Abstract
The technique of central nervous system cell implantation can affect the outcome of preclinical or clinical studies. Our goal was to evaluate the impact of various injection parameters that may be of consequence during the delivery of solute-suspended cells. These parameters included (1) the type and concentration of cells used for implantation, (2) the rate at which cells are injected (flow rate), (3) the acceleration of the delivery device, (4) the period of time between cell loading and injection into the CNS (delay), and (5) the length and gauge of the needle used to deliver the cells. Neural progenitor cells (NPCs) and bone marrow stromal cells (BMSCs) were injected an automated device. These parameters were assessed in relation to their effect on the volume of cells injected and cell viability. Longer and thinner cannulae and higher cell concentrations were detrimental for cell delivery. Devices and techniques that optimize these parameters should be of benefit.
Introduction
The transplantation of various cellular therapies into the central nervous system (CNS) is a new and innovative approach to attempt to ameliorate neurologic problems associated with such disorders as Parkinson's disease, Huntington's disease, and stroke (1,4–7,11,12,16,18–20,23,26). There are many factors that need to be considered when designing, conducting, and evaluating these preclinical or clinical trials. Among others, these include the type and concentration of cells used and the method of delivery. Some cells are implanted as grafted solid tissue (1,5–7,16,18–20,23) while others are injected as solute suspensions (4,11,12,26). Although many past transplantation studies have focused on the outcome itself, few have studied the variables that might affect these outcomes and that might be optimized before transplantation.
The goal of the present study was to evaluate the impact of various injection parameters that may be of consequence during the delivery of cells in solute suspension. These parameters include (1) the type and concentration of cells used for implantation, (2) the rate at which cells are injected (flow rate), (3) the acceleration of the delivery device, (4) the period of time between cell loading and injection (delay), and (5) the length and gauge of the needle used to deliver the cells. These parameters were assessed in relation to their effect on the volume of cells injected and cell viability.
The prototype of an automated injection device was used for all studies. Previous studies have reported the effectiveness of this device and its ability to consistently deliver a known quantity of viable cells (9). Results of the present study could provide important information regarding the optimum methods used to deliver solute suspensions of cells for CNS transplantation.
Materials and Methods
Cell Isolation
Two cell lines were used in our studies, a neural progenitor cell (NPC) line and a bone marrow stromal cell (BMSC) line. These two cell types have been used numerous times experimentally for CNS cell transplantation (2,3,13–15,17,22,27–32) and have unique, disparate characteristics that enhanced our ability to detect variations in cell delivery. BMSCs grow as a monolayer and generally do not adhere to one another. In contrast, NPCs grow as suspended clusters and tend to clump rapidly when placed in suspension.
All experiments involving the use of cells obtained from animals were approved by the Institutional Animal Care and Use Committee of the University of Pittsburgh. Both lines were isolated from adult, C57BL/6 female mice using methods previously described (8,9). Assessments for viability can also be found in Gobbel et al. (8,9).
Cell Delivery
A computer-controlled, syringe pump prototype (MEDRAD, Inc., Indianola, PA) was used for all cell injections. The device was designed to hold microliter syringes for the automated delivery of accurate volumes at standard flow rates. For this experiment, the device was set to deliver at a constant flow rate of 1 μl or 10 μl/s following an initial acceleration rate (starting from 0 μl/s flow rate) of 42 or 208 μl/s2. NPCs and BMSCs were prepared at a concentration of either 4,000 or 20,000 cells/μl in Hank's balanced salt solution (HBSS). Cells were either delivered immediately upon loading or were delivered after a 5-min delay.
A 250-μl Hamilton syringe was fitted with either a 20-gauge 3.8-cm, 20-gauge 15.2-cm, 27-gauge 3.8-cm, or a 27-gauge 15.2-cm luer-lock, stainless steel needle. To load the syringe used for a particular iteration, 350 μl of suspended cells were aliquoted into a single 1.5ml microcentrifuge tube. The remaining cells were stored for control measurements of cell number and viability at the time of each injection.
To assure that any variance in the number of cells delivered with each technique was due to variations in injection and not loading, the syringe pump device was used to load the syringe in all cases. A new syringe was used for each iteration and flushed several times with HBSS to expel all air from the device before loading. The syringe was then attached to the pump, and its tip was immersed into a 1.5-ml microcentrifuge tube loaded with 350 μl of cell suspension. The suspension was agitated just prior to loading to assure even distribution of the cells, and 110 μl was withdrawn by the device at a rate of 1 μl/s with an initial acceleration of 42 μl/s2. A needle was then attached to the syringe tip, and approximately 30 μl of cell suspension was injected by the device at a rate of 1 μl/s in order to prime the needle with cell suspension before actual injection. A total of eight iterations of three serial 10-μl injections were used to evaluate the delivery of NPCs and BMSCs by the syringe pump. The injected solutions were collected into previously weighed microcentrifuge tubes. Injection volumes were determined by measuring the weight of the injected solution with a scale calibrated to 0.1 mg (1 mg ~ 1 μl of water).
Normalized Plate Counts
A total of 40 μl of HBSS was added to the injected cell suspension, and 20 μl of this was mixed with 80 μl of either normal BMSC media or NPC media with 3% FCS (fetal calf serum). This solution was then plated into one well of an uncoated (BMSCs) or poly-D-lysine-coated (NPCs) 96-well plate. After incubating overnight at 37°C with 5% CO2, an equal volume of HBSS and 10% FCS containing 8 μM Calcein Red-Orange was added to each well, and the cells were incubated for 30 min at 37°C in a CO2 incubator to allow uptake of the dye. An equal volume of 10% buffered formalin was then added to fix the cells, and the number of cells was counted under epifluorescence at points randomly chosen from each of seven locations evenly distributed around the well. The number of cells per plate was 265 ± 25 (mean ± SD), which was the average over all conditions. When the total cell numbers within the seven locations was less than 20, the counting was repeated and summed over 14 locations to obtain a better estimate of the cell number. All measurements of cell number were converted into cell concentrations/μl within the original, undiluted volumes based on the measurements of delivered cell volume, the dilutions used, and the size of the microscopic fields used for counting. At the time of experimental cell plating, a set of control samples was pipetted, diluted, and plated to adjust for any potential loss in viability during the course of an experiment. Experimental counts of cell number were normalized to the counts within control samples. For NPC cells, at concentration of 4,000 and 20,000, the mean cells per plate was 206 ± 49 and 284 ± 81, respectively. For BMSCs, it was 278 ± 108 and 353 ± 102, respectively. The formula for the normalized plate counts was the calculated original concentration of experimental cells/original concentration of control cells.
Statistical Methods
Due to the large number of variables that were to be investigated, a fractional factorial design was employed. Thus, some, but not all, combinations of levels for each of the variables were examined. Cell survival was assessed by measuring both the injection volume of the cells and the plate count. The injection volume and plate count were not normally distributed; thus, a nonparametric Kruskal-Wallis test was employed to assess the difference between parameters. Multivariate analyses that included all the factors and their two-way interactions were carried out by general linear models and a log-rank test. All the comparisons were conducted using SAS 9.1.3 and a value of p < 0.05 was considered to be statistically significant.
Results
Injection Volume
The injection volumes were similar under different cell concentrations, flow rates, accelerations, and delays in either cell type (Table 1). The volumes were slightly higher when there was a high cell concentration, low flow rate, low acceleration, and high delay. Significant differences were observed when using different needle gauge and needle length in the BMSC cell type. The injection volume was significantly less using a 27-gauge in the BMSC group when compared to a 20-gauge needle (8.5 ± 2.7, median = 9.7 vs. 9.7 ± 1.5, median = 9.8; p = 0.031). In addition, the volume was significantly lower using the 15.2-cm needle (8.3 ± 2.9, median = 9.6 vs. 9.8 ± 0.4, median = 9.8; p = 0.004). A multivariate analysis including all factors and both cell types indicated there was no difference in injection volume between BMSC and NPC cell types. However, significant interactions were observed in concentration versus needle length, acceleration versus needle length, needle gauge versus needle length, flow rate versus needle gauge, and flow rate versus acceleration. The injection volumes did not alter much using different needle lengths when there was a lower acceleration, smaller needle gauge, or higher concentration. But the volumes decreased sharply when a longer needle was used under a higher acceleration, larger needle gauge, or higher concentration.
Injection Volumes (Microliters)

Normalized Plate Counts
Cell concentration had a significant effect on the normalized plate counts of BMSCs, where the 4,000 cells/μl group had higher plate count than the 20,000 cells/μl group (1.7 ± 0.8, median = 1.4 vs. 0.8 ± 0.5, median = 0.6; p < 0.0001). Plate counts differed for NPCs when using different flow rates (1.1 ± 0.5, median = 1.1 vs. 0.8 ± 0.4, median = 0.7; p = 0.021), accelerations (1.1 ± 0.5, median = 1.1 vs. 0.8 ± 0.3, median = 0.7; p = 0.053), and delays (0.8 ± 0.4, median = 0.7 vs. 1.1 ± 0.5, median =1.1; p = 0.046). These values correspond to flow rates of 1 versus 10 μl/s, acceleration rates of 42 versus 208 μl/s2 and delays of 0 versus 5 min. Needle length and needle gauge did not affect the plate count in either cell type (Table 2).
Normalized Plate Counts
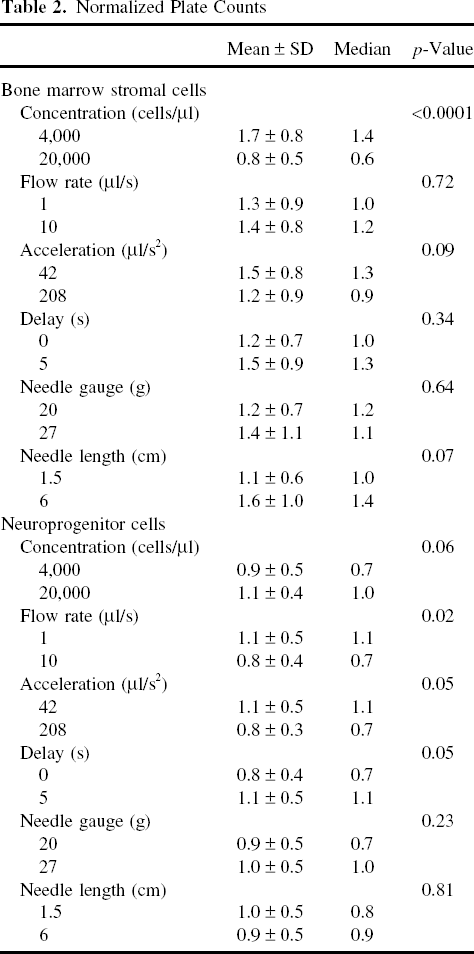
Significant interactions between cell type versus needle length and cell type versus concentration were identified in a multivariate analysis. Needle length and concentration level had no effect on the plate counts in the NPC cell type, although significant effects were found in the BMSC cell type. Interactions were also observed between needle length versus needle gauge and flow rate versus acceleration level. When there was a shorter needle, the plate counts were similar using either needle gauges, but with a longer needle, larger needle gauge yielded much higher plate counts. Further, flow rates had no effect on the plate counts under a high acceleration whereas with a low acceleration, the plate counts increased as the flow rate decreased.
Discussion
The delivery of cells for CNS transplantation can be performed in a number of ways and thus surgical techniques vary greatly (11). The use of an automated device may offer advantages over manual administration in that there could be greater control over rate and speed of delivery and cell implant volume (25). This could lead to greater efficacy of transplantation (9,10,21,24).
Injection Volume
The size and length (correspondingly the volume) of the delivery cannula appeared to affect cell delivery. Although the effect was inconsistent, the longer finer needle, in the case of BMSCs, seemed to result in the delivery of a smaller volume. The longer, finer needles can be associated with blockage of the needle path, which may explain this result. At present, long cannulae are required in human stereotactic procedures to reach from the guide stops on the stereotactic frame down to the brain target. These cannulae can be as long as 19 cm or more. Shorter needles would require direct brain exposure, although this would be more invasive. We estimate that such a length could be reduced to 8–10 cm in that situation. A larger internal cannula volume would mean that more cell suspension would be require to backload the device, with potentially more cell waste.
Although the higher flow rate did seem to result in a lower injection volume in the NPC cells, the result was not significant.
Plate Count
The present study found that injections of some cell types at high concentrations may be detrimental to cell survival. BMSCs had lower plate counts at the higher cell concentration. This finding may indicate that using cells at higher concentrations may limit their immediate viability. This may be due to the fact that cells at too high a concentration within a solution may have access to a more limited supply of nutrients within the suspension medium.
While the concentration of cells did not appear to affect NPC viability, the higher flow rate and acceleration rate caused a decrease in the viability of cells. A 5-min delay in cell delivery proved to increase the number of viable NPC cells. This may be explained by NPC's natural tendency to form clusters that may have had time to settle at the tip of the needle during the 5-min delay before injection.
Although the factors that significantly affected viability were different for BMSCs relative to NPCs, it was not feasible to directly compare the two cell lines with regard to the influence of injection parameters. The current studies focused on the influence of these various parameters and whether the parameters identified would be the same for two distinct cell types. The cell lines have clearly different morphologies and growth characteristics, as described in Materials and Methods (8,9). Each line requires a specific growth medium, indicating that these two cell types are not only morphologically but also physiologically different. Our results suggest that optimal injection parameters may depend on the cell type used for transplantation, and additional investigations will be required to determine the exact impact of cellular morphology and physiology.
Parameter Interactions
The combination of parameters used significantly altered the volume and number of viable cells injected. For example, although needle length did not appreciably affect the volume injected at low acceleration rates, at high acceleration rates, needles of length 15.2 cm substantially reduced the volume injected by −20%. Faster acceleration of fluid and cells within longer needles may be more prone to clogging, perhaps because there is less time for the cells to dislodge from transient adhesions to other cells and the injector.
Because of the large number of factors involved, the present study was not able to evaluate all combinations of parameters for potential interactions. However, the present results demonstrate that such interactions can be significant and could substantially alter the delivery of cells to a tissue. They also suggest that standardization of injection parameters may be a critical aspect of designing and comparing clinical studies of CNS transplantation.
Conclusion
The present study was designed to study the injection volume, number of cells injected, and cell viability of two cell lines over numerous parameters of concern during CNS cell transplantation. Our results demonstrate that the volume of cells delivered can be affected by needle gauge and length while the viability of injected cells is more likely affected by flow rate, acceleration, and delay between loading and injection. The use of an automated injection device allows for control of parameters not currently available when performing hand injection of solute suspensions. In order to optimize graft success, preclinical testing of the planned surgical method with the specific cell line to be used should be a component of clinical trial design.
Footnotes
Acknowledgments
This study was supported by a grant from the Pittsburgh Tissue Engineering Initiative (PTEI) and Medrad, Inc., based upon work supported by NASA under award No. NNJ06HE87G. The authors declare no conflicts of interest.
