Abstract
Stem cell transplantation is a fast-developing technique, which includes stem cell isolation, purification, and storage, and it is in high demand in the industry. In addition, advanced applications of stem cell transplantation, including differentiation, gene delivery, and reprogramming, are presently being studied in clinical trials. In contrast to somatic cells, stem cells are self-renewing and have the ability to differentiate; however, the molecular mechanisms remain unclear. SOX2 (sex-determining region Y [SRY]-box 2) is one of the well-known reprogramming factors, and it has been recognized as an oncogene associated with cancer induction. The exclusion of SOX2 in reprogramming methodologies has been used as an alternative cancer treatment approach. However, the manner by which SOX2 induces oncogenic effects remains unclear, with most studies demonstrating its regulation of the cell cycle and no insight into the maintenance of cellular stemness. For controlling certain critical pathways, including Shh and Wnt pathways, SOX2 is considered irreplaceable and is required for the normal functioning of stem cells, particularly neural stem cells. In this report, we discussed the functions of SOX2 in both stem and cancer cells, as well as how this powerful regulator can be used to control cell fate.
Introduction
Stem cell transplantation is a well-established technique that has been part of the clinical treatment strategies for both malignant (e.g., acute myeloid leukemia and Hodgkin’s lymphoma) and nonmalignant (e.g., thalassemia and sickle cell anemia) diseases and disorders since 1959 1 . In response to the increasing medical need, stem cells are now being isolated and transplanted from a variety of sources, including umbilical cord blood, placenta, amniotic fluid, dental pulp, and adipose tissue 2 –4 . In addition, the discovery of induced pluripotent stem cells (iPSCs) has dramatically broadened the field 5 . These improvements are a reflection of the unmet medical need of regenerative medicine and the limitations of drugs and medical devices 6 . Despite organ transplantation having been successfully performed for the heart, kidney, liver, lung, pancreas, intestine, and thymus, organ sources are in very limited supply 7,8 . Even if a patient receives an organ and the transplantation is considered a success, the patient is required to take antirejection drugs, which have the risk of severe side effects. As such, the development of artificial organs may represent a promising solution; however, effective and efficient techniques for tissue engineering pose numerous challenges 9 .
Stem cells and/or progenitor cells may be a good choice for compensation of the functions of target tissues and for the secretion of appropriate cytokines and growth factors, including those that are considered immunomodulatory 10 –12 . However, the majority of studies have revealed that delivered cells rarely transdifferentiate into their target type and the survival time remains insufficient 13,14 . Nonetheless, while the mechanisms remain unclear, most experimental and clinical results have shown positive results 15 –17 . As such, the technologies underlying stem cell therapy continue to improve regarding cellular function and survival duration. The most important breakthrough has been the reprogramming of somatic cells into pluripotent stem cells with the exogenous expression of certain transcription factors 18 . The potential applications of stem cells, including cellular transplantation and organ development, have been tremendously enhanced after the discovery of iPSCs 19,20 .
As a member of the SOX gene family and SOXB group, which includes SOX1, SOX2, and SOX3, SOX2 (sex-determining region Y [SRY]-box 2) encodes a 34.3 kD protein 21 . As a key regulator of self-renewal, SOX2 protein binds to octamer-binding transcription factor 4 (Oct4) and enhances the expression of Nanog 22,23 . However, Tanaka et al. indicated that SOX2 is unnecessary as an enhancer, suggesting that it modulates the expression of Oct4 24 –26 . The coupling of SOX2 to paired box protein 6 (PAX6) and BRN2 (encoded by POU3F2 in humans) has been shown to regulate eye and neural primordial cell functions 27 . Interestingly, SOX2 and/or the partner protein are not considered sufficient for transcriptional activation, but this complex is 28 . Once the complex is formed, downstream genes such as undifferentiated embryonic cell transcription factor 1 and fibroblast growth factor 4 activate and enhance embrionic stem cell development and survival 29 . Accordingly, the knockdown of Sox2 expression in mouse embryonic stem cells (ESCs) results in the failure of this self-renewal property and leads to differentiation 22 . In contrast to tumorigenesis, the expression level of SOX2 correlates with lower survival and treatment resistance 30 . Therefore, we evaluated the relationship between SOX2 and its functions in both stem and cancer cells and discovered a potential approach for improving stem cells and deteriorating cancer cells.
SOX2 Is Associated With an Enormous Expression Network
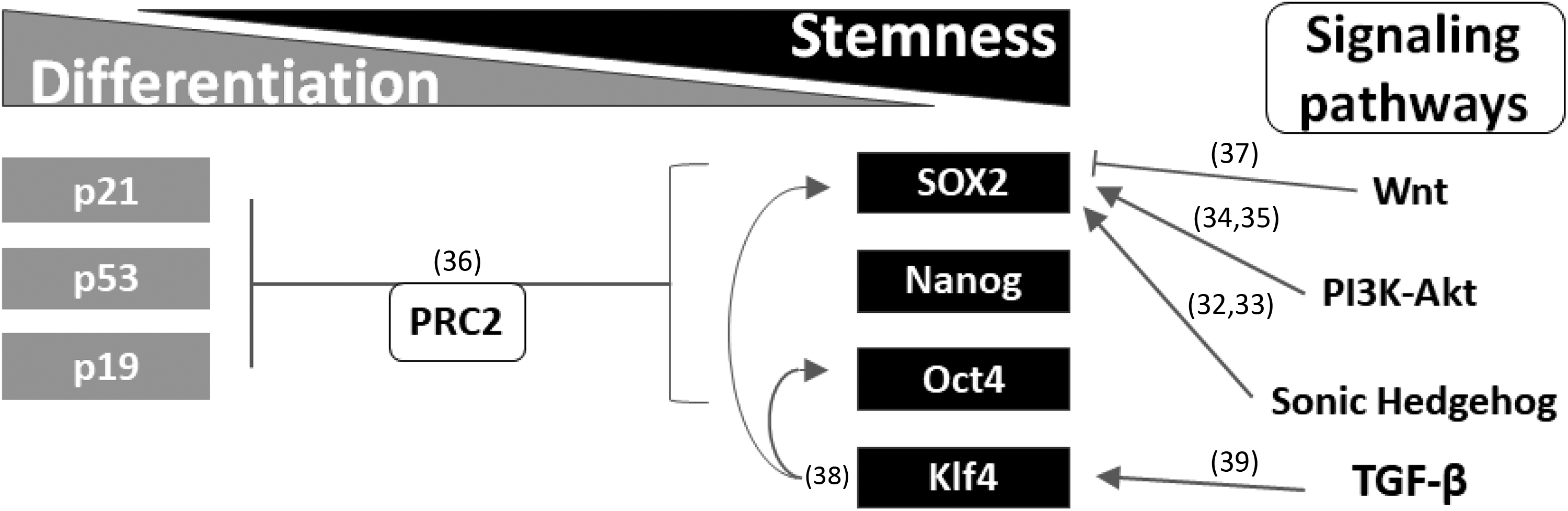
The characteristics of stemness are associated with the target genes of SOX2. In addition, stem cells possess regulatory mechanisms to maintain the appropriate expression of SOX2. For mouse ESCs, the exogenous elevated expression of Sox2 leads to differentiation of ESCs into a wide range of cell types, including neuroectoderm, mesoderm, and trophectoderm (TE) 31 . Moreover, feedback regulation involved in the Akt pathway reactivates endogenous Sox2 expression and serves to retain cellular stemness (Fig. 1) 40 . However, in comparison with iPSCs, the expression of SOX2 is artificial and lacks interactive control. Nevertheless, to reprogram cells into iPSCs, four genes, namely, Oct4, Klf-4, SOX2, and c-Myc (abbreviated to OKSM), are exogenously activated and these genes need a specific ratio to work adequately. Since the OKSM is necessary for pluripotency, other accessory factors such as Nanog and Sal-like protein 4 can only increase the efficiency of reprogramming and cannot replace SOX2 or OCT4 41,42 . For example, a ratio increase of Klf4 is recommended in one of the commercial cellular reprogramming kits. Moreover, the expression of SOX2 is activated by the VP16 transactivator and further improves reprogramming efficiency 43 . These findings indicate that the OKSM acts as a driving force in the fertilization stage and should be tightly restricted or the cells may get out of control. Thus, the upstream and downstream regions of the SOX2 coding sequence contain large untranslated regions (UTRs) or the so-called gene deserts to prevent mutations and false binding 44 . Certain enhancers, including miRNA, long-noncoding RNA, and posttranslational modification have been shown to reversely regulate both transcriptional and posttranslational activity. At least 19 miRNAs, including miR-200b and miR-145, can influence the expression of SOX2 45 . Indeed, miR-200 facilitates the reprogramming of fibroblasts into iPSCs in the presence of OKSM 46 . Moreover, miR-145 targets the 3′-UTR of SOX2 and inhibits self-renewal in human ESCs and iPSCs 47 . In other words, simply elevating the expression of SOX2 serves to attract negative regulation molecules such as the cyclin-dependent kinase inhibitor 1A (p21Cip1) 48 . Conversely, the zygote has a massive demethylation pattern compared with reprogrammed cells 49 . Although existing techniques, including single-cell RNA-Seq, methylation array, and bioinformatics, have confirmed that the methylation state changes in zygotes and somatic cells, the secrets of dedifferentiation remain a mystery 50 .
SOX2 Utilization in Stem Cell Therapy
Except for the stemness regulation of SOX2, the existence for cell differentiation and development is necessary. Notably, in the development of ESCs, when the blastocyst is formed and then divides into the TE and inner cell mass, the expression of SOX2 is decreased. However, the TE will not form if SOX2 is impaired or knocked down by siRNA 51 . This change is due to the complex formed with Oct4 and Nanog. For example, Oct4 and Nanog bind to SOX2 and regulate its functions of self-renewal and differentiation inhibition 52 . In adult humans, the olfactory nerve proliferates and is replaced every 3 to 4 weeks. The SOX2/PAX6-expressed epithelium plays an important role in maintaining the multipotency of the olfactory nerve 53 . These findings suggest further applications in the transplantation from iPSC-differentiated neural stem cells (NSCs). In particular, the in vitro-transcribed mRNA of SOX2 has been shown to induce NSC morphology in human dermal fibroblasts 54 . In addition, another study revealed that exogenous Sox2 expression in rat bone marrow–derived stem cells (BMSCs) benefits the cell transplantation treatment in a rat traumatic brain injury (TBI) model 55 . Especially, BMSCs retain their self-renewal property via the expression of Sirtuin1 (SIRT1) 56 . SIRT1 is a lysine deacetylase that contributes in maintaining SOX2 content by avoiding the acetylation and ubiquitination of SOX2 57 . Moreover, proliferation and differentiation potential is conferred by the forced SOX2 expression of BMSC 58 . Using MRI tracking, Jiang et al. found that NSCs migrate into the injury site of rats with TBI 59 . Therefore, the existence of SOX2 is essential for the maintenance of self-renewal and multipotency. These studies suggested that Sox2-positive cells may play a role in neuron regeneration, enhancing neural functions after brain injury 60 .
Direct Evidence of SOX2 Initiating Tumorigenesis
SOX2 is generally considered an oncogene; however, its role in tumorigenesis remains controversial 61,62 . As part of the same lineage of breast cancer cells, the SOX2-positive population shows a greater colony-forming ability and would be abolished by SOX2 knockdown 63,64 . SOX2 is amplified in patients with cancer, and it contributes to the same stemness property observed in stem cells of patients with lung, brain, breast, and colon tumors 65 .
The clinical implications of SOX2 and cancers vary depending on the type of cancer, influencing patient survival and prognosis 66 . These molecules and pathways include VEGF, MAPK, Notch-Shh, BMP, Jak-STAT, and others, depending on the types of tumors 67 –69 . In brief, SOX2 regulates downstream genes and microRNAs by direct DNA binding, resulting in the alteration of thousands of genes and hundreds of microRNAs 35 . Moreover, a glioma cell subset with high levels of SOX2 has been shown to be resistant to platelet-derived growth factor (PDGF)- and insulin-like growth factor 1 (IGF-1)-receptor inhibitors 70 . Conversely, SOX2 may play a role in the maintenance of PDGF and IGF-1 pathways in cancer cells as well as in stem cells and may produce dysplasia or tumor cell initiation. However, in patients with gastric cancer, the overall survival rate is lower with SOX2 methylation than with unmethylated SOX2. Moreover, the exogenous expression of SOX2 results in cell cycle arrest through cyclin-dependent kinase inhibitor 1B (p27Kip1) and Rb phosphorylation 71 . Indeed, Sox2 expression in the mouse respiratory epithelium does not cause pulmonary tumors but induces the cellular proliferation of respiratory epithelial cells 72 . Direct induced-NSCs can also be obtained by SOX2 expression in human and mouse fibroblasts without tumorigenesis 73 .
Future Prospects of SOX2 Utilization
Due to the close relationship between SOX2 and cancer, studies that have investigated SOX2-dependent gene manipulation are limited. However, cell differentiation and proliferation has been achieved, including SOX2-expressing dental pulp stem cells, which lead to the differentiation of odontoblasts 74 . SOX2 also cooperates with various cofactors, including Oct4 for stemness, BRN2 for neural differentiation, and PAX3 for melanocyte maturation. These studies suggested that SOX2 (and possibly other members in the family) is one of the masters regulating cell fate and is associated with different kinds of cofactors 75 .
An urgent question we would ask is how to take advantage of SOX2 without eliciting detrimental effects? For the nerve system, SOX2 may prove to be a useful regulator for the maintenance of progenitor characteristics, allowing the cells to retain their ability to self-renew and differentiate into neurons, astrocytes, and oligodendrocytes 76,77 . Moreover, Hagey and Muhr found that decreased expression of Sox2 resulted in the differentiation of radial glia cells into their more developed progeny, intermediate progenitor cells 78 . However, mouse NSCs and progenitors have reduced differentiation ability and SOX2 expression with the loss of the E2f3a transcription factor, indicating that the expression of SOX2 is essential for neurogenesis 79 . These studies indicated that SOX2 is required for stemness but is unfavorable for differentiation. In other words, the expression of SOX2 should be controlled or there is a risk of the cells going corrupt.
Conversely, the SOX2-induced stemness ability is devastating and problematic when it appears in tumor cells. Since the existence of SOX2 is associated with the cell membrane, specific antibodies are not effective for disrupting its function. To control the stemness and metastatic properties, Tuhin et al. found that the downregulation effect of actinomycin D induced the cell death of breast cancer stem cells 80 . Moreover, SOX2 knockdown or small molecules reduced SOX2 expression and inhibited the stemness and metastatic properties 61,81 . A significant factor in patients with cancer is the selection of SOX2+ cells after anticancer therapies, including radiotherapy and chemotherapy, and SOX2-induced drug-resistant genes have been characterized in numerous studies 82,83 . These SOX2+ cells then form a new tumor bulk, with the most well-studied type being the quiescent Sonic Hedgehog subgroup medulloblastoma, which is activated into medulloblastoma-propagating cells after antimitotic drug treatment 84 . Moreover, the inhibition of the SOX2-driven transcriptional network arrests GBM growth by treatment with mithramycin, which is an antineoplastic antibiotic 85 . Indeed, SOX2 antigen and antibody were found in small-cell lung cancer (SCLC) cell lines and sera in SCLC patients. However, neither the antigen nor the antibody of SOX2 or other SOX group B genes exist in normal sera, suggesting that SOX2 might be a potential tumor target or marker 86 . However, these effects should be precisely targeted toward tumor cells to take advantage of the attributes of SOX2.
Summary
With the use of iPSC-derived retinal pigment epithelial cells transplanted in clinical trials, a large research and development effort has been undertaken to not only evaluate the effectiveness and safety but also improve the associated techniques 87,88 . In this report, we discussed the stemness-prone properties and some possible risks of SOX2 in Table 1. In addition, the safety issue in iPSC-derived cell therapy is the most important topic; therefore, Larsson et al. established a molecular beacon to compliment SOX2 mRNA, displaying the fluorescent signal when SOX2 is expressed 89 . Although the interaction networks remain incomplete, reactions with different expression levels of SOX2 are more apparent than ever. In summary, the potential applications of SOX2 are extremely promising but precise targeting and expression in the right place and at the right dosage are crucial.
A Summary Table of SOX2 Potential in Medical Use and Their Main Concerns for Cancer Therapy and Stem Cell Transplantation.

Footnotes
Author Contributions
Writing—original draft preparation, HM Chuang; writing—review, MS Huang and YS Chen; conceptualization, HJ Harn.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Buddhist Tzu Chi Bioinnovation Center, Tzu Chi Foundation, Hualien, Taiwan (project title: Development of a small molecule regulates transcription activity of SOX2 in type I collagen synthesis in fibroblasts for treating pulmonary fibrosis, MF00A130SS01) and Ministry of Science and Technology, Taiwan (MOST 106-2320-B-303-001-MY3 and MOST 106-2320-B-303-002-MY3).
