Abstract
Current strategies for cartilage cell therapy are mostly based on the use of autologous chondrocytes or mesenchymal stem cells (MSCs). However, these cells have limitations of a small number of cells available and of low chondrogenic ability, respectively. Many studies now suggest that fetal stem cells are more plastic than adult stem cells and can therefore more efficiently differentiate into target tissues. However, the characteristics and the potential of progenitor cells from fetal tissue remain poorly defined. In this study, we examined cells from human fetal cartilage at 12 weeks after gestation in comparison with bone marrow-derived MSCs or cartilage chondrocytes from young donors (8–25 years old). The fetal cartilage-derived progenitor cells (FCPCs) showed higher yields by approximately 24 times than that of chondrocytes from young cartilage. The morphology of the FCPCs was polygonal at passage 0, being similar to that of the young chondrocytes, but it changed later at passage 5, assuming a fibroblastic shape more akin to that of MSCs. As the passages advanced, the FCPCs showed a much greater proliferation ability than the young chondrocytes and MSCs, with the doubling times ranging from 2~4 days until passage 15. The surface marker profile of the FCPCs at passage 2 was quite similar to that of the MSCs, showing high expressions of CD29, CD90, CD105, and Stro-1. When compared to the young chondrocytes, the FCPCs showed much less staining of SA-β-gal, a senescence indicator, at passage 10 and no decrease in SOX9 expression until passage 5. They also showed a much greater chondrogenic potential than the young chondrocytes and the MSCs in a three-dimensional pellet culture in vitro and in polyglycolic acid (PGA) scaffolds in vivo. In addition, they could differentiate into adipogenic and osteogenic lineages as efficiently as MSCs in vitro. These results suggest that FCPCs have stem cell properties to some extent and that they are more active in terms of proliferation and chondrogenic differentiation than young chondrocytes or MSCs.
Keywords
Introduction
The self-repair of articular cartilage is difficult when it becomes injured due to its low regenerative capacity caused by the lack of blood supply, low cellularity, and a limited number of progenitor cells (32). To replenish cells in areas with cartilage defects, surgical techniques such as the abrasion and microfracture arthroplasties have been clinically applied, but both are associated with the formation of fibrocartilage, which is weaker and less durable. Autologous chondrocyte transplantation (ACT) has also been used clinically for cartilage regeneration, showing reasonable results as a cartilage repair strategy (5,42); however, it also has shortcomings in that only a small piece of cartilage tissue can be taken from a patient, and the number of chondrocytes is very limited. In addition, the chondrogenic phenotype and redifferentiation potential can vary depending on the patient and is easily lost, particularly in aged chondrocytes during expansion in a monolayer culture (3,15,21,39).
To overcome the limitations of autologous chondrocytes, many studies have utilized mesenchymal stem cells (MSCs) isolated from various tissues, such as bone marrow, synovial membrane, fat, or cord blood (10,29,30,35). MSCs are regarded as an attractive cell source for cartilage regeneration because they have high self-renewal ability and the multipotent ability to differentiate into various cell types, including chondrocytes in vitro (43). For cartilage regeneration, MSCs are simply injected into the defect area with or without cell carriers, or they are differentiated into chondrocytes to form cartilage tissue in vitro before implantation (7,12,25,33). However, MSCs have also been shown to have limited capacity to differentiate fully into chondrocytes and form intact cartilage tissues both in vitro and in vivo. In particular, cartilage tissues formed using MSCs were mostly subject to hypertrophic changes and lost their chondrogenic properties in vivo (8,11,13,41).
Stem cells or progenitors isolated from fetal tissues such as liver, bone marrow, blood, lung, kidney, pancreas, placenta, brain, and spinal cord are another potent cell source for cell therapy and tissue engineering efforts (40). They have often been shown to have high multipotentiality and proliferation capacity, better sometimes than adult stem cells isolated from aged donors (23,28,36). Moreover, MSCs from fetal liver have been shown to elicit no alloreactive T-cell response, which suggests that fetal stem cells do not display an allogenic immune response (19,20).
Previously, several studies attempted to use fetal cartilage-derived cells in cartilage tissue engineering. Fuchs et al. reported that ovine fetal cartilage cells formed better cartilage tissue than adult chondrocytes by producing more matrix molecules in the pellet culture (17). Quintin et al. showed that human fetal femoral head cells differentiated well into chondrogenic, adipogenic, and osteogenic lineages but with less efficiency than bone marrow MSCs (44). These studies imply that fetal cartilage-derived cells could be a potential cell source for cartilage regeneration, but it is necessary to identify the characteristics and therapeutic utility of fetal cartilage-derived cells further for additional clinical applications. In this study, we examined the characteristics of fetal cartilage-derived progenitor cells (FCPCs) in comparison with bone marrow MSCs and young chondrocytes in terms of their proliferation ability over passages, their plasticity, and their chondrogenic potential in vitro and in vivo.
Materials and Methods
Cell Isolation and Culture
The study was approved by the institutional review board (IRB) of the Ajou University Medical Center (AJIRB-CRO-07-139) and was carried out with the written consent of all donors. Human fetal cartilage tissues (n = 4, M12w-a, M12w-b, F12w-c, M11w) were obtained from patients following elective termination at 12 weeks after gestation, and cells were isolated from the femoral head of the cartilage tissue. Young chondrocytes were isolated from the knee cartilage tissue of surgery patients after trauma at 11~25 years old (n = 3, M11y, M18y, M25y). Aged chondrocytes were isolated from the knee cartilage tissue of three donors undergoing total knee replacement surgery (n = 3, F45y, M54y, F56y). Cartilage tissues were minced into small pieces and treated with 0.1% collagenase type 2 (Worthington Biochemical Corp., Freehold, NJ, USA) in high-glucose Dulbecco's modified Eagle medium (DMEM; HyClone, Logan, UT, USA) containing 1% fetal bovine serum (FBS; HyClone) at 37°C under 5% CO2. After 12 h, isolated cells were cultured in DMEM supplemented with 10% FBS, 100 U/ml penicillin G, and 100 μg/ml streptomycin (Pen-Strep; HyClone) at a density of 8 × 103 cells/cm2. MSCs were isolated from the bone marrow of fractured femurs of surgery patients who were 8~25 years old (n = 3, F8y, M11y, M25y). Briefly, mononuclear cells (MNCs) were collected by Ficoll (Ficoll-Paque PLUS; Amersham Biosciences, Piscataway, NJ, USA) gradient centrifugation, suspended in α-modified Eagle's medium (α-MEM; HyClone) supplemented with 10% FBS, and plated at a density of 8 × 104 cells/cm2. After 6 days, nonadherent cells were removed, and the adherent MSCs were replenished with fresh medium. Cells were passaged at 80% confluence, where the plating density was approximately 8 × 103 cells/cm2.
Flow Cytometry Analysis
Cells at passages 2~3 were analyzed for the expression of stem cells markers on the cell surface. Cells in the suspension were incubated with anti-CD34-FITC, anti-CD29-PE, anti-CD90-FITC (BD Biosciences, San Jose, CA, USA), anti-CD105-FITC (Ancell, Bayport, MN, USA), and anti-Stro-1 (R&D Systems, Minneapolis, MN, USA) antibodies for 40 min at 4°C. For the anti-Stro-1 antibody, cells were washed with phosphate-buffered saline (PBS; Welgene, Daegu, Korea) and then incubated with goat anti-mouse IgM-FITC (R&D Systems) for 30 min at 4°C. The dilution factors of each antibody were determined according to the manufacturers' instructions. Stained cells were analyzed by flow cytometry (Becton Dickinson FACSvantage).
Analysis of mRNA Levels
The total RNA was extracted from cells using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). One microgram of RNA was used to synthesize cDNA by reverse transcription. PCR was performed using 0.5 mg of synthesized cDNA and specific primers for collagen II, SOX9, aggrecan, collagen I, and GAPDH. The sequences of the primers and the reaction conditions are listed in Table 1. Quantitative real-time PCR was performed with a CFX Connect™ PCR machine (Bio-Rad, Hercules, CA, USA) using SYBR Premix Ex Taq™ (Takara Bio Inc., Otsu, Shiga, Japan). All reactions were duplicated, and each amplified signal of the (sex-determining region Y)-box 9 (SOX9) and collagen II α 1 (COL2A1) were normalized to that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
The Primers Used for Reverse Transcription-Polymerase Chain Reaction (RT-PCR)

Cellular Senescence Assay
Senescent cells were determined by senescence-associated β-galactosidase (SA-β-gal) staining using a senescence cell histochemical staining kit (Sigma-Aldrich, St. Louis, MO, USA). Briefly, cells were first fixed for 6 min at room temperature and then incubated with a staining mixture for 8 h at 37°C without CO2. Senescent cells were observed in blue color under a microscope (TE-2000; Nikon, Tokyo, Japan).
Multilineage Differentiation
For osteogenic and adipogenic differentiation, cells were plated in six-well plates (TPP, Trasadingen, Switzerland) at densities of 2 × 103 cells/cm2 and 2 × 104 cells/cm2, respectively. After 24 h, the cells were incubated in a differentiation medium for each lineage. The osteogenic medium consisted of α-MEM supplemented with 10% FBS, 10 mM β-glycerophosphate (Sigma-Aldrich), 100 nM dexamethasone (Sigma-Aldrich), and 50 μg/ml ascorbate-2 phosphate (Sigma-Aldrich). The adipogenic medium consisted of α-MEM supplemented with 10% FBS, 1 μM dexamethasone, 10 μg/ml insulin (Sigma-Aldrich), 0.5 mM isobutyl-methylxanthine (IBMX; Sigma-Aldrich), and 0.1 mM indomethacin (Sigma-Aldrich). The control cells were incubated in α-MEM supplemented with 10% FBS for 3 weeks. After 3 weeks of differentiation, cells were stained with Alizarin red S (Sigma-Aldrich) and Oil red O (Sigma-Aldrich) to observe the degree of mineralization and the lipid droplets, respectively. For quantitative analysis, the adsorbed dyes were eluted with 3 ml of 10% acetic acid (Duksan Chemical, Seoul, Korea) and 1.5 ml of 10% ammonium hydroxide (Duksan Chemical) for Alizarin red S and with 1 ml of isopropyl alcohol (Duksan Chemical) for Oil red O. The absorbance was measured at 405 nm and 500 nm, respectively. To present the data, values from the differentiated samples were normalized by the control values of undifferentiated cells.
For chondrogenic differentiation, 3 × 105 cells were centrifuged at 500 × g for 5 min, and the cell pellet was cultured in a chondrogenic medium. The chondrogenic medium consisted of DMEM supplemented with 100 nM dexamethasone, 50 μg/ml ascorbate-2 phosphate, ITS supplement (Sigma-Aldrich), 40 μg/ml proline (Sigma-Aldrich), 1.25 mg/ml bovine serum albumin (BSA; Sigma-Aldrich), 100 μg/ml sodium pyruvate (Sigma-Aldrich), and 10 ng/ml TGF-β3 (R&D Systems). After 3 weeks of induction, the samples were fixed with 4% formaldehyde (Duksan Chemical) and embedded in paraffin wax. Sections with a thickness of 4 μm were prepared and stained with Safranin O (Sigma-Aldrich) to observe the sulfated glycosaminoglycans (sGAG).
Cartilage Tissue Formation In Vivo
To examine the potential for cartilage tissue formation, cells were seeded in a scaffold and implanted subcutaneously into the backs of BALB/c nude mice (male, 6 weeks of age; Orient Bio, Seongnam, Korea). Briefly, 5 × 106 cells were seeded in a PGA scaffold (Biofelt®; Concordia Medical, Warwick, RI, USA) with a diameter of 5 mm and a height of 2 mm. The cell–PGA constructs were cultured in the chondrogenic media mentioned above for 1 week before implantation into the nude mice. To minimize individual variations in the animals, a set of constructs from all of the different groups was implanted in a nude mouse (n ≥ 3 per group). The tissues formed were sampled at 1, 2, and 4 weeks after implantation. The experimental protocol for animal use was reviewed and approved by the Institutional Animal Experiment Committee of the Gyeonggi Institute of Science and Technology Promotion.
Histology and Immunohistochemistry
The samples were fixed with 4% formaldehyde (Duksan Chemical) and embedded in paraffin wax (Merck, Darmstadt, Germany). Sections 4 μm thick were stained with Safranin O and von Kossa staining (Sigma-Aldrich) to identify the degree of sGAG and calcium deposition, respectively. For an immunohistochemical analysis of collagen II, sections were treated with 3% hydrogen peroxide (Duksan Chemical) in methanol for 10 min and reacted with a pepsin solution (Golden Bridge International, Inc., Mukilteo, WA, USA) for 10 min. After blocking the sections with 1% BSA in PBS, they were incubated with an anti-collagen II antibody (1:100; Calbiochem, Darmstadt, Germany) or an anti-collagen X antibody (1:100; Abcam, Cambridge, MA, USA) for 1.5 h at room temperature. The sections were then incubated with a biotinylated secondary antibody against mouse IgG (SPlink HRP Detection Kit; Golden Bridge International) for 30 min and with a peroxidase-conjugated streptavidin solution (SPlink HRP Detection Kit; Golden Bridge International) for 30 min. Finally, the sections were reacted with a 3,3′-diaminobenzidine (DAB) solution (Golden Bridge International) and counterstained with Mayer's hematoxylin (YD Diagnostics, Seoul, Korea) before mounting.
Statistical Analysis
The quantitative data from the multilineage differentiation experiments were analyzed by a one-way analysis of variance (ANOVA) with Tukey's post hoc test using SPSS 12.0.1 (IBM, Armonk, NY, USA). Values of p < 0.05 were regarded as statistically significant.
Results
Isolation and Characterization of FCPCs
Human fetal cartilage tissue from the femoral condyle at 12 weeks of gestation was histologically observed after Safranin O staining to detect sGAG in comparison with young cartilage tissue. As shown in Figure 1A, the fetal cartilage tissue showed an even distribution of a large number of cells at a high density. No signs of secondary ossification and hypertrophic chondrocytes were observed. An immunohistochemical analysis showed that collagen II was highly expressed all around the fetal cartilage tissue, while collagen I was not expressed at all (Fig. 1B). When cells were isolated, the number of FCPCs per 1 g of tissue was higher by approximately 24 times compared to the number of chondrocytes isolated from the young cartilage tissue (Fig. 1C). FCPCs were more similar in terms of their morphology to chondrocytes, with both showing a triangular shape, than they were to human MSCs isolated from the bone marrow (Fig. 1D). These results suggest that fetal cartilage tissue could be a good cell source, providing a large number of chondrogenic cells from a limited amount of tissue.

Isolation of FCPCs from human fetal cartilage tissue. (A) Human fetal cartilage tissue 12 weeks after gestation (a, b) and young cartilage tissue (c, d) were stained with Safranin O/fast green staining for sulfated glycosaminoglycans and were observed under a microscope. (B) Human fetal cartilage tissue was immunostained for collagen II (upper panel) and collagen I (lower panel). (C) The yields of cells isolated from 1 g of tissue were compared between FCPCs (n = 4) and young chondrocytes (n = 3) isolated from each of the tissues. (D) The morphologies of the FCPCs and chondrocytes at 24 h and MSCs at 6 days after initial plating were observed. Y-Chon, young chondrocytes.
Phenotype Change of FCPCs Along with Passages
A decrease in cell proliferation and activity by senescence or dedifferentiation is a major challenge with regard to the therapeutic use of chondrocytes and MSCs. Therefore, we examined the proliferation ability and phenotypes of FCPCs as passages progressed. The doubling time of the FCPCs was approximately 2.2 ± 0.6 at passage 1 and did not increase significantly at least up to passage 15 (Fig. 2A). The doubling time of the human chondrocytes and MSCs increased remarkably along with passages, though these values could not be practically measured at passage 7. FCPCs showed a rapid increase in the total number of cells, reaching approximately 1.3 × 1010 cells at passage 7 and 6.9 × 1012 cells at passage 11 (Fig. 2B, left). In contrast, both the young chondrocytes and MSCs showed only approximately 1.7 × 108 cells and 1.3 × 108 cells at passage 7, respectively. The cumulative cell numbers showed very significant differences when calculated over time, with the result being 2.2 × 1010 cells in the FCPCs, 2.2 × 107 cells in the young chondrocytes, and 4.7 × 107 cells in the MSCs at 40 days (Fig. 2B, right). The proliferation ability of FCPCs was therefore clearly different from that of human young chondrocytes and MSCs, decreasing rapidly with the number of passages, as also reported in previous studies (26,31,38).

Proliferation of FCPCs with passages. (A) The doubling time of FCPCs was compared with those of human chondrocytes and MSCs until passage 15 (n = 3). Cells were subcultured at 80% confluence. The doubling time was calculated using the following formula: DT = (T1 - T0)log2/(log N1 - log N0), where T1 - T0 = the culture period in days, N0 = the plating cell number, and N1 = the harvesting cell number. (B) Accumulated cell numbers were calculated in each group with passages (left) or days (right). Y-Chon, young chondrocytes. The average values are presented with the standard deviation (SD). *p < 0.05, **p < 0.01, ***p < 0.001.
When the expression levels of chondrocyte-enriched genes were examined up to passage 5, SOX9 was highly expressed at all times. A slow decrease was noted in collagen II (COL2A1), but it was still expressed more in FCPCs than in chondrocytes (Fig. 3A). Unexpectedly, the expression of aggrecan remained high both in FCPCs and chondrocytes until passage 5. The expression of collagen I (COL1A2), a marker of chondrocyte dedifferentiation, increased along with passages in both cell types, but this occurred more rapidly in chondrocytes. These results suggest that FCPCs are highly proliferative and maintain chondrogenic phenotypes well as the passages progress. When examined in aged chondrocytes, the expressions of SOX9 and collagen II were again found to decrease rapidly along with passages according to both RT-PCR and quantitative real-time PCR analyses (Figs. 3B, C).

Gene expression analyses of FCPCs with passages. (A) Expressions of chondrocyte-specific genes in FCPCs and chondrocytes were confirmed by RT-PCR. mRNA levels of SOX9, COL2A1 (collagen II, α 1), ACAN (aggrecan), and COL1A2 (collagen I, a 2) were examined in FCPCs and young chondrocytes at passages 1–5. The mRNA level of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was measured as a housekeeping control. The mRNA levels of SOX9 and COL2A1 were examined by RT-PCR (B) or quantitative real-time PCR (C) analyses in FCPCs and in aged chondrocytes obtained from donors at 45–56 years old (FCPCs; n = 3/donor, aged chondrocytes; n = 3/donor, F45y, M54y, F56y). Y-Chon, young chondrocytes. **p < 0.01.
In morphological observation, FCPCs were changed to the fibroblastic shape at passage 5, but did not show flattened morphology of senescent cells observed in human chondrocytes and MSCs at the same passage (Fig. 4A). The fibroblast-like morphology of FCPCs was maintained without significant change until passage 15 (data not shown). Senescent cells were identified by SA-β-gal staining at passage 10 in most human chondrocytes and MSCs, while rarely detected in FCPCs (Fig. 4B).

Morphology of FCPCs with passages. (A) The morphology of FCPCs was examined in comparison with young MSCs and chondrocytes observed at passages 1 and 5. (B) Senescent cells were identified at passage 10 by SA-β-gal staining. Cells with dark stains represent the senescent cells. Y-Chon, young chondrocytes.
Surface Markers Profile of FCPCs
To determine if FCPCs have the properties of progenitor or stem cells, the expression of surface markers rich in MSCs was initially examined in FCPCs in comparison with human MSCs and chondrocytes all at passage 2 stages by flow cytometry. The results showed that all three cell types expressed CD90 and CD105 at relatively high levels and did not express CD34 (Table 2). They also showed that CD29 and Stro-1 were expressed but at varying levels, being most highly expressed in the FCPCs followed by the MSCs and chondrocytes. The expression level of Stro-1 was only 11.0 ± 2.4% in chondrocytes.
Expression of Stem Cell Markers on FCPCs, Chondrocytes, and MSCs by Flow Cytometry

Multilinage Differentiation Ability of FCPCs In Vitro
The multilineage differentiation ability of FCPCs was examined for osteogenesis, adipogenesis, and chondrogenesis in comparison with human MSCs and chondrocytes. Cells at passage 2 were differentiated into each lineage for 3 weeks in vitro. In chondrogenesis, the size of the FCPC pellets was the smallest at day 1, but increased significantly at 3 weeks such that it was comparable to that of the chondrocyte pellets in a gross observation (Fig. 5A). The size of the chondrocyte pellet was the largest at day 1, but did not increase significantly at 3 weeks, while the size of the MSC pellet rather decreased at 3 weeks. Histological images of Safranin O staining also showed strong signals for sGAG in the FCPCs, confirming that they were differentiated well into cartilage-like tissue. The chondrogenic potential of FCPCs was maintained well at passage 5, but significantly decreased at passage 10 (Fig. 5B). In osteogenesis and adipogenesis, FCPCs also showed the strongest signals in both Alizarin red S and Oil red O stains, respectively (Figs. 6A, B). MSCs were next, after the FCPCs, while the chondrocytes showed little potential for osteogenesis and adipogenesis. FCPCs were fully mineralized without individual differences in osteogenesis and showed lipid droplets in more cells than did the MSCs in adipogenesis. The staining results were confirmed statistically in quantitative data presented in the graphs. These results suggest that FCPCs have progenitor or stem cell properties and are more potent in terms of their multilineage differentiation ability compared to young bone marrow MSCs.

Chondrogenic differentiation potential of FCPCs in vitro. (A) FCPCs, MSCs, and chondrocytes all at passage 2 were prepared in a pellet conformation and differentiated into chondrogenic lineages for 3 weeks in a defined medium (n ≥ 3). In the gross images, scales are in 1 mm (top). Samples at 3 weeks were histologically stained with Safranin O (middle and bottom). (B) FCPCs at passages 2, 5, and 10 were subjected to chondrogenic differentiation as above, and Safranin O images were presented. Y-Chon, young chondrocytes.

Osteogenic and adipogenic differentiation potential of FCPCs in vitro. FCPCs, MSCs, and chondrocytes at passage 2 were differentiated into (A) osteogenic and (B) adipogenic lineages for 3 weeks and stained with Alizarin S and Oil red O to observe the degree of calcium deposition and lipid droplets, respectively (n = 3/donor). For a quantitative analysis, the adsorbed dyes were eluted, and the absorbance was measured at 450 nm for Alizarin S and 500 nm for Oil red O stains. Values normalized with the control values are presented as the mean and standard deviation (SD) from at least three independent experiments. ***p < 0.001. Y-Chon, young chondrocytes.
Cartilage Tissue Formation of FCPCs In Vivo
To evaluate chondrogenic potential of FCPCs in vivo, the three types of cells were cultured in PGA scaffolds for 1 week in vitro and were implanted in the backs of nude mice for 1, 2, and 4 weeks. In a gross observation, the construct size was nearly identical just before implantation (at 1 week in vitro), but it changed significantly over time among the different cell constructs (Fig. 7A). It increased gradually in the FCPC constructs, did not change greatly in the chondrocyte constructs and decreased in the MSC constructs. The size of the FCPC constructs at 4 weeks of implantation was twice as large as that before implantation. In particular, the FCPC constructs at 4 weeks were easily removed from the skin of the nude mice and had a very smooth surface when compared with the other constructs. Histological observations for sGAG and collagen II further confirmed that the FCPC constructs formed the highest quality of cartilage tissue in vivo. The FCPC constructs at 4 weeks showed intense accumulation of sGAG and well-developed lacunae formation throughout the construct (Fig. 7B). The MSC constructs did not differentiate well into the chondrocytes and formed a poor quality of cartilage tissue in vivo. The expression of collagen II was also observed to be significant throughout the FCPC constructs, but it occurred only around the peripheral area in the chondrocyte and MSC constructs (Fig. 7C, top). In particular, MSCs showed significant amounts of collagen X expression and calcium deposition according to von Kossa staining around the peripheral region of the constructs at 4 weeks, which represents a typical phenotype of matrix mineralization and hypertrophic changes (Fig. 7C, middle and bottom). This phenomenon was not observed in the FCPC or chondrocyte constructs.
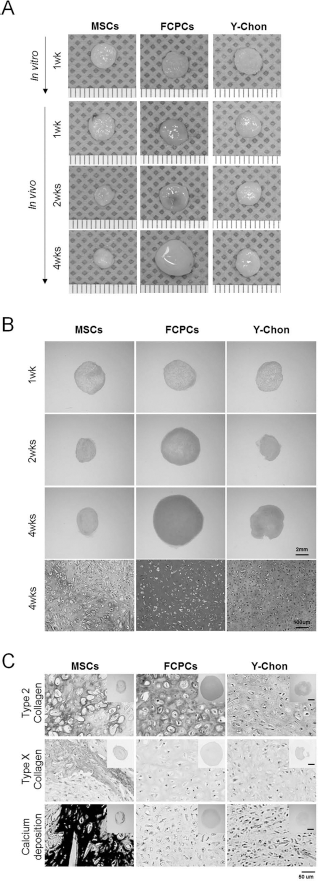
Cartilage tissue formation of FCPCs, chondrocytes, and MSCs in vivo. Cells were seeded in PGA scaffolds and differentiated initially into chondrogenic lineages for 1 week in vitro (n ≥ 3). The constructs were then implanted subcutaneously into the backs of nude mice for 1, 2, and 4 weeks before the analysis. (A) Gross images of samples at 1 week in vitro and 1, 2, and 4 weeks in vivo. Scales are in 1 mm. (B) Histological sections were prepared from the in vivo samples and stained with Safranin O. (C) The samples at 4 weeks were subjected to immune staining for collagen II (top), collagen X and von Kossa staining (bottom). Y-Chon, young chondrocytes.
Discussion
In this study, we proved the potential of human fetal cartilage-derived progenitor cells (FCPCs) as a novel cell source for cartilage regeneration. Our results showed that FCPCs could be obtained at a high yield (6.5 ± 0.95 × 107 cells per 1 g of tissue) and that they proliferate well in vitro for more than 15 passages without significant growth retardation. The results also showed a greater ability of FCPCs for multipotent differentiation into not only chondrogenic but also adipogenic and osteogenic lineages compared to human young chondrocytes and bone marrow MSCs. In particular, they were much better than bone marrow MSCs in cartilage tissue formation yet did not undergo hypertrophic development in vivo. These results suggest that FCPCs can serve as a suitable cell source at least for cartilage regeneration and tissue engineering. Cells from various fetal tissues are thought to be more primitive than young or adult cells; however, they are also considered to have a greater ability to differentiate into specific lineages than adult stem cells such as MSCs (28,36,40). The results of Quintin et al. also showed that fetal cartilage-derived cells at 14–16 weeks after gestation are more chondrogenic and less efficient than bone marrow MSCs in terms of adipogenic and osteogenic differentiation (44). However, the results of this study suggest that FCPCs at 12 weeks after gestation are more chondrogenic than MSCs, but still have better proliferation ability and plasticity for multilineage differentiation under defined conditions in vitro.
Although a small amount of cartilage tissue was obtained from the fetus, a large number of FCPCs were harvested on account of the high cellularity of this type of tissue. The number of cells obtained from the fetal cartilage (6.5 ± 0.95 × 107 cells per 1 g tissue) was much larger than that of cartilage tissue from donors who were 11~25 years old (2.7 ± 0.23 × 106 cells per 1 g tissue) in this study. Several previous studies also reported that mature cartilage had larger chondrocytes and a lower cell-to-matrix ratio than immature cartilage (22,23). We also found that the cellularity of our fetal cartilage tissue at 12 weeks of gestation appeared to be higher by approximately twofold than that at 15 weeks of gestation, as shown in the images of Hillel et al. (23). Considering that ACT is very limited when used to obtain sufficient amounts of healthy tissue, FCPCs could be a good substitute for autologous chondrocytes by providing large amounts of active and chondrogenic cells.
Autologous chondrocytes from aged patients are also limited in therapeutic applications owing to their dedifferentiation properties over passages such as growth retardation, a loss of chondrocytes phenotypes, and their low cartilage tissue formation efficiency (3,14). It is known that the proliferation rate of aged chondrocytes decreases past a donor age of 30~40 years (3). Moreover, they commonly dedifferentiate and lose their chondrogenic phenotype after an average of about five subcultures (6). In contrast, FCPCs in this study maintained their proliferation ability well, even until passage 15. The chondrogenic differentiation ability of FCPCs decreased somewhat as the passages progressed, but this was likely due at least in part to the unfavorable environment of the pellet culture. When tested in a natural three-dimensional scaffold, their chondrogenic ability showed no significant difference at passages 2 and 5 (data not shown). The expression of collagen II also decreased somewhat as the passages progressed, whereas that of SOX9 remained high with no decrease at all in FCPCs until passage 5. SOX9 is a transcription factor, which regulates the expression of chondrocyte-enriched genes such as collagen II; it is involved in cartilage formation of the axial skeletal at the development stage. During the development of endochondral bone, the expression of SOX9 is observed in the phase of mesenchymal cell condensation until chondrocyte differentiation and is downregulated with a phase change into prehypertrophic chondrocytes (45). In this study, FCPCs showed high levels of SOX9 gene expression as the passages progressed, most likely because it was isolated before the chondrocyte maturation phase. We speculate that a high level of SOX9 expression is also related to the finding that FCPCs form high-quality cartilage tissues at least until passage 5 in a pellet culture.
Fetal cartilage is an immature tissue that is not fully differentiated into cartilage, and FCPCs may possess some stem cell properties, such as self-renewal and multilineage differentiation ability. The FCPCs in this study showed multilineage differentiation ability not only into chondrocytes but also into adipogenic and osteogenic lineages. Their ability was greater than that of bone marrow MSCs derived from donors aged 8–25 years according to our results. Forced differentiation of FCPCs into other cell lineages such as neurons and hepatocytes was not as efficient (data not shown), suggesting that the ability of FCPCs is rather limited to mesengenic cell lineages such as MSCs. Many studies have reported that dedifferentiated human articular chondrocytes are capable of plasticity into multilineage cells (4,15,24). Specifically, cloned chondroprogenitors had 2.6 times more telomerase activity than nonclonal dedifferentiated chondrocytes (27), but their multipotency was highly heterogeneous across different clones (4). In our study, young chondrocytes also showed some differentiation ability into adipogenic and osteogenic lineages, which decreased with an increase in the ages of the donors (data not shown). However, their ability was not sufficiently high enough to be regarded as having multipotency.
A surface marker analysis conducted in an effort to examine the stem cell properties of FCPCs showed no significant differences among the three cell types except for CD29 and Stro-1. Of note, the expressions of CD29 and Stro-1, two well-known MSC markers, were most highly expressed in FCPCs, followed by MSCs and chondrocytes. CD29 is integrin β1, which plays a key role in cell–cell or cell–matrix interactions. It was previously suggested to mediate chondrocyte aggregation during the suspension cultivation process (18). Considering the importance of mesenchymal condensation during the early stage of cartilage development, CD29 may also play an important role in the chondrogenic differentiation and cartilage tissue formation of FCPCs. Stro-1 is known as a marker of bone marrow MSCs, and cells highly expressing Stro-1 are thought to be more primitive than nonexpressing cells (1,16,37). The significance of this needs further clarification.
MSCs are a good cell source for cartilage regeneration and tissue engineering, but still have disadvantages in terms of insufficient differentiation and a loss of chondrogenic phenotypes after long-term implantation in vivo. This process, known as hypertrophic development, involves a loss of cartilage matrix molecules, the deposition of calcium, and matrix mineralization. The cause of the hypertrophic development is not yet clear, and it was shown to be delayed to some extent by using some mechanical stimuli (11,13) or scaffold materials (8), but could not be completely overcome. In our previous studies using rabbit bone marrow MSCs, engineered cartilage with more proteoglycan showed less hypertrophic changes, and the Safranin O-negative area matched the hypertrophic area well in histological and immunohistochemical observations (8,9). In this study, FCPCs showed not only high cartilage tissue-forming ability but also no hypertrophic development until 4 weeks in vivo. Therefore, FCPCs have stem cell properties, as mentioned above, but are basically different from MSCs. We think that FCPCs share the advantages of both chondrocytes and MSCs at the same time. Quintin et al. predicted that fetal cartilage contains cells from a secondary ossification center and showed that fetal cartilage cells expressed collagen X, indicating hypertrophic chondrocyte formation in the presence of a chondrogenic induction medium (44). In this study, we also found collagen X was not expressed in our cartilage tissue formed from FCPCs in vivo. Therefore, we speculate that FCPCs would not easily become hypertrophic even after longer culturing times, considering their ability to form high-quality cartilage tissue.
For clinical applications of FCPCs, an important limitation related to immunogenicity should be resolved. Adkisson et al. reported that juvenile chondrocytes lack cell surface markers such as HLA-DR, CD80 and CD86, which are responsible for the induction of immune responses at the cellular level, and that they did not stimulate an immunologic response in vivo (1,2). CD80 and CD86 were expressed in young chondrocytes (age >20 years), but not in juvenile chondrocytes (age < 6 years). In addition, some studies reported that MSCs from various fetal tissues or fetal bone cells have low immune-related properties (19,20,37). Our further work, therefore, will focus on demonstrating the immune-related properties of FCPCs and clonal characterization to obtain more homogenous and primitive FCPCs. Another important issue with FCPCs would be ethical concerns regarding the use of fetal tissues. There have been even cases of intended conception for the donation and therapeutic use of fetal tissues or cells. We are definitely against these types of abuse of fetal tissues and claim that fetal tissues for therapeutic use should be obtained from fetuses that were found to be deceased in utero from natural causes and independent of any decisions with regard to abortion (34). In addition, the medical and technical procedures should be in accordance with the Helsinki declaration. We have well-defined guidelines and regulations concerning the use of fetuses in Korea, which are also managed well by the IRB. Securing a good source of FCPCs following the ethical and legal guidelines should be one of the next tasks for the commercialization of FCPCs.
In conclusion, we investigated the characteristics of human FCPCs in comparison with those of human bone marrow MSCs and young chondrocytes. Human FCPCs showed the characteristics of both MSCs and chondrocytes, having high proliferation and multilineage differentiation abilities yet excellent chondrogenic potential in vitro and in vivo. However, ethical considerations remain and should be discussed in depth before FCPCs can be considered for therapeutic use.
Footnotes
Acknowledgments
This study was supported by grants from the Korea Healthcare Technology R&D Project, the Ministry for Health, Welfare and Family Affairs (A091120), and the Ministry of Science, ICT and Future Planning (2011-0019730), Republic of Korea. The authors declare no conflicts of interest.
