Abstract
Stem cell therapy is under active investigation for traumatic brain injury (TBI). Noninvasive stem cell delivery is the preferred method, but retention of stem cells at the site of injury in TBI has proven challenging and impacts effectiveness. To investigate the effects of applying a magnetic field on cell homing and retention, we delivered human neuroprogenitor cells (hNPCs) labeled with a superparamagnetic nanoparticle into post-TBI animals in the presence of a static magnetic field. We have previously devised a method of loading hNPCs with ultrasmall superparamagnetic iron oxide (USPIO) nanoparticles Molday ION Rhodamine B (MIRB™). Labeling of hNPCs (MIRB-hNPCs) does not affect hNPC viability, proliferation, or differentiation. The 0.6 tesla (T) permanent magnet was placed ~4 mm above the injured parietal cortex prior to intracarotid injection of 4 × 104 MIRB-hNPCs. Fluorescence imaging, Perls' Prussian blue histochemistry, immunocytochemistry with SC121, a human-specific antibody, and T2-weighted magnetic resonance imaging ex vivo revealed there was increased homing and retention of MIRB-hNPCs in the injured cortex as compared to the control group in which MIRB-hNPCs were injected in the absence of a static magnetic field. Fluoro-Jade C staining and immunolabeling with specific markers confirmed the viability status of MIRB-hNPCs posttransplantation. These results show that increased homing and retention of MIRB-hNPCs post-TBI by applying a static magnetic field is a promising technique to deliver cells into the CNS for treatment of neurological injuries and neurodegenerative diseases.
Keywords
Introduction
Stem cell transplantation into the adult central nervous system (CNS) as cellular therapy has been suggested for treatment of neurodegenerative diseases such as Parkinson's disease (PD) and amyotrophic lateral sclerosis (ALS), CNS trauma disorders such as spinal cord injury (SCI) and traumatic brain and axonal injury, as well as stroke and brain tumors (1,5,17,30,33,45,47,48,51). In animal models of traumatic brain injury (TBI), recent studies have demonstrated transplantation of several types of stem cells for potential therapy, and stem cell-based therapies are being evaluated for safety and efficacy in humans (ClinicalTrials.gov Identifier: NCT02028104). A serious complication occurring with stem cell transplantation in TBI and in other CNS disorders is the risk of additional intracranial hemorrhage using stereotactic neurosurgery and needle injection (5,46). These procedure-related hemorrhages can result in additional permanent neurological deficits such as paralysis or cognitive disorders. An intravascular approach allows a multiple treatment paradigm with significantly lower risks and does not cause ischemia (12,38). Although intravenous (IV) delivery is minimally invasive, it has been found that most of the cells IV injected are filtered in the lung, kidney, liver, and spleen (5,25). Intracarotid injected cells enter the injured cortex on first pass and with no blockage of vessels in the brain (11), but stem cell retention at the injury site using either form of intravascular injection is inconsistent. This problem can be mitigated by magnetic cell targeting, which has been applied most frequently in the periphery. Magnetic cell targeting is ideally suited to augment cellular therapies as the external magnetic field and field gradient may guide cells to sites of injury (3,9,10,32,37,47,48).
We have designed an approach for magnetic stem cell attraction at one of the sites of injury in TBI after intracarotid delivery. This “magnetic attraction” of stem cells has the goal of enhancing entry and retention of stem cells into the brain. Following focal impact in TBI, there is a continuum of damage from the epicenter of the injury outward into three distinct regions: (1) a volume of initial contusion, (2) a “shell” penumbra surrounding the contusion, and (3) a “shell” para-penumbra surrounding the penumbra (20,52). In our model of TBI, the parietal cortex contains both a penumbra and para-penumbra (34). We have examined viability, necrosis, and inflammation in these regions and how these processes impact injected stem cells. This approach uses human neuroprogenitor cells (hNPCs) that are labeled with ultrasmall superparamagnetic iron oxide (USPIO) nanoparticles. Visualization of transplanted stem cells by magnetic resonance imaging (MRI) can also be achieved using stem cells labeled with these types of particles (3,6,19). We find that labeling of hNPCs with a USPIO does not change cell viability, proliferation, or differentiation in vitro (43). Animals are fitted with a temporary “magnetic hat” to enable magnetic retention of transplanted stem cells. Our approach shows a significant increase in delivery efficiency and retention of USPIO-labeled hNPCs at cortical sites. The magnetic-enhanced retention of hNPCs ex vivo was also confirmed with MRI. Taken together, our results show that magnetic retention of USPIO-labeled hNPCs is a promising technique for stem cell delivery in TBI, with potential for clinical translation in other neurological injuries and in neurodegenerative diseases.
Materials and Methods
Cell Culture
hNPCs were purchased from Lonza Inc. (Walkersville, MD, USA). They were derived from human fetal cortex (15 weeks gestation) and were maintained as neurospheres in growth medium [DMEM/F-12 containing 2% B27, 20 ng/ml basic fibroblast growth factor (bFGF), 20 ng/ml epidermal growth factor (EGF); Invitrogen Life Technologies, Carlsbad, CA, USA]. Medium was changed every 2–3 days. Since the cells were obtained from a commercial source, the Institutional Review Board (IRB) of the University of Maryland granted this study an exemption (IRB Number: HP-55464).
Molday ION Rhodamine B Nanoparticles and Labeling of hNPCs
The USPIO used in these studies was Molday ION Rhodamine B (MIRB), a commercial product sold by BioPhysics Assay Laboratory (CL-50Q02-6A-50, BioPAL, Worcester, MA, USA). MIRB is a dextran-coated USPIO with a hydrodynamic diameter of 35 nm and positive charge (zeta potential +31 mV). MIRB is a homogeneous fluorescent nanoparticle designed to label cells efficiently and simply without addition of a transfection agent. The optimal concentration of MIRB for labeling hNPCs is Fe 20 μg/ml for 2 days with MIRB diluted in culture medium (43). MIRB-labeled microneurospheres (diameter < 70 μm) were dissociated with Accutase (Sigma-Aldrich, St. Louis, MO, USA), concentrated, and resuspended in media at the appropriate dilution prior to injection of MIRB-hNPCs. The cell diameter of a single hNPC after dissociation and prior to injection was 15.5 ± 0.4 μm (n = 66).
Fe Measurements of MIRB-hNPCs
Iron Detection by ICP-MS
Inductively coupled plasma mass spectrometry (ICP-MS) was performed to independently quantify cellular iron uptake (UMB School of Pharmacy, Mass Spectrometry Center). Iron concentrations were determined by injecting samples into an Agilent 7700x ICP-MS (Agilent Technologies, Santa Clara, CA, USA). Iron levels were detected using He mode with a radio frequency (RF) power of 1550 W, an argon carrier gas flow of 1.0 L/min, argon make-up gas flow of 0.1 L/min, helium gas flow of 5.5 ml/min, octopole RF of 160 V, QP bias of −15 V, and OctP bias of −18 V. Samples were directly infused using a nebulizer pump speed of 0.1 rps. Iron concentrations in samples were derived from a calibration curve generated by a series of dilutions of iron atomic absorption standards (Fluka Analytical, Seelze, Germany) prepared in a manner similar to the samples. Data analysis was performed using the manufacturer-provided Mass Hunter software (Agilent Technologies).
Sample Preparation
Cell pellets of MIRB-hNPCs and control hNPCs were stored at −80°C until assayed for iron content. Cells were resuspended by vortexing and allowed to digest overnight at 65°C in 1 ml of 30% trace metal grade HNO3 (Thermo Fisher Scientific, Pittsburgh, PA, USA). MIRB-digested hNPCs (500 μl) were diluted to 2 ml in Milli-Q water (Elga Veolia, Lowell, MA, USA), from which a 100-μl aliquot was diluted to 5 ml in 2% trace metal grade HNO3 (Thermo Fisher Scientific) with a 1 ppm internal standard spike of Sc, Ge, and In. For nonlabeled hNPCs, a 20-μl aliquot was diluted to 3 ml in Milli-Q water with a 1 ppm internal standard spike.
Magnetic Field Effects on MIRB-hNPCs In Vitro
MIRB-labeled hNPCs, labeled for 2 days, were plated on six-well culture plates and magnetic discs (diameter × thickness = 1/2″ × 1/2″) were placed under the wells for 2 days or 4 days followed by propidium iodide (Sigma-Aldrich) staining for viability. For the proliferation study, the magnet was under the culture plate for 5 days followed by 5′-bromodeoxyuridine (BrdU; Sigma-Aldrich) incorporation for 6 h. For the differentiation study, the magnet was under the culture plate for 7 days, while hNPCs were in differentiation medium consisting of DMEM/F-12 containing 2% B27, without bFGF and EGF (Invitrogen Life Technologies). The magnetic surface field strength and the intensity of magnetic field at certain distances from the edge of the magnet were calculated using K&J Magnetic Field Calculator (http://www.kjmagnetics.com/fieldcalculator.asp).
Controlled Cortical Impact TBI
All surgical procedures and experiments were carried out in accordance with protocols and guidelines approved by the Institutional Animal Care and Use Committee at the University of Maryland. All animals were individually housed in the animal facility with ad libitum access to food and water. Adult male Sprague–Dawley rats (290–330 g; Taconic Biosciences, Albany, NY, USA) were anesthetized intraperitoneally [IP; 60 mg/kg ketamine (Putney, Portland, ME, USA) and 7.5 mg/kg xylazine (Bayer Healthcare LLC, Shawnee Mission, KS, USA)] and allowed to breathe air spontaneously. Core temperature was maintained at 37°C using a heating pad.
There were four groups of animals: Sham/No Magnet (n = 4), Sham/Magnet (n = 4), TBI/No Magnet (n = 5), and TBI/Magnet (n = 8). The animal's head was fixed in a stereotaxic frame (Stoelting Co., Wood Dale, IL, USA). Using aseptic techniques, a 6-mm craniectomy in the left parietal bone was made midway between bregma and lambda and abutting the sagittal suture and left lateral ridge. Impact injury was induced using a modified Feeney device, as previously described with some modifications (34). The vertical guide tube (inside diameter 6 mm) was angled −10° relative to the stereotaxic frame in the transverse plane, and the spherical impactor was gently positioned on the dura at −3.6 mm anteroposterior and +2.6 mm mediolateral to the bregma. The impact tip was advanced further to produce a brain injury of moderate severity for rats (tip diameter 4 mm, cortical contusion depth 3 mm, impact velocity 0.77 m/s). The dura was not lacerated by this procedure. The craniectomy was left open, and the scalp was closed after the hat was attached to the skull. There was no surgery-related mortality.
Placement of Magnet, Hat, and Stem Cell Transplantation
Prior to stem cell injection, two disc magnets were placed in a “hat” over the cortical lesion (Fig. 2). The hat was a Plexiglas cylinder over the lesion held in place by VetBond and sutures. The magnets were nickel-coated neodymium circular discs [D42B-N52; diameter = 1/4″ and thickness = 1/8″; the combined surface magnetic field 0.66 T; Brmax = 1.48 T; BHmax = 413.8 kJ/m3; magnetization: axially magnetized (poles on the flat ends of disc); K&J Magnetics, Plumsteadville, PA, USA]. The hat can be held in a stable position on the skull for up to 30 days (longest time tested) without distress to the animal. The output surface field at the site of injury 2.5 mm below the bottom of the hat was calculated to be 0.24 T (K&J Magnetics, KJM calculator). For ex vivo MRI studies of stem cell location, the injured cortex of the TBI/No Magnet animals was used as a control.
To prevent immune rejection of the human stem cells, cyclosporine (10 mg/kg; Novartis Pharmaceuticals, East Hanover, NJ, USA) was administered daily by IP injection. These injections started 3 days prior to the internal carotid artery injection and continued until the animals were sacrificed. Internal carotid injections were performed at 72 h after TBI (45). The external carotid artery and pterygopalatine artery on the injury side were isolated and ligated with a 3-0 silk for external carotid artery and 6-0 silk for pterygopalatine artery. The MIRB-hNPCs (40,000 cells suspended in 0.15 ml of sterile serum-free DMEM/F-12 (Invitrogen Life Technologies) were injected into the internal carotid, fully operational, through the external carotid for over 1 min using a 0.2-ml Hamilton syringe with a 30-gauge needle; a similar method was used by Chua et al. (11). With this procedure, blood flow in both the common carotid and internal carotid arteries was preserved, and only the external carotid and pterygopalatine arteries remained ligated. No morbidity or mortality was found with the internal carotid injection. No microemboli were detected following internal carotid injection of MIRB-hNPCs with or without a magnetic hat, by either immunocytochemistry with RECA-1 antibody, a marker of rat brain vasculature, or by Fluoro-Jade C (FJC) histochemistry, to detect degenerating neurons with the FJC staining at noninjury sites.
Immunocytochemistry and Histochemistry
Five days post-internal carotid injection of MIRB-hNPCs, the animals were sacrificed and perfused with paraformaldehyde (PFA, 4%, pH 7.4; Sigma-Aldrich). The dissected brains were cryostat sectioned at 40 μm. Immunocytochemistry of the brain sections was performed as previously described (42). The dissected brains were sectioned with a cryostat at 40 μm. Immunocytochemistry of the brain sections was performed as previously described (43). Briefly, the sections were incubated for 1 h in block solution consisting of 1× phosphate-buffered saline (PBS; Invitrogen Life Technologies), 0.2% Triton X-100 (Sigma-Aldrich), and 4% normal donkey serum (Jackson ImmunoResearch Laboratories, West Grove, PA, USA), then in primary antibodies diluted in block solution at room temperature overnight. After washing in PBS, the sections were incubated in secondary antibodies for 1 h. The secondary antibodies were FITC- or TRITC-coupled donkey anti-mouse or anti-rabbit IgG (Jackson ImmunoResearch Laboratories) depending on the species of the host for primary antibodies, and diluted 1:500 in 1× PBS + 0.2% Triton X-100. If necessary, the sections were counterstained with Hoechst 33342 (Sigma-Aldrich) 1 μg/ml for 30 min. Finally, the sections were mounted and coverslipped with Vectashield mounting medium (Vector Laboratories, Burlingame, CA, USA). Antibodies included RECA-1 (1:500; Serotec, Raleigh, NC, USA), SC121 (human-specific cytoplasmic antibody, 1:500; Stem Cells, Palo Alto, CA, USA), Iba-1 (1:1,000; Wako Chemicals USA, Cambridge, MA, USA), and BrdU antibody (1:400; Accurate Chemical & Scientific Corp., Westbury, NY, USA).
For FJC (EMD Millipore, Bedford, MA, USA) staining, sectioned brain tissue was mounted and dried overnight. They were then immersed in 80% ethanol + 1% sodium peroxide (Sigma-Aldrich) for 5 min, rinsed in 70% ethanol and distilled water, then incubated in 0.06% potassium permanganate (Sigma-Aldrich) solution for 10 min. Following a 2-min wash with water, the sections were stained in 0.0001% FJC solution for 10 min.
To detect MIRB labeling, either rhodamine fluorescence visualization or Perls' Prussian blue iron staining (2% HCl, 2% potassium ferrocyanide; Sigma-Aldrich) for 10 min at room temperature was performed. MIRB-hNPCs were also visualized using MRI (see below).
All microscopic images were collected using a Zeiss Axio Observe Z1 inverted microscope (Carl Zeiss Microscopy GmbH, Jena, Germany) with quantitative analysis (densitometry) performed using Zeiss Axiovision v4.8 software.
MRI Experiments
All MRI experiments were performed on a Bruker BioSpec 70/30USR Avance III 7T horizontal bore MR scanner (Bruker Biospin MRI GmbH, Karlsruhe, Germany) equipped with a BGA12S gradient system and interfaced to a Bruker Paravision 5.1 console. A Bruker four-element proton surface coil array was used as the RF receiver and a Bruker 72 mm linear-volume coil as the transmitter. For in vivo analysis, T2-weighted imaging (T2WI) was done immediately postinjury before MIRB-hNPC transplantation. Images were obtained using a 2D rapid acquisition with relaxation enhancement (RARE) sequence in the coronal plane. The acquisition parameters consisted of a repetition time (TR)/effective echo time (TEeff) of 5500/56.8 ms, RARE factor of 4, field of view (FOV) of 30 × 30 mm2, slice thickness of 1 mm, in-plane resolution of 170 × 170 mm2, number of averages = 2. The duration of the imaging session postinjury was 2 h. For ex vivo imaging following paraformaldehyde fixation, brains were immersed in a proton-free fluid (Fomblin; Solvay, Baltimore, MD, USA) during the scan. In ex vivo analysis, for data acquisition of the location and the volume occupied of MIRB-hNPCs following transplantation, T2WI was used with 3D rapid acquisition with RARE sequence. The duration of this imaging session was overnight. The acquisition parameters consisted of a TR/TEeff of 3,500/12 ms, RARE factor of 8, FOV of 20 × 20 × 28 mm3, and spatial resolution of 150 × 150 × 150 μm3, with two averages.
Image Processing
The high-resolution MR images were processed using the shading correction, volume-of-interest drawing, and statistics functions of Medical Image Processing, Analysis, and Visualization (MIPAV v5.3.1; CIT, NIH, Bethesda, MD, USA). Shading correction was first performed on all images using the Inhomogeneity N3 correction function. For the TBI rat brains, the low signal regions inside of the lesion were manually drawn. The percentage of low signal region was calculated using volume (mm3) ratios of focal low signal volume to the rat brain.
Data Analysis
For in vitro experiments, data were expressed as mean ± standard error of the mean (SEM). A total of 1,000–2,000 outgrowth cells from five random fields from nine separated neurospheres were counted. Statistical significance of the intergroup differences was assessed using Student's t-test or ANOVA followed by the Tukey's post hoc test. For nonnormal data, data were expressed as mean and interquartile range. Nonparametric data were analyzed using the Kruskal–Wallis test followed by the Mann–Whitney test. Moreover, Bonferroni's correction was applied to account for multiple testing. Student's t-test (two-tailed, unpaired) was used to compare the viability, differentiation, and proliferation of MIRB-hNPCs in the presence of a magnetic field to these properties in hNPCs without either MIRB or a magnetic field. We have previously shown that there is no difference in these properties between MIRB-hNPCs and hNPCs in the absence of a MIRB (43).
For ex vivo experiments, we analyzed the distribution of MIRB-hNPCs in the brain and peripheral tissues using Perls'-labeled or SC121-labeled stem cells (see below) in the cortex and calculated the ratio of the optical density of MIRB-hNPCs in magnet animals to the optical density in TBI animals that did not receive a magnet to confirm the targeting efficiency of magnetic retention of stem cells. In both studies, p values were calculated using Prism 5.0 software (GraphPad Software Inc., La Jolla, CA, USA); a value of p < 0.05 was considered statistically significant at the 95% confidence level.
Results
Magnetic Attraction of NPCs In Vitro
We have recently shown that hNPCs can be labeled in vitro with MIRB and that the MIRB labeling does not compromise hNPC viability, proliferation, or differentiation when compared with unlabeled hNPCs in vitro (43). Labeling of hNPCs with MIRB represents an advantageous and promising tool for stem cell transplantation in the CNS. In the current study, we investigated whether MIRB-labeled hNPCs could be targeted and retained in situ in the presence of a static magnetic field in a TBI model. First we investigated the attraction of MIRB-hNPCs in a magnetic field in vitro. When hNPCs were plated in cultureware and labeled with MIRB for 2 days, they contained 14.39 ± 1.15 pg Fe/cell (n = 6 flask cultures) and unlabeled hNPCs contained 0.014 ± 0.002 pg Fe/cell as determined by ICP-MS. Placement of neodymium magnets under the cell culture dish (dashed line indicates location of the magnets; surface magnetic field, 0.66 T) changed the cell distribution from a uniform (Fig. 1A1–A3, no magnetic field applied) to a non-uniform distribution (Fig. 1B), with the majority of the MIRB-hNPCs attracted within an effective range of 4 mm (Fig. 1B1, B2). A uniform cellular distribution was found beyond 4 mm (Fig. 1B3, surface magnetic field at 4 mm = 0.07 T), similar to Figure 1A. Semiquantitative analysis of MIRB fluorescence densitometry (Fig. 1C) indicated that the signal of MIRB-hNPCs above the magnet was 10-fold greater than the signal in the control cultures (Fig. 1C, B1). These results confirmed that MIRB-hNPCs are responsive to the presence of a magnetic field in vitro. Using the same static magnetic field, we also showed that a magnetic field did not affect cellular properties of MIRB-hNPCs, such as viability, differentiation, or proliferation in vitro (Fig. 1D–G). There were also no differences in these properties between unlabeled hNPCs and MIRB-hNPCs (43).
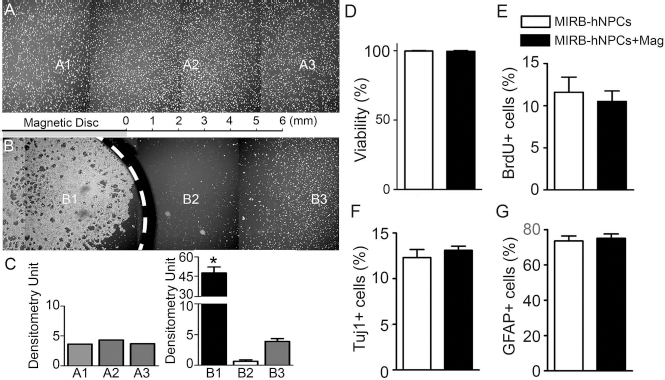
Effects of a static magnetic field on MIRB-hNPC properties in vitro. Dissociated MIRB-hNPCs were plated on regular culture plates, and magnets (0.66 T) were placed under the wells. Both fluorescent and phase images showed that in the control cultures without a magnet field, undifferentiated MIRB-hNPCs were evenly distributed in the wells (A). Placing magnets under the culture dish attracted MIRB-hNPCs with effective distance of 6 mm (B1, B2). Please note that above 6 mm of distance the magnets are not effective (B3). Semiquantitative analysis indicated that the densitometry of MIRB-hNPCs at locations beyond 6 mm is similar between magnet and nonmagnet cell populations (C). However, at less than 6 mm, the densitometry of MIRB-hNPCs above the magnet is 10-fold greater than in controls (C). Magnets (0.66 T, total) were placed under the MIRB-hNPC cultureware for 4 days. Cultures were then labeled with propidium iodide and counterstained with Hoechst dye. There was no difference in hNPC viability between hNPCs ± magnetic field, no matter whether they contained MIRB (D). To test whether a static magnetic field affected proliferation, MIRB-hNPCs cultures were pulsed with BrdU (4 μM) for 6 h after being exposed to the magnetic field for 5 days. Counting the number of BrdU-labeled cells showed that the magnetic field did not change hNPC proliferation (E). Further, the effect of the magnetic field on MIRB-hNPC differentiation was examined. Similar to the conditions used to assess hNPC proliferation, the numbers of neurons (F; Tuj1+) or glia (G; GFAP+) generated under a static magnetic field in differentiation medium showed no change in properties as compared to hNPCs in differentiation medium without a static magnetic field. The data are expressed as mean ± SEM; *p < 0.05. At least 600 cells counted for every group of data presented here.
MRI Detection Following Magnetic Retention of NPCs After TBI
An advantage of using MIRB-hNPCs is that MRI can track the location of the stem cells immediately after and following transplantation. In order to retain MIRB-hNPCs in the cortex at the site of injury following TBI, we designed a “magnetic hat” (Plexiglas cylinder of 1 cm in height) attached to the skull at the injury site that contained two neodymium magnets. The surface magnetic field was 0.66 T. The distance between the skull and the deepest part of the impacted cortex was 2.5 mm. At this distance, below the skull, the intensity of the magnetic field is 0.24 T. Three days after TBI, the hat was fitted on the injury side of the skull before intracarotid delivery of MIRB-hNPCs (Fig. 2A,B). Animals tolerated wearing the hat well without bumping their heads in cages or attempting to remove the hat. The hat and magnets stayed on the rat's head for the duration of the study (5 days following intracarotid delivery of cells).
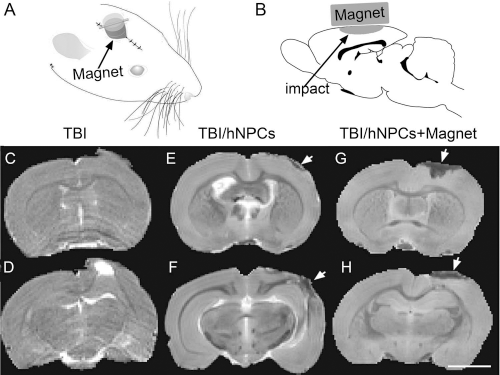
Detection of magnetic-enhanced retention of MIRB-hNPCs following TBI. Illustrations showing the magnetic hat (A) and the location of the impact injury in the CCI TBI model used (B). (C, D) In vivo T2WI of TBI rats 2 h after CCI and before MIRB-hNPC transplantation; animals were not wearing a magnetic hat. The region of increased signal contrast in (D) is due to edema. (E, F) Ex vivo T2WI 5 days after MIRB-hNPC transplantation in TBI controls not wearing a magnetic hat. A small decrease in signal contrast was seen at the site of injury. (G, H) Ex vivo T2WI in TBI rats wearing a magnetic hat. An extensive decrease in signal contrast was found. The location of the loss of signal contrast was confirmed by Perls' Prussian blue staining and with immunocytochemistry of MIRB-hNPCs with human-specific SC121 antibody. Scale bar (C–H): 4 mm.
We used T2WI, in vivo and ex vivo, to detect the location of MIRB-hNPCs in the cortex following stem cell injection. In TBI animals immediately after controlled cortical impact (CCI), but before hNPC transplantation, there was an increase in T2WI contrast detected at the site of injury in vivo, indicating edema (Fig. 2C,D). In ex vivo analyses, it was observed that T2WI at 5 days after MIRB-hNPC injection in control animals (TBI with MIRB-hNPCs injection) there was a small area with decreased T2WI contrast at the injury site (Fig. 2E,F). With the magnetic hat present during MIRB-hNPC injection, there was a noticeable decrease in T2WI contrast (Fig. 2G,H). The extent of hypointensity seen with T2WI in Figure 2C–H, was similar to the Perls' Prussian blue staining pattern observed in Figure 3.

In TBI animals, localization of transplanted MIRB-hNPCs following intracarotid injection is increased by magnetic retention. Five days after transplantation of MIRB-hNPCs, histological analysis of hNPC localization with Perls' Prussian blue staining. (A) A representative section following TBI controls, not wearing a magnetic hat (n = 5), “TBI, No Magnet,” showed low levels of USPIO-containing cells in the impact region. (B) A representative section from TBI rats wearing a magnetic hat (n = 8), “TBI + Magnet.” There were higher levels of USPIO-containing cells in the impact region. (C) A representative section from Shams, no TBI (“Sham, No Magnet”) not wearing a magnetic hat (n = 4) showing no USPIO-containing cells retained. (D) A representative section from Shams, no TBI (“Sham + Magnet”) wearing a magnetic hat (n = 4) showing no USPIO-containing cells retained. Scale bar: 1 mm.
Magnetic Retention of NPCs After TBI
Previous studies have shown the feasibility of magnetic cell targeting in vivo with mesenchymal or neural stem cells in models of peripheral vascular injury, SCI, or stroke (50). We considered whether MIRB-hNPCs could be retained in vivo in the CCI TBI model. Our technique uses a magnetic hat that can be removed at any time for MRI localization of MIRB-hNPCs and is only necessary for a short time after stem cell transplantation; 1 day posttransplantation was the shortest time tested in these studies (not shown). Five days after stem cell injection, animals were euthanized and brains were collected and prepared for histology and quantification of hNPC location.
Perls' staining showed the localization of transplanted MIRB-hNPCs following intracarotid injection in representative animals modeled with TBI (Fig. 3). Figure 3A shows low levels of MIRB-containing cells in the impact region of control rats without the magnetic hat. There was an increase above control levels in stem cell retention in rats wearing the magnetic hat (Fig. 3B), suggesting that the enhanced retention of MIRB-containing cells in the impact region is attributable to the applied external magnetic field. In sham animals with no TBI injury (Fig. 3C,D), there were no MIRB-hNPCs cells in the parietal cortex in the presence or absence of the magnetic hat. Thus, the magnetic field enhances retention of MIRB-hNPCs after CCI. These results indicate that applying a magnetic field to the area of cortex injured in CCI retained more MIRB-hNPCs in the impact region than with transplantation alone. In sham animals that had an intact blood–brain barrier (BBB; data not shown), there was no retention of MIRB-hNPCs by an external magnetic field.
Both Perls' Prussian blue staining (Fig. 3) detecting the presence of iron (ferric iron) and immunolabeling with the human-specific antibody SC121 were used to determine MIRB-hNPC location following stem cell injection. Hemorrhage associated with TBI did not interfere with quantification of SC121 immunofluorescence or Perls' Prussian blue staining. All tissue sections were pretreated with H2O2 to eliminate any artifacts. Coronal sections from bregma, −0.96 mm to −6 mm, the CCI-impacted region, were analyzed using Perls' Prussian blue staining. Since the diameter of the magnet was 6.35 mm, which covered the impact region, the sections we analyzed here were located 3 mm anterior or posterior to the center of the magnet. After Perls' Prussian blue staining, the optical density of MIRB-hNPCs retained in the parietal cortex of these sections was measured. Quantitative analysis of the densitometry readings in Figure 3 showed that more MIRB-hNPCs were found in the parietal cortex of the rats with a magnetic hat (Fig. 4A, filled circle) than in the control parietal cortices without a magnetic hat (Fig. 4B, open circle). Sham animals (non-TBI, with or without a magnetic hat) showed no retention of MIRB-hNPCs in the cortex (Fig. 4B, x symbol and open square). Figure 4B shows the scatter plot of the mean ± SEM of the densitometry measurements of individual animals injected with MIRB-hNPCs. A greater than 2.8-fold (2.83 ± 0.01) increase in mean optical density was measured. The Mann–Whitney U test indicated the difference of MIRB-hNPC retention between magnet and nonmagnet TBI animals was significant (*p < 0.05; n = 8 magnet animals; n = 5 nonmagnet animals). These results show that using a static magnetic field enhances the retention of MIRB-labeled cells in the cortex after CCI. No differences in the distribution patterns between Perls' Prussian blue staining and the immunolabeling with SC121 antibody of MIRB-hNPCs in the injured cortical area including the penumbral and para-penumbral regions (Fig. 5) were found.

Analysis of magnetic retention of MIRB-hNPCs following TBI. Sections containing impact region under the magnet were stained with Perls' Prussian blue. The sections quantitated are distributed in the distance of ± 3 mm to the center of the magnet (= 0 mm) in the rostral-to-caudal axis (A). Densitometry analysis of Perls' Prussian blue staining posttransplantation indicated that, in TBI animals, applying a static magnetic field before cell transplantation retained more MIRB-hNPCs in the injured cortex (C, n = 8, filled circle) than in the control injured cortex without a static magnetic field (C, n = 5, open hexagon). Sham animals (C, n = 4, x symbol, open square) without TBI showed no retention of MIRB-hNPCs in the cortex following transplantation with or without the magnetic hat. (B) A scatter plot of the mean densitometry and injury volume from individual animals shown in (A) (filled circle, open square) indicated that a static magnetic field enhances the retention of MIRB-hNPCs in the injured cortex. The Mann-Whitney U test indicated the difference of MIRB-hNPC retention between magnet and no magnet was significant (mean ± SEM; *p < 0.05).

MIRB-hNPCs are immunostained with both SC121 antibody and Perls' Prussian blue histochemistry. At the impact region in the parietal cortex, sequential sections from a MIRB-hNPC-transplanted TBI rat wearing a magnetic hat were stained with either Perls' Prussian blue solution (A–C) or fluorescent immunohistology labeling with SC121 antibody and rhodamine fluorescence (D–F). Arrows point to representative Perls' Prussian blue cells (C), to MIRB-labeled cells (D), or to hNPCs labeled by SC121 antibody (E). (F) Merged fluorescent image from (D, E). This demonstrates that viable MIRB-hNPCs are retained in the impact region and can be visualized by either Perls' Prussian blue staining or fluorescent immunohistology techniques. Scale bars: (A) 1 mm; (B) 100 μm; (C–F) 50 μm.
Fate of hNPCs Containing MIRB in the Penumbra and Para-Penumbra of the Impact Region in TBI Rats
We evaluated the status of the fate of hNPCs in the penumbra and para-penumbra of the impact regions, as shown in Figure 3 following MIRB-hNPC injection (34). The sequential sections were stained with Perls' Prussian blue (Fig. 5A–C), SC121 immunolabeling (Fig. 5D–F), and MIRB fluorescence (Fig. 5D–F). This staining of hNPCs demonstrated the same distribution patterns regardless of the histological methods used. Higher magnification showed that individual cells were visible in Perls' (Fig. 5C, black arrowheads), in MIRB fluorescence, and in immunolabeling with SC121 antibody (Fig. 5D, E, black arrowheads). The merged fluorescent images (Fig. 5F) and Perls' staining, which all overlap, demonstrate that at 5 days posttransplantation there are viable hNPCs in the para-penumbra of the impact region that colocalize as MIRB+, SC121+, and Perls+ cells (Fig. 5C and F, black arrowheads).
Necrosis and Inflammation of hNPCs in the TBI Penumbra and Para-Penumbra
In the sections immunolabeled with SC121 antibody (Fig. 6B) and labeled with Hoechst nuclear counterstaining (Fig. 6C), we found that MIRB-hNPCs made up a small fraction of the total cells in the region of injury (Fig. 6A–C, black arrowheads). To evaluate the effects of necrosis and of injury-induced inflammation following TBI on hNPC viability in the penumbra and para-penumbra of the impact region, we used FJC as an assessment tool for tissue necrosis (Fig. 6). The penumbra region in the parietal cortex contained FJC+ staining from the damage caused by TBI impact. It contained necrotic MIRB-hNPCs (FJC+; Fig. 6G–I, white arrowheads) and MIRB-hNPCs that were nonnecrotic (FJC–; Fig. 6D–F, black arrowheads). Necrosis of some MIRB-hNPCs was found in all animals studied.
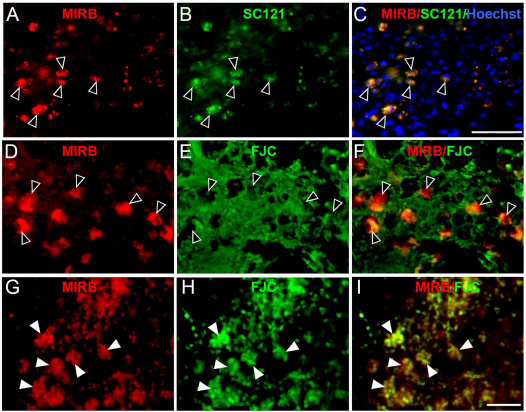
Both nonnecrotic and necrotic MIRB-containing cells are present in the penumbra region of TBI rats. (A–C) Immunocytochemical analysis with dual labeling of MIRB and human-specific SC121 antibody showed examples of nonnecrotic MIRB+/SC121+ cells in the penumbra region (black arrowheads). (D–I) Assessment of necrosis of MIRB-hNPCs in the penumbra area with FJC staining found both nonnecrotic MIRB+/FJC– cells (D–F, black arrowheads); and (G–I, white arrowheads) necrotic and swollen cells, MIRB+/FJC+ and debris. Scale bar: (A–C) 200 μm; (D–F) 50 μm; (G–O) 25 μm.
In addition to necrosis, there was increased inflammation in the penumbra and para-penumbra regions, as shown by increased immunolabeling with Iba-1, a macrophage/microglia marker (27). In a different animal from that shown in Figure 6, immunocytochemistry analysis demonstrated that in the para-penumbral region, some of the SC121+ human cells were engulfed by Iba-1+ macrophages/microglia (Fig. 7A–D, white arrows), but some of grafted hNPCs survived (Fig. 7A–D, black arrowheads) at 5 days postinjection. The most common appearance of Iba-1+ cells in the para-penumbral region is shown in Figure 7D. In these cells, phagocytosis of hNPCs did not occur at this time. Another example of both macrophage/microglial phagocytosis and survival of hNPCs is shown in Figure 7E–G; there are engulfed hNPCs (white arrows) and surviving hNPCs (black arrowheads), respectively. To confirm whether the SC121+ cells were MIRB+-labeled cells and whether MIRB was still held by hNPCs, we triple-labeled the sections with MIRB fluorescence, SC121, and Iba-1 antibodies (Fig. 7H–K). This data demonstrated 1) that there were viable MIRB-hNPCs (MIRB+ and SC121+, black arrowheads), and 2) there were dying MIRB-hNPCs engulfed by macrophages/microglia (white arrows), but these dying cells displayed cytoplasmic SC121 immunoreactivity and MIRB fluorescence. We did not detect SC121+-labeled hNPCs in the cortex without colabeling with MIRB. We also did not observe MIRB-labeled Iba-1+ cells only. Iba-1+ cells in the para-penumbra were either SC121+ (Fig. 7, black arrowheads), the minority observed, or SC121–, the majority observed. Our findings suggest that dying MIRB-hNPCs in the para-penumbra at 5 days posttransplantation undergo phagocytosis by macrophages/microglia and do not release their MIRB. Phagocytosis of MIRB-SC121+ by Iba-1+ cells was found in all animals studied.

Inflammation and inflammatory cell death of hNPCs in the para-penumbra region of TBI rats. In the para-penumbra region, there are both viable hNPCs and hNPCs undergoing phagocytosis. Dual labeling of hNPCs using human-specific antibody SC121 (A) and microglia/macrophage antibody Iba-1 (B) found SC121+ cells inside Iba-1+ cells (A–D, white arrows); other SC121+ cells were alive, not engulfed by Iba-1+ cells (A–D, black arrowheads). (D) Another field showing that the majority of Iba-1+ cells in this region have not phagocytized SC121+ cells (black arrows). Another example of the mixed population of MIRB+ cells in the para-penumbra region is shown in (E–G), where the black arrowheads point to MIRB+ cells that are not inside microglia/macrophages (Iba-1–), and the white arrows indicate MIRB+ cells inside Iba-1+ cells. To confirm that MIRB+ cells inside microglia/macrophage are also SC121+ cells, a triple immunofluorescent image is shown (H–K). In this image, MIRB+ cells Rhodamine B+, SC121 cells are immunostained with a fluorescein secondary, and Iba-1+ cells are immunostained with an AMCA secondary. Both SC121+/MIRB+ cells inside Iba-1+ microglia/macrophages (white arrows) and SC121+/MIRB+ cells not inside Iba-1+ cells are present (black arrowheads). Prior to transplantation, none of the Iba-1+ cells express SC121. Figures 6 and 7 are from different animals. Scale bar: (A–K) 25 μm.
Our studies confirm the findings in previous studies in TBI about the fates of stem cells following injection and also confirm that the loss of signal contrast seen on MRI following MIRB-hNPC transplantation is not solely due to the location of live hNPCs, but also contains dying hNPCs (6,7,29). Current studies are aimed to investigate these processes.
Discussion
Cellular therapy for TBI has been suggested to replace dead cells, restore growth factor homeostasis, repair damaged circuits, and improve functional recovery (4,16,31). Stem cell therapy for TBI has been given by intracerebral injection, IV injection, lumbar puncture, and intracarotid injection (1,26,49,54). However, the efficacy of each of these techniques has not been addressed thoroughly. We aimed to develop a less invasive procedure for enhancing cellular therapy for TBI. A serious complication occurring with stem cell transplantation in the CNS is the risk of intracranial hemorrhage, when carried out via stereotactic neurosurgery and needle injection. These procedure-related hemorrhages can result in permanent neurological deficits such as paralysis or cognitive disorders and hemorrhage-related inflammatory events (5,46).
Previous studies and our data support the lack of stem cell entry into the brain unless the BBB has been disrupted (10,25). In the studies investigating cellular therapy for models of stroke and TBI, intracarotid injection was found to deliver the greatest number of stem cells to the brain. We chose to deliver hNPCs by intracarotid injection 3 days after TBI based upon studies that determined the optimal administration protocol for stem cell injection (5,25,35,41). For certain CNS injuries and conditions, an intravascular approach allows for a multiple treatment paradigm with significantly lower risks and does not cause ischemia (12,38). TBI is one CNS injury in which the BBB is compromised (2). Following TBI, cells delivered by intracarotid injection gain access to injured tissue not only because of the opening of the BBB but also because of decreased blood flow around the injury site (28,39). Although IV delivery is minimally invasive, it has been found that most of the cells IV injected are filtered into the lung, liver, and spleen (5,26). Intracarotid injected cells enter the injured cortex with minimal blockage of brain vessels, unless the cell diameter is large (12,17). hNPCs are small cells with a mean diameter of about 15 μm (39).
The potential effectiveness of cell-based therapies on the brain is dependent upon homing/retention of transplanted cells to sites of injury. We have shown that there is enhanced hNPC homing/retention to the damaged cerebral cortex following TBI using magnetic attraction of MIRB-hNPCs. Magnetic attraction of USPIO-loaded cells has been used to enhance the delivery of stem cells to a wide range of target tissues, including liver, muscle, joint, heart, and retina as well as the brain (3,9,11,18,24,32,37,47,50,53). Cell types assessed in previous studies include mesenchymal stem cells, hematopoietic stem cells, cardiac stem cells, and human and rodent NPCs (10,21,22). Both static magnets as well as electromagnets have been employed with magnetic field strengths ranging from 0.2 to 5 T. Peripherally, magnetic retention of stem cells has been reported following vascular injury and for cartilage repair (18,37). Successful outcomes have included increased number of cells in the target tissue as well as physiological improvement.
Two published studies have evaluated magnetic attraction of stem cells to the brain. Both studies utilized a rodent stroke model with the rationale that stem cells enter the brain from the systemic circulation in this model, presumably due to BBB damage. The study by Song et al. showed that an external magnet (0.32 T) mounted on the skull of rats for 1 week increased the number of USPIO-loaded hNPCs after IV injection over threefold to the area of infarction under the magnet, with a significant reduction in infarct size (47). Li et al. used endothelial progenitor cells loaded with USPIO and similar methods and magnet strength for delivering cells following infarction (23). Our data supports the feasibility of intracarotid delivery including a demonstration that application of a magnetic field can significantly improve the homing/retention of USPIO-loaded stem cells after intracarotid injection in a model of disease where the BBB is compromised, but our method is not TBI specific. Other neurological diseases where BBB permeability occurs or transiently opening the BBB using MRI-guided focused ultrasound prior to stem cell therapy would allow for this method of magnetically enhanced stem cell homing/retention to be applied (8). This method is ideally suited to augment cellular therapies as the external magnetic field and field gradient retain cells at sites of injury. In addition, USPIO-labeled cells can be tracked in vivo in the CNS using MRI (6).
We have previously shown that labeling hNPCs with MIRB does not affect their viability, proliferation, and differentiation as compared with unlabeled hNPCs in vitro. Once labeled, hNPCs hold MIRB up to 3 weeks, which was the longest time we examined under differentiation conditions (43). Now we have shown that in the presence of a static magnetic field, MIRB-hNPCs show normal viability, proliferation, and differentiation properties (Fig. 1). Using our method of assessing magnetic attraction, we have determined that with our labeling protocols, the effective range for magnetic attraction on MIRB-hNPCs in vitro was 4 mm. At this distance, the magnetic field decreased to 0.07 T. In TBI animals with the magnet placed on the skull above the impact region, the sections we analyzed were in the effective range of the magnetic field. This field strength is much greater than the field strength needed to attract and retain MIRB-hNPCs in vitro (Fig. 1). Indeed, analysis of Perls' stained coronal sections containing the CCI impacted region demonstrated the enhanced retention of MIRB-hNPCs (Figs. 3, 4).
Having established no toxic or deleterious effects from either labeling hNPCs with MIRB or on its properties in a maintained magnetic field, we explored whether an applied magnetic field changes properties of hNPCs in vivo. We only explored the initial properties 5 days postinjection in the present study and found no toxic or deleterious effects of either maintaining MIRB inside of hNPCs or effects caused by application of a magnetic field on MIRB labeling of hNPCs. These findings confirm previous findings using a variety of different stem cells labeled with MIRB (28,30,36,40). Although potential clumping of cells after intra-arterial injection is of concern because of the risk of vessel occlusion and ischemia, our data (RECA-1 immunocytochemistry–Perls' Prussian blue histochemistry) do not show intravascular clumping of transplanted cells.
We found that MIRB-hNPC magnetic retention occurs after 1 day and can be seen with MRI (data not shown). The strength of the magnetic field decreases dramatically as the distance from the magnet discs increases. In our present study, we used a small magnet due to the size of the animal model. In humans, we would have to build a magnetic hat to maintain enough field strength to retain MIRB-labeled cells at deeper regions. Using a Halbach magnet, it should be possible to maintain sufficient field strength (37). USPIO had no deleterious effect on cell viability or differentiation, consistent with other studies and our own published work (13,14,43). We found that MIRB-hNPCs without magnetic attraction do not accumulate significantly at the site of injury. Our findings (Figs. 3, 4) are similar to those of Lundberg et al., when a magnetic field is not present during transplantation (26).
MRI also detected magnetic retention of MIRB-hNPCs (Fig. 2). It occurred in the same cortical location as the Perls' Prussian blue staining and SC121+ immunoreactivity (not shown). The ex vivo T2WI detection of MIRB-hNPCs at 5 days postinjection using magnetic retention was greater than the T2WI detection of MIRB-hNPCs without magnetic retention (54) (Fig. 2). In our studies, we observed that the BBB is opened postinjury with this type of TBI (2,15), allowing magnetic retention at the site of injury. There was no magnetic retention of MIRB-hNPCs in sham animals with or without a magnetic hat (Figs. 3, 4).
The location of the retained MIRB-hNPCs following TBI determines their survival. In the parietal cortex, both the penumbra, surrounding the initial contusion, and the para-penumbra, surrounding the penumbra, are present (34). In the penumbra and para-penumbra, native neurons can exhibit cleaved caspase 3 consistent with apoptosis, and will also undergo degeneration marked by Fluoro-Jade staining (20), consistent with necrosis. Similar changes are also expected in hNPC viability as seen in Figures 5, 6, and 7. Following TBI, there is tissue loss due to necrosis, inflammation, and apoptosis. An important issue concerning use of USPIO in stem cells is what happens to the nanoparticles when the hNPCs die. In culture, we considered whether a MIRB-labeled cell releases its MIRB into the extracellular space when it dies. We induced apoptotic cell death by treatment of hNPC cultures with H2O2 (500 μM) overnight and found that MIRB is not released into the medium from labeled hNPCs; it still resides within the cell as the cell undergoes apoptosis (data not shown). In vivo, we found that some transplanted MIRB-hNPCs underwent necrosis and inflammatory death (Figs. 6, 7). MIRB was present in necrotic hNPCs (Fig. 6G–I). In addition, following transplantation of MIRB-hNPCs, we did not observe an uptake of MIRB alone into neighboring astrocytes or Iba-1+ macrophages/microglia. Microglia were found that were unlabeled with MIRB; microglia were also found that were MIRB-labeled along with SC121+ immunoreactivity (Fig. 7). This finding is similar to studies of microglial phagocytosis of viable neurons in vivo and in vitro described previously (8,32). Our current studies are designed to further investigate this process of hNPC phagocytosis by microglia following TBI. In addition, our study again found that the MRI signal (Fig. 2) is not an accurate measurement of viable hNPC location.
How can this technique be scaled up for clinical use? We are currently investigating increasing the magnetic moment of transplanted MIRB-hNPCs by increasing the iron loading/cell, increasing the strength of the magnetic field (and field gradient) applied by changing the type of magnet employed, such as using a Halbach array and delivering stem cells into deeper regions of the rat brain, on the injured side (37,39,41). An increase in the magnetic intensity does not always lead to an increase in the functional benefit of stem cell therapy (44). Several concerns about our method need be addressed: 1) What happens to the transplanted stem cells when the magnet is no longer present? The minimum amount of time the magnet has to be in place for stem cell attraction in TBI has not been determined, but we found that at 1 day after transplantation, MIRB-hNPCs were retained at the site of the injury. In an ischemia model, Li et al. used endothelial progenitor cells loaded with USPIO and similar methodology and magnetic field strength (23). Application of the magnetic field for only 2 h resulted in a threefold increase in stem cell accumulation in the underlying infarction along with significant anatomical and behavioral improvement. 2) Is there increased intravascular clumping of hNPCs after intracarotid injection in the presence of an elevated magnetic field? Although potential clumping of hNPCs after intracarotid injection is of concern, our data (RECA-1 immunohistochemistry–Perls' Prussian blue staining) did not show intravascular clumping of transplanted cells. However, in another study using magnetic retention of mesenchymal stem cells for cell therapy in a rat model of ischemia/reperfusion, too high of a magnetic intensity resulted in microembolization and undermined the functional benefits of cell transplantation (44).
Study Limitations
Although we described a new and promising method for stem cell delivery following TBI, the present study has a number of limitations. 1) The retention of “magnetic” stem cells at different depths below the cortical surface must be optimized for magnetic intensity, and the optimized magnetic intensity obtained for the small animal studies must be extrapolated to larger animals. 2) The minimum length of time the magnetic hat must be in place for successful stem cell retention must be determined. 3) In TBI during the time period chosen for stem cell transplantation, the BBB is permeable, allowing non-invasive delivery (6). In other CNS disorders, where the BBB is not permeable, opening the BBB transiently must be achieved, for example, by using focused ultrasound for our method to deliver cell-based therapy.
Conclusion
Our studies of cellular therapy for TBI demonstrate that magnetic field intensity does not affect the hNPC viability, proliferation, and differentiation in vitro, when hNPCs are loaded with iron oxide nanoparticles. In vivo, in a rat model of TBI, using an externally applied magnetic field, it is possible to retain MIRB-hNPCs at the site of injury following transplantation. T2WI detects the region of MIRB-hNPC retention; at this site of injury, MIRB-hNPCs can exist as viable cells, necrotic cells, or be phagocytized by Iba-1+ microglia/macrophages. We are currently assessing the long-term changes in hNPC viability and differentiation following magnetic retention and the ability of transplanted cells to enhance functional recovery after TBI.
Footnotes
Acknowledgments
We acknowledge Dr. Ernest Groman, BioPAL, for helpful discussions, and Dr. Stéphan Vigues for graphics. This work was supported by grants from the Maryland Stem Cell Research Fund (P.Y.), the US Department of Veterans Affairs (P.S.F., P.Y.), and the US National Science Foundation (1306208, S.L.J.M.). The authors declare no conflicts of interest.
