Abstract
Stem cell-based therapy is considered to be a new approach for the treatment of cerebral palsy (CP). Given the potent anti-inflammatory activity and high regenerative potential of M2 macrophages, these cells may be an alternative source for cell transplantation. To evaluate the safety and efficacy of autologous M2 macrophages, we conducted a pilot clinical trial in 21 children with severe CP. The primary outcome measure was safety, which included assessment of mortality of any cause, immediate adverse reactions, and serious adverse effects and comorbidities during 5-year follow-up. The secondary outcome measure was functional improvement in Gross Motor Function Measure (66-item GMFM) test, Peabody Developmental Motor Scale-Fine Motor (PDMS-FM) test, Ashworth scale, MRC scale, and an easy-to-understand questionnaire for evaluation of cognitive functions in our modification. Intradural injection of M2 cells (in mean dose of 0.8 × 106/kg) into the lumbar spinal area did not induce any serious adverse events. No cases of mortality, psychomotor worsening, exacerbation of seizures, and long-term comorbidities, including tumors, were observed during a 5-year follow-up. After 3 months, GMFM score increased from 13.7 ± 7.8 to 58.6 ± 14.6, PDMS-FM score improved from 0.76 ± 0.42 to 5.05 ± 0.97, and the Ashworth score decreased from 3.8 ± 0.21 to 3.3 ± 0.24. Along with gross and fine motor function enhancement, an improvement of cognitive activity (from 1.62 ± 0.41 to 4.05 ± 0.64, according to questionnaire assessment) and reduction of seizure syndrome were registered as well. The neurological improvements did not diminish during the 5-year follow-up period. The data obtained suggest that cell therapy based on M2 macrophages is safe, does not induce early adverse effects and long-term comorbidities, and is accompanied with a significant improvement of motor and cognitive activities in severe CP patients. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Cerebral palsy (CP) is a heterogeneous group of disorders that are defined as a chronic nonprogressive motor disability due to a problem in the developing brain (30). It may be closely associated with natal development abnormalities and result from brain injury occurring during the prenatal, perinatal, or postnatal periods (2,31,42). Since this disease does not substantially reduce the life span, CP is an important social and economic problem and may exert a heavy burden on their families and society (32).
Currently there are no effective treatments for CP (29) that can substantially improve motor and cognitive functions in patients with moderate to severe CP (16,29), and most CP children require lifelong medical and physical care. In this aspect, cell-based therapy is considered to be a new perspective approach for treatment of CP (4,8,30). There are many potential cell sources that demonstrate benefit in experimental models of brain injury. Most of them include various types of stem cells or their derivatives (7,22). The promising results of stem cell therapy in CP have been demonstrated in the first clinical trials using olfactory ensheathing cells, neural progenitor cells, umbilical cord blood stem cells, and neural stem cell-like cells (9,10,23,27). However, it is now clear that along with stem cells, the capacity to promote neural regeneration is also demonstrated for immune cells, in particular, for macrophages, known to play a major role in tissue repair. The role of macrophages in central nervous system (CNS) pathology is controversial, since these cells can promote both injury and repair (41). These opposite effects may be caused by distinct macrophage subsets. Actually, there are at least two distinct functional macrophage subpopulations: “classical” proinflammatory (M1) and anti-inflammatory (M2) macrophages. M1 cells support inflammation, mediate resistance to intracellular pathogens and tumor resistance, but are tissue destructive. In contrast, M2 cells display anti-inflammatory/immunoregulatory activity, stimulate angiogenesis, and activate tissue repair (14,24,28). In addition, M2 macrophages induce predominantly T helper 2 cell (Th2) response that is particularly beneficial in the repair of the CNS (15). Recent investigations in the spinal cord injury (SCI) model have shown that only M1 macrophages are neurotoxic, and M2 macrophages promote axon growth and functional improvement (18). Similarly, experimental studies in a stroke model have demonstrated that M2 cells protect neurons from apoptosis (17). Given the fact that adoptive transfer of macrophages improves neurological recovery in CNS injury models, and infiltrating monocyte-derived macrophages play a major role in promoting the M2 response (35), transplantation of M2 cells seems to be a prospective strategy for CP treatment.
The major promise of M2 macrophages is that they can downregulate the inflammatory response known to play a major role in the pathogenesis of perinatal brain injury (5,39). Actually, proinflammatory mediators can stimulate a cascade of secondary responses, including fluid– electrolyte imbalance, regional blood flow alterations, calcium-mediated cellular injury, glutamate-induced excitotoxicity, disturbances of mitochondrion function, and disruption of the blood–brain barrier resulting in cell apoptosis (25). Another aspect is the capacity of M2 cells to produce a wide range of cytokines (11) capable of promoting neuroprotection and activating angiogenesis and neuroregeneration (40).
Recently, we have designed a new protocol for the generation of M2-like macrophages in low growth factor conditions. The cells obtained had low antigen-presenting and proinflammatory activity and possessed considerable regenerative potential [in particular, produced high amounts of insulin-like growth factor (IGF-1) and vascular endothelial growth factor (VEGF)] (11). In the current study, we analyzed the safety of M2 cell transplantation in severe CP patients, including immediate adverse reactions and possible long-term side effects and comorbidities during 5-year follow-up, and evaluated the efficacy of transplanted M2 cells in neurological improvement.
Materials and Methods
Study Design and Patient Enrollment
An open-label, phase I/II, noncontrolled clinical study of chronic children who had severe CP was conducted. The study was initiated in May 2008 and the follow-up was for 5 years. The purpose of this study was to assess the safety and therapeutic efficacy of M2-like macrophages for treatment of CP patients. A written informed consent was obtained from the patient's parents before the therapy. Permission for the present study was granted from the Institutional Academic Board and Institutional Review Board (Local Ethics Committee). Rehabilitation intervention like physiotherapy was continued after the stem cell therapy.
According to the study protocol, the inclusion criteria were 1) age ≥12 months and ≤8 years; 2) diagnosis: CP; 3) performance status: Gross Motor Function Classification System: level IV–V; 4) parental consent. The exclusion criteria were 1) autism and autistic spectrum disorders without motor disability; 2) progressive neurologic disease; 3) HIV or uncontrolled bacterial, fungal, or viral infections; 4) impaired renal or liver function (as determined by serum creatinine >1.5 mg/dl and/or total bilirubin >1.3 mg/dl); 5) genetic disease or phenotypic evidence of a genetic disease on physical examination; 6) requires ventilatory support; 7) unable to obtain parental consent. The patients did not pay for their inclusion or treatment in this study.
Generated macrophages were injected via lumbar puncture. All patients were examined before cell therapy, after 3 months, and followed up for the following 5-year period.
Macrophage Generation and Introduction
The generation of macrophages from plastic-adherent cells obtained from 20–50 ml of peripheral blood (depending on patient weight, and it never exceeded 5% of blood volume) was performed according to a previously developed protocol (11). In brief, adherent cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 (Sigma-Aldrich, St. Louis, MO, USA) with supplements (2 mM l-glutamine, 50 μM 2-mercaptoethanol, 100 mkg/ml gentamycin, 10 mM HEPES, 2 mM sodium pyruvate, 1% nonessential amino acids; all reagents from Sigma-Aldrich) at 37°C with 5% CO2. To obtain M2-like macrophages, we used recombinant human granulocyte macrophage colony-stimulating factor (rhGM-CSF, 50 ng/ml, R&D Systems, Minneapolis, MN, USA) and serum deprivation conditions (low percent of autologous plasma). In 7 days, the macrophages were harvested by using ethylenediaminetetraacetic acid (EDTA, Sigma-Aldrich) in Hank's balanced salt solution (Sigma-Aldrich), washed, and counted. The generated M2-like macrophages were resuspended in 2 ml 0.9% sodium chloride (BioloT, St. Petersburg Russia) and infused into the spinal cord fluid of the patient. For this, the thecal sac was punctured in the L4–L5 lumbar space, and cells were introduced through a spinal needle.
Measurement of Safety and Efficacy
The primary outcome measure was safety, which included assessment of mortality of any cause and immediate adverse reactions and long-term side effects and comorbidities during 5-year follow-up. The secondary outcome measure was functional improvement in neurological scales, including Gross Motor Function Measure (66-item GMFM) test, Peabody Developmental Motor Scale-Fine Motor (PDMS-FM) test, Ashworth scale, Medical Research Council (MRC) Weakness scale, and an easy-to-understand unified questionnaire for evaluation of cognitive functions in our modification. Gross motor abilities were characterized by six functions (head controlling, rolling, crawling, sitting, standing, and walking). Fine motor development was evaluated on five functions (hand movement in the midline, active grasping of objects, shifting the objects from one hand to the other, pinching of tiny objects, and “eye–hand” coordination). The five-point Ashworth scale was used for evaluating degree of spasticity, and the six-point MRC Weakness Scale served for muscle strength estimation. Cognitive functions were estimated based on six functions (understanding addressed speech, speaking of single words, aggression, contact with outsiders, recognizing relatives, meaningful glance).
To evaluate the influence of M2 therapy on endogenous neurotrophic factor production, the samples of patient serum obtained before and a month after macrophage introduction were collected and frozen at −80°C until the measurement. The concentration of cytokines and growth factors was determined using ELISA following the instructions of manufacturers: interferon (IFN)-γ, interleukin (IL)-17, IL-4 (all from Protein Contour, St. Petersburg, Russia), brain-derived neurotrophic factor (BDNF; R&D Systems), and VEGF (Invitrogen Corp., Carlsbad, CA, USA).
Statistical Analysis
The data were expressed as medians and interquartile range (in brackets). Statistica 6.0 software for Windows (StatSoft, Inc., Tulsa, OK, USA) was used for analysis of data. Exact Fisher test was used to determine relationships between categorical variables; Wilcoxon matched pairs test and Mann–Whitney U-test were used to compare nonparametric values.
Results
As shown in Table 1, the study group included 21 children with severe CP (including 15 boys and 6 girls) with mean age of 4.9 years. The examined children formed a fairly homogenous group with predominantly spastic forms of CP (spastic quadriplegia was revealed in 16/21 children). The majority of children had the fifth level of movement abnormalities estimated by the Gross Motor Function Classification System. These children did not keep their head and back straight, all motor functions were limited, and these defects were not compensated by adaptive equipment and assistive technology. The GMFM-66 score at entry was 13.7 ± 7.8. Evaluating degree of spasticity based on Ashworth scale was evidenced with a considerable (4–5 points) increase in muscle tone in 19 out of 21 (90.5%) of CP children with an average Ashworth score of 3.8 ± 0.21. One child had difficulties even in passive movements, and spasticity achieved five points.
Baseline Characteristics of Participants

BW, body weight; GMFM, Gross Motor Function Measure; MRC, Medical Research Council Scale; qpl, quadriplegia; dpl, diplegia.
The MRC weakness score reflecting muscle strength in forearms was 2.0 ± 0.19, which indicated the marked reduction in muscle strength in all children at baseline. Seven children had epileptic seizures requiring anticonvulsant therapy. Mental faculties were impaired in practically all patients. Actually, 19 patients (90%) were unable to speak, and 16 children (76%) could not understand the addressed speech.
All patients received one grafting of autologous M2-like macrophages generated from peripheral blood according to protocol (see Materials and Methods). Mean cell yield was 77.1 ± 13.4 × 103 from 1 × 106 peripheral blood mononuclear cells. The dose of injected M2-like macrophages ranged from 0.18 to 2.15 × 106/kg and was on average 0.8 ± 0.12 × 106/kg (Table 2). The viability of obtained cells in all cases was never less than 93%. Patients were evaluated for neurological changes 3 months after therapy and further monitored up to 5 years.
Early and Long-Term (During the 5-year Study Period) Adverse Events and Comorbidities

Endolumbar administration of M2-like cells was generally well tolerated. Although 14 (67%) patients experienced fever and one to two episodes of vomiting, these cell therapy-related reactions were observed during the first 2 days and were easily controlled with Dexasone (Nycomed, Linz, Austria) and Metoclopramide (Polpharma, Warsaw, Poland). No cases of immediate severe adverse events such as allergic reactions, local hematoma, or infections at the site of lumbar puncture, meningeal reactions, and systemic inflammatory or toxic complications were noted after cell introduction. At the 3-month follow-up, no signs of neurological worsening, appearance of new seizure episodes, or increase in the frequency of existing episodes as well as other serious comorbidities (dyspepsia, infections, etc.) were observed. However, one child demonstrated the exacerbation of atopic dermatitis. To the end of the 5-year follow-up period, all patients were alive and had no long-term comorbidities and tumors.
After 3 months, a significant improvement in most of the evaluated functional measures was registered. Overall therapeutic responses are shown in Tables 3 and 4. It is evident from Table 3 that 3 months after cell therapy a significant decrease in spasticity was revealed, while the muscle strength as well as gross and fine motor activities were enhanced (p < 0.01). In addition, cell therapy resulted in clear improvement of cognitive functions (p < 0.01).
Functional Scores of CP Children Over a 3-Month Observation Period

Values are median and interquartile range;
p < 0.05,
p < 0.01 (Wilcoxon matched pairs test).
Improvement in Motor and Mental Activities Over a 3-Month Observation Period
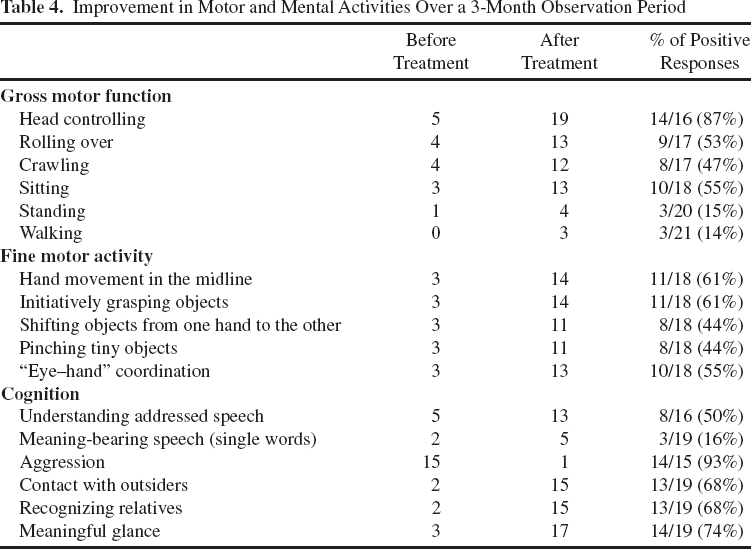
Apparent clinical improvements were noted in 15 out of 21 cell-grafted CP children (71.4%; responding group). With the cell-based therapy, 14 children out of 16 who were initially unable to independently hold their head in the vertical position became able to consistently execute this function (Table 4). The majority of CP children enrolled in our investigation (17/21; 81%) had no capacity to crawl or roll over. With M2 therapy, eight children (47%) became able to crawl/move on their back, and nine children (53%) showed a clear ability to roll over on their abdomen from a supine position. Moreover, CP children treated with M2 cells displayed an obvious improvement in object (toy) manipulations, grasping, and visual–motor integration.
The beneficial effects of cell therapy appeared not only in motor activities but also in cognitive/mental functions. In fact, we observed a decrease in aggression (14/15) and improvement of contact with outsiders (17/19). Eight of 16 children began to understand addressed speech, while three of five improved their understanding of the addressed speech, and 5 of 19 children showed the appearance of meaning-bearing speech. Of importance, three of eight children (37.5%) with seizure syndrome (up to 10 episodes per month) experienced seizure arrest, which persisted after discontinuation of anticonvulsants; another three children exhibited significantly reduced seizure frequency (from 7–10 to 1 episode per month).
It should be noted that studied children were characterized by prolonged episodes of low-grade temperature not always associated with respiratory infections. After cell therapy, the parents of most children (75%) noticed a decline in the number of respiratory infections (up to their complete disappearance in 47%) or reducing the periods of low-grade temperature.
The improvement of motor functions and mental abilities appeared quite early (from several days to 1 month) and was clearly manifested at 3 months and did not decline during the 5-year follow-up period.
By comparing children that responded and did not respond to cell therapy, we revealed that the better outcomes did not depend on some demographic parameters (namely, age or sex) and severity of the neurological deficit as well as the number of input cells. However, the children who responded to cell therapy showed the higher rate of cytokine reactions, which were observed two times more often (in 85% vs. 38%, p = 0.07) than in the nonresponding children.
Since clinical improvements in the majority of patients were observed in a month after therapy, we hypothesized such quick effects were probably mediated by endogenous neurotrophic factors. To test this, we examined some cytokines and growth factors in the serum of CP children 1 month after M2 therapy. The data summarized in Table 5 showed that the macrophage introduction was not accompanied by an increased production of IFN-γ, IL-17, and IL-4. At the same time, such a therapy resulted in significantly higher levels of brain-derived neurotrophic factor (BDNF; pU = 0.015) and a strong tendency to increasing VEGF (pU = 0.07). It is of great importance that these changes were the most pronounced in the responding group.
Cytokine and Growth Factor Levels in the Serum of 16 Children With Severe CP

Values are median and interquartile range. BDNF, brain-derived neurotrophic factor; VEGF, vascular endothelial growth factor; IFN, interferon; IL, interleukin; <OOR, out of range (the minimum detectable does of IL-17 is 5 pg/ml).
p < 0.05 (Wilcoxon matched pairs test).
Discussion
Recent studies have demonstrated that cell-based therapy can improve neurological functions in various neuropathologies including CP (1,8,37). Along with stem cells and their derivatives, a major role of M2 anti-inflammatory macrophages in promoting neural repair was documented (19,35). The idea of a beneficial role of macrophages in CNS repair was firstly proposed by the Schwartz group, who demonstrated that implantation of the macrophages into transected rat spinal cord stimulated tissue repair and partial recovery of motor function (33). Later, this group initiated a pilot Phase I study of M2-like macrophage implantation in patients with acute complete SCI. The injection of activated macrophages into the patient's spinal cord was concluded to be well tolerated (20), but a phase II randomized controlled multicenter trial failed to show a clinical improvement (21). This fact could be partially explained by the invasiveness of the implanting procedure. Nevertheless, this clinical trial was a major achievement to transfer macrophage-based therapy into the clinic. Immunomodulation by M2 macrophages may partially underlie the effect of stem cell-based therapy, since many transplanted cells die from apoptosis, and engulfment of apoptotic cells induces M2 polarization (38). Another possibility is that transplanting mesenchymal stem cells can “educate” macrophages toward an immunoregulatory phenotype (12).
The present study provides the first evidence for the possible application of M2 macrophages for treatment of CP. We utilized our original approach to obtained M2 cells using low growth factor conditions (11) and have shown that M2 macrophages may be successfully generated in children with severe CP. Introduction of these M2 macrophages via lumbar puncture was shown to be safe, well tolerated, and did not result in immediate adverse reactions and long-term side effects and comorbidities. Aggravation of atopic dermatitis was only registered in one person. We attribute this to the possible Th2-stimulating capacity of M2 cells (26). Certainly, this fact requires a careful examination of patients for allergic diseases and may be exclusion criteria for M2 macrophage application in children with severe and diffuse forms of atopic pathology. On the other hand, exacerbation of atopic dermatitis in only 1 out of 21 cases evidenced that endolumbar application of macrophages did not obviously induce systemic activation of Th2 response. Indeed, we did not observe any enhancement of IL-4 serum level after introduction of M2 macrophages. The important result of the present work is the analysis of long-term adverse effects and comorbidities after 5-year follow-up. The data obtained have shown that there was not any case of neurological worsening, appearance of new episodes or enhancement of seizures, or development of tumor during the 5-year follow-up.
In our phase I study, there was no control group, and the only possibility to evaluate the effect of transplanted cells was comparison of motor function and cognitive abilities before and after therapy. M2 macrophage implantation was accompanied by significant decrease in spasticity and enhancement of muscle strength and GMFM scores. In addition, we demonstrated a marked improvement in cognitive functions. Enhancement of mental functions was manifested by appearance of the capacity to understand (in half of the patients) and improvement of speaking (in 26% of patients). In addition, irritability and aggressiveness as well as improvement in communication were found to be decreased in virtually all children. Importantly, functional improvement appeared quite early (from some days to 1 month), which was consistent with the observations of other researchers (9,10,23). Once they appeared, the improvements did not decrease with time and were observed throughout the 5-year follow-up period. Given that the majority of patients recruited to our clinical trial had the fifth level of disability, did not display any improvement after 3–6 months of standard rehabilitation training before cell therapy, were older than 3 years in 66%, and displayed early manifestation of the improvement, we suggested that the positive effects were not the result of rehabilitation therapy or age-related development, but were primarily mediated by the transplanted cells.
Of importance is the fact that M2 therapy ameliorates seizure syndrome. This is confirmed by full arrest of seizures in three persons and pronounced decrease in the rate of seizure episodes in another three children out of eight patients having seizure syndrome. Another interesting observation (according to parents' opinion) was that most of the children following therapy significantly reduced the rate of virus respiratory infections and/or duration of low-grade fever episodes.
Our results concerning the possibility of cell therapy to improve psychomotor functions in CP are consistent with others. So Chen et al. showed that olfactory ensheathing cells derived from aborted fetal tissue and injected into the bilateral corona radiata in the frontal lobes resulted in significant increase in a GMFM score and improvement of mental functions according to the caregiver questionnaire scale score in CP (10). Later, Luan et al. demonstrated similar results utilizing intracerebral implantation of allogeneic neural progenitor cells (23). The efficacy of cord blood stem cells was recently demonstrated by Min et al., who showed amelioration of motor and cognitive impairments following infusion of allogeneic umbilical cord blood cells potentiated with recombinant human erythropoietin (rhEPO) (27).
Currently, several mechanisms of cell-based therapy, which can improve functions in damaged brain including CP, are postulated. These comprise releasing growth and differentiation factors enhancing the local trophic milieu, neuronal regeneration, angiogenesis, decrease inflammation, and decrease neuronal apoptosis. According to our previous data, M2 cells are capable of spontaneous production of BDNF, IGF-1, epidermal growth factor, basic fibroblast growth factor, granulocyte colony-stimulating factor, erythropoietin, and VEGF (11) that stimulate endogenous neuroregenerative mechanisms, including activation of the “silent” synapses (6,18,19,36,40). The developing brain is considered to have a significant number of synapses with normal anatomic structures that are kept in a silent state, and their activation upon stimulation by neurotrophic factors may underline quick neurological improvement and explain stronger plasticity of child brain compared with adults. In addition, we do not rule out completely the role of neuroprotection, since neuroinflammation resulting in cell damage and apoptosis plays a major role in the pathogenesis of CP (25). Stimulation of CNS repair in ischemic brain damage may also be the result of increased angiogenesis and vasculogenesis (3,43). Therefore, our findings concerning an increase in serum levels of BDNF and VEGF after cell therapy can argue this hypothesis. Recent findings also showed the capacity of monocytes/macrophages to differentiate into endothelium-like cells and function as precursors of endothelial cells (34) participating in vascular repair (13). So both macrophages themselves and endotheliumlike cells as their derivatives may participate in activating angiogenesis, which is closely related to neurogenesis. Finally, given the major role of inflammatory mediators in pathogenesis of CP (25), functional improvement may be mediated by the immunomodulatory activity of M2 cells.
The advantage of M2 macrophages is safety and feasibility of procedure, since it provides the utilization of autologous cells generated from peripheral blood, and endolumbar delivery that is less invasive compared to intracerebral implantation of candidate cells. Another privilege is that M2 macrophage transplantation has a stabilizing effect and may be carried out in children with seizure syndrome. However, to better define the therapeutic effects of M2 macrophages and to elucidate the mechanism of M2 cells in CP, further studies should be performed.
Footnotes
Acknowledgments
This study was funded by Russian Foundation for Basic Research (N 13-04-00113). The authors declare no conflicts of interest.
