Institutes of Health Stroke Scale (NIHSS) score ≥7 (at the stroke onset) with persistent neurological deficits (NIHSS ≥4 points before cell therapy); and (5) a signed informed consent. The exclusion criteria were as follows: (1) history of previous stroke; (2) seizures; (3) thrombophilias or primary hematological disease; (4) malignancies; (5) autoimmune diseases; (6) impaired renal or liver function (as determined by serum creatinine >1.5 mg/dl and/or total bilirubin >1.3 mg/dl); (7) hemodynamic or respiratory instability; (8) human immunodeficiency virus (HIV) or uncontrolled bacterial, fungal, or viral infections; (9) pregnancy; and (10) participation in other clinical trials.
A total of 13 patients were enrolled in this ongoing study and were subjected to cell transplantation therapy (study group). Thirteen matched case-control stroke patients who were conventionally treated at the same time and at the same clinical hospital were included in the clinical analysis as a control group. Selection of case-control pairs was performed taking into account age, stroke type and severity, and time since the stroke.
Macrophage Generation
Autologous peripheral blood mononuclear cells (PBMNCs) were obtained using density gradient centrifugation (Ficoll-Paque; Sigma-Aldrich, Munich, Germany) of heparinized whole-blood samples. The generation of M2 macrophages from plastic-adherent cells was performed according to a previously developed protocol 25 . In brief, adherent cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 medium supplemented with human granulocyte macrophage colony-stimulating factor (GM-CSF) (rhGM-CSF, 50 ng/ml; R&D Systems, Minneapolis, MN, USA) in low serum deprivation conditions at 37°C and 5% CO2. On day 7, the macrophages were harvested using ethylenediaminetetraacetic acid (EDTA; Sigma-Aldrich) in Hank's balanced salt solution (HBSS; Sigma-Aldrich), washed, resuspended in saline solution, and counted. Quality control criteria for the release of cell product included the evaluation of bacterial sterility, cell viability [by trypan blue (Sigma-Aldrich) exclusion], and immunophenotyping to count CD14+ cells.
Cell Administration
All subjects were hospitalized during the administration of M2 macrophages. Single intrathecal injection by lumbar puncture was performed after a premedication with dexasone (8 mg, intravenously; KRKA d.d., Novo Mesto, Slovenia). Patients were given local anesthesia, and lumbar puncture was performed in the lateral decubitus position under sterile conditions. The needle was introduced into the lumbar cistern. After removal of 2 ml of cerebrospinal fluid, 2 ml of cell suspension in saline was injected. On average, 21.9 × 106 [interquartile range (IQR), 13–29 × 106] of ex vivo-generated autologous M2 macrophages were injected.
Patient Evaluation and Follow-Up
All patients were evaluated according to the study protocol before and 6 months after the cell therapy. The primary outcome was safety, namely, the absence of adverse events (AEs; including allergic reactions, hematoma or local infection, neurological worsening, new/recurrent ischemic or hemorrhagic stroke, seizures, and intrahospital mortality) during cell introduction and within 72 h following the injection. For this purpose, general clinical and neurological examinations, as well as routine blood tests, were performed before and 3 days after cell therapy. Additionally, the peripheral blood of 10 of 13 consecutively recruited patients in the study group was subjected to Bio-Plex cytokine analysis (Bio-Rad Laboratories, Hercules, CA, USA) before and after cell injection to evaluate the systemic inflammatory response. Any AEs that occurred during the 6-month follow-up period were also documented. The secondary outcome measures were NIHSS score, the difference between NIHSS scores pre- and posttreatment, and a modified Rankin scale (mRS) score 6 months after the therapy.
Blood Cell Cytokine Production
For cytokine analysis, the samples of whole peripheral blood supernatants were collected before and 3 days after macrophage administration. For this, 0.5 ml of heparinized peripheral blood was added to 1.5 ml of RPMI-1640 and incubated without any supplements or with concanavalin A (Con A; 15 μg/ml; Sigma-Aldrich) for 24 h. The samples were then temporarily cryopreserved at −80°C until the measurement. The concentration of 26 cytokines was determined using the multiplex analysis using Bio-Plex Pro Human cytokine assay (Bio-Rad Laboratories) following the manufacturer's instructions. The Bio-Plex human cytokine assay included the following groups of cytokines: (1) pro- and anti-inflammatory cytokines (IL-1β, TNF-α, IL1-ra, IL-10, and IL-13); (2) immunoregulatory cytokines (IL-2, IFN-γ, IL-12, IL-4, IL-5, IL-15, and IL-17); (3) growth factors [granulocyte colony-stimulating factor (G-CSF), IL-7, IL-9, fibroblast growth factor-β (FGF-β), PDGF, VEGF, and IL-6]; (4) CXC chemokines [IL-8 and IFN-γ-inducible protein 10 (IP-10)] and CC chemokines [monocyte chemoattractant protein-1 (MCP-1), macrophage inflammatory protein-1α (MIP-1α), MIP-1β, Rantes, and Eotaxin]. Results were reported as spontaneous and mitogen-stimulated cytokine production (in pg/ml) and the stimulation index (SI), which was calculated as the fold increase in Con A-stimulated cytokine concentration over the background.
Statistical Analysis
The data were expressed as medians and IQR. Statistica 6.0 software for Windows (StatSoft Inc., Tulsa, OK, USA) was used for data analysis. The differences between groups were compared by the Mann–Whitney U test and Wilcoxon matched pairs test. Nominal variables were analyzed using Fisher's exact test. Spearman's rank correlation coefficient was used to measure the statistical dependence between two continuous variables.
Results
Table 1 summarizes baseline characteristics of the patients enrolled in the study. The study group consisted of 12 males and 1 female with a median age of 63 years (ranging from 42 to 77 years) diagnosed with a history of ischemic (n = 10) and hemorrhagic (n = 3) stroke. The control group was composed of 10 males and 3 females with a median age of 63 years (from 52 to 74 years), including 10 patients with ischemic stroke and 3 patients with hemorrhagic stroke. Baseline stroke severity in the study group showed the NIHSS score ranged from 8 to 22 points (median, 15; IQR, 13–17), with 8 patients having more than 14 points, indicating “moderate” and “moderate to severe” stroke. In the control group, the NIHSS score varied from 7 to 19 at stroke onset, with a median of 16 (IQR, 11–18) points, and similarly to the study group, most of them had an NIHSS score above 14. The median time since stroke onset in the study and control groups was 107 and 86 days, respectively, and did not differ significantly. At the start of the cell therapy (in the study group) and at an analogous time period after the stroke onset for the control group, the patients in both groups still demonstrated pronounced neurological impairments with identical NIHSS scores of 11 points. The modified Rankin score was 4 in both groups, consistent with considerable disability of the patients. Thus, the patients of the study and control groups were very similar and did not differ significantly in age, sex, type of stroke, severity of neurological impairments, functional activity, or stroke risk factors.
Baseline Characteristics of Patients

Data are presented as median and interquartile range (in parentheses) unless otherwise stated. The differences between groups were compared by Mann–Whitney U test for continuous values and Fisher exact test (FET) for categorical values.
Mean NIHSS scores before cell therapy for the study group, and NIHSS scores at an analogous time period after the stroke onset for the control group.
All the patients in the study group received one injection of autologous M2 macrophages. The median dose of transplanted cells was 22.0 × 106 (13–29 × 106), of which 85% (72–93%) were CD14+ cells. The viability of the cells obtained was above 93% in all cases.
Intrathecal injection of autologous M2 macrophages was followed by febrile or subfebrile fever in two and three cases, respectively. Two patients suffered from headache, and three patients complained of back pain. Nevertheless, we did not observe any serious short-term AEs during the first 3 days following cell administration. No cases of allergic reactions, local hematoma, or infections at the site of the lumbar puncture; meningeal reactions; systemic inflammatory complications; or complications related to toxicity were noted after the injection. There were no cases of neurological worsening, new/recurrent ischemic or hemorrhagic stroke, or seizures. There were no statistical differences in the blood parameters tested before and 3 days after the cell treatment (Table 2). Taken together, these data indicate that intrathecal cell transplantation was safe and did not induce cell-related AEs.
Blood Parameters in Cell-Treated Patients

Data are presented as median and interquartile range (in parentheses). The differences between groups were compared using Wilcoxon matched pairs test. WBC, white blood cells; RBC, red blood cells; Hb, hemoglobin; PLT, platelets.
One (7.7%) patient in the study group and two (15.4%) patients in the control group died during the 6-month follow-up period due to recurrent stroke. Neurological examination of the surviving patients demonstrated a significant decline of the NIHSS score from 11 (6.5–13) before cell therapy to 6 (3–10) (p = 0.007) at 6 months after therapy in the study group. The patients in the control group showed a less pronounced neurological improvement, which was manifested as a trend (the NIHSS score decreased from 11 to 8, p = 0.07). Figure 1 shows individual changes in NIHSS scores in the patients of the study and control groups at the start of observation and at the end of the 6-month follow-up period. Cell therapy was also accompanied by decreases in the mRS scores from 4 (3–4) to 3 (2–4), although these changes were not statistically significant (p = 0.07). The patients in the control group did not improve functional independence [mRS scores before and after treatment were 4 (3–5) and 4 (4–5), respectively, p = 0.75].
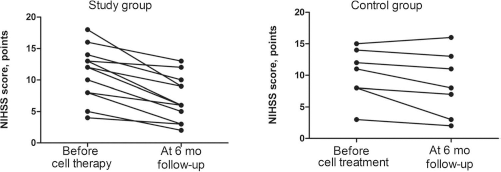
Individual changes in NIHSS scores before and 6 months after M2 macrophage injection. The data are presented as individual values of NIHSS score before and 6 months after cell therapy in surviving patients including 12 cell-treated patients and 11 case-control patients. Study group: two patients had an NIHSS score change from 5 to 2. Control group: two patients had an NIHSS change from 12 to 11.
The clear positive neurological response (the improvement of the NIHSS score by at least 3 points) in surviving patients was noted in 9 of 12 (75%) patients in the study group, whereas only 2 of 11 (18%) patients in the control group had similar neurological improvement (pFET = 0.03).
It is important to note that we did not find any differences in age, sex, or time since stroke onset, stroke severity, or the number of injected M2 cells in responder and nonresponder patients (Table 3).
Baseline Characteristics of Responder and Nonresponder Patients

Data are presented as median and interquartile range (in parentheses). The differences between groups were compared using Mann–Whitney U test and Fisher exact test (for categorical values). Nonresponders included the patients who died or displayed NIHSS improvement <3.
To investigate whether intrathecal injection of M2 macrophages influenced the cytokine production, multiplex analysis of cytokines produced by the patients' PBCs before and after cell transplantation was performed. Injection of M2 macrophages was not found to significantly affect the spontaneous and Con A-stimulated production of proinflammatory cytokines, Th1/Th2 immunoregulatory cytokines, chemokines, or growth factors (Table 4).
Results of MultiPlex Analysis of Cytokines Produced by Patients' Blood Cells Before and After Cell Transplantation
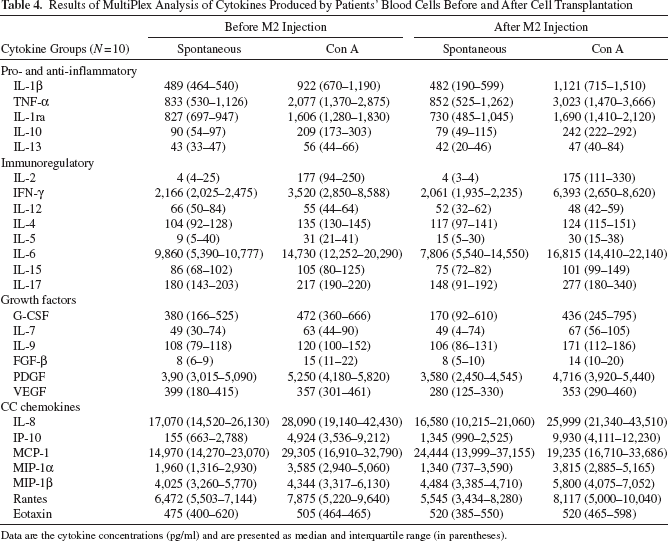
Data are the cytokine concentrations (pg/ml) and are presented as median and interquartile range (in parentheses).
To understand whether there is any relationship between injury severity and cytokine levels, we evaluated correlations between the concentration of various cytokines and the NIHSS scores before therapy. No significant correlations between spontaneous cytokine production and stroke severity were observed. Nonetheless, NIHSS scores were found to be strongly correlated with Con A-stimulated production of proinflammatory chemokine IL-8 and inversely correlated with anti-inflammatory cytokines IL-10 and IL-4 (Fig. 2). Of note, these relationships were not influenced by the duration of the poststroke period, since we did not find any correlation between cytokine concentrations and time after stroke onset (data are not shown).

Correlations between the Con A-stimulated production of IL-10, IL-4, and IL-8 and NIHSS score before the cell introduction. Spearman's rank correlation coefficient was used to measure the statistical dependence between two variables in 10 cell-treated patients.
Although intrathecal macrophage transplantation did not induce any significant changes in cytokine production by blood cells, the analysis comparing responder patients with nonresponders revealed considerable differences in the pretreatment levels of several cytokines. Blood cells of responder patients (Table 5) had lower spontaneous production of anti-inflammatory cytokine IL-10 and several growth factors, including FGF-β, PDGF, and VEGF. At the same time, the SIs of IL-1β, TNF-α, IFN-γ, and IL-6 in responder patients were significantly higher, indicating the increased reactivity of blood cells in terms of production of proinflammatory and immunoregulatory cytokines.
Cytokine Production by Patients' Blood Cells in Responders and Nonresponders Before M2 Cell Transplantation

Data are the cytokine concentrations (pg/ml) and are presented as median and interquartile range (in parentheses). The patients who decreased their NIHSS scores by 3 points and more were considered as responders. The differences between the groups were compared by Mann–Whitney U test.
p < 0.05 was taken to indicate statistical significance.
To describe the balance of pro- and anti-inflammatory activities of blood cells, as well as the Th1/Th2 ratio, we compared the ratios of TNF-α/IL-10 and IFN-γ/IL-4 levels in responder and nonresponder patients. The responder patients had a higher ratio of TNF-α/IL-10 (p = 0.08) and IFN-γ/IL-4 (p = 0.05) in Con A-stimulated blood cell cultures.
Additionally, we performed a correlation analysis between cytokine concentrations before M2 administration and the responsiveness to cell therapy (calculated as NIHSS score 6 months after cell therapy – NIHSS score prior to cell therapy). There was a significant inverse correlation between the responsiveness to cell therapy and the spontaneous production of IL-10 (rS = −0.82, p = 0.002), FGF-β (rS = −0.79, p = 0.004), PDGF (rS = −0.87, p = 0.007), and VEGF (rS = −0.64, p = 0.046). Furthermore, we found a direct relationship between the clinical response and the SI of IFN-γ (rS = 0.7, p = 0.02), IL-6 (rS = 0.79, p = 0.009), and the TNF-α/IL-10 ratio (rS = 0.74, p = 0.02).
Discussion
Accumulating evidence suggests that resident microglia and infiltrating monocyte-derived macrophages play a major role not only in neuroinflammation but also in subsequent repair processes27–29. In addition to the phagocytosis of cellular and fibrillar debris resulting from brain damage and inactivation of toxic and inhibitory molecules, the microglial cells and macrophages produce a variety of neurotrophic, proangiogenic, and immunomodulatory factors that are involved in the regulation of remodeling and repair in the brain30,31. Hence, monocytes/macrophages may provide a promising alternative to stem cell transplantation for the stimulation of neural repair.
Actually, studies of the Schwartz group demonstrated partial recovery of motor function following the implantation of macrophages into transected rat spinal cords 19 . Later, Knoller et al. conducted a pilot clinical trial showing safety of activated macrophages in patients with acute SCI 32 . However, a randomized, controlled multicenter study using macrophages failed to demonstrate clinical improvement compared to the control group 33 , which could be partially explained by the invasiveness of the transplantation procedure.
Simultaneously, several independent groups have demonstrated that hUCB-MNCs improve functional recovery in models of ischemia34–36 and that neuroprotective effects of human cord blood were largely mediated by monocytes 14 . Moreover, Glod et al. found that CD14+ peripheral blood monocytes participate in the repair of the vascular barrier after brain injury 12 . In turn, Marchetti et al. revealed a rescue effect of transplanted UCB CD14+ cells in a model of ischemic retinopathy. According to their study, human UCB CD14+ cells differentiated into type 2 macrophages, which promoted a physiological angiogenesis, reduced inflammatory processes in the retina, and mediated neurotrophic and antiapoptotic activities in retinal neurons 37 .
In our study, we first evaluated the safety and therapeutic potential of autologous macrophages in the treatment of nonacute stroke patients. To do so, we used M2 macrophages generated in low growth factor conditions25,38. The data obtained suggest that intrathecal administration of autologous M2 macrophages generated ex vivo and transplanted into 13 patients with nonacute stroke was safe and did not result in any significant AEs. According to the recommendations for early phase clinical studies 39 , we formed a heterogeneous group of stroke patients including subjects with both ischemic and hemorrhagic stroke, with a poststroke period from 1 to 7 months and a baseline NIHSS score ranging from 7 to 19 points. All of the patients showed good tolerability of cell injection with only minor AEs (temporal fever, headache, and back pain in some patients). We also did not find any statistical difference in blood cell counts (red blood cells, white blood cells, and platelets), hemoglobin levels, or C-reactive protein (CRP) concentration before the treatment and 3 days after cell injection. There were no cases of intrahospital mortality, neurological worsening, or seizures related to the cell transplantation. However, during the 6-month follow-up period, 1 of the 13 cell-treated patients developed recurrent vascular episodes and died. We do not attribute this event to cell injection, since 13.9% of stroke survivors suffer from recurrent stroke 40 and 2 of 13 control patients died from recurrent stroke during the 6-month follow-up.
Intrathecal administration of cells is a minimally invasive procedure as compared to intracerebral injection. In addition, Lim et al. demonstrated that intrathecal injection of MSCs in a stroke rat model resulted in a significantly higher number of cells that migrated to the ischemic area as compared to the intravenous cell introduction. Moreover, intrathecal administration of MSCs significantly reduced the ischemic damage in contrast to the intravenously treated group 41 . In addition, Ogata et al. reported on the migration of intrathecally injected macrophages to the injured portion of the spinal cord 42 . Our data support the idea that intrathecal delivery of macrophages can be considered a useful and feasible treatment approach for brain injuries, and are consistent with other reports that demonstrate the safety of intrathecal administration of various cell types including BM-MNCs and CD34+ hematopoietic stem cells (HSCs) in stroke patients43–45.
The current study was designed to primarily assess the safety and tolerability of the intrathecal injection of M2 macrophages. Nevertheless, a comparative clinical analysis demonstrated better neurological recovery in the study group than in the case-control patients. A remarkable neurological improvement in the treated group was registered for 75% of patients, whereas only 18% of patients from the control group demonstrated such an improvement during the 6-month follow-up.
Although cell therapy seems to be a promising new approach in the treatment of stroke, the efficacy of stem cell transplantation is not yet fully understood 10 . Several phase I studies demonstrated neurological improvement in stroke patients following the transplantation of autologous BM-MNCs46–48 and MSCs 49 . Nevertheless, the improvements do occur naturally in the patients with stroke, and since the cells were delivered during the acute or subacute phase of stroke, it was difficult to distinguish between the effects of cell therapy and spontaneous recovery. However, two studies using BM-MNCs and CD34+ HSCs showed improved outcomes in the patients with a chronic phase of stroke when spontaneous regenerative responses are largely limited44,50. Moreover, two controlled studies demonstrated a significant improvement in MSC- and CD34+-treated groups compared with control groups50,51. Our research supports the therapeutic potential of cell therapy in stroke treatment and indicates that along with the stem cells, M2 macrophages may improve neurological recovery in nonacute stroke patients.
In the present study, we attempted to answer the question of whether intrathecal administration of macrophages influenced systemic immunity. The comparison of cytokine production before and after cell administration did not reveal significant differences in either spontaneous or Con A-activated secretion levels of various pro- or anti-inflammatory cytokines (IL-1β, TNF-α, IL1-ra, IL-10, and IL-13), immunoregulatory Th1/Th2 cytokines (IL-2, IFN-γ, IL-12, IL-4, IL-5, IL-6, IL-15, and IL-17), growth factors (G-CSF, IL-7, IL-9, FGF-β, PDGF, and VEGF), and chemokines (MCP-1, MIP-1α, MIP-1β, Rantes, and Eotaxin). These data indicate that the intrathecal administration of M2 macrophages does not influence a systemic production of cytokines by blood cells.
A growing body of literature data provide evidence that inflammatory mechanisms contribute directly to the pathogenesis and progression of primary and secondary stroke lesions52,53. Inflammation is considered to be a critically important factor for stroke outcome, contributing to the development of secondary injury. Actually, the severity of neurological impairments and poor outcome are associated with higher levels of CRP, TNF-α, IL-1β, IL-6, and IL-10 when measured in an acute phase of stroke54–58. In contrast to these studies, we evaluated cytokine production in the nonacute phase of stroke. Given that only a restricted quantity of cytokines are present in the plasma or serum of acute stroke patients, we measured cytokine concentrations in blood cell cultures. Additionally, we tested numerous different cytokines in a single blood cell sample using multiplex methodology.
Our results demonstrate a significant positive correlation of stroke severity with Con A-stimulated production of IL-8 and an inverse correlation with anti-inflammatory cytokines IL-10 and IL-4. To our knowledge, this is the first study reporting the association of cytokines with neurological impairments during the nonacute phase of stroke. Our finding concerning the negative correlation between NIHSS score and Con A-stimulated production of IL-10 differs from the results of Chang et al., who demonstrated a direct association of stroke severity with serum IL-10 in acute stroke patients 55 . The relevance of IL-8 and IL-4 in stroke severity was also demonstrated for the first time. The differences observed may result from the use of distinct sources of cytokines in our research and other studies (blood cell cultures vs serum), but what is more likely is that these differences are related to the diverse roles of various cytokines in the pathophysiology of acute and nonacute stroke phases.
Finally, we demonstrated that in the study group the neurological improvement was associated with the pretreatment levels of several cytokines. Thus, the patients who significantly improved following the cell therapy had a lower spontaneous production of anti-inflammatory cytokine IL-10 and several growth factors including FGF-β, PDGF, and VEGF, and higher SIs of IL-1β, TNF-α, IFN-γ, and IL-6. Moreover, we found an inverse correlation between cell therapy responsiveness and spontaneous production of IL-10, FGF-β, PDGF, and VEGF and a direct relationship between clinical response and SIs for IFN-γ, IL-6, and TNF-α/IL-10 ratio. This indicates that M2 macrophages are more effective in the patients with lower production of growth factors, lower endogenous immunosuppressive mechanisms, and increased proinflammatory activity. On the other hand, these results provide evidence for the potential benefits of an anti-inflammatory strategy. Our data also suggest that the detection of cytokines, as markers of poststroke immune response, may be useful for the prediction of cell therapy responsiveness.
Footnotes
Acknowledgments
We thank Nataliya V. Krystova and Nataliya I. Shevchuk (Clinical Department, Institute of Fundamental and Clinical Immunology, Novosibirsk, Russia) for their assistance in laboratory studies. The authors declare no conflicts of interest.
