Abstract
Mesenchymal stromal cells (MSCs) have been shown to exert therapeutic effects by modulating various cell types, including vascular endothelial cells, immune cells, and resident stem cells, at sites of tissue injury. Recent studies have highlighted the importance of MSC–macrophage interactions in suppressing inflammation and promoting tissue regeneration; however, the underlying mechanisms remain poorly understood. In this study, we administered MSCs intratracheally to mice with bleomycin (BLM)-induced lung injury and investigated their effects on alveolar macrophages (AvMs), defined as CD64High/F4/80High/SiglecfHigh/CD11bNegative. MSC transplantation alleviated loss of AvMs observed after BLM treatment. Transcriptomic profiling of AvMs revealed increased expression of genes associated with cell survival, oxidative stress resistance, and efferocytosis. Among them, Sesn2 was notably upregulated.
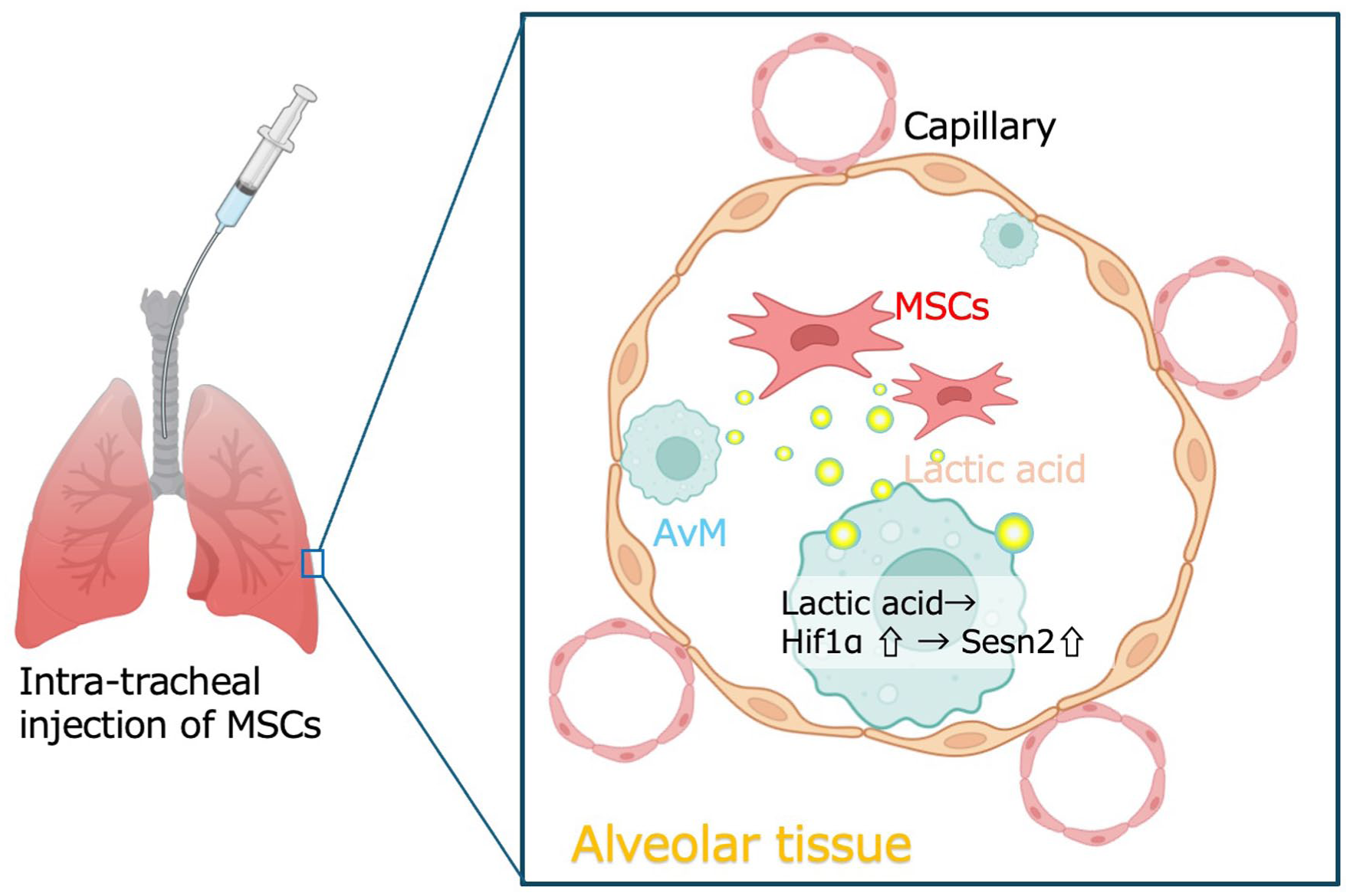
Introduction
Mesenchymal stromal cell (MSC) transplantation has been applied to a variety of chronic inflammatory diseases and has shown remarkable therapeutic efficacy1–6. Due to their broad biological functions including anti-inflammatory and regenerative effects and safety characteristics, over 1000 clinical trials involving MSCs have been registered to date. In addition, 12 MSC-based products have been approved by regulatory authorities worldwide, including the Food and Drug Administration (FDA) (United States), European Medicines Agency (EMA) (Europe), and Pharmaceuticals and Medical Devices Agency (PMDA) (Japan) as of 2023 7 . However, the precise mechanisms by which MSCs influence cells in injured tissues remain incompletely understood.
Among the proposed mechanisms, increasing attention has been directed toward the interaction between transplanted MSCs and macrophages, which are thought to be critical mediators of the immunomodulatory and regenerative effects of MSCs8–11. Macrophages are major phagocytic cells in the host innate immune system and are a principal source of inflammatory cytokines and growth factors. They play a pivotal role in determining whether tissue damage progresses toward chronic inflammation or transitions to regeneration 12 . Therefore, elucidating how MSCs modulate macrophage behavior is essential for understanding the therapeutic potential of MSC-based interventions.
While it is generally accepted that MSCs exert their effects via the secretion of cytokines and extracellular vesicles (EVs)13–16, it is also well documented that their
However, studying direct MSC–macrophage interactions
Our objective in this study was to investigate the interaction between MSCs and tissue-resident macrophages in the lung using a transbronchial delivery model, which allows the direct administration of MSCs into the airways20,21. This model facilitates the delivery of a defined number of MSCs into the lung, an environment that is partly isolated from external influences and contains a limited spectrum of macrophage types and differentiation stages. In this setting, interactions can be clearly examined between MSCs and tissue-resident macrophages, specifically alveolar macrophages (AvMs). AvMs constitute a key population of tissue-resident macrophages in the lung and serve as the first line of immune defense in the respiratory system22,23. In our approach, MSCs were directly delivered into the lung via a micro-sprayer, enabling localized administration while minimizing confounding effects such as systemic redistribution or immune cell infiltration secondary to vascular injury. Using this strategy, we found that MSC transplantation enhanced the expression of stress resistance and survival-related genes in AvMs, with Sestrin2 (Sesn2) emerging as a prominent transcriptional marker. Further analysis revealed that Sesn2 expression was at least partly regulated by Hif1a, a transcription factor known to be induced by lactate that is a metabolite abundantly secreted by MSCs. Given the central role of macrophages in orchestrating inflammation and regeneration, these findings suggest that MSCs may promote tissue repair by modulating macrophage viability through a lactate–Hif1a–Sesn2 signaling axis. This mechanism provides a plausible explanation for the therapeutic effects of MSCs, even in the absence of sustained engraftment.
Materials and methods
Ethical consideration for animal studies
All procedures related to animal handling, care, surgery, and sacrifice were approved by the Institutional Animal Care and Use Committee of Kindai University as the approved research project number KAME2025-044 and performed in accordance with institutional guidelines and regulations. All methods are reported in accordance with ARRIVE guidelines.
Animal care and experimental procedures
All animal experiments were conducted in accordance with the ARRIVE guidelines and approved by the Institutional Animal Care and Use Committee of Kindai University. Male C57BL/6 mice (8–10 weeks old) were housed under specific pathogen-free (SPF) conditions with controlled temperature (22 ± 2°C), humidity (50%–60%), and a 12-h light/dark cycle. Animals had free access to standard rodent chow (CLEA Japan, Osaka, Japan) and water ad libitum.
For experimental procedures, anesthesia was induced and maintained with isoflurane (2%–3% in oxygen) delivered via inhalation. At the designated time points, animals were euthanized by cervical dislocation under deep anesthesia. All efforts were made to minimize animal suffering.
Preparation of mouse bone marrow mesenchymal stromal cells
All mice used in this study were purchased from CLEA Japan, Inc. The mice were bred and maintained under controlled conditions in accordance with institutional and international guidelines for the care and use of laboratory animals. Eight-week-old EGFP-transgenic C57BL/6 (B6-EGFP, Japan SLC Inc., Shizuoka, Japan) male mice were used. Following euthanasia with an overdose of isoflurane and subsequent cervical dislocation, cleaned femurs and tibias were cut into small pieces and treated with 0.1% collagenase type II (Wako, Tokyo, Japan) for 30 min. Single-cell suspensions containing bone marrow mesenchymal stromal cells (BMMSCs) were collected, washed twice with phosphate buffered saline (PBS), and plated on cell culture dishes (Sumitomo Bakelite Co. Ltd., Tokyo, Japan) in αMEM (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 200 mM
Intratracheal administration of bleomycin and MSC transplantation
Eight- to 10-week-old male C57BL/6J (B6) mice were used. Under anesthesia with isoflurane, a micro-sprayer was slowly inserted into the airway under direct visualization using a laryngoscope. Once the carina was reached, 4 mg/kg of bleomycin hydrochloride (Nippon Kayaku Co., Ltd., Tokyo, Japan) diluted in 100 µL sterile saline was sprayed into the trachea. Control mice received an equal volume of sterile saline. MSC transplantation was performed 48 h after bleomycin administration. The mice were re-anesthetized with isoflurane, and 1 × 106 MSCs, passed through a cell strainer and diluted in 10.0 µL PBS, were intratracheally delivered using the same micro-sprayer (Fine Particle Sprayer /FPS-050-A1/ Toray Precision Co., Ltd., Shiga, Japan) and laryngoscope setup. Control animals received an equal volume of sterile saline. To ascertain whether the effects of MSCs were mediated through lactate, intratracheal instillation of the lactate transporter inhibitor AZD3965 reconstituted in dimethyl sulfoxide (DMSO) was performed simultaneously with MSC administration. Lungs were harvested 48 h post-treatment for subsequent cellular analyses. To ensure procedural consistency and minimize animal distress, all administrations were performed by a technically experienced operator. Prior to the experiments, spraying performance was validated using India ink to confirm uniform and stable delivery to the distal airways.
Masson–Goldner trichrome staining
Paraffin-embedded tissue sections (3 µm) were deparaffinized in xylene and rehydrated through a graded ethanol series to distilled water. Nuclei were stained with Weigert’s iron hematoxylin for 10 min, followed by rinsing in running tap water for 10 min. Sections were then stained with Ponceau red–acid fuchsin solution for 10 min, rinsed briefly in distilled water, and differentiated in phosphomolybdic acid–orange G solution (all were purchased from Sigma-Aldrich, St. Louis, MO, USA) for 5 min. Then, sections were transferred to light green SF solution and stained for 5 min. Finally, sections were dehydrated through graded ethanol, cleared in xylene, and observed.
Immunofluorescence
Paraffin-embedded lung tissue sections (3 µm) were deparaffinized, rehydrated, and subjected to antigen retrieval in citrate buffer (pH 6.0) at 95°C for 15 min. Sections were blocked with 10% BlockAce for 1 h and incubated overnight at 4°C with anti-αSMA (1:200, L19D14, Selleck chemicals, Yokohama, Japan). After washing, sections were incubated with fluorophore-conjugated secondary antibodies (Donkey Anti-IgG(H+L), Rabbit CF555, 1:1000; Biotium, Fremont, CA, USA) for 1 h at room temperature in the dark. Nuclei were counterstained with DAPI (Dojindo Laboratories, Kumamoto, Japan).
Detection of transplanted GFP-MSCs in the lung
The GFP-MSC transplanted mice were sacrificed at 15 min, 24, 48, and 72 h, and at 1 week after transplantation, and lungs were harvested. The tissues were washed 3 times with PBS, finely minced with scissors, and digested with 0.1% collagenase for 30 min at 37°C. After two washes with 5% Fetal Calf Serum–Dulbecco’s Modified Eagle Medium (FCS-DMEM), the cells were subjected to flow cytometric analysis or sorting using an FACS Aria II cell sorter (BD Biosciences, San Jose, CA, USA).
Isolation of AvMs
Mice were euthanized by cervical dislocation, followed by immediate exsanguination and lung excision. The excised lungs were gently washed in chilled PBS, and surrounding connective tissue and trachea were carefully removed. The lung tissue was then minced with fine scissors and digested with 0.1% collagenase (Worthington Biochemical Corporation, NJ, USA) for 30 min at 37°C. Following digestion, the cell suspension was washed twice with DMEM supplemented with 2% FCS and HEPES. Cells were then incubated with fluorophore-conjugated antibodies against CD64 (BioLegend, Inc., San Diego, CA, USA: PE anti-mouseCD64, Clone: X54-5/7.1), F4/80 (BioLegend: FITC anti-mouse F4/80, Clone: BM8), CD11b (BioLegend: Brilliant Violet 421 anti-mouse/human CD11b, Clone: M1/70), and Siglecf (BioLegend, Inc., APC anti-mouse CD170, Clone: S17007L) at 4°C for 30 min in the dark. After staining, the cells were washed twice with the same DMEM/FCS/HEPES buffer and immediately subjected to flow cytometric analysis or sorting using an FACSAria II cell sorter. In this study, monocytes/macrophages isolated from bone marrow tissue (BM) were used as controls to confirm the characteristic marker expression of AvMs in the transcriptome analysis. Briefly, bone marrow tissue was harvested from 6-week-old male B6 mice using a 26-G needle and digested with 0.1% collagenase for 30 min. The resulting cells were incubated with anti-CD45 and anti-CD11b antibodies for 30 min, and the double-positive population was subsequently isolated using an FACSAria cell sorter (BD Biosciences).
In vitro coculture of macrophages with human MSCs and lactate supplementation
To investigate the effects of human MSCs on macrophages, primary cultures of macrophages were prepared. AvMs isolated from B6 mice were expanded in RPMI 1640 medium (Thermo Fisher Scientific) supplemented with 5% FCS and 20 ng/ml M-CSF (Proteintech, Rosemont, IL, USA). For coculture experiments, 1 × 105 macrophages were seeded together with 5 × 104 human MSCs (PT-2501, Lonza, Tokyo, Japan) in 12-well plates and cocultured for 48 h. For lactate supplementation, 5 × 104 human MSCs were treated with lactate at a final concentration of 20 mM, and gene expression analysis was performed after 24 h. In addition, AZD3965, dissolved in DMSO, was applied at a final concentration of 2 μM for 24 h.
Sample preparation for proteome analysis
Proteins were extracted using radio-immunoprecipitation assay (RIPA) buffer and the concentration was determined using a Bicinchoninic Acid (BCA) assay. Protein digestion was performed using trypsin at a 1:50 ratio overnight at 37°C. The resulting peptides were desalted using C18 columns and dried using a vacuum centrifuge.
Chromatography and mass spectrometry
The peptide mixtures were reconstituted in 0.1% formic acid and subjected to liquid chromatography using the Vanquish Neo UHPLC system (Thermo Fisher Scientific) equipped with an Aurora Ultimator column (IonOpticks, Fitzroy, VIC, Australia). Mass spectrometric analysis was performed on an Orbitrap Exploris 480 (Thermo Fisher Scientific). Mass spectrometry data were processed using Thermo Proteome Discoverer version 2.5.0.400. The raw files were searched against the UniProt Mouse database using the SEQUEST algorithm. Peptide and protein identifications were filtered for a false discovery rate (FDR) of <1% at both the peptide and protein levels. Label-Free Quantification (LFQ) was performed to assess the relative abundance of proteins across samples.
RNA amplification and microarray hybridization for pico-scale microarray analysis
Total RNA was extracted from FACS-sorted alveolar macrophages using the TRIreagent according to the manufacturer’s instructions. Transcriptome profiling was performed using the GeneChip™ WT Pico Reagent Kit (Thermo Fisher Scientific), following the manufacturer’s protocol. Briefly, 100 pg to 10 ng of total RNA was reverse-transcribed into double-stranded cDNA using primers containing a T7 promoter sequence. The resulting cDNA was subjected to low-cycle polymerase chain reaction (PCR) amplification and
Quantitative RT-PCR
Total RNA was extracted from MSCs using TRI Reagent® (Molecular Research Center Inc., Cincinnati, OH, USA) and reverse-transcribed with the PrimeScript® RT Master Mix Kit (TAKARA Bio Inc., Shiga, Japan). Quantitative reverse transcription-polymerase chain reaction (qRT-PCR) was performed using the Perfect Real-Time SYBR Green II kit (TAKARA Bio Inc.) according to the manufacturer’s instructions. The following primer sequences were used:
Treatment of AvMs with miRNA mimic or siRNA
Sorted AvMs from healthy B6 mice were transfected with miRNA mimic or siRNA using Lipofectamine® RNAiMAX (Thermo Fisher Scientific) following the manufacturer’s instructions and cultured in 5%FCS-DMEM for 24 h. The siRNA blocking Hif1a were synthesized with the following sequences (Japan Bio Services Co., LTD., Saitama, Japan): sense sequence- GCU UUU UUC UCA GAA UGA AdTdT and antisense sequence- UUC AUU CUG AGA AAA AAG CdTdC.
Statistical analysis
All statistical analyses were performed using JMP Pro 18 (SAS Institute Inc., Tokyo, Japan). All animal experiments and
Results
Intratracheally transplanted MSCs transiently engraft in the lungs and decline in over time
To evaluate whether spray-based intratracheal transplantation enables the delivery of viable MSCs into the lung, we first assessed the efficiency of cell engraftment and distribution. A total of 1 × 106 EGFP-labeled MSCs derived from EGFP-transgenic mice were transplanted intratracheally using a spray device. Approximately 50,000 cells reached the distal periphery legion of the lung (Fig. 1a–c). Although the engraftment efficiency was limited, this result indicates that the transplanted MSCs reached the distal regions of the bronchial tree. To determine the retention of transplanted cells over time, the lungs were harvested at 24, 48, 72 h, and 1 week after transplantation. Following collagenase digestion, the remaining GFP-positive cells were quantified by flow cytometry. The number of engrafted cells declined to approximately 31,000 cells at 24 h, 1600 cells (0.16% of the transplanted MSCs) per 1 × 106 cell analysis at 48 h after cell transplantation. At 72 h after cell transplantation, the number of remaining cells further decreased to approximately 20 cells per 1 × 106 cell analysis, and then, only six cells were detectable in the lung at 1 week post-transplantation (Fig. 1c). To confirm whether the remaining MSC was viable, GFP-positive cells were isolated by FACS at 48 h post-transplantation and cultured

Confirmation of MSC delivery and retention in the lung after transplantation. (a) Region analyzed by flow cytometry. Cells were collected from the area outlined by the white dotted line and analyzed. (b) GFP-positive MSCs recovered from the periphery region of the lung. To evaluate the distribution of transplanted cells, mice were sacrificed 15 min after transplantation. (c) Retention of transplanted cells in the lung. Cells were isolated from approximately 50% of the lung tissue after removal of the major bronchi and connective tissue. Scores represent the mean ± SD of three independent biological replicates. The vertical axis is shown on a logarithmic scale. Asterisks denote statistically significant differences between groups at
Taken together, these results demonstrate that the spray-mediated intratracheal transplantation delivers viable MSCs to the lung including the alveolar regions, although the transplanted cells persist only for a limited period.
Intratracheal transplantation of MSCs attenuates the BLM-induced lung fibrosis
To evaluate the therapeutic efficacy of intratracheal MSC transplantation in a BLM-induced pneumonia model, we first established the model by intrapulmonary administration of BLM using a spray device. Lungs were harvested 3 weeks after MSC transplantation. Five mice were used: three were analyzed by histological examination, and the remaining two were subjected to proteomic analysis. To confirm the onset of fibrosis in the BLM-treated model, peripheral lung tissues were collected from three groups of mice: healthy controls, technical controls administered saline 48 h after BLM treatment, and MSC-treated mice administered MSCs 48 h after BLM treatment. The tissues were fixed in formalin and subjected to histological staining and immunofluorescence with an anti-αSMA antibody for microscopic evaluation. Histological analysis using Masson’s trichrome staining revealed that BLM-treated lungs exhibited typical fibrotic changes. In contrast, lungs from MSC-transplanted mice also displayed some degree of fibrosis compared with healthy controls; however, the staining intensity in fibrotic areas was reduced, indicating an attenuation of fibrotic progression (Fig. 2a). Moreover, in the BLM-treated model, proliferating fibrotic cells with high αSMA expression were observed. In MSC-transplanted mice, although fibrosis was still evident compared with healthy controls, the number of αSMA-positive cells was markedly reduced relative to saline-treated controls (Fig. 2b).

Development of fibrosis in BLM-treated lungs and suppression of inflammation/fibrosis markers by intratracheal MSC transplantation. (a) Representative lung histology at 3 weeks after bleomycin (BLM) treatment and MSC transplantation. Masson’s trichrome stained samples are shown; blue staining indicates deposition of fibrotic extracellular matrix. NTC refers to the non-treatment control. (b) Expression of the fibrosis marker αSMA in the lungs 3 weeks after treatment in healthy controls, saline-treated mice following BLM administration, and MSC-treated mice following BLM administration. The lower panels show magnified views of the areas outlined by white boxes in the upper panels. (c) Proteomic analysis of inflammation- and fibrosis-related markers in lungs 3 weeks after BLM treatment. The y-axis represents the relative expression level normalized to healthy lung (average of two normal samples are set as 1.0). Data represent the mean of two independent samples (N = 2). The BLM+TP group (dark blue bars) refers to lungs that received MSC transplantation 48 h after BLM treatment.
Proteomic comparison of lung tissues revealed that in BLM-treated lungs, known fibrosis-associated markers such as Fn1, Itgb2, and Casp1, as well as inflammation- and fibrosis-related proteins including Ptprc, and Syk, were significantly upregulated. In contrast, lungs from MSC-transplanted mice showed overall reduced expression of many of these signature markers compared with untreated BLM lungs. Although some proteins, such as Vav1 and Ctsz, remained elevated at more than 2-fold compared with healthy controls, the overall trend indicated suppression of inflammatory and fibrotic responses. These findings are consistent with previously reported anti-inflammatory and anti-fibrotic effects of MSC therapy (Fig. 2c).
MSC transplantation suppresses inflammatory gene expression and enhances stress resistance and proliferation-related pathways in alveolar macrophages
To investigate the effects of MSC transplantation on AvMs, we isolated AvMs by FACS at 1 and 3 weeks after BLM treatment. AvMs were identified by the expression of surface markers CD64, F4/80, and Siglecf (Fig. 3a). To characterize the population defined by CD64High/F4/80High/SiglecfHigh/CD11bNegative, we observed gene expression profile using microarray. We isolated CD64High/F4/80High/SiglecfHigh/CD11bNegative cells from the lung and compared their transcriptional profile with that of CD45+/CD11b+ monocytes/macrophages isolated from the bone marrow tissue as controls.

MSC transplantation attenuates the expression of pro-inflammatory and pro-fibrotic genes in alveolar macrophages. (a) Isolation and analysis of alveolar macrophages using CD64, F4/80, CD11b, and Siglecf as its surface markers. (b) Transcriptomic profiling of the CD64High/F4/80High/SiglecfHigh/Cd11bNegative population using pico-scale microarray analysis. CD45HighCD11bHigh monocytes isolated from bone marrow tissue were used as controls. Numbers in the heatmap indicate log2 expression values. (c) Transcriptomic profiles of alveolar macrophages (AvMs) were analyzed at 1 and 3 weeks after BLM treatment. Comparisons were made between saline-treated (MSC−) and MSC-transplanted (MSC+) groups. The table summarizes genes related to inflammation and fibrosis, including pro-inflammatory mediators, regulators of macrophage activation, and other inflammation-associated markers. In the MSC+ group, suppression of inflammation- and fibrosis-related genes was already evident at 1 week and persisted through 3 weeks. (d) Proportion of AvMs among total live cells at 96 h, 1 week, and 3 weeks after BLM treatment. The MSC-treated group showed a higher proportion at 1 week, but no difference was observed at 3 weeks. Scores represent the mean ± SD of three independent biological replicates. Asterisks indicate statistically significant differences between groups at
The CD64High/F4/80High/SiglecfHigh/CD11bNegative population exhibited lower expression of canonical monocyte and leukocyte markers, but showed high expression of AvM-specific markers previously reported (Fig. 3b). These findings support the validity of analyzing CD64High/F4/80High/SiglecfHigh/CD11bNegative cells as AvMs.
To investigate the direct effects of MSC transplantation on AvMs, we isolated AvMs and examined the expression of genes with a log2 fold change greater than 1 in AvMs from BLM-treated, untreated mice compared with healthy controls. These genes were associated with inflammation, fibrosis, and macrophage activation. By 3 weeks post-BLM treatment, expression levels of all these genes had decreased. Importantly, in MSC-transplanted mice, most of these pro-inflammatory and pro-fibrotic genes were already suppressed by 72 h post-transplantation (1 week after BLM treatment), and this suppression was maintained through 3 weeks after BLM treatment (Fig. 3c), indicating a sustained immunomodulatory effect of MSCs on AvMs.
Quantification of AvMs as a proportion of total lung cells revealed a marked reduction in AvM frequency in BLM-treated mice. By 96 h post-BLM treatment, AvMs had declined to approximately 10% of levels observed in healthy control lungs. This proportion gradually recovered to 50% at 1 week and the proportion of AvMs relative to total macrophages was restored to 65% of healthy lungs in the MSC-transplanted group at 3 weeks post-BLM treatment. In contrast, MSC-non transplanted mice showed a slower recovery (Fig. 3d, e).
MSC-derived lactate enhances Hif1a expression and promotes stress resistance in alveolar macrophages
To clarify the protective effects of MSC transplantation on AvM cell numbers, we analyzed gene expression changes related to macrophage survival, including oxidoreductase activity, pyroptosis, granzyme-mediated programmed cell death, and efferocytosis. Pyroptosis and granzyme-mediated cell death are forms of regulated cell death that negatively impact inflammation regulation. In contrast, efferocytosis, the process by which dead cells are engulfed, promotes anti-inflammatory responses and supports macrophage proliferation. At 1 week following BLM administration, transcriptomic analysis revealed that 22 out of 29 genes associated with oxidoreductase activity exhibited greater than 5-fold upregulation, suggesting a substantial enhancement of oxidative stress response pathways. With respect to pyroptosis-related genes, eight genes displayed more than a 2-fold change in expression in the MSC-transplanted group. Among these,

Lactic acid-induced Sesn2 may underlie the phenotypic changes observed in AvMs following MSC transplantation. (a) Effects of MSC transplantation on gene expression in AvMs at 1 week after BLM administration. MSC transplantation altered gene expressions associated with oxidative stress resistance, pyroptosis, and efferocytosis as defined by GO enrichment analysis. (b)
Transcriptomic analysis at 72 h post-MSC transplantation revealed a modest 1.35-fold increase in the expression of the lactic acid transporter
To investigate whether lactic acid regulates the stress-adaptive and pro-survival phenotype of AvMs, we treated FACS-sorted AvMs with 20 mM lactic acid for 24 h. Under these conditions, both Hif1a and Sesn2 expression levels were upregulated, and this effect was attenuated by treatment with the lactate transporter inhibitor AZD3965 (Fig. 4b). To confirm that lactic acid–induced upregulation of
Transplanted MSCs exhibited enhanced lactate-producing capacity and regulated AvMs through lactate as a mediator
To test the hypothesis that lactate functions as a mediator of the therapeutic effect of MSC transplantation, we first examined whether transplanted MSCs retained their lactate-producing capacity by assessing the gene expression of lactic acid dehydrogenases and a lactate exporter protein Mct4. The GFP-MSCs were transplanted into BLM-treated mice, and GFP-positive cells were isolated by FACS at 24 and 48 h post-transplantation for PCR analysis. At 24 h, transplanted MSCs showed increased expression of

Addition of the lactate transporter inhibitor AZD3965 suppresses MSC-induced expression of Hif1a and Sesn2. (a) Expression of lactate production- and export-related genes in transplanted MSCs. GFP-MSCs were isolated by FACS before transplantation (Pre-TP) and at 24 and 48 h post-transplantation, followed by qPCR analysis. Data represent the mean ± SD of three independent biological replicates. Asterisks indicate statistically significant differences between groups at
Discussion
MSC transplantation has already been tested in several clinical trials for severe pneumonia, with favorable therapeutic outcomes reported16,27–35. Moreover, intratracheal cell delivery has been clinically applied, and the scientific validity of this approach has been partially demonstrated and reproduced in previous studies36–39. In this study, we found that MSCs administered into the lungs act on AvMs via lactate as a mediator, thereby enhancing their viability and stress resistance.
AvMs play a pivotal role in maintaining lung homeostasis by eliminating pathogens and noxious particles without provoking inflammation; rather, they are thought to contribute to the resolution of inflammation and the maintenance of immune tolerance through the expression of anti-inflammatory mediators22,40,41. In infectious diseases, depletion of AvMs has been well documented. At this stage, AvM cell death plays a dual role: not only does it help restrict pathogen spread by eliminating infected host cells 42 , but it also provides apoptotic cells as substrates for efferocytosis, thereby indirectly suppressing excessive inflammation 43 . From this perspective, physiologically tolerable reduction in AvM numbers may be necessary. However, excessive depletion of AvMs has been shown to impair the therapeutic efficacy of granulocyte-macrophage colony-stimulating factor (GM-CSF) during infection 42 and to promote excessive neutrophil infiltration 44 , leading to heightened inflammation and lung injury. Therefore, preserving AvM numbers within a controllable range may be critical as a therapeutic strategy to prevent acute lung damage. Moreover, the occurrence of pyroptosis in AvMs poses a particularly detrimental threat to lung tissue integrity45,46.
A central question of this study was to elucidate the mechanisms by which MSCs influence tissue-resident macrophages. In the present study, we observed that MSC transplantation attenuated the reduction in AvM numbers. Emerging evidence has demonstrated that the death of alveolar macrophages plays a critical role in the progression of lung inflammation by modulating the activity of other immune cell populations within the pulmonary environment
47
. Given that AvMs are not highly proliferative cells, it is likely that MSC transplantation suppressed acute-phase cell death in AvMs. Transcriptomic analysis of AvMs 1 week after BLM treatment revealed increased expression of genes associated with stress resistance and cell survival. Among the most significantly altered genes, Sesn2 exhibited particularly prominent upregulation. Sesn2 is well recognized as a key regulator of cellular stress resistance48–51 and defense against pyroptosis52,53. In macrophages, Sesn2 not only protects from cell death
54
but also plays a critical role in multiple phagocytosis processes
55
, thereby contributing to the maintenance of macrophage homeostasis under inflammatory conditions56,57. Previous studies have demonstrated that Sesn2 protects AvMs and alleviates lung injury in lipopolysaccharide (LPS)-induced acute lung inflammation models
55
. Sesn2 is capable of regulating all of the responses observed in this study, including resistance to oxidative stress, regulation of programmed cell death, and enhancement of phagocytic activity. However, the mechanisms by which MSCs upregulate Sesn2 in target cells remain poorly understood. Both our current findings and previous reports indicate that the survival and persistence of MSCs
One plausible mechanism linking lactic acid to the upregulation of Sesn2 involves the transcription factor Hif1a. Hif1a is widely recognized as a key transcriptional regulator activated under hypoxic conditions. Interestingly, several studies have demonstrated that lactic acid can upregulate Hif1a expression even in normoxic environments66,67. Hif1a, in turn, has been identified as a transcriptional regulator of Sesn2, suggesting a potential mechanism by which MSC-derived lactic acid may promote Sesn2 expression in target cells49,51,68. The role of Hif1a as a key regulator of Sesn2 was further demonstrated in this study, as siRNA-mediated silencing of
Importantly, the MSC-induced upregulation of
Limitations of this study
This study has at least three limitations. First, we defined AvMs based on CD64+/F4/80+/Siglecf+ markers; however, previous studies have suggested that interstitial or monocyte-derived macrophages may transiently acquire AvM-like characteristics during lung repair. Thus, the marker set used in this study may have captured a heterogeneous population of AvMs comprising cells at different stages of differentiation or with distinct phenotypic characteristics. To more directly analyze the effects of MSCs on AvMs, further investigation, including single cell–level analyses, will be required to determine whether these heterogeneous populations contribute to the MSC-induced protective phenotype.
Second, our observation period was limited to 3 weeks, and it is likely that,
Third, the mechanism demonstrated in this study is limited to the interaction between MSCs and AvMs. Although MSCs have shown efficacy in other inflammation-related diseases, such as Crohn’s disease and spinal cord injury, the characteristics of tissue-resident macrophages that regulate inflammation vary across organs. It is, therefore, possible that macrophages in different tissues respond to MSCs in distinct ways. Moreover, MSCs are known to exert therapeutic effects through multiple pathways, including cytokine secretion and the transfer of microRNAs via extracellular vesicles. Thus, it is important to acknowledge that the anti-inflammatory and anti-fibrotic effects of MSCs are not mediated by lactate alone, but rather result from a complex interplay of diverse molecular signals.
Conclusions
In conclusion, intratracheal transplantation of MSCs exerts a protective effect on alveolar macrophages via lactic acid transfer and subsequent upregulation of hypoxia-inducible factor 1-alpha (HIF-1α). Given that lactic acid is rapidly metabolized
Footnotes
Acknowledgements
We thank Mr. Atsuhiro Tatemizo, Mr. Kentaro Egawa, and Mr. Nobuyuki Mizuguchi, Life Science Institute in Kindai University, who provided technical support for animal studies. We also thank Mr. Shinji Kurashimo, Life Science Institute in Kindai University, who provided technical support for FACS analysis.
Ethical Considerations
All procedures related to animal handling, care, surgery, and sacrifice were approved by the Institutional Animal Care and Use Committee of Kindai University as the approved research project number KAME2025-044 and performed in accordance with institutional guidelines and regulations.
Author Contributions
All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by MEXT/JSPS KAKENHI Grant-in-Aid for Scientific Research (B) 23H03034 (T.Teramura).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
