Abstract
CTLA4Ig has therapeutic potential for rheumatoid arthritis patients unresponsive to methotrexate (MTX) or TNF-α blockers. However, recombinant CTLA4Ig proteins are short acting and expensive. Adipose tissue-derived mesenchymal stem cells (ASCs) present an ideal stem cell source for practical regenerative medicine due to their abundant availability and their beneficial properties including immunomodulation, homing activity, paracrine effects, and differentiation ability. Therefore, we aimed to determine whether CTLA4Ig and human ASCs show synergistic effects on immunomodulation and clinical improvement of sustained severe rheumatoid arthritis in a mouse model. hASCs overexpressing CTLA4Ig (CTLA4Ig–hASC) were serially transplanted into mice with collagen-induced arthritis. Arthritic mice were subjected to four treatments based on their arthritis score on day 62 postimmunization: control (C group), hASC (H group), CTLA4Ig–hASC (CT group), and MTX (MTX group). A group of healthy mice was used as a normal control (N). Mice in the N and C groups were infused with 150 μl saline, and 2 × 106 hASCs or CTLA4Ig–hASCs in 150 μl of saline were intravenously administered to those in the H and CT groups, respectively, on days 63, 70, 77, and 84 after CII immunization. About 1 mg/kg of methotrexate was intraperitoneally administered to the MTX group three times a week for 4 weeks. Serial hASC and CTLA4Ig–hASC transplantation modulated various cytokines and chemokines related to the development of rheumatoid arthritis. Both treatments protected against destruction of cartilage, with CTLA4Ig–hASCs being most effective. Serum levels of CII autoantibodies and C-telopeptide of type II collagen were significantly low in the group transplanted with CTLA4Ig–hASCs. In vitro, ASC and CTLA4Ig–hASC treatment significantly decreased T-bet and GATA-3 expression in splenocytes from arthritic mice, and CTLA4Ig–hASC treatment significantly increased the ratio of Treg/Th17 (CD4+CD25+FoxP3+/CD4+CD25+RORγt) cells. Serial hASC and CTLA4Ig–hASC transplantation offers promising treatment for rheumatoid arthritis, and CTLA4Ig–hASCs showed stronger therapeutic effects than nontransduced hASCs.
Keywords
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disease characterized by chronic inflammatory disorder of the synovial joint, destruction of articular cartilage, and bone erosion (20,40). To study the underlying disease mechanism and develop potential treatments, animal models have been used (28). Furthermore, future successful application of stem cells in human medicine depends on the establishment of preclinical animal studies (42).
The autoantigen for RA is unknown, but type II collagen, a major component of joint cartilage, may be a target antigen (45). Collagen-induced arthritis has been established in genetically susceptible mouse strains by immunization with type II collagen in complete Freund's adjuvant containing Mycobacterium spp., followed by subcutaneous booster injection with type II collagen emulsified in incomplete Freund's adjuvant 2 or 3 weeks later (18,46). The collagen-induced arthritis mouse model is widely used for studying disease pathology and developing new therapeutics for human RA (2).
Cytokines including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), IL-6, and IL-17 may mediate the initiation and progression of inflammation and tissue destruction in RA (1,31,36,50). Methotrexate (MTX) and TNF-α blockers are widely used nonbiological (36) and biological (13) disease-modifying antirheumatic drugs (DMARDs), respectively. Other biological agents that have been developed to treat RA include IL-1 receptor antagonists and anti-IL-6 antibodies (26). Cytotoxic T-lymphocyte-associated protein 4 (CTLA4) is a protein receptor that negatively regulates T-cell activation. Fusion proteins comprising the extracellular domain of CTLA4 and the CH2-CH3 domains of immunoglobulin (CTLA4Ig) have been assessed in clinical trials (27,41), which indicated that CTLA4Ig is potentially therapeutic for patients unresponsive to methotrexate. Furthermore, CTLA4Ig is the first approved agent that shows efficacy and safety in patients with an inadequate response to TNF-α blockers or methotrexate. However, recombinant proteins are short acting and expensive (9).
Mesenchymal stem cells (MSCs) have many beneficial properties such as immunomodulation, homing activity, paracrine effects, and differentiation ability (15,39,47). MSCs are being considered for treatment of inflammatory and immune-mediated diseases due to their immunomodulatory capacity and paracrine effects through the secretion of various soluble factors such as prostaglandin E2, indoleamine 2,3-dioxygenase, and leukemia inhibitory factor (32). Adipose tissue-derived mesenchymal stem cell (ASC) transplantation has been shown to control experimentally induced graft-versus-host disease in mice (48) and to alleviate various diseases including colitis, acute kidney injury, rheumatoid arthritis, autoimmune hearing loss, and systemic lupus erythematosus (11,18,32).
ASCs present an ideal MSC source for practical regenerative medicine because they are abundant, and their use is associated with minimal ethical considerations (7,11,12,44,49). Furthermore, their capacity for proliferation and differentiation is less likely to be affected by aging than bone marrow-derived MSCs (BMMSCs) (5).
Recently, genetic modification of MSCs has been explored to determine whether their efficacy can be increased by overexpressing or suppressing target genes. Syngeneic TGF-β-transduced BMMSCs suppressed the immune response and ameliorated collagen-induced arthritis in mice, whereas nontransduced BMMSCs delayed the onset of clinical signs, but did not prevent the eventual development of severe arthritis (38). According to our previous study on experimental dogs with established autoimmune thyroiditis, the addition of supernatant from CTLA4Ig-expressing ASCs suppressed peripheral blood mononuclear cell proliferation stimulated by the autoantigen thyroglobulin more effectively than the addition of supernatant from wild-type ASCs (8).
In this study, we aimed to ascertain whether CTLA4Ig and human ASCs show synergistic effects in immune modulation and specific targeting because of the homing effect of the hASCs (4,15,47). Therefore, we introduced CTLA4Ig-overexpressing hASCs (CTLA4Ig–hASC) in a DBA/1 mouse model with sustained severe collagen-induced arthritis.
Materials and Methods
Experimental Animals
Two hundred 6-week-old male DBA/1 mice were purchased from Orient Bio (Sungnam, Korea) and were acclimatized for 2 weeks before the study's commencement. The mice were maintained in a specific pathogen-free (SPF) environment. The study was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Samsung Biomedical Research Institute, accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International, and conducted abiding by the guidelines of the Institute of Laboratory Animal Resources.
hASC Preparation
hASCs were prepared from surplus of frozen, banked stem cells (K-STEMCELL, Seoul, Korea). In brief, human abdominal subcutaneous fat tissue was obtained by simple liposuction with informed consent from the donor. hASCs were isolated from the fat stromal vascular fraction based on their ability to adhere to plastic and were cultured under Good Manufacturing Practice conditions (K-STEMCELL) as described previously (10). De-identified hASCs were used in this study under an institutional review board (IRB)-approved exemption (Samsung Medical Center IRB).
Preparation and Validation of CTLA4Ig–hASCs
We constructed a therapeutic gene comprising the extracellular domain of mouse CTLA-4 (GenBank accession No. NM_009843, nt 258-629) and the CH2-CH3 domains of the mouse immunoglobulin γ 1 constant (IGHGC) region (AB097849.1, nt 772-1,452). The human oncostatin M (NM_020530.3) signal sequence (nt 53-127) was cloned along with the therapeutic gene to enable protein secretion into body fluid. The construct was transduced into hASCs using a ViraPower™ Lentiviral Expression System (Invitrogen, Carlsbad, CA, USA) as described previously (10). The preparation and assessment of the CTLA4Ig–hASCs were performed as described previously (10).
Immunophenotypic Characteristics and Differentiation Capacity
Immunophenotypic characterization of hASCs and CTLA4Ig–hASCs was performed as described in our previous study (10). Differentiation of the ASCs was assessed using the MSC Adipogenesis Kit (Millipore, Darmstadt, Germany) and NH OsteoDiff medium (Miltenyi Biotec GmbH, Bergisch Gladbach, Germany) according to the manufacturer's instructions.
Induction of Collagen-Induced Arthritis
For the induction of collagen-induced arthritis, chicken type II collagen (2 mg/ml, CII; Sigma-Aldrich, St. Louis, MO, USA) was dissolved overnight in 0.05 M acetic acid (Sigma-Aldrich) at 4°C. The CII solution was emulsified with complete Freund's adjuvant (F5881; Sigma-Aldrich) using an electronic homogenizer (30,000 rpm, 3 min, Polytron PT3100D; Kinematica, Bohemia, NY, USA) in an ice water bath. One hundred microliters of the emulsion (100 μg CII) was injected intradermally into the base of the tail for primary immunization. Three and 7 weeks later, the mice were given a booster injection of 100 μg CII in incomplete Freund's adjuvant (F5506; Sigma-Aldrich).
Treatments
Experiment I
Collagen-induced arthritis was established in 90 mice; 10 mice were used as a normal control group (N). On the basis of their arthritis score on day 62 postimmunization, 45 arthritic mice were selected for the in vivo study and divided into four treatment groups: control (C, n = 13), hASC (H, n = 12), CTLA4Ig–hASC (CT, n = 13), and MTX (n = 7). Each group contained a similar proportion of arthritis scores. Before treatment (day 62 post-immunization), the mean arthritis scores of the C, H, CT, and MTX groups were 6.2 ± 0.6, 6.5 ± 0.7, 6.8 ± 0.7, and 6.4 ± 0.7, respectively. Mice in the N and C groups were infused with 150 μl of saline, and those in the H and CT groups were intravenously administered 2 × 106 hASCs/150 μl saline and 2 × 106 CTLA4Ig–hASCs/150 μl saline, respectively, on days 63, 70, 77, and 84 after CII immunization. The MTX group was intraperitoneally administered 1 mg/kg of MTX (Medac, Hamburg, Germany) three times a week for 4 weeks. One week after the last transplantation, all mice were killed, and blood and tissue samples were harvested.
Experiment II
To compare the therapeutic effects of recombinant CTLA4Ig and CTLA4Ig–hASC transplantation in vivo, collagen-induced arthritis was established in all but 10 (normal group, N) of the remaining 100 mice, as described above. The mice were divided into four treatment groups: normal (N, n = 10), control (C, n = 10), Orencia (O, n = 10), and CTLA4Ig–hASC (CT, n = 10). Mice in the N, C, and CT groups were treated identically to the corresponding groups in experiment I. Approximately 100 μg of recombinant CTLA4Ig protein (abatacept, Orencia®; Bristol-Myers Squibb, New York, NY, USA) was intraperitoneally administered to the mice of group O on days 63, 70, 77, and 84 after CII immunization.
Arthritis Severity Assessment
Three times a week, each mouse paw was scored on a graded scale from 0 (no arthritis) to 4 (severe arthritis) by two independent observers. The mean scores of the four paws were summated, resulting in a maximum possible score of 16 per mouse. The clinical scores were estimated for each mouse for up to 90 days postimmunization
Detection of Anti-Type II Collagen Antibody
Mouse CII antibodies were measured using a commercial mouse anti-mouse CII ELISA kit (Astarte Biologics, Bothwell, WA, USA) following the manufacturer's instructions.
Serum C-Telopeptide of Type I Collagen (C-Telopeptide I) and C-Telopeptide of Type II Collagen (C-Telopeptide II) Levels
C-telopeptide I in the serum was measured using the RatLap™ EIA kit (Immunodiagnostic Systems, Boldon, Tyne & Wear, UK). This kit can detect certain C-telopeptide degradation products from type I collagen released during osteoclastic bone resorption. Serum C-telopeptide II levels were measured using a mouse C-telopeptide of type II collagen ELISA kit (CUSABIO, Wuhan, Hubei Province, China) for quantification of cartilage degradation. Both assays were performed following the manufacturer's instructions.
Flow Cytometric Determination of the Fraction of Regulatory T Cells (Tregs) and CD138 Cells
Mouse spleen cells were analyzed for Treg markers using antibodies to CD4, CD25, and FoxP3 [three-color analysis, FITC rat anti-mouse CD4 (1 μg/test, 5 × 105 to 106 cells/test, a final volume of 20 μl), APC rat anti-mouse CD25 (0.2 μg/test), and PE rat anti-mouse FoxP3 (0.1 μg/test); BD Biosciences, San Jose, CA, USA]. Additionally, the fraction of CD138+ cells was analyzed [experiment I; single-color analysis, PE rat anti-mouse CD138 (0.2 μg/test), BD Biosciences].
Flow Cytometric Determination of the T Helper Cell Subset (Th1, Th2, Th17, and Treg) in the Spleen
In Vitro Study
Nine days after the third immunization, the mice were sacrificed, and their spleens were removed. A total of 6 × 106 splenocytes (six-well plate, 106 cells/well) were cultured in RPMI-1640 medium (Sigma-Aldrich) supplemented with 10% fetal bovine serum and 1× antibiotic–antimycotic (Nunc, Roskilde, Denmark) with denatured CII (400 μg/ml) in the absence (control) or presence of CTLA4Ig (10 μg/ml), ASCs (ASC/splenocytes = 1:5), or CTLA4Ig–hASCs (CTLA4Ig–hASC/splenocytes = 1:5). The plates were incubated at 37°C in a humidified atmosphere. For analysis of the fraction of the T helper subsets (Th1, Th2, Th17, and Treg), the splenocytes were immunostained with antibodies to CD4 and CD25 [FITC rat anti-mouse CD4 (1 μg/test, 5 × 105 cells/test, a final volume of 20 μl) and APC rat anti-mouse CD25 (0.2 μg/test); BD Biosciences]. The cells were washed and fixed. Intracellular staining was performed using the FoxP3/transcription factor staining buffer set (eBioscience, San Diego, CA, USA) and intracellular antibodies [PE rat anti-mouse FoxP3 (0.2 μg/test, 5 × 105 cells/test, a final volume of 100 μl); BD Biosciences and PE anti-RORγt (0.4 μg/test), PE anti-T-bet (0.4 μg/test), and PE anti-GATA-3 (0.048 μg/test); eBioscience] according to the manufacturer's protocols. Flow cytometric analyses were carried out using a FACSAria flow cytometer (BD Biosciences).
Experiment II
Between 91 and 94 days after the first immunization, splenocytes were collected from each mouse, and the fraction of each T helper cell subset was analyzed (n = 10 per group): Th1 cells (CD4+CD25+T-bet+), Th2 cells (CD4+CD25+GATA-3+), Th17 cells (CD4+CD25+ROR-γt+), and Tregs (CD4+CD25+FoxP3+). The staining procedure was performed as described above.
ELISA of Multiple Cytokines in Spleen Cell Culture Supernatants, Sera, and Joint Extracts
The spleen cells of each mouse were isolated and seeded in 96-well flat-bottom plates (Nunc, Roskilde, Denmark) at 2 × 105 cells/well in a final volume of 200 μl medium with or without denatured CII (100 μg/ml), ConA (2.5 μg/ml; Sigma-Aldrich), and LPS (2.5 μg/ml; Sigma-Aldrich). The plates were incubated at 37°C in a humidified atmosphere. After 48 h, the cell culture supernatant from each well was collected and stored at −70°C until use. The samples were assayed using a multiplex cytokine ELISA kit for TNF-α, interferon-γ (IFN)-γ, IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-10, IL-12p70, IL-15, IL-17, keratinocyte chemoattractant (KC), monocyte chemotactic protein 1 (MCP-1), macrophage inflammatory protein 2 (MIP-2), and “regulated upon activation normal T cell expressed and presumably secreted” (RANTES) (Millipore, Bedford, MA, USA). Serum samples and joint extracts from all mice were assayed using the same multiplex cytokine kit.
Histopathology
For histopathological examination, the knee joint tissues were fixed with 10% neutral buffered formalin (Sigma-Aldrich) and subsequently decalcified with rapid decalcifier (Thermo Scientific, Cheshire, UK) on a shaker at room temperature for 24 h. The solution was changed twice. Decalcification was considered completed when the bone tissue could easily be cut with a razor blade. Following decalcification, the tissues were dehydrated and embedded in paraffin. To evaluate histopathological damage, the paraffin-embedded tissues were sliced into 4-μm sections, deparaffinized in xylene, rehydrated in graded alcohol, and stained with hematoxylin and eosin (H&E; Dako, Carpinteria, CA, USA) and safranin O (IHC World, Woodstock, MD, USA). The severity of cartilage damage was scored on a graded scale from 0 (no damage) to 4 (severe damage).
Microcomputed Tomography (Micro-CT) Imaging
To examine the morphological deformity of paw joints, the mice were sacrificed, and paws were fixed with 4% formalin for micro-CT imaging, using a micro-CT scanner (Inveon Preclinical CT; Siemens Healthcare, Hoffman Estates, IL, USA), at 40-μm slice thickness, with an exposure time of 0.6 s, photon energy of 70 keV, and a current of 400 μA. Projection images were reconstructed into three-dimensional images with the IRW software (Siemens Healthcare, Malvern, PA, USA). The severity of morphological deformity was scored on a graded scale from 0 (no damage) to 4 (severe damage).
Biodistribution of Transplanted ASCs
Three mice from the H and CT groups were injected with hASCs and CTLA4Ig–hASCs that were fluorescently labeled with conjugated red fluorochrome CellTracker™ CM-DiI (Life Technologies, Carlsbad, CA, USA) according to the manufacturer's instructions regarding identification in histopathological sections. An additional animal study was conducted for confocal microscopic examination; normal mice (n = 3, age-matched DBA/1) were injected with hASCs fluorescently labeled with CM-DiI. The presence of CM-DiI-labeled cells was examined in various tissues, including the spleen, draining lymph node, kidney, liver, lung, and the heart, using a laser scanning confocal microscope (LSM 700; Carl Zeiss, Jena, Germany). The cells were counterstained with 10 μl of mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI) (1.5 μg/ml; Vector Laboratories, Burlingame, CA, USA). Bone samples of the knee (femorotibial) joint were fixed overnight with 4% paraformaldehyde in phosphate-buffered saline (PBS) (Biosesang, Seongnam, Korea) at 4°C and decalcified with 5% EDTA (Sigma-Aldrich) in PBS (pH 7.4) at 4°C for approximately 21 days. The samples were dehydrated with a graded series of sucrose solutions (Sigma-Aldrich), embedded in OCT compound (Sakura/Tissue-Tek, Torrance, CA, USA), and examined for the presence of CM-DiI-labeled cells.
Statistical Analysis
All results are expressed as the mean ± the standard error of the mean (SEM). The means of treatment groups—except for cytokine data—were compared using one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison post hoc tests. Cytokine data were analyzed using a Kruskal–Wallis test followed by Mann–Whitney U-tests with Bonferroni correction. The paired t-test and Wilcoxon signed ranks test were used to compare means from two related samples (before vs. after treatment). Confidence levels of 95% or higher were considered significant (p < 0.05). All statistical analyses were performed using SPSS version 21.0 (IBM, Armonk, NY, USA).
Results
Immunophenotypic Characteristics and Differentiation Capacity
The immunophenotypic characterization of hASCs and CTLA4Ig–hASCs revealed that both cell groups strongly expressed CD29, CD44, CD73, CD90, and CD105 but not CD31, CD34, CD45, or HLA-DR surface markers (Fig. 1A). The features indicating differentiation of the ASCs and CTLA4Ig–hASCs into adipocytes and osteocytes are presented in Figure 1B.
Immunophenotypic characteristics and differentiation capacity of human adipose tissue-derived mesenchymal stem cells (hASCs). (A) Immunophenotypes of ASCs and CTLA4Ig-overexpressing human ASCs (CTLA4Ig–hASC). (B) hASCs and CTLA4Ig–hASC that had differentiated into adipocytes contained Oil red O-positive lipid droplets (top) and hASCs and CTLA4Ig–hASC that had differentiated into osteocytes stained with Alizarin red S (bottom).
Experiment I
Assessment of the Arthritis Severity
The arthritis index was significantly lower in the H and CT groups than in the saline-treated C group after three injections (H: observed on days 84, 85, 87, and 90 postimmunization, CT: observed on days 78, 80, 84, 85, 87, and 90 postimmunization). The MTX and C groups showed similar arthritis index patterns (Fig. 2A). At day 90 post-immunization, the average thickness of the hindpaws in the N, C, H, CT, and MTX groups was 1.9 ± 0.04 mm, 2.58 ± 0.13 mm, 2.18 ± 0.08 mm, 2.28 ± 0.10 mm, and 2.49 ± 0.17 mm, respectively.
Arthritis scores and serum levels of anti-mouse type II collagen antibody, C-telopeptide I, and C-telopeptide II of experimental groups in experiment I. Collagen-induced arthritis was introduced in 90 mice; 10 mice were used as a normal control group (N). Of the 90 arthritic mice, 45 were selected based on their arthritis score on day 62 postimmunization and divided into four groups: control (C, n = 13), hASC (H, n = 12), CTLA4Ig–hASC (CT, n = 13), and MTX (n = 7). All mice in the N and C groups were infused with 150 μl of saline, and those in the H and CT groups were intravenously administered 2 × 106 hASCs and CTLA4Ig–hASCs/150 μl saline, respectively, on days 63, 70, 77, and 84 after CII immunization. The MTX group was intraperitoneally administered 1 mg/kg of methotrexate three times a week for 4 weeks. Data are expressed as the mean ± SEM. (A) Severity of arthritis was scored on a graded scale from 0 (no arthritis) to 4 (severe arthritis) on the basis of the degree of inflammation. The group means were compared using one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison post hoc tests. *p < 0.05 (vs. C). (B) Serum levels of anti-mouse type II collagen antibody. The paired t-test (†) and Wilcoxon signed-ranks test (‡) were used to compare the means from two related samples (before vs. after treatment). “†” and “‡” indicate significant differences (p < 0.05). (C) Serum levels of C-telopeptide I. (D) Serum levels of C-telopeptide II. The intergroup analysis was performed using ANOVA followed by Tukey's multiple comparison post hoc tests. *p < 0.05. SEM, standard error of the mean.
Anti-Type II Collagen Antibody
Anti-mouse CII autoantibodies were significantly lower after therapy than before therapy in the CT group. Anti-mouse CII autoantibodies were higher after therapy than before in the C and MTX groups (Fig. 2B).
Serum C-Telopeptide I and C-Telopeptide II Levels
The serum level of C-telopeptide I did not significantly differ among the C, H, CT, and MTX groups (Fig. 2C). The serum level of C-telopeptide II was significantly lower in the N and CT groups than in the C group (Fig. 2D).
Flow Cytometric Determination of the Fraction of Tregs and CD138 Cells
The fraction of Tregs in spleen cells was estimated. The fraction of CD4+FoxP3+ cells was 5.7 ± 1.1%, 3.4 ± 0.4%, 3.4 ± 0.3%, 6.3 ± 4.6%, and 4.4 ± 3.3% in the N, C, H, CT, and MTX groups, respectively. The fraction of CD4+CD25+FoxP3+ cells was 1.2 ± 0.1%, 1.7 ± 0.4%, 1.2 ± 0.2%, 2.4 ± 0.5%, and 2.5 ± 2.0% in the N, C, H, CT, and MTX groups, respectively. Treg expansion in the spleen did not significantly differ between the treated and control mice. The fraction of CD138 cells was 13.8 ± 3.0%, 18.8 ± 1.9%, 7.2 ± 0.6%, 7.7 ± 0.6%, and 17.2 ± 2.4% in the N, C, H, CT, and MTX groups, respectively. The fraction of CD138 cells was significantly lower in the H and CT groups than in the C group.
Cytokine Levels in Sera and Joint Extracts
Cytokine Levels in Serum and Knee Extracts From the Experimental Groups (pg/ml, Experiment I)

Data obtained from groups were compared using a Kruskal–Wallis
p < 0.05) followed by Mann–Whitney U-test with Bonferroni correction.
p < 0.05. N, normal group; C, control group (saline treated); H, hASC treated; CT, CTLA4Ig–hASC treated; MTX, methotrexate treated.
The level of RANTES in joint extracts was significantly lower in the H, CT, and MTX groups than in the C group, and the MIP-2 level was significantly lower in joint extracts from the H and CT groups than in that from the C group. The level of IL-1β in the joint extracts was significantly lower in the CT than in the C group.
Cytokine Levels in Splenocyte Culture Supernatants and Draining Lymph Node Cells
Cytokine Levels in Spleen Cell Culture Supernatants From the Mice With Collagen-Induced Arthritis (pg/ml, Experiment I)

Data obtained from groups were compared using a Kruskal–Wallis (C, H, CT and MTX
p < 0.05) followed by a Mann–Whitney U-test with Bonferroni correction.
p < 0.05. C, control group (saline treated); H, hASC-treated; CT, CTLA4Ig–hASC treated; MTX, methotrexate treated; Spleen-CII, Spleen-ConA, Spleen-LPS, cytokine levels in spleen cell culture supernatants after type II collagen, ConA, or LPS stimulation, respectively.
The level of IL-10 in the culture supernatants of lymph node cells stimulated with ConA was significantly higher in both the H and CT groups than in the C group (N: 51.91 ± 23.04, C: 35.19 ± 6.34, H: 94.71 ± 36.96, CT: 62.04 ± 8.31, MTX: 23.11 ± 4.08 pg/ml, respectively).
Histological Evaluation of Collagen-Induced Arthritis
The joints of mice from both the H and CT groups showed decreased destruction of articular cartilage compared to the C group, and CTLA4Ig–hASC transplantation provided greater protection of cartilage (Fig. 3A, B). The C and MTX groups showed severe cartilage destruction. This result is concordant with that of the serum C-telopeptide II analysis.
Knee joint histopathology and paw joint micro-CT images (experiment I). (A) Histopathology of knee joints: representative joint sections of mice of each treatment group at 13 weeks after administration of primary immunization. Mice from the normal control group (N; no arthritis induced) were sacrificed on the same day and examined. Original magnification: 40×, arrowhead: severe cartilage destruction. (B) The severity of cartilage damage (knee joints) on a graded scale from 0 (no damage) to 4 (severe damage). (C) Representative micro-CT images from each group: in order to examine the morphological deformity of the paw joints, the paws were scanned with micro-CT at 40-μm slide thickness, with an exposure time of 0.6 s, photon energy of 70 keV, and a current of 400 μA. (D) The severity of morphological deformity (paw joints) was scored on a graded scale from 0 (no deformity) to 4 (severe deformity). The intergroup analysis was performed using ANOVA followed by Tukey's multiple comparison post hoc tests. *p < 0.05 (vs. C). N, normal group, C, control group, H, hASC-treated group, CT, CTLA4Ig–hASC-treated group, MTX, methotrexate treated.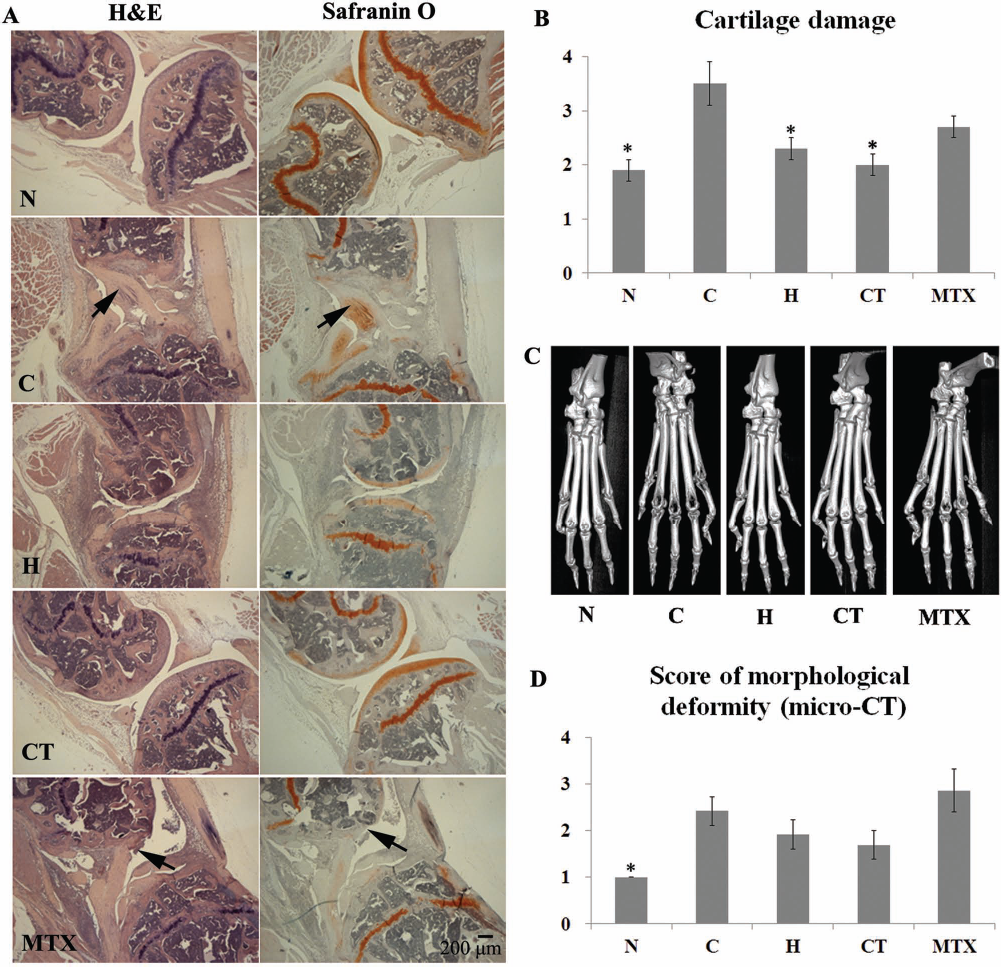
Paw Micro-CT Imaging
The micro-CT scans of the paws revealed that 4 out of 12 mice in the C group, 2 out of 12 mice in the H group, 2 out of 13 mice in the CTLA4Ig–hASC group, and 4 out of 7 mice in the MTX group showed severe morphological deformity of the paw joint. Representative images and the deformity scores are presented in Figure 3C and D.
Biodistribution of Transplanted ASCs
Multiple hASCs and CTLA4Ig–hASCs labeled with CM-DiI red fluorescent tracker dye were present in the spleen and lungs, and some were evident in the lymph nodes and liver (Fig. 4). Very few fluorescently labeled cells were observed in the kidney and heart, but some were observed in joint-containing bone samples (Fig. 4). When hASCs labeled with CM-DiI were injected into normal DBA/1 mice, relatively more cells were observed in the lungs than into arthritic DBA/1 mice (Fig. 4).
Confocal microscopic examination of CM-DiI-labeled human adipose tissue-derived mesenchymal stem cells (hASCs) and CTLA4Ig-overexpressing hASCs (CTLA4Ig–hASCs) (experiment I). Three mice from the H and CT groups were treated with hASCs and CTLA4Ig–hASCs that were fluorescently labeled (conjugated red fluorochrome CellTracker™ CM-DiI) for identification in histopathological sections. Additional normal mice (n = 3) were injected with hASCs fluorescently labeled using CM-DiI on the same schedule (Normal-hASC). Representative images for each treatment group are shown. Original magnification: 200×. BM, bone marrow.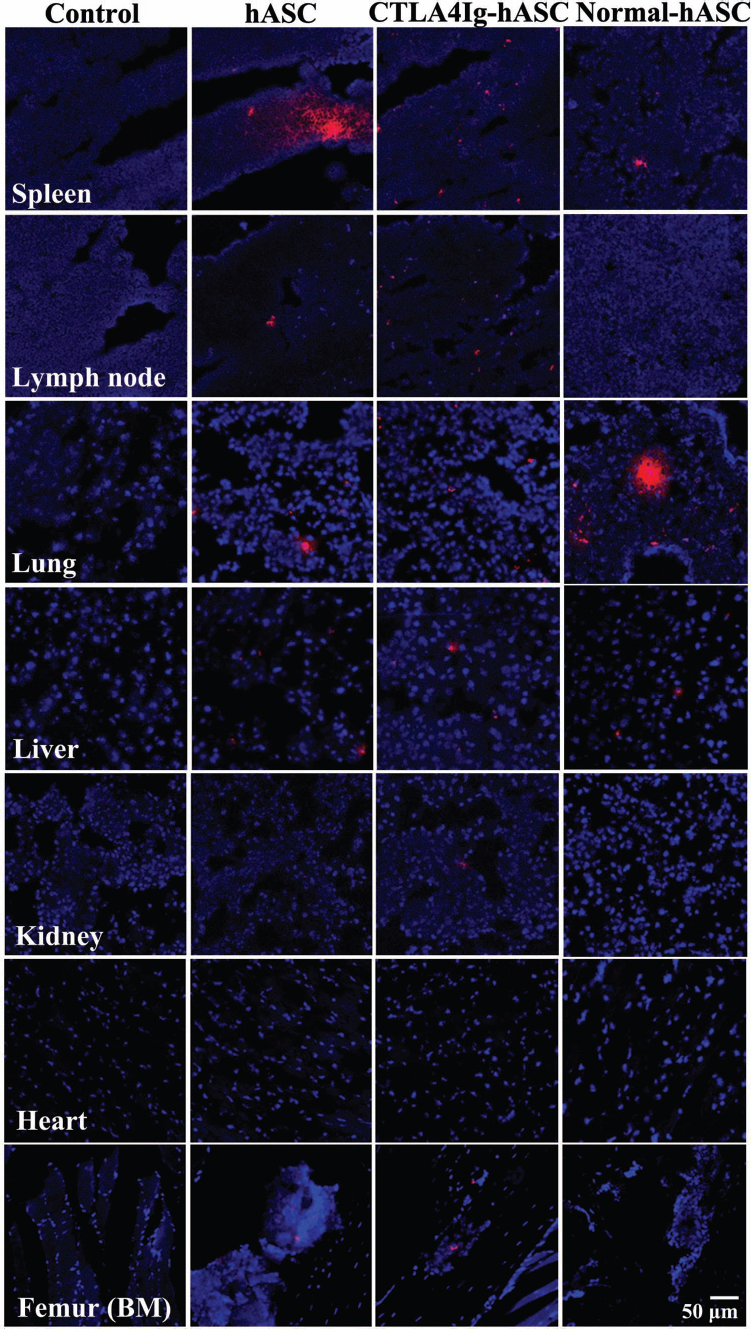
Experiment II
Determination of Anti-Type II Collagen Antibody, Serum C-Telopeptide I, and C-Telopeptide II Levels
The level of anti-mouse CII autoantibodies was significantly lower after therapy than before therapy in the CT group. After therapy, the CII autoantibody level was significantly lower in the CT than in the C group (Fig. 5A). The serum levels of C-telopeptides I and II were significantly lower in the N and CT groups than in the C group (Fig. 5B, C). The CII autoantibody and C-telopeptide II serum levels in the O group showed a decrease, but were not significantly different from those in the C group.
Serum levels of anti-mouse type II collagen antibody, C-telopeptide I, and C-telopeptide II in the experimental groups (experiment II). (A) Serum levels of anti-mouse type II collagen antibody: data obtained from groups were compared using ANOVA followed by Tukey's multiple comparison post hoc tests. A paired t-test (†) was used to compare the means from two related samples (before vs. after treatment). †p < 0.05. (B) Serum levels of C-telopeptide I. (C) Serum levels of C-telopeptide II. The intergroup analysis was performed using ANOVA followed by Tukey's multiple comparison post hoc tests. *p < 0.05 (vs. C). SEM, standard error of the mean; N, normal group; C, control group; O, recombinant CTLA4Ig-treated group; CT, CTLA4Ig–hASC-treated group.
In Vitro Study of Splenic T Cells Expressing the Master Regulators of Th1, Th2, Th17, and Treg
The expression of T-bet and GATA-3 was significantly lower in splenocytes grown in the presence of hASCs or CTLA4Ig–hASCs than in the controls. The ratio of the Treg/Th17 master regulators (CD25+FoxP3+/CD25+RORγt) in CD4+ T cells grown with CTLA4Ig–hASCs was significantly higher than that in the control wells (Fig. 6).
Flow cytometric determination of the T helper cell subset (Th1, Th2, Th17, and Treg) in the spleen (in vitro). Nine days after the third immunization, immunized male DBA/1J mice were sacrificed, and their spleens were removed. A total of 6 × 106 splenocytes (six-well plate, 106 cells/well) were cultured in medium with denatured CII (400 μg/ml) in the absence (control well, X) or presence of CTLA4Ig (10 μg/ml, CTLA4Ig), ASC (hASC/splenocytes = 1:5, hASC) or CTLA4Ig–hASC (CTLA4Ig–hASC/splenocytes = 1:5, CTLA4Ig–hASC). After 3 days, the splenocytes were immunostained using antibodies to CD4, CD25, FoxP3, ROR-γt, T-bet, and GATA-3 and analyzed by FACS. Graphs and representative flow cytometric gating analysis images from each well are presented.
Flow Cytometric Determination of the T Helper Cell Subset (Th1, Th2, Th17, and Treg) in the Spleen
The CT group showed the highest fraction of Tregs (CD4+ CD25+FoxP3+). The fractions of Th1 cells (CD4+CD25+T-bet+) and Th17 cells (CD4+CD25+RORγt+) of the CT group showed the lowest in the arthritic groups; however, the differences were not significant. The ratio of Treg/Th17 was also highest in the CT group (Fig. 7).
Flow cytometric determination of the T helper cell subset (Th1, Th2, Th17, and Treg) in the spleen (experiment II). At 91-94 days after the first immunization, splenocytes from each mouse were collected, and the fraction of the T helper cell subset was analyzed (n = 10 per group). N, normal group; C, control group; O, recombinant CTLA4Ig-treated group; CT, CTLA4Ig–hASC-treated group. Graphs and representative flow cytometric gating analysis images from each group are presented.
Histological Evaluation of Collagen-Induced Arthritis
The severity scores of the joint cartilage damage in the mice from the CT group were significantly lower than in those from the C group (Fig. 8A, B).
Knee joint histopathology and paw joint micro-CT images (experiment II). (A) Histopathology of knee joints: representative joint sections of mice of each treatment group at 13 weeks after administration of primary immunization. Mice in the normal group (N; no arthritis induced) were sacrificed on the same day and examined. Original magnification: 40×; arrowhead: severe cartilage destruction). (B) The severity of cartilage damage (knee joints) was scored on a graded scale from 0 (no damage) to 4 (severe damage). (C) Representative micro-CT images from each group: in order to examine the morphological deformity of the paw joints, the paws were scanned with micro-CT at 40-μm slide thickness, with an exposure time of 0.6 s, photon energy of 70 keV, and a current of 400 μA. (D) The severity of morphological deformity (paw joints) was scored on a graded scale from 0 (no deformity) to 4 (severe deformity). Intergroup analysis was performed using ANOVA followed by Tukey's multiple comparison post hoc tests. *p < 0.05 (vs. C). N, normal group; C, control group; O, recombinant CTLA4Ig-treated group; CT, CTLA4Ig–hASC-treated group.
Paw Micro-CT Imaging
The scores for paw deformity in the CT group were lower than those in the C group, although the difference was not statistically significant. The scores for paw deformity in the N group were significantly lower than those in the C group (Fig. 8C, D).
Discussion
We evaluated the therapeutic effects of CTLA4Ig-expressing ASCs on established collagen-induced arthritis (advanced stage of disease) in comparison with those of treatment with nontransduced ASCs, methotrexate, and recombinant CTLA4Ig. CTLA4Ig expression in the ASCs was established by transduction of a lentiviral vector construct. Lentiviral systems might be advantageous for treatment of autoimmune diseases or diseases related to gene defects based on cell-based gene therapy (7,10). They can be used in a broad range of cell types and have a capacity for long-term expression (35). Indeed, transduction of the ASCs with the lentiviral vector resulted in long-term expression of the therapeutic gene. Furthermore, we confirmed that the concentration of murine CTLA4 in the culture supernatant of the CTLA4Ig–hASCs was sustained at least until passage 19. Antigenic stimulation of T cells in the absence of costimulation signals leads to aborted T-cell proliferation and T-cell clonal anergy (6,33). CTLA4Ig can block the B7-CD28 costimulatory pathway owing to the comparatively higher binding affinity of CTLA4 for CD28, which results in the induction of T-cell clonal anergy and the amelioration of autoimmune disease (6,33).
The arthritis scores were significantly lower in the groups that received repeated injection (once every week, four times) of hASCs and CTLA4Ig–hASCs than in the saline-treated control group at 3-4 weeks after treatment. These results indicated that aggravation of arthritis was effectively prevented by serial transplantation of hASCs and CTLA4Ig–hASCs, even in mice with sustained, severe collagen-induced arthritis. In contrast, arthritis progressed rapidly in mice treated with saline or MTX. Previously, a single injection of murine BMMSCs at 7 weeks postimmunization has been shown to delay the onset of clinical signs for approximately 2 weeks; however, severe disease eventually developed in all mice, and arthritis scores were similar to those of PBS-treated control mice at 3 weeks after treatment (38).
In the in vivo experiment, the fraction of Tregs in the spleen did not significantly differ among the treatment groups. These results were in agreement with those of a previous study using BMMSCs (38). The percentage of Th1 (CD4+CD25+T-bet+) and Th2 (CD4+CD25+GATA-3+) cells in spleens from CTLA4Ig–hASC-treated mice showed a subtle, though insignificant, decrease. In the in vitro study, treatment with ASCs or CTLA4Ig–hASCs significantly decreased T-bet and GATA-3 expression in splenocytes, and CTLA4Ig–hASC treatment significantly increased the ratio of Treg/Th17.
The fraction of CD138 was significantly lower in spleens of mice transplanted with hASCs and CTLA4Ig–hASCs than in those of control mice. Furthermore, CII autoantibodies decreased significantly after CTLA4Ig–hASC therapy in the arthritic mice. CD138 is expressed on plasma cells and B-cell precursors. Plasma cells generated during autoimmune responses are responsible for autoantibody production, and humoral responses are essential for the development of chronic autoimmune diseases (29).
A major clinical feature of RA is abnormal and degraded cartilage in affected joints. As type II collagen is a specific product of cartilage, its degradation product (C-telopeptide II) in serum, urine, and synovial fluid provide a measure of cartilage degradation (21) and are useful biomarkers for monitoring the effects of chondroprotective drugs (30,37). In our histological study of the joints, the mice treated with saline or MTX showed severe cartilage destruction. By contrast, serial transplantation of either hASCs or CTLA4Ig–hASCs decreased the destruction of articular cartilage; CTLA4Ig–hASC transplantation was more effective in cartilage protection than hASCs alone as indicated by the histopathological findings and serum levels of C-telopeptide II. It has been shown that intra peritoneal administration of MTX (1 mg/kg, three times a week) prior to the onset of arthritis completely prevents the development of clinical and pathological signs of collagen-induced arthritis (36). However, in our study, the same dose of MTX did not ameliorate established arthritis. In a previous study, recombinant CTLA4Ig treatment at an early stage significantly suppressed the development of arthritis compared to a saline-treated control group (25). The researchers had intraperitoneally administered 100 μg of CTLA4Ig at 7, 15, and 18 days after primary immunization and examined arthritic symptoms until day 63. In this study, booster injections were given at 21 and 49 days after primary immunization, to sustain severe arthritis. Recombinant CTLA4Ig treatment at an advanced stage (63, 70, 77, and 84 days after primary immunization, 100 μg/body) improved the serum levels of CII autoantibody and C-telopeptide II, although the results were not significant.
The CTLA4Ig–hASCs decreased the serum levels of IL-12p70 and knee-joint extract levels of MIP-2 more effectively than the hASCs. IL-12 plays a critical role in the regulation of immune responses in various autoimmune disease models; treatment with anti-IL-12 antibodies ameliorates established inflammation in experimental colitis (16), and IL-12 in combination with type II collagen induces severe arthritis in DBA/1 mice (17). The median level of circulating IL-12p70 in patients with RA was significantly higher than that in healthy controls (23). After DMARD treatment, the improved group showed a more significant decrease in IL-12 than the non-improved group (23). MIP-2 (CXCL2) is secreted by monocytes and macrophages and is chemotactic for polymorphonuclear leukocytes. Macrophages accelerate disease via the acute phase response network, production of TNF-α, and chronic activation of circulating monocytes (24). Therefore, a decrease in IL-12p70 and MIP-2 by treatment with CTLA4Ig–hASCs can ameliorate RA. The level of IL-10 in culture supernatants of spleen cells stimulated with CII was significantly higher in both the H and CT groups than in the C group. The levels of IL-17 and MCP-1 were significantly lower in the CT than in the C group. MCP-1 is one of the key chemokines that regulate migration and infiltration of monocytes/macrophages (14). Thus, a decrease in MCP-1 by treatment with CTLA4Ig–hASCs also can ameliorate RA. Furthermore, activation of Th1 and Th17 cells has been implicated in the development of autoimmune arthritis (3,22,51), and Th2 cells are protective in human RA and collagen type II-induced RA animal models (19,34,43). Therefore, decreased IL-17 and increased IL-10 levels following hASC or CTLA4Ig–hASC transplantation may be involved in improving the symptoms of RA. Together, the modulation of cytokines and chemokines by transplantation of hASCs or CTLA4Ig–hASCs can prevent the recruitment of neutrophils, monocytes, and macrophages, and regulate the activation of Th cells, which may be involved in therapeutic effects on collagen-induced arthritis.
To determine their biodistribution, hASCs and CTLA4Ig–hASCs were labeled with CM-DiI cell tracker. We found that both cell types were present in the spleen and lungs, and some were evident in the lymph nodes, liver, and joint-containing bone samples of the mice with established arthritis.
Taken together, our results indicated that serial hASC or CTLA4Ig–hASC transplantation can improve the condition of established autoimmune arthritis by immune modulation. CTLA4Ig–hASCs showed stronger therapeutic effects by decreasing autoantibodies and cartilage damage more than nontransduced hASCs. Insertion of therapeutic or inducible regulatory genes into ASCs by nontoxic and effective new gene transfer systems can offer a novel method for immune modulation in autoimmune diseases.
Footnotes
Acknowledgments
The authors thank the staff of the Laboratory Animal Research Center, Samsung Biomedical Research Institute for their technical support and assistance. This study was supported by grants from the Samsung Biomedical Research Institute (SBRI fund AB20011/SMX1131211/SMX11 40141) to E. W. Choi. E. W. Choi is listed as an inventor on a pending patent that describes the “Composition and kit for the prevention or treatment of arthritis, and methods for preventing or treating arthritis using the same.” Since E. W. Choi and her institute stand to profit from this work, E. W. Choi has a conflict of interest in this capacity, which has hereby been officially disclosed. The other authors declare no conflicts of interest.
