Abstract
Hepatocyte-transplantation is a therapeutic approach for diverse acute and chronic liver diseases. As availability of primary cells is limited, there is an increasing demand for hepatocyte-like cells (e.g., neohepatocytes generated from peripheral blood monocytes). The aim of this study was to evaluate the effects of six different human AB sera, fetal calf serum, or autologous serum on production of neohepatocytes. The yield and quality of neohepatocytes varied considerably depending on the different sera. Using autologous sera for the whole production process we constantly generated the highest amount of cells with the highest metabolic activity for phase I (e.g., CYP1A1/2, CYP3A4) and phase II enzymes (e.g., glutathione-S-transferase). Moreover, similar effects were seen examining glucose and urea metabolism. Especially, glucose-6-phosphatase and PAS staining showed distinct serum-dependent differences. The role of macrophage activation was investigated by measuring the secretion of TNF-α, TGF-β, and RANKL, MMP activity, as well as mRNA levels of different interleukins in programmable cells of monocytic origin (PCMO). Our data clearly demonstrate that the use of autologous serum reduced initial macrophage activation in PCMOs and subsequently improved both yield and function of differentiated neohepatocytes. The autologous approach presented here might also be useful in other stem cell preparation processes where cell activation during generation shall be kept to a minimum.
Keywords
Introduction
Worldwide, millions of patients are suffering from chronic liver disease, with clinical manifestations leading from fibrosis over cirrhosis to hepatocellular carcinoma. The main causes of chronic liver disease in industrialized countries include viral infections, alcohol abuse, and nonalcoholic steatohepatitis (NASH). So far the only definite treatment for end-stage liver disease is liver transplantation, which is often associated with long-term intensive care of the patients. The major limitation of this technique, however, is donor organ scarcity. Thus, prevention or treatment of liver damage has a high medical need in our society because treatment costs are soaring. Hepatocyte transplantation has been used successfully in order to bridge patients to whole organ transplantation (11,12,38,40), to decrease mortality in acute liver failure (32), or for treatment of metabolic liver disease (38). The main advantage of cell therapeutic approaches to treat liver disease is that transplantation of cells is less invasive than whole-organ transplantation and can be performed repeatedly.
Although promising results have been achieved with primary hepatocytes, further application of this technology is hampered mainly due to limited availability of fresh hepatocytes (23). A wider use of this technique will not be possible until adequate numbers of cells for transplantation become more readily available. Hence, several efforts have been made to generate hepatocyte-like cells from other sources (e.g., monocyte-derived neohepatocytes) (21,26,30,36,41,43,44). Recently, we have shown that injection of monocyte-derived hepatocyte-like cells can increase the survival in rats after extended liver resection (16). Although neohepatocytes seem to be an ideal cellular system suitable for therapy of liver failure, the yield of generated cells is too small to perform repeated injections in patients. Furthermore, functional hepatocyte characteristics (e.g., phase I and II drug metabolism) were shown to be significantly lower in neohepatocytes compared to primary human hepatocytes (14). Thus, the aim of this study was to optimize the yield and function of neohepatocytes for autologous cell therapy (9).
We propose that macrophage activation of mononuclear cells by allogenic sera interferes with the dedifferentiation step of programmable cells of monocytic origin (PCMOs) as well as with the differentiation into hepatocyte-like cells. Therefore, we investigated the effects of six different human AB sera, fetal calf serum (FCS), and the donor's own autologous serum on macrophage activation of PCMOs as well as the yield, urea and glucose metabolism, and phase I and II enzyme activities of the corresponding neohepatocytes. In the experimental set-up, the use of autologous serum might be especially interesting for using these neohepatocytes for autologous cell therapy such as during acute liver failure or visceral bleeding.
Materials and Methods
Human recombinant fibroblast growth factor (FGF)-4, interleukin (IL)-3, macrophage colony-stimulating factor (M-CSF), and active transforming growth factor (TGF)- β1 were from Peprotech (London, UK); cell culture medium and supplements were from PAA Laboratories (Cölbe, Germany), and chemicals were obtained from Sigma (Munich, Germany).
Generation and Culture of Neohepatocytes
Briefly, peripheral blood monocytes (PBMCs) were isolated by density gradient centrifugation from whole blood donations (obtained from the Blood Donor Service, Bavarian Red Cross, Munich, Germany). Mononuclear cells (100,000 cells/cm2) were cultured in RPMI-1640 medium (10% serum, 2 mM L-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 5 ng/ml M-CSF, 0.4 ng/ml IL-3). After 6 days medium was changed to RPMI-1640 medium (10% serum, 2 mM L-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 3 ng/ml FGF-4). Culture medium was replaced every third day (14,36). Prior to all experiments cells were serum starved overnight.
Isolation of Primary Human Hepatocytes
Human liver tissue was obtained according to the institutional guidelines from liver resections of tumor patients with primary or secondary liver tumors (Technische Universität Munich, MRI, Munich, Germany). Written informed consent was obtained from all patients. Human hepatocytes were isolated by a two-step collagenase perfusion technique followed by Percoll gradient centrifugation for purification (31. Hepatocyte viability (trypan blue exclusion) was consistently above 90%. Hepatocytes were cultured on collagen-coated culture plates in Williams' E medium [10% FCS, 1 mM insulin, 15 mM HEPES, 0.8 μg/ml fortecortin (9-fluor-11β,17,21-trihydroxy-16α-methyl-1,4-pregnadien-3,20-dion), 100 U/ml penicillin, and 100 mg/ml streptomycin]. Prior to all experiments cells were serum starved overnight to avoid interference between serum components and the substrates.
Urea Measurement
Cells were washed three times with PBS before incubation for 24 h with DPBS (1 mM MgCl2, 1 mM Na-pyruvate ± 5 mM NH4Cl ± 1 mM ornithine). Supernatant (80 μl) was incubated with 60 μl 0.0002% O-phthaldehyde solution (0.03% Brij-35, 7.4% H2SO4) and 60 (0.l NED reagent [0.0006% N-(1-naphthyl)ethylenediamine dihydrochloride, 0.5% boric acid, 0.03% Brij-35, 22.2% H2SO4] for 2 h at 37°C. Absorbance was measured at 505 nm and compared to standard samples (22).
Glucose Measurement, Periodic Acid Schiff (PAS), and Glucose-6-Phosphatase (G6Pase) Staining
Cells were washed three times with PBS before incubation for 24 h with DPBS (30 mM KCl, 1 mM MgCl2, 1 mM Na-pyruvate ± 10 mM Na-L-lactate). Supernatant (100 μl) was incubated with 150 μl GLOX solution (250 mM Tris, 0.2 mM EDTA, 0.04% glucose-oxidase, 0.007% peroxidase, 0.01% O-dianisidine, pH 8.0) for 2 h at 37°C. Absorbance was measured at 420 nm and compared to standard samples.
For PAS and G6Pase staining, cells were first fixed with 3.7% formaldehyde for 5 min. Then cells were incubated with 0.1% periodic acid solution for 5 min, followed by incubation with Schiffs reagent for 15 min for color (pink) formation. For G6Pase staining, cells were incubated with G6Pase staining solution (100 mM Tris, pH 6.5, 0.06% G6Pase, 0.08% lead-II-nitrate) for 45 min, followed by incubation with 1% ammonium sulfide solution for 30 s for color (brown/black) formation. Pictures were taken with an inverted microscope (26).
Phase I and II Enzyme Activity Assays
Fluorescence-based P450 assays were performed by incubation of intact cells with selected substrates as reported. Briefly, cells cultured on a 96-well plate were serum starved overnight prior to measurement. For measurement culture medium was replaced with 100 μl reaction buffer [1 mM Na2HPO4, 137 mM NaCl, 5 mM KCl, 0.5 mM MgCl2, 2 mM CaCl2, 10 mM O-(+)-glucose, 10 mM HEPES, pH 7.4) containing the fluorogenic substrate as summarized in Table 1. Fluorescence was measured every 10 min over a period of 2 h with the help of a microplate reader (BMG Labtech, Offenburg, Germany). Afterwards cells were fixed for protein quantification by sulforhodamine B (SRB) staining as previously described (14). Results are given as pmol of fluorescent product formed (phase I enzyme activities) or fluorescent substrate reduced (for phase II enzyme activities) per minute normalized to total protein content in mg. Methanol fixed cells were used as negative control.
Reaction Conditions for Phase I and II Enzyme Activities

CYP, cytochrome p450; AMMC, 3-[2-(N,N-diethyl-N-methylamino)ethyl]-7-methoxy-4-methylcoumarin; AHMC, (3-[2-(N,N-diethyl-N-methylam-monium)ethyl]-7-hydroxy-4-methylcoumarin; UDP, uridine diphospho; CHC, 7-hydroxy-3-cyanocoumarin; EFC, 7-ethoxy-4-trifluoromethylcoumarin; HFC, 7-hydroxy-4-trifluoro-methylcoumarin; MFC, 7-methoxy-4-trifluoromethylcoumarin; BFC, 7-benzyloxy-4-trifluoromethylcoumarin.
TGF-fi, Tumor Necrosis Factor-α (TNF-α), and Receptor Activator for Nuclear Factor κB Ligand (RANKL) Measurement
PCMOs were washed three times with DPBS before incubation with serum-free culture medium (RPMI-1640, 2 mM L-glutamine) for 48 h at 37°C. TGF-β levels were measured by TGF-α reporter cells (MFB-F11), provided by Dr. Tesseur, as previously described (42). Briefly, MFB-F11 cells were cultured for 48 h with PCMO culture supernatants. Resulting secreted alkaline phosphatase (SEAP) activity in the culture supernatant was measured according to the manufacturer's instructions with the Great EscAPe™ SEAP fluorescent detection kit. TNF-α and RANKL levels were measured by ELISA according to the manufacturer's protocol (Peprotech, London, UK).
Zymography
PCMOs were washed twice with DPBS before incubation with serum-free medium for 48 h. Culture supernatant was mixed 1:3 with loading buffer [2% sodium dodecyl sulfate (SDS), 10% glycerol, 0.01% bromophenol blue] and transferred to a 12% (bis)acrylamide gel according to Lämmli with slight modification (0.3% gelatin, 0.1% SDS). Following electrophoresis (150 V in 25 mM Tris, 0.1% SDS, 200 mM glycine), the gel was washed twice with 2.5% Triton X-100 and 4–5 times with ddH2O. After incubation in developing buffer (50 mM Tris, 100 mM NaCl, 10 mM CaCl2, 0.02% Brij-35) overnight, the gel was stained with Coomassie, according to the manufacturer's instructions. Densitometric analysis was performed using the ImageJ software (NIH software http://rsbweb.nih.gov/ij/download.html).
Conventional RT-PCR
Total cellular RNA was isolated with Trifast (Peqlab, Erlangen, Germany) according to the manufacturer's protocol. First-strand cDNA was synthesized from 1 μg RNA according to the manufacturer's instructions using the Transcriptor High Fidelity cDNA synthesis kit (Roche, Mannheim, Germany). Primer sequences and the corresponding annealing temperatures are summarized in Table 2. Products, resolved by gel electrophoresis in a 1.5% (w/v) agarose gel, were visualized with ethidium bromide.
Primer and PCR Conditions

IL, interleukin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Statistics
Results are expressed as Whiskers graphs showing median, 25th and 75th percentile, and extreme values. Data sets were compared by Kruskal-Wallis followed by Dunn's multiple comparison test. A value of p < 0.05 was taken as minimum level of significance.
Results
Highest Yield of Neohepatocytes Generated with Autologous Serum
Monocytes were dedifferentiated to PCMOs for 6 days in the presence of six different human AB sera, FCS, or autologous serum. The following 14 days cells were cultured with differentiation medium supplemented according to the original protocol with 10% FCS (33) or autologous serum for the all-autologous setting (Fig. 1A). The yield of the generated neohepatocytes was determined by staining surface proteins with SRB. For all six donors (N = 6; n = 12) investigated constantly, the all-autologous setting reached highest cell numbers, while the lowest cell numbers were obtained by culture in FCS-supplemented medium. The yield of cells obtained from the different AB sera varied strongly from donor to donor (Fig. 1B). The yield of mononuclear cells obtained per milliliter blood varied between 0.8 and 1 × 106 cells/ml blood in the case of the blood donations, which is significantly lower compared to freshly drawn blood where the yield is between 2 and 2.5 × 106 cells/ml blood (10). Thus, taking 50 ml blood as starting volume the expected yield of neohepatocytes in the all-autologous setting would be between 8.1 × 106 and 10.1 × 106 cells in the case of the blood donation and between 20.2 × 106 and 25.2 × 106 cells when the blood is processed immediately.

Use of autologous serum improves yield and urea metabolism of neohepatocytes. (A) Experimental setup, showing use of different human AB sera, fetal calf serum (FCS), and autologous serum throughout generation of neohepatocytes. (B) Sulforhodamine B (SRB) staining of neohepatocytes (N = 6; n = 12) generated with six different AB sera (AB1–AB6), FCS, and autologous serum (A+F and A+A). ***p < 0.001 compared to all autologous setting (A+A); ooop < 0.001 compared to A+F. (C) Basal urea formation and NH4Cl metabolism in neohepatocytes (N = 6; n = 4) generated with six different AB sera (AB1–6), FCS, and autologous serum (A+F and A+A) compared to primary human hepatocytes (N = 4; n = 4). Additional supplementation with ornithine to improve NH4Cl metabolism. ***p < 0.001 compared to the corresponding sample of primary human hepatocytes; ooop < 0.001 compared to the corresponding sample of the all-autologous setting (A+A).
All-Autologous Setting Improved Urea Formation and NH4Cl Metabolism in Neohepatocytes
To remove residual urea from the culture medium, cells were washed twice with DPBS. To determine basal levels of urea formed, cells were incubated with buffer for 24 h. To measure the ability of the cells to metabolize ammonium, the buffer was supplemented with NH4Cl or NH4Cl and ornithine. All neohepatocytes (N = 6, n = 4) generated with the different sera formed and secreted urea. The amount, however, was significantly lower compared to primary human hepatocytes, especially when cultured in medium containing FCS. The results obtained when culturing the cells with different AB sera varied strongly from donor to donor. Use of autologous serum significantly improved the ability of neohepatocytes from all donors to form urea, reaching basal levels comparable to primary human hepatocytes. Addition of NH4Cl or NH4Cl and ornithine increased urea formation in all settings (Fig. 1C).
All-Autologous Setting Increased Glucose Formation and Metabolism in Neohepatocytes
To remove residual glucose from the culture medium, cells were washed twice with DPBS. To determine basal levels of glucose formed from glycogen storage, cells were incubated with buffer for 24 h. All neohepatocytes (N = 6, n = 6) generated with the different sera formed and secreted glucose. The amount of glucose formed was significantly lower compared to primary human hepatocytes. Similar to urea formation, the results obtained using the different AB sera varied strongly between the different donors while use of autologous serum constantly improved the ability of neohepatocytes to store and produce glucose (Fig. 2A). This was confirmed by more intense PAS and G6Pase staining in the autologous settings (Fig. 2B, C). Interestingly, for the all-autologous setting the G6Pase staining was more uniform, comparable to primary human hepatocytes, while culture with AB sera and FCS results in a more local staining at the membrane border of the cells (white arrow). To measure the ability of the cells to perform gluconeogenesis, the Na-pyruvate-containing incubation buffer was additionally supplemented with Na-L-lactate. Addition of Na-L-lactate increased glucose formation in all settings (Fig. 2A).

Highest glucose production of neohepatocytes generated with autologous serum. (A) Glucose formation from stored glycogen (basal) and pyruvate and lactate (gluconeogenesis) in neohepatocytes (N = 6; n = 6) generated with six different AB sera (AB1–6), FCS, and autologous serum (A+F and A+A) compared to primary human hepatocytes (N = 4; n = 4). ***p < 0.001 compared to the corresponding sample of primary human hepatocytes; ooop < 0.001 compared to the corresponding sample of the all-autologous setting (A+A). Representative picture of Periodic acid Schiff (PAS) (B) and glucose-6-phosphatase (G6Pase) (C) staining in neohepatocytes generated with human AB serum, FCS, and autologous serum (A+FCS and A+A) compared to primary human hepatocytes.
Highest Phase I and II Enzyme Activities of Neohepatocytes with All-Autologous Setting
As reported previously, neohepatocytes show phase I and II enzyme activities; however, levels are significantly lower compared to primary human hepatocytes (14). We investigated the effect of the different sera on the activity of six different cytochrome P450 isoforms (1A1/2, 2A6, 2B6, 2D6, 2E1, and 3A4) and six different phase II enzymes (e.g., UDP-glucuronosyl-transferase and glutathione-S-transferase). The activities measured in cells cultured with the different AB sera varied strongly between the different donors. In general, neohepatocytes generated with FCS showed lowest enzyme activities. For all donors the use of autologous serum improved the enzyme activities significantly, reaching levels close to primary human hepatocytes especially for phase I enzymes (Fig. 3A–L).

Highest phase I and II enzyme activities of neohepatocytes in the all-autologous setting. (A–F) Measurement of phase I, cytochrome p450 1A1/2 (CYP1A1/2), CYP2A6, CYP2B6, CYP2D6, CYP2E1, and CYP3A4, and (G–L) phase II, uridine diphospho-[UDP]-glucuronosyltransferase, glutathione-S-transferase, AHMC, CHC, HFC, and 7HC conjugation, enzyme activities in neohepatocytes (N = 6; n = 3) generated with six different AB sera (AB1-AB6), FCS, and autologous serum (A+F and A+A) compared to primary human hepatocytes (N = 4; n = 4). Activities were normalized to total protein contents measured by SRB staining. AHMC, 3-(2-(N,N-diethyl-amino)ethyl)-7-hydroxy-4-methyl-coumarin; CHC, 3-cyano-7-hydroxycoumarin; HFC, 7-hydroxy-4(trifluoromethyl)-coumarin; 7HC, 7-hydroxycoumarin. *p < 0.05, **p < 0.005, ***p < 0.001 compared to primary human hepatocytes; op < 0.05, oop < 0.005, ooop < 0.001 compared to all-autologous setting (A+A).
Less Macrophage Activation by Autologous Serum Compared to other Settings
To determine macrophage activation of PCMOs we measured secretion of TNF-α, TGF-β, RANKL, and matrix metalloproteinases (MMP). Cells were washed twice with DPBS before incubation with serum-free medium for 48 h at 37°C. In the culture supernatant we measured TNF-α and RANKL levels by ELISA (Fig. 4A, B). TGF-β levels were measured using MFB-F11 reporter cells (Fig. 4C) and proteolytic activity of MMPs was detected by zymography (Fig. 4D). Consequently, for all four factors the highest levels were observed in culture supernatants of PCMOs generated with FCS, indicating the most macrophage activation. As expected, the lowest levels of all four factors were observed in culture supernatants of PCMOs generated with autologous serum, while the levels measured in culture supernatants of PCMOs generated with the different AB sera varied strongly between the different donors.
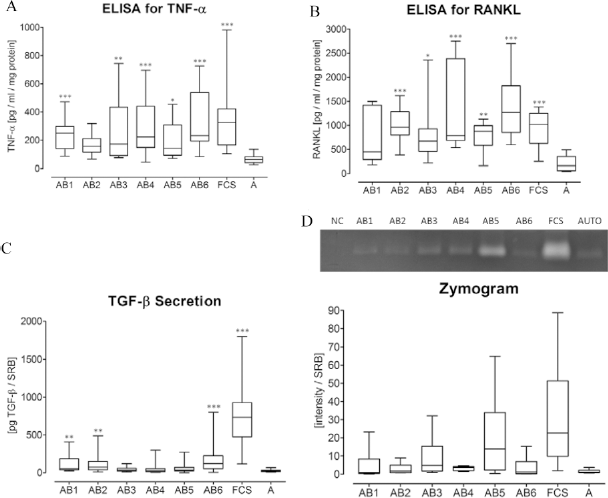
Tumor necrosis factor-α (TNF-α), transforming growth factor-β (TGF-β), and receptor activator for nuclear factor κB (RANKL) expression and matrix metalloproteinases (MMP) activity as measure of macrophage activation. Programmable cells of monocytic origin (PCMOs) (N = 6; n = 3) generated with six different AB sera (AB1-AB6), FCS, and autologous serum (A) were cultured for 48 h with serum-free medium. Resulting culture supernatants were analyzed for (A) TNF-α and (B) RANKL levels by ELISA, (C) TGF-β levels with the help of MFB-F11 reporter cells, and (D) MMP activity by gelatin zymography representative of N=6. Densitometric analysis was performed using the ImageJ software. Levels were normalized to total protein contents determined by SRB staining. *p < 0.05, **p < 0.005, ***p < 0.001 compared to autologous serum.
Furthermore, as measure of macrophage activation, we analyzed mRNA levels of IL-1α, IL-1β, IL-6, IL-8, and IL-10 in PCMOs by RT-PCR (Fig. 5). As positive control we used monocytes treated for 24 h with LPS. Similar to TNF-a, TGF-β, RANKL, and MMP activity, IL mRNA levels were highest in PCMOs generated with FCS and lowest for PCMOs generated with autologous serum, confirming macrophage activation in FCS-treated cells. Interleukin levels of PCMOs generated with the different AB sera varied strongly between the different donors.
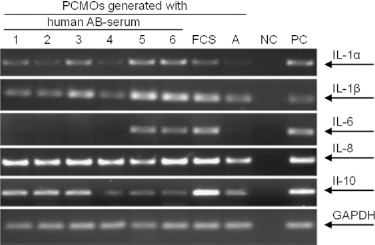
Highest interleukin mRNA levels in PCMOs cultured with FCS. Representative picture (N = 6) of RT-PCRs for interleukin (IL)-1α, IL-1β, IL-6, IL-8, and IL-10 in PCMOs cultured with six different human AB sera (1–6), FCS, or autologous serum (A). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as housekeeping gene. mRNAs were isolated with Trifast according to the manufacturer's instructions. PCR products were resolved in a 1.5% agarose gel and visualized by ethidium bromide.
Discussion
Hepatocyte transplantation is a promising alternative to whole-organ transplantation either to bridge patients awaiting a donor liver or to correct at least temporarily genetic deficiencies of the liver (19,27,29,33,34,38,40). Nevertheless, this procedure is limited by a critical shortage of organs suitable for the isolation of primary human hepatocytes. A number of investigators attempt to overcome this deficit by generating hepatocyte-like cells using stem or precursor cells (12,21,25). The plasticity of human or mouse embryonic stem cells (ESCs) allows successful hepatic differentiation (1,2), but raises ethical concerns. Adult stem cells, on the other hand, are ethically more accepted but are limited in their differentiation potential. In the last years, there have been reports about hepatocyte-like cells derived from mesenchymal stem cells from bone marrow (28), fat tissue (3,5), liver (30), umbilical cord matrix stem cell (8), and human skin fibroblasts (26). Although these cell types show good proliferation in vitro, to provide sufficient cell numbers for transplantation isolation often requires invasive methods, bearing risks for the patient. Furthermore, often after requiring some hepatic features, the differentiation remained incomplete. Inducing a pluripotent state in somatic cells by overexpressing transcription factors similar to ESCs yield promising results concerning the differentiation potential of these so-called iPSCs (15,24). As transcription factors are usually delivered via viral particles, this technique is still far from clinical application.
In this study we focus on improving yield and function of monocyte-derived hepatocyte-like cells, as they can be obtained easily from patients (minimal invasive) and we were able to show that injection of these cells can increase the survival of rats after extended liver resection (16). Nevertheless, up to now, therapeutic approaches are mainly hampered by a low yield and poor function of the hepatocyte-like cells compared to human hepatocytes (31). We here demonstrate that the yield and function of monocyte-derived neohepatocytes significantly improves when cells are cultured in the presence of autologous serum instead of human AB or FCS. This improvement has a significant impact for autologous hepatocyte-like cell therapy in patients either waiting for an organ or are not eligible for orthotropic liver transplantation.
Our data suggest that the significant improvement of yield and function of neohepatocytes is mainly due to a reduced activation of monocytes/macrophages during the crucial dedifferentiation step to PCMOs and the subsequent differentiation phase into hepatocyte-like cells. There are several hypotheses supporting our assumption that monocyte/macrophage activation by serum component(s) may play a crucial role in this event. One likely reason during the dedifferentiation step could be simply described by a graft versus host reaction (7,11) of the cells towards the applied human type AB sera or FCS. Under these circumstances an inflammatory reaction would be initiated and lead to an increased monocyte/macrophage activity, hereby secreting TNF-α, RANKL, TGF-β, various interleukins, and MMPs.
Another possibility leading to monocyte/macrophage activation is the role of M-CSF. One role of M-CSF consists in the stimulation of proliferation and differentiation of myelomonocytic progenitors, resulting in the production of blood monocytes and macrophages (18). On the other hand, it is widely accepted that M-CSF can activate mitogen-activated protein kinase (MAPK) by elevating c-Fos and phospho-CCAAT (cytidine-cytidine-adenosine-adenosine-thymidine)/enhancer bnding proteins (phospho-C/EBP) (45), via the transcription factor phosphorylation of extracellular signal-related kinases 1/2 (ERK1/2) and thereby stimulate monocytes (6,10,17,20). This activation may interfere with the dedifferentiation step of monocytes towards PCMOs and further differentiation to neohepatocytes and may lead to cell interaction with contaminating cells, such as T cells or granulocytes after the first isolation step, and the secretion of cytokines.
A third reason for monocyte/macrophage interaction might be directly linked to serum components. When serum is heat inactivated there is still a considerable amount of active soluble factors such as monocyte chemotactic protein (MCP), regulated upon activation of normal T-cell expressed, and presumably secreted protein (RANTES), Il-2, Il-6, or IL-16 (4). Most of these factors can trigger an inflammatory reaction, resulting in monocyte/macrophage activation.
Due to the high need of hepatocyte-like cells for cell transplantation in end-stage liver diseases, several approaches have been made to generate these cells from various sources (12,25). Campard et al. have used native umbilical cord matrix stem cells (UCMSC) (8), which were obtained postnatal from patients. Access to umbilical cords is rather difficult, especially as preserving stem cells for potential therapeutic application for the newborn baby has been widely promoted by media, thus persuading many parents to this step (25,37). Another promising source for generation of hepatocyte-like cells might be fat tissue. Human mesenchymal stem cells from adipose tissue showed better integration into the host livers after predifferentiation into hepatocyte-like cells in vitro compared to undifferentiated cells (3). Nevertheless, in order to gain fat tissue for cell differentiation, the patient has to undergo surgery. This might be a rate-limiting step of this method (5,45), in particular if the patient is too sick to wait for an organ transplantation, resulting in intraoperative complications. Miyazaki and coworkers differentiated a rat bone marrow-derived stem cell line to hepatocyte-like cells with good results regarding quality and yield of cells (28). However, without differentiation these cells were not able to prevent or reverse acute liver failure (35).
These selected examples show that there are several approaches for generating hepatocyte-like cells for autologous cell therapy. But considering availability of the progenitor cells as well as quality and yield of gained hepatocyte-like cells, most of the approaches come up with some restrictions. Therefore, differentiation of PBMCs to neohepatocytes using an all-autologous serum approach not only would provide a high amount of cells with an increased quality but also reduce the need for immune suppression after transplantation.
Although there are already some reports of patients awaiting a donor organ who have been bridged by transplantation with various liver cell types (19,27,39,40), it still remains elusive which liver diseases are possibly treatable by cell transplantation. As end-stage liver disease has several different geneses (e.g., metabolic, viral, autoimmune, toxic), it needs to be evaluated which kind of liver disease is suitable for transplantation. Moreover, we need to identify the disease stages at which cell transplantation is effective in spite of disease-depending altered hepatic architecture.
In summary, transplantation of neohepatocytes with excellent quality might help patients suffering from acute or acute-on-chronic liver disease, bridging the time for whole-organ transplantation or support the regeneration of the liver. Recently, we could show that neohepatocytes might have a survival or proliferation advantage compared to primary human hepatocytes due to reduced Smad1 and Smad3 expression (13).
Above all, liver recovery of patients who underwent extended hepatectomy could be supported by transplantation of these cells. For clinical application the all-autologous setting seems to be the most promising approach. Besides prevention of exposure to unknown infectious agents potentially present in donor blood, it provides the highest yield of cells with an excellent metabolic quality analyzed by a broad screening of activity tests and, above that, it could reduce the need for immune-suppressive drugs, thus supporting a faster recovery of the patient.
Footnotes
Acknowledgments
This work was partially supported by the Federal Ministry of Research (BMBF-0315208E; BMBF-01GN0984). The authors declare no conflicts of interest.
