Abstract
Standard treatment for large burns is transplantation with meshed split skin autografts (SSGs). A disadvantage of this treatment is that healing is accompanied by scar formation. Application of autologous epidermal cells (keratinocytes and melanocytes) may be a suitable therapeutic alternative, since this may enhance wound closure and improve scar quality. A prospective, multicenter randomized clinical trial was performed in 40 adult patients with acute full thickness burns. On two comparable wound areas, conventional treatment with SSGs was compared to an experimental treatment consisting of SSGs in combination with cultured autologous epidermal cells (ECs) seeded in a collagen carrier. The primary outcome measure was wound closure after 5–7 days. Secondary outcomes were safety aspects and scar quality measured by graft take, scar score (POSAS), skin colorimeter (DermaSpectrometer®) and elasticity (Cutometer®). Wound epithelialization after 5–7 days was significantly better for the experimental treatment (71%) compared to the standard treatment (67%) (p = 0.034, Wilcoxon), whereas the take rates of the grafts were similar. No related adverse events were recorded. Scar quality was evaluated at 3 (n = 33) and 12 (n = 28) months. The POSAS of the observer after 3 and 12 months and of the patient after 12 months were significantly better for the experimental area. Improvements between 12% and 23% (p ≤ 0.010, Wilcoxon) were detected for redness, pigmentation, thickness, relief, and pliability. Melanin index at 3 and 12 months and erythema index at 12 months were closer to normal skin for the experimental treatment than for conventional treatment (p ≤ 0.025 paired samples t-test). Skin elasticity showed significantly higher elasticity (p = 0.030) in the experimental area at 3 months follow-up. We showed a safe application and significant improvements of wound healing and scar quality in burn patients after treatment with ECs versus SSGs only. The relevance of cultured autologous cells in treatment of extensive burns is supported by our current findings.
Keywords
Introduction
Burn wounds often heal with serious scarring, even though progress has been made in the treatment modalities (e.g., better intensive care, antibacterial treatment, wound dressings). For many years the “gold standard” for treating wounds of burn patients has been transplantation with an autologous split skin graft. In patients with extensive burn wounds donor sites may be limited. In order to cover all the wounds, the patients often need multiple operations and/or the skin had to be expanded as much as possible. However, the current different expansion techniques and treatments [e.g., mesh and Meek-Wall (21)] frequently lead to scar formation, especially in the large mesh interstices.
The rate of wound closure depends on how quickly epidermal cells migrate out of the meshed autograft and/or wound edges to close the wound. Accelerating reepithelialization could potentially improve the outcome of the healing process in terms of reducing granulation tissue formation, reducing the healing time, and thereby reducing the risk of colonization and infection, as well as scar formation.
Cultured epidermal cells have been used in burn treatment since the 1980s. Confluent epithelial sheets (29) or cultured epithelial autografts (CEAs) could restore epidermal coverage and have previously been lifesaving for patients suffering from extensive burns (26). However, both short-term (e.g., variable take rates, risk of contamination because of extended wound healing times) and long-term results (e.g., blister formation due to lack of restoration of anchoring fibrils, limited mechanical resistance) were below expectations (39,40). The disappointing results, accompanied by long culture times, difficulties in handling due to fragility of the sheets, and high costs, lead to the search for further improvements in therapy for this specific category of large and deep burn wounds.
To overcome some of these drawbacks, new methods were developed in which proliferating keratinocytes were transferred to the wound bed. These proliferating cells showed a better take rate than confluent sheets (17). In addition, the use of pre-confluent keratinocytes decreased culture time and therefore costs. Animal and clinical studies showed that transfer of these cells to the wound bed could be accomplished by a variety of carrier systems such as fibrin (27), polyurethane membranes (28), porous synthetic carriers (32), fibrin glue suspension (20), and aerosol sprays (24). Such keratinocyte delivery systems are easier to handle than the epidermal sheets. For the coverage of large burn wounds, this technique can be used in combination with meshed skin autografts, which certifies the supply of epidermal stem cells. Zhu et al. showed that keratinocytes cultured on a plasma polymer surface in combination with a meshed split skin autograft (SGG) accelerated reepithelialization and showed improved healing with a less visible mesh pattern (41). Magnusson et al. showed accelerated wound closure and decreased transepidermal water loss as a result of the “application of cultured keratinocytes in suspension” onto a standardized partial thickness wound (donor site wounds) (22).
Autologous skin cell sprays would have a theoretical advantage in that no culture time would be needed (14,38). However, harvesting larger quantities of proliferating cells for treatment of extensive burns would still require sufficient donor material and/or expansion by culture. Thus the advantage of sprays as a treatment modality for larger burns is still limited.
We developed a culture method for epidermal cells in which we took all the above-mentioned limitations into account. In addition to autologous keratinocytes, autologous melanocytes are cocultured and amplified to provide sufficient numbers of cells to cover large wounds within 2 to 3 weeks, importantly without the need of a mouse 3T3 feeder layer (7,13), or components such as bovine pituitary gland extracts. In an animal model, we have shown that wound closure could be accelerated by applying cultured keratinocytes seeded on a carrier (32). Subsequently, we modified our protocols so that they were fully compliant with Dutch regulations for advanced therapy medicinal products (ATMPs) and confirmed the stability during transport. We also demonstrated the viable transfer of ECs from the carrier to de-epidermized dermis in vitro including the subsequent formation of a reconstructed epidermis containing melanocytes in the basal layer (36).
Nowadays, application of cultured human cells in clinical treatment is subjected to strict regulations in terms of culture methods (use of clinical-grade reagents) and location (quality assured production in a clean room facility). Therefore, clinical trials involving ATMPs require advanced logistics and handling capacity. Taking all of these requirements into account, we set out to study if autologous cultured proliferating epidermal cells, harvested from a small donor site, seeded in a collagen carrier (MatriDerm®), could enhance the wound healing rate for patients with large and deep burns and whether this therapy could lead to improved long-term scar quality.
Materials and Methods
Study Design
This prospective randomized multicenter intrapatient comparative trial was performed in the three specialized burn centers in the Netherlands: the Red Cross Hospital in Beverwijk, the Maasstad Hospital in Rotterdam, and the Martini Hospital in Groningen. The study protocol was approved by the Dutch Central Committee on Research Involving Human Subjects (CCMO) (NL19048.000.07), the ethical committee of the three hospitals, registered at Clinical Trials (NCT00832156), and received an Eudract number (2007-004296-19).
Study Population
All patients were checked for eligibility according to the inclusion and exclusion criteria (Table 1). Immunocompromised patients, based on known medical history about congenital or acquired immunodeficiency, were excluded to participate in our trial in order not to miss a potential reaction to the carrier and/or increased inflammatory reactions due to an a priori malfunctioning of the immune system of the recipient patient. Patients were included during their admission in the burn centers between June 2008 and September 2012. A delay in study progress was experienced due to the reconstruction of the culture facility in Amsterdam, which needed to be adapted due to new regulatory demands from the health inspection authorities for the production of the cultured epidermal construct. Because of these activities, no patients could be included in the trial from September 2010 until October 2011.
Study Inclusion and Exclusion Criteria

The patients or their legal representatives gave written informed consent before inclusion in the study. The patients were anonymized using codes including consecutive numbers and the first letter of the city of the hospital in which the patient was admitted. To transfer these codes to the randomization paper inside the envelope, carbon paper was used. The clinical researchers (M.B., R.M., K.G.) made the randomization envelopes, included the patients, and assisted during the randomization procedure. The total study period for an individual patient was approximately 13 months, including the 3 and 12 months follow-up in the outpatient clinic.
Biopsy and Preparation of the Carrier
After informed consent was given, a skin biopsy of approximately 3 cm2 was harvested from nonaffected skin of the patient. Before transport to the Good Manufacturing Practice (GMP) clean room facility (A-SkinBV, Amsterdam, The Netherlands), the skin biopsy was wrapped in a gauze with saline solution and placed in a sterile cup. Culture and preparation of the ECs for transplantation were performed exactly as described previously (36). In short, in the laboratory, the ECs were isolated from the biopsy and cultured to increase the cell numbers. After communication between the laboratory and the performing hospital regarding the timing of surgery, the expanded ECs were seeded at a density of 50,000 cells/cm2 onto a collagen carrier with a size of 148 × 105 × 1 mm (MatriDerm®, MedSkin Solutions, Billerbeck, Germany) and further cultured for 2–5 days before the operation. The collagen carrier MatriDerm® is a highly porous 1-mm-thick membrane consisting of a native bovine collagen type I, III, and V template coated with a 3% elastin hydrolyzate, which was derived from the bovine nuchal ligament. After 2–5 days of culture of EC-MatriDerm® product, four 3-mm punch biopsies were taken from different areas of the product for quality assurance. One biopsy was embedded in paraffin and tissue sections were analyzed to confirm the presence of individual (not multilayered) ECs on MatriDerm® (36). Three biopsies were used to assess metabolic activity/viability of the product by MTT assay as previously described (36). An optical density (OD) at 550 nm with a reference wavelength of 650 nm of above 0.05 indicates mitochondrial activity and cell viability. The average OD obtained for n = 53 products: 39 patients and 14 Blanco test runs was, respectively, 0.202 ± 0.109 (SD) or ±0.017 (SEM) and 0.041 ± 0.010 (SD) or ±0.002 (SEM), indicating that in all products viable cells were present.
Maximally, 24 h before the operation the carrier with ECs was prepared for transport by rinsing once in Hanks' balanced salt solution (HBSS) (Lonza, Verviers, Belgium) followed by removal of the HBSS. The product was packaged in a sterile tissue culture dish, sealed with Petri seal, and placed in a Styrofoam transport box and transported by a courier to the hospital under ambient conditions and preserved at room temperature until use. The mean culture time from start of biopsy to transfer to the patient was 13 days.
Operation Procedure
Wounds were treated with topical silver sulfadiazine 1% (Farmacie Medisch Centrum Alkmaar, the Netherlands) or cerium-silver sulfadiazine cream (Sinclair Pharmaceuticals Ltd, Chester, UK) preoperatively, in line with the burn center treatment protocols. Before excision, the wounds were scrubbed with chlorhexidine (Added Pharma BV, Oss, the Netherlands) and cleaned with saline. Thereafter, the wound beds were prepared through tangential excision and/or hydrosurgery followed by adequate hemostasis. After this procedure the final wound beds were judged comparable for both study wounds. Subsequently, the wounds were defined according to a preestablished randomization schedule A (right, proximal, cranial, ventral) or B (left, distal, caudal, dorsal). Then the randomization envelope was opened, including the allocation of both wound areas. Meanwhile, thin split-thickness skin autografts were harvested with a dermatome (Zimmer® Inc., Dover, OH, USA) and expanded to the ratio 1:3 using the Mesh technique. Both wounds were transplanted with a meshed split-thickness skin autograft, and the experimental wound was subsequently covered with the collagen carrier containing the cultured autologous ECs (Fig. 1). Prior to application on the split-thickness skin autograft, the carrier with ECs was rinsed in saline to remove any remaining traces of culture medium. The grafts and carrier with ECs were fixed in place with staples and the transplanted wounds were covered with a nonadhesive dressing. Photographs were taken to facilitate localization of the exact experimental and control area during follow-up. Since the presence of the collagen carrier is visible during the first days, blinding the clinician and patient was not possible during this period.

Culture procedure including the collagen carrier. (A) Incoming skin biopsy 3 cm2. (B) Keratinocyte and melanocyte culture (EC): dashed arrow shows melanocytes growing as single dendritic cells, solid arrow shows keratinocyte colony. (C) ECs are seeded at a density of 50,000 cells/cm2 onto a collagen carrier and further cultured for 2–5 days. (D) EC-MatriDerm construct is placed on top of meshed split thickness autograft during surgery. For extensive culture and transport details, see Waaijman et al. (36).
Outcome Parameter
Wound Healing: Primary Outcome.
Wound healing is defined as the percentage of epithelialization of the wound at a given point in time. At the first dressing change, 5 to 7 days postsurgery, the wound epithelialization of the autografts was measured by an experienced burn clinician. Bloemen et al. have shown that one experienced observer can evaluate the reepithelialization rate in a reliable and effective way (4). Wound inspection was performed during the clinical admission period up until complete wound healing.
Graft Take and Complications.
The graft take is defined as the percentage of the graft that appears to be vital and shows good adherence to the wound bed. This was measured at the same time as the wound healing during the first dressing change by an experienced burn clinician.
Bacterial contamination of both study areas was monitored using pre- and postoperative wound swabs (twice weekly during the clinical admission). Wound swabs containing “no growth of pathogens,” “resident skin flora,” or “mixed Gram-positive flora” were noted as negative cultures. Positive cultures contained species such as Staphylococcus aureus, Pseudomonas aeruginosa, Acinobacter, Klebsiella, Aspergillis niger, Escherichia coli, Enterococcus faecium, Candida parapsilosis, or Bacillus cereus.
Scar Quality.
In the outpatient clinic, the scar quality was assessed at 3 and 12 months postsurgery using different measurement instruments. First, the Patient and Observer Scar Assessment Scale (POSAS) was used. The POSAS is a reliable and validated scar assessment scale (10,31,33). It consists of two numeric scales: the Patient Scar Assessment Scale (patient scale) and the Observer Scar Assessment Scale (observer scale). The six parameters of the observer scale are vascularization, pigmentation, thickness, relief, pliability, and surface area. The six parameters of the patient scale are pain, itching, color, stiffness, thickness, and surface irregularity. Responses were gathered into a score ranging from 1 to 10 for each parameter. For both scales, the summed outcome range (total scar score) can be between 6 and 60, respectively, representing “normal skin” and “worst imaginable scar.”
Second, to measure the scar color and pigmentation, the DermaSpectrometer® (Cortex Technology, Hadsund, Denmark) was used. It is a validated instrument to measure scar vascularization (erythema) and pigmentation (melanin) by a narrow band simple reflectance meter (9).
Finally, scar elasticity was measured with a validated instrument, the Cutometer® (Courage & Khazaka GmbH, Cologne, Germany) (8). The vertical deformation of the skin was measured in millimeters when the skin was pulled by means of a controlled vacuum into a defined circular opening.
Statistical Analyses
The data were analyzed with the Statistical Package for the Social Sciences (SPSS) for Windows version 21.0 (IBM, Armonk, NY, USA). Sample size calculation was based on the results obtained from a study in which a keratinocyte cell lysate was used to stimulate closure of a meshed skin autograft on burn wounds (11). Given a power of 0.90, a significance level of 0.05 and including compensation for the risk of complications or lost-to-follow-up, a sample size of 40 patients was calculated.
Normality was tested by calculating the skewness and kurtosis. The normally distributed numeric data were tested with the paired samples t-test (mean and SD data values). For the POSAS scar assessment scale, statistical analysis was performed using the nonparametric Wilcoxon test. Wound contamination was tested using cultures and analyzed tested with the chi-square test.
Results
A total of 40 adult patients were included in this multicenter study. The study flowchart is shown in Figure 2. The baseline characteristics of all included patients are described in Table 2. Most patients were male (n = 25), and most patients suffered from flame burns (90%). The mean age at time of the accident was 50 years (SD 19) with a maximum of 80 years. The total body surface area (TBSA) of the patients varied from 6% to 51% with a mean of 24% (SD 13) of which 13% were full-thickness burn wounds. In most cases, the groin was used as a donor site for the biopsy. From the initial 40 patients participating in our study, four patients were excluded. Biopsies were taken from these patients; however, one patient was deceased before the transplantation procedure was planned, one patient healed spontaneously and did not require an operation in the study areas, and the keratinocyte culture of another patient could not be transplanted due to a fungal contamination. The last patient was excluded due to slow cell division, and this patient died 1 month postburn. In total, five patients died during the study period and follow-up. These were reported as serious adverse events to the medical ethical commission and were judged as not related to the treatment. No adverse events were identified as related to the experimental treatment. Other adverse events are described in the Graft Take and Complications section.

Study flowchart. The flowchart of this study is based on the CONSORT statement 2010 Flow Diagram and shows the eligibility of the patients assessed for this clinical trial. *Most important reasons were language barrier of the patient, logistic reasons, participation in other trials, refusal of participation in a clinical trial, psychiatric disorders, infected wounds. †Deceased patient. n, number of patients.
Baseline Characteristics of the Included Patients (n = 40)

The treatment characteristics for the 36 patients are described in Table 3. The mean culture time of the autologous ECs was 13 days (SD 2.1) and at a mean of 16 days (SD 2.2) postburn, the operation was performed. Two follow-up evaluations were performed at 3 months postoperatively with a mean time of 13 weeks (SD 5.9) and at 12 months with a mean time of 56 weeks (SD 13.8).
Characteristics of the Treatment (n = 36)

UrgoTul SSD® (Urgo Medical BV, Cuijk, The Netherlands), Mepilex® Ag (Mölnlycke Health Care AB, Göteborg, Sweden), SurfaSoft® (Taureon, Rijswijk, The Netherlands), Adaptic® (Systagenix Wound Management Limited, North Yorkshire, UK), Cuticell® (BSN Medical GmbH, Hamburg, Germany).
Wound Healing: Primary outcome
Wound epithelialization on days 5–7 postoperatively in the experimental wounds (mean = 71% closed) was statistically significantly higher (p = 0.034, Wilcoxon) compared to the standard treatment (mean = 67% closed), as indicated in Table 4.
Wound Healing Characteristics of Study Patients (n = 36)

Values are mean with SD in parentheses.
Two values missing.
Five values missing.
Significant difference (nonparametric Wilcoxon test).
Graft Take and Complications
The take rates of the meshed autologous split skin autografts (SSGs) of the experimental and standard treated wounds did not show significant differences (Table 4). No significant differences were found in wound contamination from swabs representing experimental and standard treated wounds separately. However, separate swabs were not evaluated in all cases for standard versus experimental treatment (data not shown). Overall, more patients had bacterial colonization in their wounds postoperatively (61%, 20 patients, n = 33) than preoperatively (31%, 11 patients, n = 36). It should be noted that some of these wounds were already colonized with bacteria (in 10 out of 20 patients) before the procedure. This also led to a slightly, but not significantly, longer stay in hospital (51 vs. 42 days, p = 0.278).
Reoperation of some parts of the experimental and/or standard treated area occurred in total in nine patients. However, this is not unusual for burn patients, since these patients had larger burn areas (mean TBSA 29%) and were treated with meshed grafts with an expansion of 1:3. Specifically, in three patients, only the experimental treated area was reoperated and in one patient only the standard treated area. In the other cases, the reoperation occurred in both treated areas. Reasons to reoperate were insufficient graft take, due to bacterial problems, dislocation of (parts of) the graft, or hematoma formation.
Scar Quality
An impression of the clinical application and follow-up appearance of the EC-treated versus standard-treated wounds is shown in Figure 3. Total scores of the Observer scale from the POSAS were significantly better for the experimental versus standard treatment at 3 months follow-up (p = 0.001, Wilcoxon), and the Patient scale showed a similar trend at 3 months follow-up (p = 0.084, Wilcoxon) (Table 5). The items pliability (p = 0.076) and relief (p = 0.057) were responsible for this trend in the Patient scale. In the Observer scale, all items contributed to the difference: redness (p = 0.011), pliability (p = 0.010), pigmentation (p = 0.006), thickness (p = 0.004), and relief (p = 0.010) were all significantly better for the experimental treatment in comparison to the standard treatment.

Treatment of a patient with proliferating epidermal cells (ECs) seeded on a collagen matrix as a carrier. The autologous EC-containing carrier was applied on top of a widely meshed split thickness skin autograft. Black dashed line: experimental area (EC-MatriDerm construct). (A and B) Application of the EC-containing carrier onto the wound. (C) EC-treated wound 6 days after application. (D) Standard treated wound without cultured ECs 6 days after application. (E) Three months follow-up of the experimental area and the standard treatment area contralateral. (F) Twelve months follow-up of the experimental area and the standard treatment area contralateral.
Mean Specific Items POSAS Score

POSAS, Patient, and Observer Scar Assessment Scale. Different items are scored on a scale 1–10, with 1 representing normal skin and 10 representing worst imaginable scar. n, number of patients.
Two measurements missing.
One measurement missing.
Significant difference (nonparametric Wilcoxon test).
At 12 months postsurgery, the total scores of the POSAS reported by both the observer and the patient were significantly better for the experimental treatment (p = 0.020 for the Observer scale, p = 0.024 for the Patient scale), as shown in Table 5. The items redness (p = 0.008), pliability (p = 0.002), and thickness (p = 0.015) were scored significantly better by the observer for the experimental treatment (Table 5). The patients scored their scar treated with cultured ECs significantly better for the item itching (p = 0.027), color (p = 0.048), and pliability (p = 0.016) in comparison to the standard treatment (Table 5). These results indicate that the experimental treatment versus the standard treatment shows an improvement of 14% to 33%.
The erythema index of the EC treatment, as assessed with the DermaSpectrometer®, was significantly more comparable to nonaffected skin than that of the standard treatment at 12 months (p = 0.025, paired samples t-test) but not at 3 months follow-up (Table 6). The melanin index for the experimental treatment was significantly more similar to normal skin when compared to standard treatment, both at 3 months (p = 0.002) and at 12 months follow-up (p = 0.011).
Scar Color and Pigmentation by DermaSpectrometer Analyzing
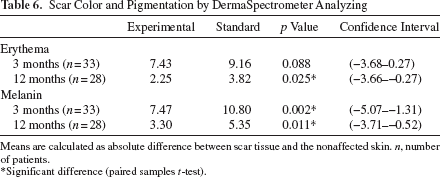
Means are calculated as absolute difference between scar tissue and the nonaffected skin. n, number of patients.
Significant difference (paired samples t-test).
Table 7 presents an overview of the different elasticity parameters as measured by the Cutometer®. Overall, all parameters were higher—indicating better elasticity characteristics—for the EC-treated wounds versus the standard-treated wounds, although not all parameters were statistically significantly different. At 3 months postoperatively, differences were greater than at 12 months. A higher elasticity ratio in the experimental group was found in the parameters: pliability (p = 0.039, paired samples t-test) and elasticity (p = 0.030) at 3 months follow-up.
Scar Elasticity by Cutometer Analyzing

Values represent the ratio between scar tissue and nonaffected skin. n, number of patients; Uf, extension; Ua, pliability; Ue, elasticity; Ur, retraction; Uv, visco-elasticity.
Significant difference (paired samples t-test).
Discussion
This prospective randomized multicenter intrapatient comparative trial was designed to determine the effect of autologous cultured ECs in combination with a meshed split skin autograft (SSG) on wound healing and scar quality of burn patients. Proliferating autologous ECs were seeded onto a collagen carrier, which was applied (EC side down) on top of the 1:3 meshed SSG. Autologous cultured ECs in combination with a meshed SSG were found to be an improved therapy compared to the treatment with only the conventional SSG and were safe to use. The primary outcome parameter “wound healing” is of interest since the general belief is that this is fundamental in determining short- and long-term functional and cosmetic scar quality. Therefore, an important finding in our study was that the rate of epithelialization of the experimental treatment was statistically significantly better compared to the standard treatment (an SSG without the carrier and ECs), despite the relatively high level of bacterial colonization that was noted, and which is not uncommon in these larger burn wounds (16). Furthermore, no related adverse events were noted. This was not surprising, since similar products such as CEAs have been used safely, but with less consistent effects on wound healing and scarring, over the past decades.
One of the important disadvantages of application of CEAs is the long culture time (23,25). Therefore, instead of using fully differentiated confluent sheets of cells, we used undifferentiated proliferating keratinocytes. This allows a potential reduction of culture time. Indeed, we were able to transfer ECs to the patient already 13 days after harvesting the biopsy, which is quite short compared to similar techniques. The required culture time is related to the area to be treated and the size of the biopsy that is used as source of the autologous cells. To guarantee that the carrier containing ECs would be ready within 2 weeks, a biopsy of approximately 3 cm2 was taken from the patient, and the experimental area to be covered was limited to 300 cm2.
Using a well-accepted collagen matrix MatriDerm® as a carrier for the cells, we were able to apply predominantly proliferating ECs to the wound bed instead of predominantly terminally differentiated keratinocytes as used in confluent sheets. Animal studies as well as clinical studies have shown that this commercially available class III device is well tolerated by the body when implanted as a dermal substitute (3,35). Furthermore, the carrier solved the problem of dealing with fragile sheets of cells during transport and operation. The carrier was grafted EC side down, resulting in direct contact of the ECs with the wound, and additionally, the carrier functioned as a wound dressing.
Several outcome parameters of short- and long-term functional and cosmetic quality of the scar were measured with validated instruments. At the short-term follow-up of 3 months, the observer reported significantly better scores on the POSAS items redness, pliability, pigmentation, thickness, and relief. The patient reported an improvement on the item color, pliability, thickness, and relief. However, none of these differences were statistically significant. The melanin index determined with the DermaSpectrometer® and the scar elasticity measured with the Cutometer® were significantly more comparable to the unaffected skin of the patient for the experimental area compared to the standard treatment at this time point.
Importantly, we found that the POSAS scores of both the observer and the patient at the long-term follow-up of 12 months were significantly better for the experimental treatment. In line with these findings, the melanin and the erythema indices measured with the DermaSpectrometer® were significantly more comparable to the unaffected skin of the patient for the experimental treatment.
The healing process of burn wounds and scar formation differs between patients. Even within patients, diversity in wound healing and scar formation in different body areas is likely. Therefore, this intrapatient observer trial provides valuable outcomes, limiting interpatient bias. It is therefore remarkable that the patients themselves were able to establish a difference between the two scar areas as reflected in the Patient scale of POSAS.
We established an improved pigmentation of the wounds treated with cultured ECs, both by observers at 3 months follow-up, patients at 12 months follow-up and DermaSpectrometer® at 3 and 12 months. We have shown that during our culture process both keratinocytes and melanocytes are viable and amplified and, furthermore in vitro, are able to produce a pigmented reconstructed epidermis (13,36). Since we compared the experimental treatment to the standard treatment with SSG in the same patient, it is likely that the improved pigmentation was due to the transplanted autologous EC construct.
The observation that the rate of epithelialization increased in the wounds where cultured ECs were added is not surprising, since in these wounds more epidermal cells were present than in the standard treated wounds. It does provide an indication that the cultured ECs actually survived and contributed to wound closure.
The improved outcomes regarding functional scar quality such as elasticity is more striking, since this suggests that other cells and processes are stimulated by the autologous keratinocytes. Faster wound closure in general is associated with better outcomes, so this could be one important aspect. Another potential contribution to the improved healing process could come from stimulating growth factors and cytokines that are produced by the cultured cells. Indeed, we have previously shown that keratinocytes in our culture system secrete factors that stimulate fibroblasts to produce an extensive cocktail of wound healing mediators (30). The question remains whether or not the same or similar results could have been achieved by using allogeneic keratinocytes. To our knowledge, there have been no randomized clinical trials performed that directly compared the origin of cultured keratinocytes (autologous vs. allogeneic) and the clinical treatment effect in burn patients.
A theoretical benefit of using allogeneic keratinocytes resides in the fact that these cells could be “ready to use.” Cryopreserved cells would only need one final culture step before transplantation, which takes approximately 1 week (5). Compared to our culture system this would greatly simplify logistics as patient biopsies would not need to be transferred to the clean room.
Furthermore, the creation of large master and working stocks of a single batch of cells would greatly reduce costs compared to personalized medicine. Necessity for safety screening and the ultimate immunological rejection of allogeneic keratinocytes, which only provide a temporary cover, are clear disadvantages. However, beneficial effects on deep partial thickness wounds and donor sites of using allogeneic keratinocytes for the treatment of full- and partial thickness burns have been described (2,5).
From an ethical point of view we found it undesirable to mesh the split skin graft to a larger scale than absolutely necessary. The outcome in terms of scarring is better for the patient if the extension of the graft is limited. Although we could expect that the differences in scar quality of the two different therapies would be more apparent if the graft would be expanded to a higher ratio than 1:3, we considered this ratio as the maximum extension for the standard of care sites, and therefore this extension was chosen for both treatment modalities.
It is evident that additional costs are associated with the use of cultured autologous keratinocytes. A comparative cost effectiveness study of the experimental and standard treatment is necessary to justify the use of cultured autologous ECs. Nevertheless, it is important to realize that the largest cost component in burn care is hospital stay (1,19). We could hypothesize that a reduction of hospital stay is achievable, since the rate of epithelialization of the experimental treatment was statistically significantly better compared to the standard treatment. Furthermore, a reduction in need of reconstructive surgery and rehabilitation could be expected. Although most costs to burn patients are accrue during their treatment the first 2 years after injury, the subsequent rehabilitation process and reconstructive therapies are still a high economic burden (18).
The most important limitation of this trial is the lack of a study arm in which MatriDerm® was used without cultured cells. Therefore, we cannot state that only the cultured cells are responsible for the observed effects on wound healing and scar improvement. It could be that also the presence of the carrier added to that effect. However, we chose not to include a third study arm in this trial for reasons of complexity of the study. Therefore, additional research is needed to clarify the role of MatriDerm® as a wound dressing. Although Wood et al. have previously reported on the use of a synthetic collagen sheet as wound dressing, they did not present data from (objective) measurements as an outcome in this study population (37).
Additional research in burn wound treatment and the improvement of scar quality will always be the highest priority for burn patients. Scar quality is of great importance for patients (34). Recently, Burnett et al. showed in a qualitative study among patients that the greatest dissatisfaction of their scar, after receiving a split thickness skin graft, was decreased elasticity followed by increased fragility (6).
Reviewing the literature, an important innovative aspect of our clinical trial is the multicenter setup in combination with the clinical application of an ATMP. Dealing with regulatory issues and logistics related to the transport of the biopsy and eventually the carrier with autologous ECs were challenging as described by Green (15). During the last decade, progress has been made within the development of culture methodologies and implementation of autologous cells used in clinical therapy (12).
In conclusion, proliferating cultured autologous ECs applied in a carrier system for the treatment of large, deep burns can be applied safely, reduce wound healing time, and will improve short-term and long-term functional and cosmetic scar quality. Our research project concerning this clinical trial made several noteworthy contributions to the field of regenerative medicine in burn wound treatment. First, we have shown that it is possible to conduct a multicenter clinical trial that follows all regulatory requirements and handles the logistics regarding an ATMP that positively contributes to the treatment of burn patients. Second, our trial is one of few that included both subjective and objective measurements on outcome of an ATMP on burn patients. Finally, the statistically significant positive findings on wound healing and scar quality using a carrier with cultured autologous ECs in combination with an SSG implicates an additional future treatment option for patients with deep dermal and full-thickness burn wounds.
Footnotes
Acknowledgments
We would like to extend our grateful thanks to the patients and their families for their contribution and participation in our study. A grant to support this study was received from the Dutch Burns Foundation (grant No. 07.116). Part of this work was supported by the European Commission within the 7th framework programme (grant agreement No. FP7-HEALTH-279024). Prof. S. Gibbs is cofounder of the university spin off company A-Skin BV. We also thank the additional members of the Dutch Outback Study Group (Outcome for treatment of burns with autologous cultured keratinocytes):
Burn center Beverwijk: R.R.H. Tempelman, A.F.P.M. Vloemans, P.P.M. van Zuijlen, R.S. Breederveld.
Burn center Groningen: G.I.J.M. Beerthuizen, M.K. Nieuwenhuis, J. Hiddingh.
Burn center Rotterdam: J. Dokter, M.E. van Baar, H. Hofland, N. Trommel.
Department of Dermatology, VU University Medical Center: M. Breetveld.
A-Skin BV, Amsterdam, The Netherlands: M. Thon, M.S.P. Haasjes.
