Abstract
Regulatory T (Treg) cells have anti-inflammatory functions and heighten immune tolerance. The proportion and functions of Treg cells are perturbed in rheumatoid arthritis (RA), contributing to the excessive immune activation associated with this disease. We therefore hypothesized that supplementation with foreign Treg cells could be used to treat RA. To investigate the therapeutic effects of exogenous Treg cells on RA and its mechanism, we used human Treg cells to treat collagen-induced arthritis (CIA) in a rat model to observe whether exogenous Treg cells can treat the disease across species. Successful treatment would indicate that Treg cell transplantation in humans is more likely to affect RA. In the present study, human Treg cells were collected from healthy human peripheral blood and culture-expanded in vitro. Induced human Treg cells were injected into CIA rats via the tail vein. The rats’ lymphocyte subtypes, cytokines, and Th1/Th2 ratios were measured using flow cytometry. In the rats, following injection of the human Treg cells, the severity of CIA was significantly reduced (P < 0.01), the proportion of endogenous Treg cells increased in the peripheral blood and spleen (P = 0.007 and P < 0.01, respectively), and the proportion of B cells decreased (P = 0.031). The IL-5 level, IL-6 level, and Th1/Th2 ratio in the peripheral blood were decreased (P = 0.013, 0.009, and 0.012, respectively). The culture-expanded human Treg cells were also cultured with synovial fibroblast cells from RA patients (RASFs). After coculture with Treg cells, RASFs showed reduced proliferation (P < 0.01) and increased apoptosis (P = 0.037). These results suggest that exogenous and induced Treg cells can produce a therapeutic effect in RA and CIA by increasing endogenous Treg cells and RASF apoptosis and reducing B cells, the Th1/Th2 ratio, and secretion levels of IL-5 and IL-6. Treg cell transplantation could serve as a therapy for RA that does not cause immune rejection.
Introduction
The main manifestation of rheumatoid arthritis (RA) is chronic, systemic joint inflammation. Abnormal proliferation of a large number of RA synovial fibroblast (RASF)-like cells, infiltration of inflammatory cells, abnormal secretion of inflammatory cytokines, thickening of the synovial membrane, and destruction of articular cartilage and bone are observed in the diseased joints 1 . Currently, the pathogenesis of RA is not entirely clear.
Dysregulation of the naïve T cell (Tn) population is an important characteristics of RA pathogenesis 2 . Due to the influence of cytokines, Tn cells differentiate into a variety of subsets with different functions. Interleukin (IL)-2 and a high concentration of tumor growth factor beta (TGF-β) induce Tn cells to differentiate into CD4+CD25+ regulatory T (Treg) cells 3 , which secrete the anti-inflammatory cytokines IL-10 and TGF-β to inhibit inflammatory responses 4 . Treg cells interfere with the overactivation of T cells through direct contact with effector T cells 5 . Moreover, Treg cells inhibit effector T cell proliferation by competing with them for IL-2 binding, and together with IL-2, TGF-β enhances Treg cell polarization 6 . A decrease in the proportion of Treg cells results in a decrease in these functions and can lead to unrestrained autoimmune responses. Previous studies have shown that the number of Treg cells in RA patients is negatively correlated with the Disease Activity Score 28, the C-reactive protein level, and the erythrocyte sedimentation rate 7 . Furthermore, RA patients have fewer Treg cells, and the immune suppressive function of these cells is defective 8 . The number of Treg cells in the peripheral blood of RA patients is significantly decreased, and their immunosuppressive function is lower than normal; these two features are especially important during the active stage of RA disease. After antirheumatic treatment of RA patients, the number of Treg cells in the peripheral blood increases significantly 9,10 . We recently found that Xuebijing, a traditional Chinese medicine injection, can treat RA and CIA and achieve good results by elevating endogenous Treg cell proportions 11 . The proportion of Treg cells is also robustly decreased in collagen-induced arthritis (CIA) rats 12 . The inflammation associated with CIA is aggravated in CD4+CD25+ Treg cell–deficient rats 13 . The injection of bone marrow–derived inhibitory cells increases the in vivo expression of Foxp3 and restores the Treg cell population to normal levels in CIA rats, resulting in a reduction in the severity of arthritis 14 . In mice, depletion of Treg cells results in the onset of a variety of autoimmune diseases, including arthritis, while replenishment of Treg cells alleviates arthritic symptoms 15 . Treg cells maintain homeostasis of the immune system, limit the magnitude of effector responses, and allow for the establishment of immunological tolerance 16 . In the context of RA pathogenesis, Treg cells play a suppressive role, and artificially increasing the number of Treg cells could restore immunosuppressive function and might therefore be a potential therapy for RA.
There are two major Treg cell subsets: natural CD4+ Treg cells, which develop in the thymus; and induced Treg cells, which develop from conventional CD4+ T cells in the periphery. Induced Treg cell therapy has great potential to treat autoimmune disease. Dall’Era et al. reported the first case of a patient with systemic lupus erythematosus treated with induced Treg cell therapy 17 . In this study, Treg cells were induced to amplify from healthy human peripheral blood in vitro. The induced human Treg cells and RASFs were cocultured so that the effects of Treg cells on RASFs could be observed. The culture-expanded human Treg cells were also injected into CIA rats via the tail vein to assess the therapeutic effects of exogenous human Treg cells on these rats. Once the cell transplantation across species was successful, it indicated that injection of induced Treg cell therapy in human was more likely to obtain therapeutic effect within species. The purpose of this study was to explore the therapeutic effects of foreign Treg cells on RA through in vitro cytological experiments and in vivo animal experiments.
Materials and Methods
Tissue Collection
Human synovial tissue samples were collected from RA patients (n = 7, four females, 30–68 years old, mean age of 54) during knee joint arthroscopic synovectomy procedures. The diagnosis was made according to the revised criteria of the American College of Rheumatology. The RA patients were medicated with nonsteroidal anti-inflammatory drugs to help reduce pain and swelling in the joints and to decrease stiffness. These patients had received treatment with low-dose corticosteroids and disease-modifying antirheumatic drugs, but they were not treated with any immunobiological treatment, such as anti-TNF or another target-specific treatment. Patients were enrolled between June 2018 and October 2019 at The Affiliated Hospital of Qingdao University. All of the patients signed written informed consent statements for participation in the study. The study protocol was approved by the Ethical Committee of the Affiliated Hospital of Qingdao University (approval number: 20190306), Qingdao, China.
Induced Amplification of Treg Cells
Healthy human (n = 9) peripheral blood was aseptically collected. All of the donors (19–32 years old, mean age of 24) provided written informed consent to participate in the study. Peripheral blood mononuclear cells (PBMCs) were isolated using density gradient centrifugation and were then plated in a culture flask coated with 5 µg/ml anti-human-CD3 antibody (Sungene, Shanghai, China). Treg cell differentiation and amplification were induced in Dulbecco’s modified Eagle’s medium (DMEM; HyClone, Logan, UT, USA) containing TGF-β (PeproTech, Rocky Hill, NJ, USA) (5 ng/ml), recombinant human IL-2 (T&L, Beijing, China) (2,000 IU/ml), anti-human CD28 antibody (Sungene) (100 ng/ml), and 10% fetal bovine serum (FBS; Gibco, Grand Island, NY, USA). Fresh growth medium was added every 2 days, and the first round of amplification was completed after 6 days of culture. The cells were transferred to a culture flask without the antibody and further cultured for 2 days in DMEM containing recombinant human IL-2 (2,000 IU/ml) and 10% FBS. After 2 days, the cells were transferred to a culture flask coated with anti-human CD3 antibody (5 µg/ml) and were amplified following the same method used for the first amplification cycle. The entire process required 14 days.
Treg cell suspension (1 × 105) samples with a volume of 100 µl each were collected after 14 days of amplification, and anti-human CD25-fluorescein isothiocyanate (FITC; BioLegend, San Diego, CA, USA) (2 µl per 1 × 105 cells in a 100-µl volume) and anti-human CD4-PerCP (2 µl per 1 × 105 cells in a 100-µl volume) (BioLegend) antibodies were added. The samples were then incubated in the dark for 30 min at 4°C. The cells were washed twice with phosphate-buffered saline (PBS) and then resuspended in PBS to form single-cell suspensions. Flow cytometry (ACEA Biosciences, San Diego, CA, USA) was used for detection. This method was designed based on Schmidt’s study 18 .
Primary Culture of RASFs
RA synovial tissue samples were aseptically disrupted; next, DMEM containing 4% type II collagenase (Solarbio, Beijing, China) was added, and the samples were incubated in an atmosphere of 5% CO2 at 37°C for 4 h until the tissue pieces were dispersed into a cell suspension. The cell suspension was filtered through a 70-µm cell strainer and then centrifuged at 1,200 rpm for 5 min. The cells were resuspended in DMEM containing 10% FBS and incubated at 37°C in a 5% CO2 incubator to obtain primary RASFs. Following three to eight passages, the RASF was preserved, and other cells, such as T cells and B cells, in the synovial membranes were removed. Cells passaged for three to eight generations were used in the subsequent experiments. The cell number was calculated using a cell counter. This method has been used in many studies 19,20 .
Assessment of RASF Amplification via the Cell Counting Kit-8 Assay
A single-cell suspension of RASFs (1 × 104) was seeded into 96-well plates. The induced mature Treg cells (1 × 104) were washed with PBS and seeded into 96-well plates at a 1:1 ratio of RASFs to Treg cells. These cells were cocultured in DMEM containing 10% FBS for 72 h. At the end of the coculturing period, the suspended Treg cells were washed away with PBS, and Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan) solution (10 μl) was added to the plates, which were then incubated for 3 h at room temperature. Independent cultures of RASFs without Treg cells under the same culture condition were used as controls. The OD450 values were measured using a microplate reader (BioTek, Winooski, VT, USA). The data were statistically analyzed using an independent samples t-test. This experiment was repeated three times.
Assessment of RASF Apoptosis Using Annexin V-Propidium Iodide/FITC
A single-cell suspension of RASFs (1 × 107) was seeded into six-well plates. Induced mature Treg cells (1 × 107) were seeded into six-well plates at a 1:1 ratio of Treg cells to RASFs, and these cells were cocultured for 48 h. At the end of the coculturing period, the suspended Treg cells were washed away with PBS, and a single-cell suspension of RASFs was collected and resuspended in binding buffer. FITC-conjugated antibody (50 μg/ml) and propidium iodide (PI)-conjugated antibody (BioLegend) (50 μg/ml) were added, and the samples were then mixed well and incubated at room temperature for 15 min in the dark. RASFs that were independently cultivated without Treg cells under the same culture conditions were used as controls. Apoptosis was assessed by flow cytometry. The data were statistically analyzed using an independent samples t-test. This experiment was repeated three times.
Assessment of Cytokine Levels in the Coculture Using Flow Cytometry
A single-cell suspension of RASFs was seeded into six-well plates. Induced mature Treg cells were seeded into six-well plates at a 1:1 ratio of Treg cells to RASFs, and the cells were cocultured for 48 h. After the coculturing period, the supernatant was collected, centrifuged at 1,500 rpm for 10 min, and assayed for changes in cytokine levels using a human Th1/Th2 cytokine assay kit (Cell-Genebio, Hangzhou, China). The data were statistically analyzed using one-way analysis of variance (ANOVA).
Injection of Treg Cells into CIA Rats
A total of 30 male rats (6 weeks old) were purchased from Shandong Laboratory Animal Center (Jinan, China) and randomly divided into a normal control (NC) group, a CIA group, and a Treg treatment group (n = 10 rats per group). The CIA rat model was established in all of the groups except for the NC group. Bovine type II collagen (Chondrex, Redmond, WA, USA) (2 mg/ml) was mixed with complete Freund’s adjuvant (5 ml) (Sigma, St. Louis, MO, USA) in equal amounts, and the mixture was fully emulsified. The initial immunization with 0.2 ml of emulsion per rat was performed by intracutaneous injection into the root of the tail. One week later, bovine type II collagen (2 mg/ml) was mixed with incomplete Freund’s adjuvant (Sigma) (5 ml) in equal amounts, and the mixture was fully emulsified. A booster immunization was performed via intracutaneous injection of 0.2 ml of emulsion per rat into the root of the tail. The rats in the Treg treatment group were injected with the amplified human Treg cells (107 Treg cells/kg) via the tail vein simultaneously with the booster immunization, and the cell injection was repeated again 1 week later. The NC and CIA groups were injected with the same volume of PBS at the same time points. All mice were humanely euthanized with a lethal dose of ketamine and xylazine. This method was designed based on Kelchtermans et al. 21
The breeding and operation of the experimental animals were carried out in accordance with the Helsinki Convention on Animal Protection and the Regulations of the People’s Republic of China on the Administration of Experimental Animals. The study protocol was approved by the Medical Ethical Committee of The Affiliated Hospital of Qingdao University (Approval number: 20190306).
Evaluation of the General CIA Conditions
After the start of treatment, the degree of ankle joint swelling was measured with a Vernier caliper, and the joints were photographed every 3 days. At the end of the experiment, an inflammation curve was plotted based on the degree of joint swelling over time. The data were statistically analyzed by repeated-measures ANOVA.
Histopathological Examinations
The experiment was terminated on the 20th day after the booster immunization or the cell injection. Tissue samples located 0.5 cm around the knee joint were collected, fixed in 4% paraformaldehyde (Solarbio) for 48 h, decalcified with ethylenediaminetetraacetic acid, and embedded in paraffin. Pathological changes in the joint tissue samples were observed under light microscopy after hematoxylin–eosin (HE) staining.
Assessment of the Lymphocyte Subsets in the Peripheral Blood and Spleen of Rats
The experiment was terminated on the 20th day after treatment. The animals were anesthetized via intraperitoneal injection of 30 mg/kg 3% sodium pentobarbital. For sample collection, each rat was fixed on its back; the abdominal cavity was then opened, and a blood sample was collected from the inferior vena cava. The spleen was removed and placed in 2 ml PBS, where it was then cut into pieces and filtered through a strainer to obtain a single-cell suspension. Samples of both the peripheral blood and the spleen single-cell suspensions were collected after treatment using red blood cell lysis buffer (Solarbio). Anti-rat CD3-allophycocyanin (APC, BioLegend) and anti-rat CD161-FITC (BioLegend) were used for the detection of natural killer (NK) cells, and anti-rat CD3-APC (BioLegend) and anti-rat CD45RA-PE (BioLegend) were used for the detection of B cells. The samples were incubated at 4°C for 30 min in the dark, washed twice with 500 µl PBS, and examined after resuspending in PBS. The data were statistically analyzed using one-way ANOVA.
Detection of Rat Treg Cells
APC anti-rat CD4 and FITC anti-rat CD25 antibodies (BioLegend) or APC IgG2a and FITC IgG2a isotype control (BioLegend) were added to the blood samples with anticoagulation treatment (1 µg per 106 cells in 100 µl volume). The mixture was incubated at room temperature for 15 min in the dark. Then, 1× red blood cell lysis/fixation solution (BioLegend) was added, and the mixture was incubated at room temperature for 15 min. After centrifugation, the cell staining buffer (BioLegend) was added to resuspend the pellet, and the mixture was centrifuged at 300×g for 5 min. The cell pellet was then fixed with fixation buffer (BioLegend) and incubated for 20 min at room temperature. Following centrifugation, the fixed cells were resuspended in permeabilization wash buffer (BioLegend), and Perkin-Elmer (PE, CA, USA) anti-rat FOXP3 antibody or PE IgG2a isotype control (BioLegend) was added (1 µg per 106 cells in 100 µl volume). The mixture was incubated at room temperature for at least 30 min. The mixture was centrifuged and resuspended in 1× flow cytometry staining buffer. CD4+ CD25+ FOXP3+ Treg cells were detected by flow cytometry (ACEA Biosciences, NovoCyte D2040 R), and the data were analyzed using FlowJo software BD (Franklin Lake, NJ, USA)).
Assessment of Cytokine Concentrations in Rats
The obtained blood samples were coagulated at room temperature for at least 30 min and then centrifuged at 3,000 rpm for 20 min at 4°C to obtain serum. Changes in cytokine expression levels in the peripheral blood were detected by flow cytometry using a rat Th1/Th2 cytokine assay kit (BioLegend). Th1 cells are characterized by interferon gamma (IFN-γ) secretion, and Th2 cells mainly secrete IL-4. The Th1/Th2 ratio was calculated based on the average fluorescence intensities detected for IFN-γ and IL-4. The data were statistically analyzed using one-way ANOVA.
Statistical Analysis
Normal and variance homogeneity tests were performed using SPSS 17.0 software (Chicago, IL, USA), and data that met the test criteria are represented as x ± s. Independent and paired sample t-tests were used to determine significant differences between two groups. One-way ANOVA was used to determine significant differences between multiple groups. The least significant difference method or the Tamhane method was used for pairwise comparisons. P < 0.05 was considered statistically significant.
Results
Amplification of Human Treg Cells
Fourteen days after induction of PBMC differentiation with anti-human CD3 and anti-human CD28 antibodies, IL-2 and TGF-β, the culture-expanded Treg cell phenotypes were examined by flow cytometry. The percentage of CD4+CD25+ Treg cells in the cultures increased from 6.85% (unamplified) to 74.33% after amplification, indicating successful amplification of Treg cells (Supplemental Fig. 1).
Effects of the Induced Treg Cells on RASFs
The induced Treg cells were cocultured with RASFs for 24, 48, and 72 h and then washed with PBS to remove the suspended Treg cells. The proliferation of the adherent RASFs was measured using CCK-8 assays. Compared with the results obtained for the controls (in which the RASFs were cultured alone) group, RASF proliferation was significantly reduced after 24 and 48 h of coculturing (1.17 ± 0.15 versus 0.83 ± 0.14, P < 0.01; and 1.30 ± 0.47 versus 0.10 ± 0.12, P < 0.01, respectively), suggesting that the induced Treg cells inhibit RASF amplification (Fig. 1A).

Detection of RASF activities following coculture with induced Treg cells. (A) RASF cell proliferation using the CCK-8 assay. (B) Apoptosis of RASFs using annexin V-PI/FITC double staining. (C) Statistical analysis of the apoptotic percentage. NC indicates RASF culture alone, and RASF + Treg indicates cell coculture. *P < 0.05 and ***P < 0.001. CCK-8: Cell Counting Kit-8; FITC: fluorescein isothiocyanate; PI: propidium iodide; RASF: RA synovial fibroblast; Treg: regulatory T.
The induced Treg cells were cocultured with RASFs for 48 h, and the apoptosis of the RASFs was measured using flow cytometry. Compared with the rate of apoptosis in the control group (in which the RASFs were cultured alone), the apoptosis rate of the RASFs cocultured with the culture-expanded Treg cells was significantly increased (1.73 ± 1.21 versus 10.31 ± 4.69, P = 0.035), suggesting that coculture with Treg cells promotes RASF apoptosis (Fig. 1B, C).
Effects of the Induced Treg Cell Treatment on CIA
Rats were treated with the induced Treg cells simultaneously with the second booster immunization. One week later, joint swelling was obvious in the rats in the CIA group compared with those in the healthy rats, which were not injected with collagen II. In contrast, the joint swelling in the rats injected with the human Treg cells was mild (Fig. 2A). HE staining showed that the joint structure of the rats in the healthy group was intact, the joint cavity was clean, the articular cartilage on both sides was even and smooth, and the synovial membrane showed no obvious hyperplasia. The CIA rats showed detached necrotic tissue masses in their joint cavities. Furthermore, local calcification basophilic enhancement was accompanied by infiltration of many inflammatory cells, consisting mainly of neutrophils and a small number of pus cells. The articular cartilage and bone tissue were absent locally, and a single layer of inflammatory cells was attached to the surface of the cartilage. There was clear synovial hyperplasia, along with an increase in the number of capillaries, which was accompanied by obvious vasospasm. In CIA rats injected with the human-induced Treg cells, histochemical staining showed few cartilage changes, synovial hyperplasia, and a small amount of inflammatory cell infiltration (Fig. 2B). The inflammation curve showed that the degree of joint swelling in the CIA rats injected with human Treg cells was significantly lower than that in the rats without cell injection (F = 5.023, P < 0.01, Fig. 2C).

The effects of human-induced Treg cells on CIA rats. (A) Degree of rat joint swelling. (B) Hematoxylin–eosin staining of rat joint tissues. (C) Inflammation curve. NC indicates the rats were treated with PBS alone. ***P < 0.001. CIA: collagen-induced arthritis; PBS: phosphate-buffered saline; Treg: regulatory T.
The peripheral blood and spleen lymphocyte subsets of the rat models were examined by flow cytometry following the induced human Treg cell treatment. The proportions of endogenous rat Treg cells in peripheral blood and spleen of the animals were significantly lower in the CIA group than in the healthy group (P = 0.020; P < 0.01). After treatment with the induced human Treg cells, the proportion of the endogenous rat Treg cells increased significantly in the injected CIA rats compared with the uninjected animals (P = 0.007; P < 0.01). The proportions of NK cells in the peripheral blood and spleens of the animals in the CIA group were higher than those of the animals in the healthy group (P = 0.012; P = 0.017), and the proportions of NK cells in the animals in the Treg cell treatment group were even higher (P = 0.003; P = 0.001). However, there was no significant difference between the animals in the CIA group and those in the Treg cell injection group. The proportion of peripheral blood B cells in the animals in the CIA group was significantly greater than that in the animals in the healthy group (P = 0.004). Moreover, the proportion of B cells in the animals in the Treg cell injection group was lower than that in the animals in the CIA group; however, the difference was not statistically significant. The proportion of B cells in the spleen was the highest in the animals in the CIA group, and the difference was statistically significant compared with the B cell proportions in the healthy and Treg treatment groups (P = 0.036; P = 0.031, Fig. 3).

The effects of human-induced Treg cells on lymphocyte subsets in CIA rats. (A) Peripheral blood lymphocyte subsets. (B) Lymphocyte subsets in the spleen. NC indicates the rats that were treated with PBS alone. *P < 0.05, **P < 0.01, and ***P < 0.001. CIA: collagen-induced arthritis; PBS: phosphate-buffered saline; Treg: regulatory T.
Flow cytometry was used to measure cytokine levels in the peripheral blood of the rats treated with Treg cells. The levels of TNF-α, IFN-γ, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-4, IL-5, IL-6, IL-10, and IL-13 were higher in the peripheral blood of the rats in the CIA group compared with the levels detected in the animals in the healthy control group (P = 0.007, 0.019, 0.016, 0.043, 0.002, 0.002, 0.002, and 0.030, respectively). The peripheral blood levels of IL-5, IL-6, and IL-10 in the Treg cell injection group were lower than the levels detected in the animals in the CIA group (P = 0.013, 0.009, and 0.006, respectively). There were no statistically significant differences between the levels of these cytokines in the animals in the Treg treatment group and those in the healthy controls, suggesting that IL-5, IL-6, and IL-10 had recovered to normal levels (Fig. 4A). We also analyzed the Th1/Th2 ratio based on IFN-γ and IL-4 levels. The Th1/Th2 ratio in the animals in the CIA group was higher than that in the animals in the healthy group. After Treg cell treatment, the Th1/Th2 ratio of the treated animals was significantly lower than that of the animals in the CIA group (P = 0.012), and there was no significant difference in the Th1/Th2 ratio compared with the healthy controls (Fig. 4B).
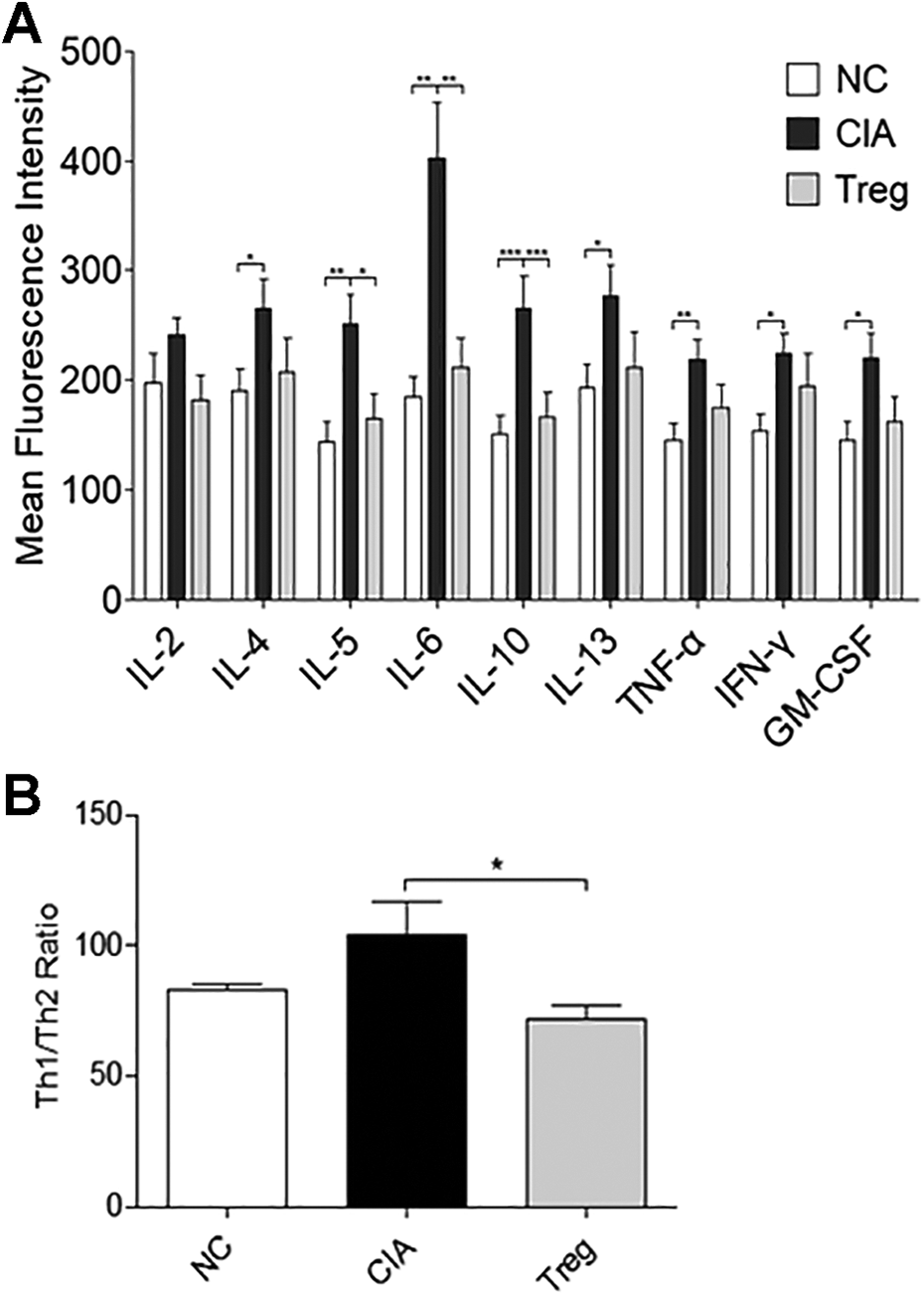
The effects of human-induced Treg cells on cytokine levels in the peripheral blood of CIA rats. (A) Cytokine levels in the peripheral blood of the rat models. (B) Th1/Th2 ratio in the peripheral blood of the rats. NC indicates the rats that were treated with PBS alone. *P < 0.05, **P < 0.01, and ***P < 0.001. CIA: collagen-induced arthritis.
Discussion
In the present study, CIA rats were injected with induced human Treg cells obtained by in vitro amplification from healthy volunteers. Both the inflammatory curve and histochemical results demonstrated significant alleviation of arthritis in the CIA rats following the injection, indicating successful treatment with exogenous cell therapy. The animal experiment also revealed that the proportions of endogenous Treg cells in the peripheral blood and spleen were lower in CIA rats than in normal rats. Furthermore, the number of rat Treg cells was much higher in the animals in the Treg cell treatment group than in the animals in the CIA group. Treg cells can induce the transformation of CD4+ T cells into Treg cells 6,22 . Therefore, we suggest that exogenous human Treg cells could stimulate rat models to produce additional Treg cells.
In addition to antibody production, B cells promote the activation and proliferation of T cells, stimulate cytokine production, participate in cell–cell interactions, and promote RA pathogenesis 23 . Our results showed that there were fewer B lymphocytes in the spleens of animals in the Treg cell–treated group than in the spleens of animals in the CIA group, suggesting that exogenous human Treg cells may also play a therapeutic role by reducing B cell levels. Other studies have reported that coculture of Treg cells with B cells inhibits B cell activation and proliferation 24 , which was consistent with our results.
RA pathogenesis is closely related to immune system disorders. Disruption of the Th1/Th2 cell ratio can play an important role in the development of RA 25 . Th1 cells secrete cytokines such as IFN-γ, IL-2, TNF-α, and GM-CSF to mediate cellular immunity and are mainly characterized by IFN-γ secretion. Th2 cells secrete IL-4, IL-5, IL-6, IL-10, IL-13, and other cytokines to mediate fluid immunity and are mainly characterized by IL-4 secretion 26 . Th1 cells are typically elevated in RA 27 . Both clinical studies and animal experiments support the idea that a decrease in the Th1/Th2 ratio can favorably contribute to RA treatment 28 . In the present study, the Th1/Th2 ratio was higher in CIA rats compared to the animals in the healthy control group, indicating a relatively low abundance of Th2 cells, in accordance with the findings of others. After the injection of exogenous human Treg cells, the Th1/Th2 ratio significantly decreased and approached that of the animals in the healthy control group. This result indicates that Treg cell injection can exert a therapeutic effect against CIA by restoring a normal Th1/Th2 balance, although the mechanism was not clear. Naïve CD4+ T cells differentiate into subsets of effector cells, including Th1, Th2, and Th17 cells. Regulation of Th2 cells by Tregs and vice versa were exerted by the interplay between the IL-4-activated STAT6/GATA3 pathway and Foxp3 29 . Tumor cells overexpressing programmed cell death-ligand 1 converted Th1 cells into FOXP3+ Treg cells in vivo in a human-into-mouse xenogeneic graft-versus-host disease model 30 .
In present study, the level of IL-6 was significantly decreased after cell injection, although IL-6 levels in the peripheral blood were higher in the CIA rats than in the healthy animals. IL-6 is an important inflammatory factor in RA that is significantly elevated in the serum of RA patients, and it can damage inflamed joints 31 . Treg cells can inhibit macrophage production of TNF-α and IL-6 inflammatory cytokines, directly contact dendritic cells, and inhibit the expression of MHC II molecules to reduce their antigen presentation abilities 32 –34 . The decreased IL-6 level observed in the animal model following the human Treg cell injection further demonstrated that exogenous human Treg cells have therapeutic effects on CIA.
The results of this study showed that IL-5 and IL-10 levels were elevated in the peripheral blood of CIA rats and significantly reduced in the animals in the Treg cell treatment group. A clinical dynamic monitoring study revealed a significant increase in the serum IL-5 level in patients during the progression from joint pain to RA, which was absent in patients who did not develop RA 35 . Injection of anti-IL-5 antibody into heart-transplanted mice can reduce collagen deposition and eosinophil infiltration in the graft and prevent a rejection response 36 . The present study showed that the IL-5 level was significantly lower following Treg cell therapy, which corresponded to the findings of others. Although the IL-10 level was lower after Treg cell therapy, the Th1/Th2 ratio was simultaneously decreased, and the CIA disease symptoms were robustly alleviated.
Abnormal RASF proliferation and apoptosis are important pathological features of RA 1,37 . In this study, we extracted RASFs from the synovial tissue of RA patients. The RASFs were collected following three to eight passages. The RASFs were preserved, and other cells, such as infiltrating T cells and B cells, were removed following multiple passages. Many studies have used this protocol 19,20 . RASFs were cocultured with human Treg cells derived from in vitro amplification, and the induced Treg cells inhibited RASF proliferation and promoted apoptosis, suggesting that exogenous Treg cells inhibit the excessive activation of RASFs in RA pathogenesis. In particular, we detected significant inhibition of RASF proliferation following 48 h of coculture with exogenous human Treg cells. To date, extremely few reports have addressed Treg cell–induced inhibition of RASF proliferation. Treg cells secrete anti-inflammatory cytokines such as IL-4, IL-10, and TGF-β 38 . Inhibition of the TGF-β/Smads signaling pathway produces antiproliferative effects on synovial fibroblasts in RA 39 . Research has shown that IL-4 and IL-10 can significantly inhibit the production of IL-1β, TNF-α, IL-6, and IL-8 in RA synovial tissue cells 40 . Moreover, in the current study, we detected increasing apoptosis in RASFs after 48 h of coculture; thus, increasing apoptosis was another factor responsible for decreased RASF proliferation. We also detected slight inhibition of RASF proliferation at 72 h following coculture with Treg cells, although the observed effects were not statistically significant. IL-2, IL-4, IL-7, and IL-15 maintain the optimal regulatory function of human CD4+ CD25+ T cells in a PI3K-dependent manner 41 . Cultured RASFs secrete IL-6 42 , and IL-6 reduces the Treg population 43 . Following 72 h of coculture, Treg cell secretion might decrease to levels insufficient to inhibit RASF proliferation. Thus, it is possible that Treg cells with superior suppressive potency were unable to overcome the increased secretion of pathogenic cytokines by RASFs and could not affect RASF proliferation.
The numbers of NK cells in peripheral blood and spleen and IL-4, IL-13, TNF-α, IFN-γ, and GM-CSF levels in the peripheral blood were significantly higher in the CIA group than in the healthy control group but did not differ significantly between the Treg cell–injected animals and the healthy controls. These results indicate that the therapeutic effect of exogenous human Treg cells on CIA is not exerted via regulation of the number of NK cells or the levels of IL-4, IL-13, TNF-α, IFN-γ, or GM-CSF.
It is generally believed that human Treg cells cannot survive in wild-type rodents except immunocompromised mice. However, human ex vivo expanded Treg cells have been used for clinical purposes in some clinical studies. One study injected tonsillar CD4+CD25+ Treg cells from patients with IgA nephropathy into male Sprague–Dawley rats. Transplantation of these Treg cells ameliorated disease progress by coordinating immune balance in the rats. IL-2, IL-4, and IL-6 in splenic cells and plasminogen activator inhibitor type 1, IL-6, and TGF-β1 expression in renal mesangial cells of the rats were dramatically decreased following the cell injection. The transferred tonsillar CD4+CD25+ Treg cells migrated predominantly to secondary lymphoid organs and not to the kidneys 44 . Treg cells have also been shown in animal models to promote transplantation tolerance and curb autoimmunity following adoptive transfer 45 . One study found that injection of TGF-β-induced CD4+CD25+ cells induced antigen-specific immune tolerance in vivo in male C57BL/6 (B6, H-2b), DBA/2 (D2, H-2d), C3 H (H-2 k), and TGF-β RII KO (C57BL/6 strain) mice. An increased abundance of CD4+CD25+ cells in recipients contributes to this immune tolerance. Adoptive transfer of TGF-β-induced CD4+ CD25+ cells did not result in significant toxicity or side effects in the recipients 46 . In another study, heparinized blood from Dark Agouti rats was injected into naïve Lewis rats prior to liver allograft transplantation, and that study found that donor-specific blood transfusion induced the time-dependent formation of CD4+ Foxp3+ Treg cells that potently suppressed alloantigen-induced activation of naïve Lewis rat T cells in vitro and liver allograft rejection in vivo 47 . In a different investigation, CD4+ CD25+ T cells were transferred into mice exhibiting arthritis symptoms. The transfer of Treg cells markedly slowed disease progression. Furthermore, these Treg cells could be found in inflamed synovium soon after they were transferred, indicating that regulation may occur locally in the joint 48 . The above studies by others not only support our findings regarding induced human Treg injection to treat CIA but also indicate that exogenous Treg cells possess suppressive activity on immune activation. For the same reason, we did not observe immune rejection with induced human Treg cell injection in the CIA model.
In this study, we used induced human Tregs to treat CIA rats. The CIA model is one of the standard and most commonly used RA models used in preclinical research. The CIA model shares several pathological features with RA, such as synovial inflammatory cell infiltration, synovial hyperplasia, cartilage destruction, and bone erosion. However, the CIA model cannot entirely recapitulate the clinical pathology of RA. CIA is an acute animal model, and the joint arthritis gradually disappears 30 days after the first collagen injection, while RA is chronic disease; CIA generates antibodies targeting collagen, and RA has rheumatic factors and anti-CCP autoantibodies (anticyclic citrullinated peptide antibody). In addition, certain pathogenic features, such as synovial angiogenesis, are not as strong for CIA as for human RA 49,50 .
In the current study, we did not conduct experiments to trace either phenotypic and functional characteristics of the culture-expanded human T cells in vitro and in vivo or cell fate and distribution after transplantation, although certain studies have shown that human or other exogenous Treg cells can function in wild-type rats. In our study, the preparation of culture-expand Treg cell was designed based on Schmidt’s study 19 . A similar protocol was used in many studies 45 . This protocol results in reproducible expression of FOXP3 and other Treg signature genes, and it enables the study of general FOXP3-regulatory mechanisms. The induced Tregs generated by these protocols can be used for functional assays, such as investigations of these cells’ suppressive functions. In the present study, we detected alleviated CIA in rat models, suggesting that the regulatory phenotypes and/or functions of the injected human Treg cells were preserved, although additional exploration of the detailed mechanism of this phenomenon is necessary.
The major bottleneck of the Treg adoptive therapy is maintaining the in vivo stability of the Treg cells. There is unexpected plasticity between both Tregs and T effector cells, especially between Treg and Th17, Treg, and Th1 6,37,51,52 . Various reagents have been proposed to upregulate and stabilize Foxp3 expression. Recent progress in genome editing technology has provided a new solution for this bottleneck. Safari et al. have discussed the potential application of clustered regularly interspaced short palindromic repeats–mediated Treg genome editing in personalized therapy for RA 53 .
In conclusion, the results of the current study showed that Treg cells can inhibit RASF proliferation while promoting their apoptosis. The injection of induced human Treg cells into CIA rats effectively alleviated CIA symptoms by increasing the number of endogenous Treg cells and reducing the number of B cells, the Th1/Th2 ratio, and the secretion levels of IL-5 and IL-6. The current study provides a novel therapeutic strategy for RA by transplantation of exogenous Treg cells. Since trans-species cell transplantation can achieve therapeutic effects, Treg cell therapy within ethnic groups should achieve therapeutic success without causing immune rejection.
Supplemental Material
Supplemental Material, Additional_file_1_of_S41 - Therapeutic Effect of Exogenous Regulatory T Cells on Collagen-induced Arthritis and Rheumatoid Arthritis
Supplemental Material, Additional_file_1_of_S41 for Therapeutic Effect of Exogenous Regulatory T Cells on Collagen-induced Arthritis and Rheumatoid Arthritis by Shutong Li, Hongxing Wang, Hui Wu and Xiaotian Chang in Cell Transplantation
Footnotes
Ethical Approval
All of the patients and healthy subjects signed written informed consent statements for participation in the study. The study protocol was approved by the Ethical Committee of the Affiliated Hospital of Qingdao University (Approval number: 20170306), China. All animal experiments were carried out with ethical approval from the Laboratory Animal Care and the Use Committee of Qingdao University.
Statement of Human and Animal Rights
The breeding and operation of the experimental animals were carried out in accordance with the Helsinki Convention on Animal Protection and the Regulations of the People’s Republic of China on the Administration of Experimental Animals. The study protocol was approved by the Medical Ethical Committee of The Affiliated Hospital of Qingdao University at Qingdao, China (Approval number: 20190306).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Shandong Provincial Key R&D programs (2017CXGC1202 and GG201703080038). The funding supported all experiments, data analysis, and English editing.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
