Abstract
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease that selectively affects motor neurons in the cortex, brain stem, and spinal cord. The precise pathogenic mechanism remains unknown, and there is currently no effective therapy. We evaluated the therapeutic effects of human adipose tissue-derived stem cells (ASCs) in an animal model of ALS. Human abdominal subcutaneous fat tissues were obtained by simple liposuction from donors, and ASCs were isolated from the fat stromal vascular fraction. ASCs were found to differentiate into adipocytes, chondrocytes, osteocytes, and neurons. SOD1G93A ALS mice were divided into three groups: sham, intravenous (IV), and intracerebroventricular (ICV) groups. Human ASCs were transplanted in the ALS mice at 70 postnatal days before the appearance of clinical symptoms. Behavior of transplanted animals was assessed by rotarod test, paw grip endurance (PaGE), and reflex index. Mice in every group were sacrificed after 4 weeks posttransplantation. Transplanted ASCs were identified in the lumbar spinal cords with an antihuman mitochondria antibody and cell type-specific markers for neurons or astrocytes. Delayed onset of clinical symptoms (26 days) and extended survival of animals (24 days) were observed in ALS mice transplanted with ASCs via ICV route. ASCs were found to secrete high levels of neurotrophic factors such as NGF, BDNF, IGF-1, and VEGF. Reduction of apoptotic cell death by these factors was confirmed in cultured CNS cells and in the ALS spinal cord. These results indicate that transplantation of ASCs in ALS mice provides neuroprotective effects by production of cytokines/growth factors, delays disease progression, and prolongs the life span of ALS mice.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is an adult onset neurodegenerative disease characterized by degeneration and progressive loss of motor neurons, leading to muscle wasting, weakness, and eventually to death within 5 years after the onset of clinical symptoms (4,37). The proposed pathogenetic mechanisms of ALS, albeit not fully elucidated, include oxidative stress, protein aggregation, mitochondrial dysfunction, impaired axonal transport, glutamate-mediated excitotoxicity, and insufficient production of neurotrophic factors (2). To date there is no effective treatment for patients suffering from ALS.
During the past 20 years, stem cell-based cell therapy for neurological diseases such as Parkinson's disease, Huntington's disease, ALS, stroke, and spinal cord injury has been investigated intensively because of its therapeutic potential (10,12,22,23,25,27). Recent studies have indicated that motor neurons could be generated from stem cells such as embryonic stem cells (ESCs), embryonic germ cells (EGCs), induced pluripotent stem cells (iPSCs), and neural stem cells (NSCs) and transplanted in ALS animal models resulting in behavioral improvement and extended life span (14,19,30,43). Motor neurons were also generated from iPSCs isolated from an ALS patient (6). Recently human spinal cord neural stem/progenitor cells were transplanted into the spinal cord of superoxide dismutase 1 G93A mutation (SOD1G93A) transgenic rats, and the results indicated that the neurological function of transplanted animals was improved, but no improvement in disease onset and the overall animal survival was found (15).
More recently, a phase I trial of intraspinal injections of fetal brain-derived NSCs in ALS patients was conducted in the US using the same human NSC preparations as the rat ALS study, and there was no evidence of clinical improvement (36).
Mesenchymal stem cells (MSCs) derived from bone marrow have been transplanted in ALS patients because of their autologous cellular source and ability for multilineage differentiation in vivo, but no clear clinical benefits were detected in these patients (9,29). Recently, adipose tissue-derived stem cells (ASCs) have also been proposed as an alternative to bone marrow-derived MSCs. ASCs are an abundant, accessible, and replenishable source of adult stem cells that can be isolated from liposuction waste tissue. They exhibit characteristics of multipotent adult stem cells, which are capable of differentiating along the adipocyte, chondrocyte, osteoblast, and neuronal lineages. Preclinical use of ASCs has been reported (8,33,39,44). ASCs were transplanted in animal models of ischemic brain damage and spinal cord injury to regenerate the damaged central nervous system (CNS) tissue (5,11,20). Furthermore, ASCs are capable of secreting a large number of growth factors, such as vascular endothelial growth factor (VEGF) and insulin-like growth factor 1 (IGF1), which exhibit protection against neuronal cell death from injury (38,40,41).
In the present study, we investigated the therapeutic potential of human ASC transplantation in a mouse model of ALS.
Materials and Methods
Human ASC Culture
Use of human ASCs in this study was approved by the Chung-Ang University Ethics Committee on Human Subjects (Certificate #09–0041), and informed consent was obtained from the donors. Human abdominal subcutaneous fat tissues were obtained by simple liposuction from three donors (age: 64, 69, and 84; two males, one female), and ASCs were isolated from the fat stromal vascular fraction by their adherence to plastic and were cultured in RKCM medium (RmedicaStemCell, Seoul, Korea) under Good Manufacturing Practice (GMP) conditions as described previously (34). The stem cell potency of the cultured ASCs was confirmed by their differentiation into adipocytes, chondrocytes, osteocytes, and neurons (data not shown). Adipogenic, osteogenic, or chondrogenic differentiation was induced by growing the cells for 2 weeks in a 12-well plate (BD-Falcon Labware, Franklin Lakes, NJ, USA) with a StemPro Adipogenesis Differentiation Kit, StemPro Osteogenesis Differentiation Kit, or StemPro Chondrogenesis Differentiation Kit (Life Technologies), respectively. Differentiation of the ASCs was evaluated by Oil Red O (for adipocytes; Sigma, St Louis, MO, USA), Alizarin Red S (for osteocytes; Sigma), or Alcian blue (for chondrocytes; Sigma). To confirm neurogenic differentiation, ASCs were cultured in Dulbecco's modified Eagle's medium (DMEM, Sigma) supplemented with 10% fetal bovine serum (Life Technologies), basic fibroblast growth factor (bFGF; 100 ng/ml, Peprotech, Rock Hill, NJ, USA), and 1 μm retinoic acid (Sigma) for 2 weeks. The induced cells were stained with antibodies specific for β-tubulin III (1:800; TubIII; Millipore, Billerica, MA, USA), nestin (1:500; Millipore), or glial fibrillary acidic protein (GFAP; 1:800; Millipore) (data not shown).
Transplantation of Human ASCs Into ALS Mice
Animal experiments were approved by the Chung-Ang University Animal Care Committee (Certificate 11–0033) and were in compliance with the US National Institutes of Health guidelines for the care and use of laboratory animals. Female transgenic mice overexpressing human superoxide dismutase-1 (SOD1) carrying G93A mutation (13) (Jackson Lab, Bar Harbor, ME, USA) were divided into three groups [n = 9 in each group: sham (phosphatebuffered saline; PBS), intravenous (IV), and intracerebroventricular (ICV) groups]. Each mouse in the IV and ICV groups received a graft of 1 × 106 cells intravenously (in 100 μl PBS via tail vein) or 2 × 105 cells into lateral ventricle (in 6 μl PBS; 0.22 mm posterior, 0.4 mm lateral, and 3 mm ventral from bregma), respectively. The cells transplanted in this study were given by three donors who gave informed consent for research use (Tables 1 and 2). Animals were anesthetized with a 3:2 mixture of zoletil (30 mg/ml, Bayer Korea, Seoul, Korea) and rompun (10 mg/ml, Bayer Korea) at 70 days of age, and human ASCs were delivered intravenously in PBS (for IV, 1 × 106 cells) or stereotaxically (for ICV, 2 × 105 cells; 0.2 mm posterior, 0.4 mm lateral, and 3.0 mm ventral to bregma) using a 10-μl Hamilton Gastight syringe (Hamilton, Reno, NV, USA) with an attached 26-gauge needle.
ASC Donors and Transplantation Results via ICV Route

ASC, adipose tissue-derived stem cell; ICV, intracerebroventricular; PaGE, Paw grip endurance; PBS, phosphate-buffered saline.
ASC Donors and Transplantation Results via IV Route
IV, intravenous.
Behavioral Tests
Motor strength and motor coordination were evaluated on a rotarod rotator (Daejong instrument, Seoul, Korea) after a 1-week learning period. The running time of the mice was measured on a rotarod rotating with constant speed of 15 rpm, and 300 s was selected as the arbitrary cutoff time. The animals performed the test twice a week until they were no longer capable of performing the task. Paw grip endurance (PaGE) was used as the index of grip strength of the animals. The wire lid was gently shaken to prompt the mouse to hold onto the grid before the lid was swiftly turned upside down. The latency of the mouse to let go of the grid with at least both hind limbs was measured. Each mouse was allowed up to three attempts to hold on to the inverted lid for an arbitrary maximum 200 s, and the longest latency was recorded. Extension reflex was evaluated by the following scoring system: 4 for balanced normal extension reflex of both hind limbs, 3 for imbalance of extension, 2 for the extension reflex of only one hind limb, 1 for the absence of any hind limb extension, and 0 for total paralysis. The time of death was defined as the date on which the mice could no longer roll over within 30 s after being placed on their sides. Disease onset was assessed by a 15% loss of performance on the PaGE and rotarod tests and by a reduction of one point in extension reflex (16).
Immunohistochemistry
Mice in every group were sacrificed after 4 weeks posttransplantation with ASCs. The lumbar segment of the spinal cord was removed and cut at 20-μm thickness by cryostat. Sections were doubly stained with an antihuman mitochondria antibody (hMit, 1:500; Millipore) to detect the engrafted human ASCs and antibodies for neuron-specific markers microtubule-associated protein-2 (MAP2, 1:250; Millipore) or neurofilament-M (NF, 1:250; Millipore) or an antibody for astrocyte-specific marker GFAP (1:500; Millipore) according to previously published methods (21). Apoptotic cell death in the spinal cords of ASC-transplanted mice was evaluated by terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining using the In Situ Cell Death Detection Kit (Roche Diagnostics, Mannheim, Germany). All stained sections were observed via Olympus fluorescence microscopy (Seoul, Korea).
RT-PCR
Total RNA was extracted from ASCs using a Trizol reagent (Life Technologies), and cDNA was synthesized from 2 μg of total RNA using oligo (dT) primers and TOPscript reverse transcriptase (Enzynomics, Seoul, Korea) with the manufacturer's instructions. cDNA was amplified for 35 cycles of PCR with the previously designed primers (Table 3).
Sequence of Primers

NGF, nerve growth factor; BDNF, brain-derived neurotrophic factor; NT3, neurotrophin 3; GDNF, glial cell line-derived neurotrophic factor; IGF1, insulinlike growth factor 1; CNTF, ciliary neurotrophic factor; HGF, hepatocyte growth factor; VEGF, vascular endothelial growth factor; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
ELISA
The amount of cytokine secretion in the culture supernatant of ASCs and in spinal cord homogenates was determined using enzyme-linked immunosorbent assay (ELISA) kits. To examine in vitro production of cytokines, 1 × 106 ASCs were cultured in DMEM with 10% FBS for 24 h, and the conditioned medium was collected. Ten milligrams of spinal cord was removed from the ASC-transplanted ALS mouse and the PBS-injected ALS mouse. Collected tissues were homogenized with lysis buffer [137 mM NaCl, 20 mM Tris, pH 8.0, 1% Nonidet P-40, 1 mM phenylmethanesulfonyl fluoride (PMSF), 10 mg/ml aprotinin, 1 mg/ml leupeptin, and 0.5 mM sodium vanadate; Cell Signaling, Danvers, MA, USA] and diluted in the dilution buffer provided by the ELISA kits. Concentration of cytokines of in vitro and in vivo samples was measured by ELISA kits specific for human nerve growth factor (NGF; R & D Systems, Minneapolis, MN, USA), human brain-derived neurotrophic factor (BDNF; R & D Systems), human neurotrophin 3 (NT3; Abcam, Cambridge, UK), human glial cell line-derived neurotrophic factor (GDNF; R&D Systems), human insulinlike growth factor 1 (IGF1; R&D Systems), human ciliary neurotrophic factor (CNTF; R&D systems), human hepatocyte growth factor (HGF; R&D Systems), and human vascular endothelial growth factor (VEGF; R&D Systems) according to the manufacturer's instructions.
Analysis of Cell Viability
Cell viability in mouse neural cells was determined by Cell Counting Kit-8 (CCK8, Dojindo Lab, Kumamoto, Japan) with the manufacturer's instructions. The primary neural cell cultures were prepared from B6SJL newborn mice (Jackson Lab) spinal cords by the procedure reported previously (24). Spinal cord cells were seeded in 96-well plates (BD Biosciences, San Jose, CA, USA) at a density of 5 × 103 cells/well and incubated for 24 h prior to experimental treatments. To assess the antiapoptotic effects of cytokines released by ASCs, apoptosis was induced by 200 μM H2O2 (Sigma) and treated with NGF (1.3 ng/ml), BDNF (1.29 ng/ml), NT3 (0.13 ng/ml), GDNF (0.45 ng/ml), IGF1 (3.43 ng/ml), HGF (0.97 ng/ml), VEGF (5.82 ng/ml), or the conditioned medium (CM) of ASCs. The dose of cytokines was equalized with the concentration in CM measured by ELISA. After 24 h of incubation in the presence of cytokines/growth factors, CCK8 substrate was added to each well, and the cells were incubated for 3 h at 37°C, and absorbance was measured at 450 nm using an absorbance microplate reader (Biotek Instruments, Winooski, VT, USA).
Tunel Staining
To evaluate the prevention effects of cytokines secreted by ASCs, primary mouse spinal cord cells were cultured on gelatin-coated 9-mm Aclar coverslips (Honeywell, Morristown, NJ, USA) and cell apoptosis was induced by 200 μM H2O2 with or without treatment of NGF, BDNF, NT3, GDNF, IGF1, HGF, VEGF, or CM of ASCs for 24 h. Apoptotic cell death was detected by TUNEL stain assay using the In Situ Cell Death Detection Kit with the manufacturer's instructions.
Cell Count
The total number of hMit-positive ASCs in spinal cord sections was determined by stereological estimation. Actual counting was performed in whole spinal cord, and the counting areas were divided into the gray matter and the white matter. In total, 30 sections taken from the serial sections with an equal distance (1 mm) from cervical to lumber levels of spinal cord were counted. An estimate of the total number of hMit-positive cells was calculated using the optical fractionator's formula (42).
Statistical Analysis
All values are expressed as means ± SEM. Statistical significance (p < 0.05 for all analyses) was determined by one-way ANOVA among experimental groups, and Student–Newman–Keuls analyses to determine survival. In survival graphs, Kaplan–Meier curve analysis was employed. SPSS (v12.0; IBM, Armonk, NY, USA) was used to calculate significance.
Results
Transplantation of ASCs Results in Improved Behavior, Disease Onset Delay, and Life Extension
To demonstrate therapeutic effects, human ASCs were transplanted into the lateral ventricle of SOD1G93A mutant mice at 70 postnatal days before the appearance of clinical symptoms. A significant delay in disease onset was observed in ALS mice transplanted with ASCs via ICV route compared with ASCs injected via the IV route or PBS (IV and ICV)-treated mice, as assessed by rotarod test (117, 102, and 91 days, respectively, p < 0.01) (average of results with three donors). There were 26 days of delay in clinical onset in ALS mice transplanted ASCs via the ICV route, and delay of 11 days in mice treated via the IV route. The disease onset delay found following ASC transplantation via ICV route was greater compared with the IV route or PBS (IV and ICV)-treated mice (Fig. 1, Tables 1 and 2). The disease onset delay assessed by PaGE was 96, 95, and 91 days, respectively (p < 0.05) for ASC-ICV, ASC-IV, and PBS-IV-ICV (Fig. 1, Tables 1 and 2), and by extension, reflex was 110, 95, and 84 days, respectively (p < 0.01).
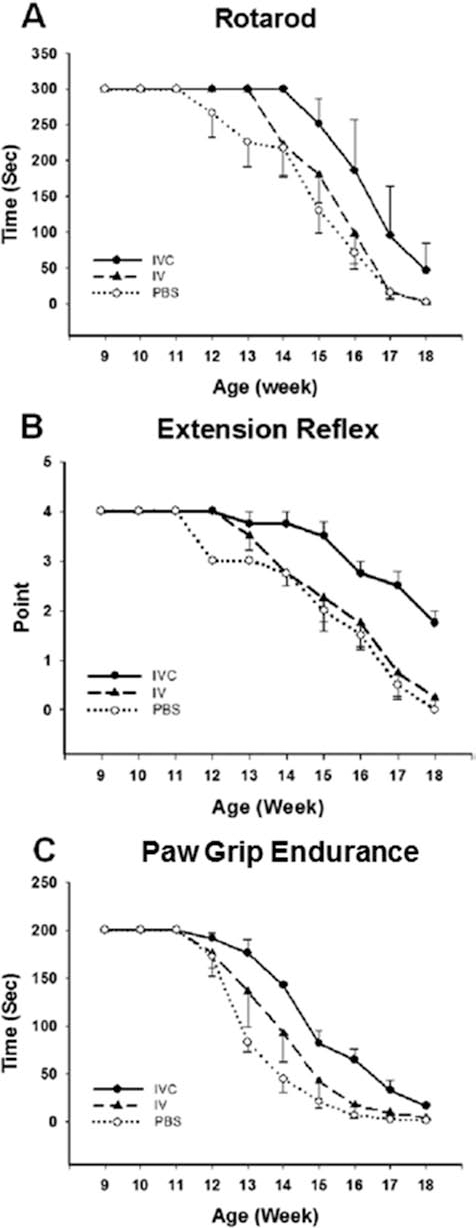
Transplantation of ASCs delays motor performance decline. The graphs display the cumulative results of the onset of motor deficits demonstrated by the rotarod test (A), the extension reflex (B), and paw grip endurance (C). Amyotrophic lateral sclerosis (ALS) mice transplanted with adipose tissuederived stem cells (ASCs) or phosphate-buffered saline (PBS) by IV or ICV injection showed a significant delay on motor performance decline.
The animals in the ICV group showed a significant improvement in life extension with a 24-day increase in average survival compared to the control group (survival of 148 days vs. 124 days; p < 0.05) (Fig. 2, Table 1). ASC transplantation in ALS mice via IV route showed a smaller, 9-day increase in average survival compared to the PBS-treated controls (130 days vs. 121 days) (Fig. 2, Table 2). Thus, it is clear that the transplantation of ASCs via the ICV route results in a superior outcome in disease onset delay and extension of life span in ALS mice compared to ASC injection via the IV route.

Transplantation of ASCs induces life extension in ALS mice. Substantially prolonged survival was revealed in mice treated with ASCs by ICV injection compared to the PBS-treated control (148 days vs. 124 days, in median survival). A smaller increase was observed, and mice injected ASCs by IV route also showed a prolonged survival (130 days vs. 121 days, in median survival).
Survival and Neuronal Differentiation of the Grafted ASCs
At 4 weeks posttransplantation, the number of surviving ASCs transplanted via the ICV route was determined in immunohistochemically stained ALS spinal cord sections using an anti-hMit-specific antibody. In whole spinal cord sections, 6.8% of total injected cells (13,600) was found, and 0.7% of total cells (1,400) was localized in the gray matter (Table 4). In order to confirm the differentiation of the grafted cells (a process crucial to the replacement of host neural cells), the cord sections were double stained by anti-hMit with anti-MAP2, NF, or GFAP antibody (Fig. 3). Only 1.1 (with MAP2), 0.8 (with NF), and 0 (with GFAP) cells per section were found to be double positive cells. hMit-positive cells were also found in the spinal cord of the IV group (Fig. 4). Only 0.25 cells per section of hMit/MAP2 double positive cells were found. There were no double positive cells for hMit with NF or GFAP in the spinal cord sections of IV group.

Immunofluorescence microscopy of the transplanted ASCs in the spinal cord of the ICV-injected group. Double immunofluorescence staining was performed with anti-human mitochondria (hMit) antibody and anti-microtubule-associated protein 2 (MAP2), neurofilament (NF), or glial fibrillary acidic protein (GFAP) antibodies. Although most hMit-positive cells were located in the white matter, a small number of cells was also located in the gray matter, and these cells were MAP2, NF, or GFAP negative. The hMit/GFAP double positive cells were not visible in the sections. Images of fluorescence microscopy were merged in column direction. The white box indicates the magnified area. Scale bars: 200 μm.

Immunofluorescence microscopy of the transplanted ASCs in the spinal cord of the IV-injected group. Double immunofluorescence staining was performed with anti-hMit antibody and anti-MAP2, NF, or GFAP antibody. Only a small number of hMit-positive cells was found in the spinal cord, and one hMit/MAP2-double positive cell is noted in the gray matter. However, there are no double positive cells for hMit/NF or hMit/GFAP in the sections. Images of fluorescence microscopy were merged in column direction. The white box indicates the magnified area. Scale bars: 200 μm.
Survival Ratio of Transplanted ASCs

hMit, human mitochondria.
Production and Secretion of Cytokines by ASCs
In the pathogenesis of neurodegenerative disease, neurotrophic growth factors that regulate neuronal proliferation and survival are involved in a particularly important role (7,35). Thus, to determine whether ASCs release cytokines/growth factors as survival factors to prevent apoptosis of host cells, gene expression of ASCs was screened by RT-PCR (Fig. 5). We selected NGF, BDNF, NT3, GDNF, CNTF, IGF1, HGF, and VEGF because the cell death protection effect of these cytokines was reported by previous studies (7,32). Transcripts of these cytokines were detected in ASCs by RT-PCR, and we measured the cytokine levels in vitro and in vivo by ELISA assay (Fig. 5). In a conditioned medium of ASCs (106 cells in culture for 24 h), 1.30 ng/ml NGF, 1.29 ng/ml BDNF, 0.13 ng/ml NT3, 0.45 ng/ml GDNF, 3.43 ng/ml IGF1, 0.98 ng/ml HGF, and 5.82 ng/ml VEGF were detected. Although the transcript of CNTF was detected in ASCs by RT-PCR, the level of CNTF in CM was below the detectable level by ELISA.

RT-PCR and ELISA analyses of cytokine/growth factors in ASCs. (A) The transcripts of nerve growth factor (NGF), brain-derived growth factor (BDNF), neurotrophin 3 (NT3), glial cell line-derived neurotrophic factor (GDNF), insulin-like growth factor 1 (IGF1), ciliary neurotrophic factor (CNTF), hepatocyte growth factor (HGF), and vascular endothelial growth factor (VEGF) were detected in ASCs by RT-PCR. M indicates the size marker (100 bp ladder presents 1,000 bp to 100 bp). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as internal standard. (B) Enzyme-linked immunosorbent assay (ELISA) demonstrates that ASCs secrete NGF, BDNF, NT3, GDNF, IGF1, HGF, and VEGF (but not CNTF) into the culture medium. Concentration of each growth factor is indicated as a number on the bar (ng/106 cells/day). N.D., not detectable. *p < 0.05 compared with the control. (C) Expression levels of NGF, BDNF, NT3, GDNF, IGF1, CNTF, HGF, and VEGF in spinal cord of the ASC transplanted mouse were also determined by ELISA assay. Concentration of each factor is indicated as a number on the bar (ng/g tissue). *p < 0.05 compared with PBS-injected mouse spinal cord tissue extracts.
In homogenates of 10 mg of spinal cord samples removed from the ICV group mice, levels of cytokines/growth factors were 41.95 pg/g of NGF, 35.56 pg/g of BDNF, 5.6 pg/g of NT3, 29.45 pg/g of GDNF, 92.67 pg/g of IGF1, 3.6 pg/g CNTF, 2.33 pg/g of HGF, and 120.38 pg/g of VEGF by ELISA assay.
Neuroprotective Effect of Cytokines Produced by ASCs
Apoptosis of primary spinal cord cells was induced by H2O2, and then spinal cord cells were treated with cytokines/growth factors at the levels found in ASC-conditioned medium (CM) (Fig. 6). Neuroprotective effects of cytokines/growth factors and CM were determined via cell viability assay and TUNEL stain. Treatment with CM that contains all of the cytokines showed the best protective effect, suggesting that the cytokines in CM induced neuroprotection of ASCs synergistically. Neuroprotection of host spinal cord cells mediated by grafted ASCs was also confirmed in the spinal cord of ALS mice (Fig. 7). In the ICV group, a marked reduction of TUNEL-positive cells (60 ± 2 cells per section) was found compared to the PBS control group (601 ± 71 cells per section) or the IV-injected group (460 ± 11 cells per section) at 4 weeks post-transplantation. These results suggest that the multiple ASC-derived cytokines/growth factors are effective neuro-protective agents to block apoptotic cell death of motor neurons and other neural elements in the ALS mouse spinal cord.
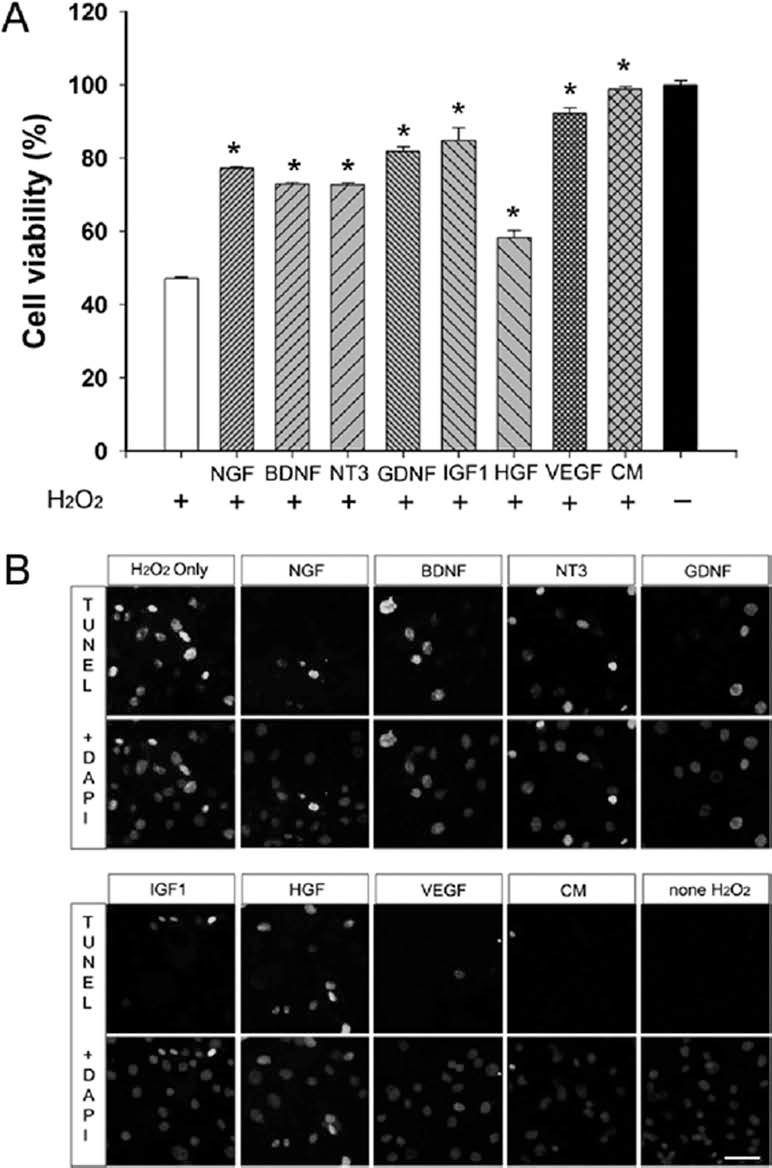
Cytokines detected in ASCs induce increased survival of primary mouse spinal cord cells exposed to H2O2. The spinal cord cells were exposed to 200 μM H2O2 and were treated with NGF, BDNF, NT3, GDNF, IGF1, HGF, VEGF, or ASC-conditioned medium (CM). Cell viability was evaluated by Cell Counting Kit-8 (CCK8) (A). Although all cytokines significantly increased the survival of mouse spinal cord cells (*p < 0.05 compared with the negative control, white bar), IGF1 and VEGF treatments were found to be most potent. Treatment with CM that contains all the cytokines induced the highest cell viability. Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining also illustrates that cytokines neuroprotect mouse spinal cord cells and that IGF1, VEGF, and CM significantly reduce the number of TUNEL-positive cells (B). Scale bar: 20 μm.

Transplantation of ASCs reduces neuronal cell death in the spinal cord of ALS mouse. Cell death in the ventral horn of the spinal cord was evaluated by TUNEL staining. A considerable reduction of TUNEL-positive cells is demonstrated in the mice transplanted with ASCs via the ICV route compared to the IV-injected group or PBS-treated control group. The white box indicates the magnified area. Scale bar: 200 μm.
Discussion
In this study, transplantation of human ASCs via the ICV route significantly delayed disease onset (26 days) and prolonged the survival of mouse SOD1G93A ALS model (24 days). Our results suggest that this treatment modality of ICV transplantation of human ASCs might be of value in the treatment of ALS patients without significant adverse effects.
At 4 weeks posttransplantation, the number of surviving ASCs transplanted via the ICV route was determined to be 6.8% of total injected cells in total spinal cord. This figure does not seem to reflect a real cell number of surviving cells in the spinal cord. Current delivery protocols using direct needle injection result in poor cell viability. In a previous study, it was demonstrated that during the actual cell injection process, mechanical membrane disruption resulted in 59% acute cell loss (1). With this figure in mind, better than 10% of total cells could have survived in spinal cord. Among the ASCs found in the spinal cord, only a small number of ASCs was found to differentiate into MAP2-positive or NF-positive neurons and none into GFAP-positive astrocytes. Cell count in the spinal cord sections indicates that a considerable number of ASCs transplanted via the ICV route migrated down to the spinal cord but did not appreciatively differentiate into neurons or astrocytes. It appears that the ASCs selectively migrated to the spinal cord but did not replace lost neurons or glia. Rather, ASCs appeared to serve as providers of growth factors such as VEGF and IGF1, which exhibit protection from cell death of host spinal cord neurons.
In this study, we used hydrogen peroxide (H2O2) to address the antiapoptotic effects of ASC-conditioned medium (CM) and cytokines/growth factors released by ASCs, reflecting on earlier reports indicating the presence of high levels of H2O2 in ALS mouse spinal cords (28) and apoptotic cell death of motor neurons in vitro by free radical treatment (17). Our in vitro study shows that addition of the CM from ASC culture and cytokines/growth factors produced from ASCs effectively block apoptotic cell death of spinal cord cells caused by H2O2 (Fig. 6). Our in vivo TUNEL assay also shows a significant decrease in neuronal cell death in the ASC-transplanted ALS mouse spinal cord (Fig. 7). Among the cytokines/growth factors examined, IGF-1 and VEGF are proven to be the major trophic factors to block neuronal cell death found in CM of ASCs (Figs. 5 and 6). Our results are in agreement with previous studies that the administration of VEGF or IGF1 in ALS mice provided improved motor function and extended survival in ALS mice (16,18). Production and secretion of multiple angiogenic and antiapoptotic cytokines by ASCs were reported earlier (35).
Previous studies have presented successful delivery of NSCs using an IV injection route in animal models of neurological diseases including ALS (3,26,31), we tested both IV and ICV injection of ASCs in ALS mice. Unfortunately, behavioral tests showed poorer motor performance of the IV group compared to the ICV group. These results suggest that additional protocols such as neuronal induction of ASCs prior to transplantation, treatment with drugs that promote increased survival of engrafted cells, or titration of cell number and injection frequency before the ASC transplantation via IV injection route is widely adopted.
In conclusion, we demonstrated in this study that the transplantation of ASCs resulted in delayed disease onset and the extended life of ALS mice and that the ASCs were capable of secreting multiple cytokines/growth factors (NGF, BDNF, NT3, GDNF, IGF1, HGF, and VEGF) that provide neuroprotection of host spinal cord neurons and behavioral improvement.
Footnotes
Acknowledgments
This study was supported by the Korea Healthcare Technology R&D Project (A12021412010000100, H.J.L.) and Canadian Myelin Research Initiative (S.U.K.). The authors declare no conflicts of interest.
