Abstract
Ginger (Zingiber officinale Roscoe) is one of the most commonly used medicinal plants and is extensively used for the treatment of arthritic patients in Traditional Korean Medicine (TKM) due to its various pharmacological properties. In this study, we evaluated the therapeutic effects of ginger on rheumatoid arthritis (RA), particularly focusing on the regulation of Th1, Th2, and Th17 cytokines and the inhibition of matrix metalloproteinase (MMP) release in mice with collagen-induced arthritis (CIA) and primary synovial fibroblasts. RA was induced in male DBA/1J mice via immunization with type II collagen (CII). A ginger extract was prepared in water. The ginger extract (100 and 200 mg/kg) or Mobic (50 mg/kg), as a reference drug, was orally administered to CIA mice once daily for 14 days after arthritis induction. Primary fibroblasts were isolated from the synovial tissues of osteoarthritis patients and then were stimulated with IL-1β and treated with the ginger extract at different concentrations. IL-4, IFN- γ, and IL-17 levels were measured in the serum or spleen and paw tissues of CIA mice and culture media via enzyme-linked immunosorbent assay (ELISA). The mRNA expression of IL-17, MMP-1, MMP-3, and MMP-13 was also detected in paw tissues and synovial fibroblasts through reverse transcription polymerase chain reaction (RT-PCR). Histological changes in the knee joints were observed via hematoxylin and eosin (H&E) and safranin-O staining. The major compounds in the ginger extract were analyzed using high-performance liquid chromatography (HPLC). Treatment with the ginger extract at 100 or 200 mg/kg significantly decreased the levels of IL-4, IFN-γ, and IL-17 and inhibited the expression of IL-17 in the spleen and paw tissues of CIA mice. Ginger extract inhibited the expression of MMP-1, MMP-3, and MMP-13 in the paw tissues of CIA mice and reduced inflammatory bone destruction in joint tissues. In IL-1β-stimulated synovial fibroblasts, the ginger extract significantly decreased the production of IFN-γ and IL-17 via inhibition of mRNA expression. The ginger extract also suppressed the expression of MMP-1, MMP-3, and MMP-13 mRNA. Vanillylacetone, 6-gingerol, 6-shogaol, and 1,4-cineol were identified as the main compounds in the ginger extract. These results indicate that ginger can prevent RA progression by inhibiting the secretion of Th1/Th2 and Th17 cytokines and MMPs, which are involved in the pathogenesis of RA.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic, systemic, inflammatory autoimmune disease that primarily affects joints and is characterized by swelling, inflammation, pain, stiffness, and muscle wasting. 1 Although the exact cause of RA is unknown, complex pathobiological events in inflammation and tissue destruction as well as various immune cells and modulators, including inflammatory cytokines, and signaling pathways are known to be involved in RA onset. 2 The goals of RA treatment are to stop pain and inflammation, relieve symptoms, and prevent joint and organ damage. To these ends, non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and disease-modifying anti-rheumatic drugs (DMARDs) are prescribed. Despite this range of drugs for managing pain and slowing the progression of RA, there is no drug that cures the disease completely. Furthermore, long-term use of these medications causes deleterious side-effects. Thus, the use of complementary and alternative medicine (CAM) to relieve chronic pain and slow joint destruction is increasing. 3 In the clinical practice of CAM, herbs with a variety of biological activities have been widely used to treat bone-related diseases such as RA for thousands of years and will undoubtedly continue to be employed by traditional users as cost-effective alternatives to commercial pharmaceutical products. 4
Zingiber officinale Roscoe (Zingiberaceae), commonly known as ginger, is one of the most commonly used medicinal plants. This herb is extensively used as an integral component of many traditional medicines in Asian and Middle Eastern countries for the alleviation of various ailments.5,6 The properties of ginger have been extensively reviewed in pharmacology and medicine. 7 Ginger possesses pharmacological activities such as anti-type 2 diabetes,8,9 anti-oxidation, gastroprotection,10,11 anti-inflammation,12–14 immune-modulatory (in allergic diseases), 15 and anti-arthritis16,17 properties. Ginger contains phytochemicals such as 6-gingerol, 6-shogaol, paradol, isoshogaol, gingerdione, and diarylheptanoids and gingerol-related compounds.5,18 In particular, gingerols and shogaols exhibit therapeutic benefits in bone diseases through the inhibition of arthritic inflammation and autoimmune responses.7,16,17
T-cell-mediated autoimmune responses are critical in the pathogenesis of RA. To increase the specificity of RA therapeutics, emphasis has shifted to targeting the imbalance of Th1/Th2 cytokines. The Th1-type cytokine such as IFN-γ tend to product the pro-inflammatory responses responsible for killing intracellular parasiters and for perpetuating autoimmune responses. 19 The Th2-type cytokine such as IL-4 is a positive regulator of inflammatory arthritis. 20 Th17 cells have been classified as a third subset of Th cells associated with the Th1/Th2 paradigm, and IL-17 release from Th17 cells is critical in synovitis in the pathogenesis of many inflammatory and autoimmune diseases, including RA. 21 The activation of matrix degradation enzymes such as matrix metalloproteinases (MMPs) in synovial fibroblasts is mediated by pro-inflammatory cytokines and plays an important role in the degradation of cartilage matrix in RA. Thus, recent therapeutic strategies have focused on controlling the Th1/Th17 response and MMPs release in RA.
In this study, we therefore investigated the therapeutic effects of ginger in RA, particularly focused on the modulation of Th1/Th2 and Th17 cytokines and the inhibition of MMPs (MMP-1, MMP-3, and MMP-13) expression, which directly influence bone degradation in mice with collagen-induced arthritis and primary synovial fibroblasts isolated from osteoarthritis (OA) patients.
Materials and methods
Preparation of ginger extract
The roots of commercially available ginger (Zingiber Rhizomes) were purchased from the Herbal Medicinal Materials Company (Gwangmyungdang Co., Ulsan, Korea) and authenticated by Prof. Y.-K. Park, a botanist at the College of Korean Medicine (CKM), Dongguk University, Republic of Korea. A voucher specimen (G-WE/2012) was deposited at the Herbalium of CKM. To prepare the extract, ginger roots (200 g) were chopped, ground in a mechanical grinder, and extracted by boiling in water for 3 h. The extract was then filtered through Whatman’s Grade 1 filter paper, concentrated under a vacuum rotary evaporator and lyophilized in a freeze-dryer (IlShin BioBase Co., Yangju, Korea). The obtained ginger power (yield of 21.2%) was stored at 4°C until use, at which time it was dissolved in 1× PBS (ginger extract). For cellular experiments and HPLC analysis, the ginger extract was filtered through Corning syringe filter (0.45 μm pore size; Sigma–Aldrich, St Louis, MO, USA) before the treatment of cells.
Experimental animals
Male DBA/1J mice at 6 weeks of age were purchased from SLC Inc. (Shizuoka, Japan). All animals received food and water ad libitum and were housed under standard laboratory conditions at an ambient temperature of 22°C ± 3°C, with humidity of 60% ± 5%, under a daily 12 h/12 h light/dark schedule. All mice were cared for and used according to the guidelines of the Institutional Animal Care and Use Committee (IACUC) of Dongguk University.
Induction of arthritis and drug administration
For arthritis induction in mice, bovine type II collagen (CII; Sigma–Aldrich) was dissolved overnight at 4°C in 50 mM acetic acid to a concentration of 2 mg/mL. Mice were administered CII emulsified in complete Freund’s adjuvant via intradermal injection at the base of the tail. Two weeks later, the mice were given a booster intradermal injection of an equal volume of CII emulsified in incomplete Freund’s adjuvant in the hind paw. The ginger extract or Mobic (Meloxicam; Boehringer Ingelheim GmbH & Co., Germany), as a reference drug, was orally administered once daily from day 22 to day 35 after the induction of arthritis; mice in the normal group received an equal volume of water using the same procedure. The CIA mice were randomly divided into five groups of five animals each: a CIA control group, two ginger extract (100 and 200 mg/kg body weight)-treated groups of CIA mice, and a Mobic (50 mg/kg)-treated group of CIA mice as a reference NSAID group.
Serological analysis
Blood samples were collected via cardiac puncture under isoflurane anesthesia, and the mice were then sacrificed on day 36 after the primary immunization. The blood was clotted for 2 h at room temperature and centrifuged at 5000 r/min for 10 min at 4°C to obtain serum. The levels of cytokines (IL-4, IFN-γ, and IL-17) were measured using commercially available Mouse Quantikine ELISA kits according to the manufacturer’s recommendations (R&D Systems, Minneapolis, MN, USA). The concentration of IL-4 (sensitivity: 2 pg/mL), IFN-γ (2 pg/mL), and IL-17 (5 pg/mL) was calculated from the standard curve for each recombinant protein in the ELISA kits.
Preparation of spleen and paw tissue lysates
The spleens were harvested from each mouse, and paw tissues were prepared by removing the skin and separating the limb below the ankle joint. Each tissue was minced, placed in round bottom microtube, snap frozen in liquid nitrogen, and immediately homogenized with an electric homogenizer in five volumes of extraction buffer (100 mM Tris, pH 7.4, 150 mM NaCl, 1 mM EGTA, 1 mM EDTA, 1% Triton X-100, and 0.5% sodium deoxycholate). The tissue lysates were placed on a shaker at 4°C for 1 h, and centrifuged at 10,000×g for 5 min. The supernatants as a soluble protein extract were harvested and stored at −80°C. The concentrations of IL-17 in the supernatants were determined by an ELISA kit (R&D Systems) according to the manufacturer’s instructions.
Histological analysis
On day 36, all mice were sacrificed via anesthesia after serum collection. The hind paws and knee joints were removed from the mice for histological examination. The joints were fixed in 4% paraformaldehyde solution, decalcified with Calci-Clear Rapid (National Diagnostics, Atlanta, GA, USA), embedded in paraffin, and longitudinally cut into 5-µm serial sections. The sections were then stained with hematoxylin and eosin (H&E) and safranin-O (Sigma) to assess cartilage damage.
Isolation of primary synovial fibroblasts
Synovial tissues were obtained from OA patients during arthroplasty at Dongguk University Gyeongju Hospital after the patients previously signed an informed consent. The tissues were finely minced, digested for 30 min at 37°C in 1× PBS containing 0.1% trypsin (Sigma–Aldrich), and subsequently digested in 0.1% collagenase P (Boehringer Mannheim, Mannheim, Germany) in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10% FBS, penicillin (100 units/mL), and streptomycin (100 μg/mL) for 2 h at 37°C in a 5% CO2 incubator. The cell suspension was filtered, and the cells were collected via centrifugation at 5000 r/min for 5 min. The cells were subsequently maintained in primary culture for 7 days in DMEM containing 10% FBS, 25 mM HEPES, penicillin (100 units/mL), streptomycin (100 μg/mL), and 2.5 μg/mL amphotericin B (Gibco BRL, Eggenstein, Germany) in 25 T flasks. Non-adherent cells collected on days 1, 3, 5, and 7 were used to isolate synovial fibroblasts. For the following experiments, primary synovial fibroblasts were cultured with the ginger extract (1 and 2 mg/mL) for 30 min and then stimulated with IL-1β (10 ng/mL) for 5 h for reverse transcription polymerase chain reaction (RT-PCR) analysis or for 24 h for ELISA.
RT-PCR assay
Total RNA was isolated from paw cells and synovial fibroblasts using the TRIzol reagent (Invitrogen Co., CA, USA) according to the manufacturer’s recommendations. Total RNA was reverse transcribed using 5 units of M-MLV Reverse Transcriptase (Promega Co., WC, USA) and 2 μM oligo-dT primer for 1 h at 42°C. PCR was then conducted with the cDNA template, Taq polymerase (Takara Bio Inc., Shiga, Japan) and specific primers (Table 1) over 30 cycles at 94°C for 30 s (denaturation), 58°C–62°C for 30 s (annealing), and 72°C for 60 s (extension). The amplified DNA products were visualized in 1% agarose gels and photographed under UV. Band intensity was quantified via densitometric analysis (Digital Image Analysis System; GenoMxTM Vision, BioGenex, San Ramon, CA).
Primers for RT-PCR.
m: mouse; h: human.
HPLC analysis
To identify the constituents of the ginger extract, high-performance liquid chromatography (HPLC) was conducted. The HPLC apparatus was a Shimadzu HPLC equipped with a SIL-20A Autosampler, a SPD-20A UV/VIS detector (Shimadzu, Japan). An Optimapak C18 (4.6 × 250 mm2; RStech Co., Korea) column was employed. Chromatographic separation was performed using a gradient solvent system consisting of acetonitrile (HPLC grade; J. T. Baker, NJ, USA) (B) and water with 0.1% phosphoric acid (A). The gradient program was as follows: 0 min, 0%B; 12 min, 5%B; 35 min, 45%B; 43 min, 50%B; 50 min, 55%B; and 75 min, 90%B. The column eluent was monitored at UV 205 nm. All solvents were subsequently degassed with a micro-membrane filter (PTFE; Advantec., Japan). Chromatography was performed at room temperature with a flow rate of 1.0 mL/min, and a 10 μL volume was analyzed.
Statistical analysis
All data were analyzed with GraphPad PRISM 5.0 software. Data are expressed as mean ± standard error of the mean (SEM). The significance of treatment effects was determined using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc analysis. The null hypotheses of no difference were rejected for P-values less than 0.05.
Results
Effect of ginger extract on the production of IL-4, IFN-γ, and IL-17 in CIA mice and in IL-1β-stimulated synovial fibroblasts
To investigate the effect of the ginger extract on the production of Th1 and Th17 cytokines, we measured the levels of IL-4, IFN-γ, and IL-17 in the sera of CIA mice via ELISA. The serum levels of IL-4 (P < 0.001, Figure 1(A)), IFN-γ (P < 0.001, Figure 1(B)), and IL-17 (P < 0.001, Figure 1(C)) were significantly increased in the CIA control group compared to the levels in the normal group. Serum IL-4 (Figure 1(A)), IFN-γ (Figure 1(B), and IL-17 levels (Figure 1(C)) were significantly reduced by treatment with 100 and 200 mg/kg ginger extract compared to the CIA control group. Mobic also significantly inhibited the production of IFN-γ (Figure 1(B)) and IL-17 (Figure 1(C)) but had no effect on the level of IL-4.

Effect of ginger extract on the levels of inflammatory cytokines in CIA mice. Ginger extract (100 and 200 mg/kg) or Mobic (50 mg/kg), as a reference drug, was orally administered to CIA mice once daily from days 21 to 37. The levels of (A) IL-4, (B) IFN-γ, and (C) IL-17 were measured in the sera of mice via ELISA. Data are expressed as mean ± SEM (n = 5 mice per group). *P < 0.05; **P < 0.01; ***P < 0.001 vs (a) normal group or (b) CIA control group.
To examine the responsible mechanism of the ginger extract in the effects observed in CIA mice, we measured the production of IFN-γ and IL-17 and their mRNA expression in IL-1β-stimulated synovial fibroblasts using ELISA and RT-PCR, respectively. The concentration of ginger extract for treatment in synovial fibroblasts was determined by MTT assay, and used the non-toxic range (data not shown). The levels of IFN-γ and IL-17 were significantly increased by IL-1β stimulation (P < 0.001) and significantly (P < 0.01) decreased by treatment with the ginger extract at 1 and 2 mg/mL (Figure 2(A)). The ginger extract significantly inhibited not only the expression of IFN-γ (P < 0.05) and IL-17 (P < 0.05) mRNA (Figure 2(B)) in IL-1β-stimulated synovial fibroblasts. These results indicate that ginger extract can inhibit the inflammation in RA through downregulation of the transcription level of Th1 and Th17 cytokines.

Effect of ginger extract on the levels of inflammatory cytokines in IL-1β-stimulated synovial fibroblasts. Synovial fibroblasts were isolated from RA patients, treated with ginger extract at 1 or 2 mg/mL for 30 min, and then stimulated with IL-1β or without for 5 h. (A) The concentration of each cytokine was measured in culture supernatants via ELISA and (B) the mRNA expression of the cytokines was detected through RT-PCR. GAPDH was used as an internal control. The histogram was analyzed with a densitometer. Data are expressed as mean ± SEM of three independent experiments. *P < 0.05; **P < 0.01; and ***P < 0.001 vs (a) normal or (b) IL-1β-stimulated cells.
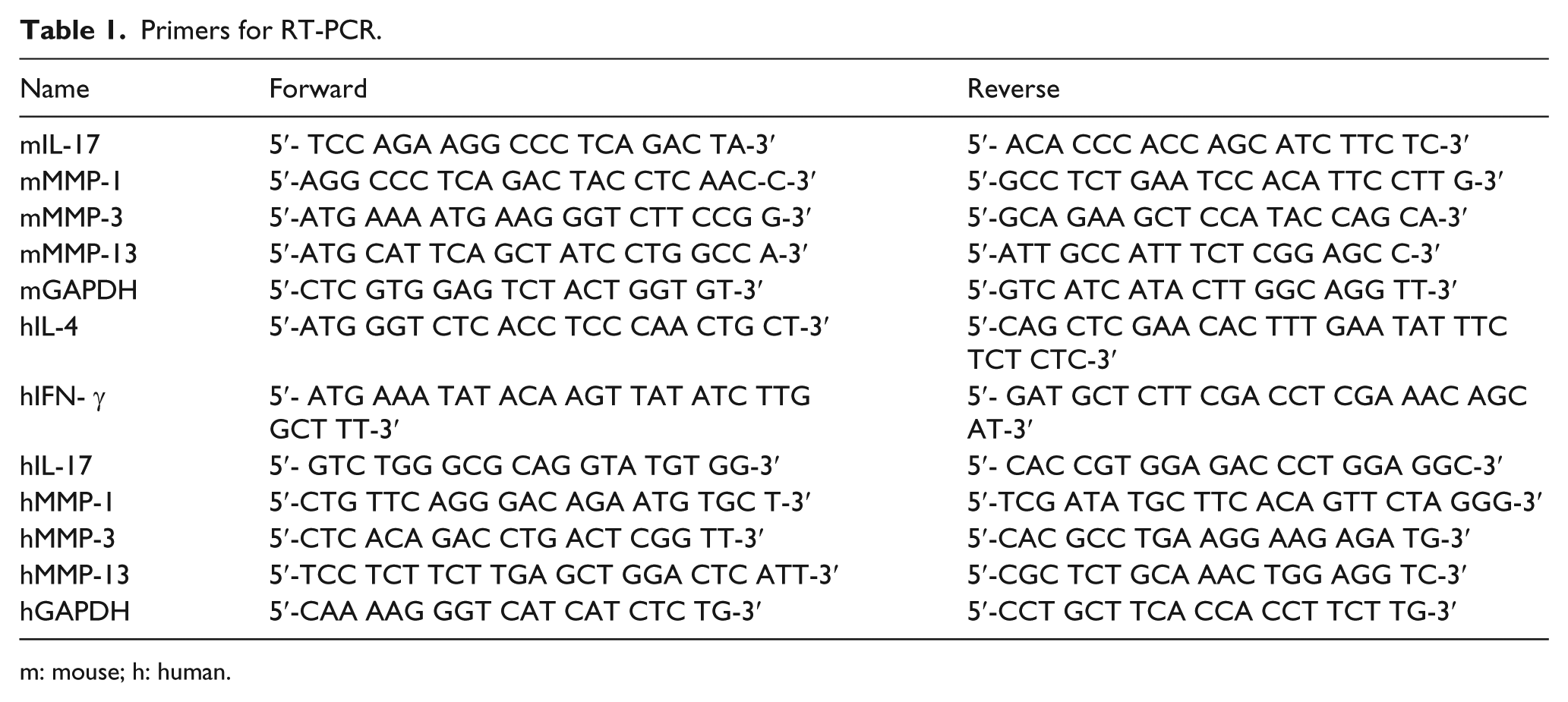
Effect of ginger extract on the level of IL-17 in the spleen and paw tissues of CIA mice
Next, we measured IL-17 levels in the tissues of arthritic spleen and paw of CIA mice via ELISA. IL-17 levels were significantly increased in spleen (P < 0.05, Figure 3(A)) and paw (P < 0.001, Figure 3(B)) of the CIA control group compared to the levels in the normal group. Treatment of CIA mice with 200 mg/kg ginger extract significantly decreased IL-17 levels in the cells of spleens (P < 0.05) and paws (P < 0.001) compared to the CIA control group. The ginger extract and Mobic drastically decreased IL-17 levels in paw compared to those in the spleen.
Effect of ginger extract on the levels of IL-17 in the spleens and paws of CIA mice. The levels of IL-17 were measured in the (A) spleen and (B) paw tissues of the mice through ELISA. (C) The expression of IL-17 was determined in the paw tissues of the mice through RT-PCR. GAPDH was used as an internal control. (D) The histogram was analyzed with a densitometer. Data are expressed as mean ± SEM (n = 5 mice per group). *P < 0.05; ***P < 0.01; and ***P < 0.001 vs (a) normal group or (b) CIA control group.
We also investigated the mRNA expression of IL-17 in the tissues of arthritic paw of CIA mice by RT-PCR. The expression of IL-17 was significantly (P < 0.01) increased in paw of the CIA control group compared to the levels in the normal group (Figure 3(C)). Treatment of CIA mice with 200 mg/kg ginger extract significantly decreased IL-17 levels in the cells of paws (P < 0.05) compared to the CIA control group. These results indicate that ginger extract can inhibit IL-17-mediated inflammation in RA.
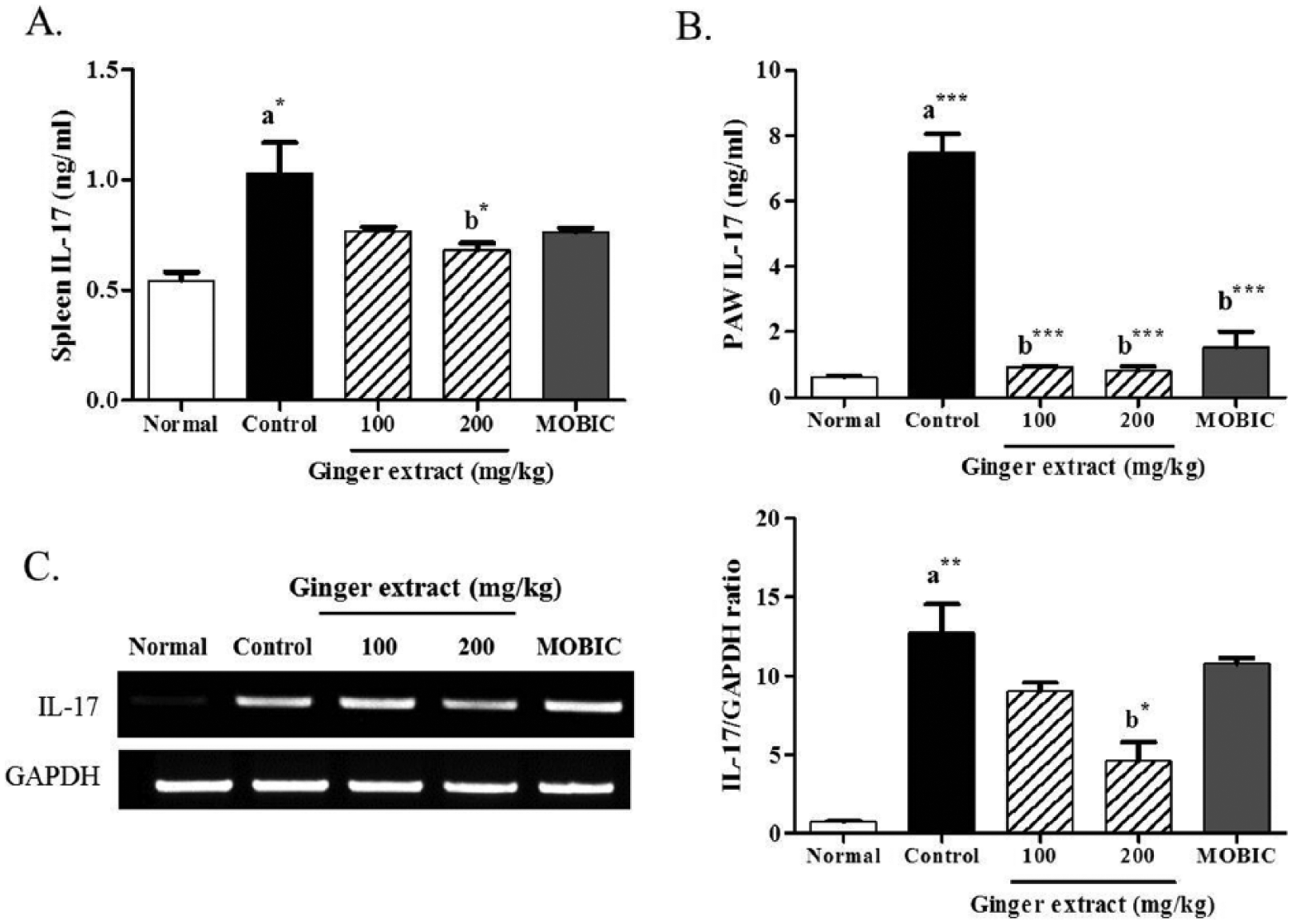
Effect of ginger extract on the expression of MMP-1, MMP-3, and MMP-13 in the paw tissues of CIA mice and in IL-1β-stimulated synovial fibroblasts
We measured the mRNA expression of MMP-1, MMP-3, and MMP-13, which are responsible for T-cell-mediated joint degradation with IL-17 release22,23 in the paw tissues via RT-PCR. The expression of MMP-1 (P < 0.01), MMP-3 (P < 0.01), and MMP-13 (P < 0.001) mRNA was significantly increased in the CIA control group compared to those in the normal group (Figure 4). The expression of MMP-1 (P < 0.05), MMP-3 (P < 0.001), and MMP-13 (P < 0.001) in CIA mice was significantly decreased upon treatment with 200 mg/kg ginger extract. In addition, 200 mg/kg ginger extract significantly (P < 0.05) inhibited IL-17 and MMP mRNA expression in the paw tissues of CIA mice.

Effect of ginger extract on the levels of IL-17 in the paws of CIA mice. The expression of IL-17 was determined in the paw tissues of the mice through RT-PCR. GAPDH was used as an internal control. The histogram was analyzed with a densitometer. Data are expressed as mean ± SEM (n = 5 mice per group). *P < 0.05; ***P < 0.01; and ***P < 0.001 vs (a) normal group or (b) CIA control group.
We also investigated the expression of MMP-1, MMP-3, and MMP-13 mRNA in IL-1β-stimulated synovial fibroblasts using RT-PCR. The ginger extract significantly inhibited the expression of IL-3 (P < 0.05, respectively) and MMP-13 (P < 0.01 and P < 0.001) mRNA by treatment with the ginger extract at 1 and 2 mg/mL (Figure 5). However, the ginger extract did not affect MMP-1 expression. These results indicate that ginger extract can prevent the joint degradation in RA through downregulation of the transcription levels of MMP-3 and MMP-13.
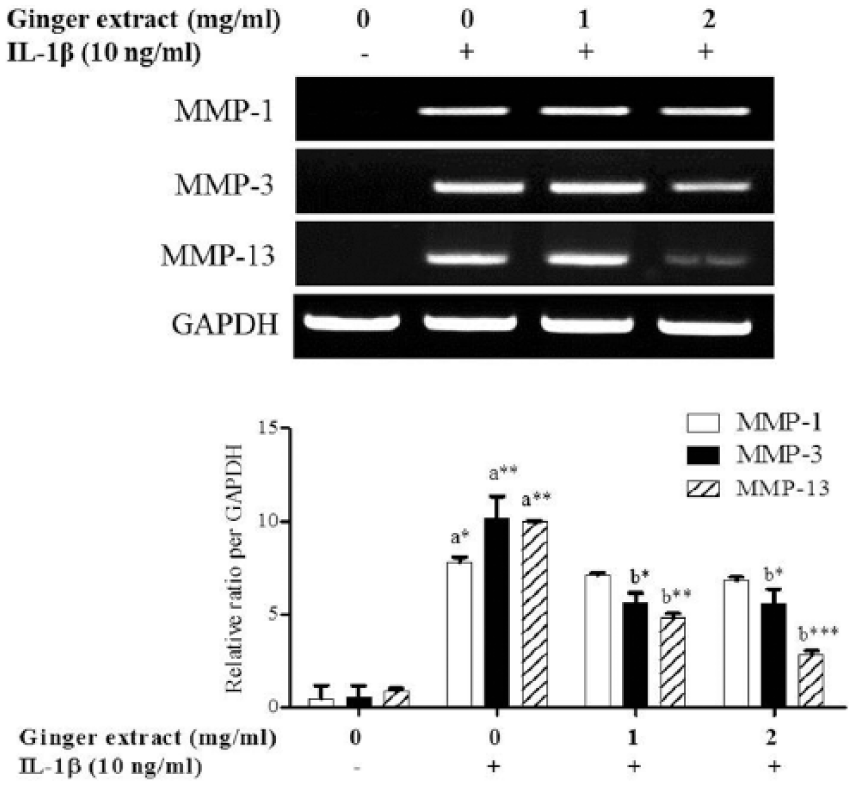
Effect of ginger extract on the expression of MMPs in IL-1β-stimulated synovial fibroblasts. Synovial fibroblasts were isolated from RA patients, treated with ginger extract at 1 or 2 mg/mL for 30 min, and then stimulated with IL-1β or without for 5 h. The mRNA expression of MMP-1, MMP-3, and MMP-13 was detected through RT-PCR. GAPDH was used as an internal control. The histogram was analyzed with a densitometer. Data are expressed as the mean ± SEM of three independent experiments. *P < 0.05; **P < 0.01; and ***P < 0.001 vs (a) normal or (b) IL-1β-stimulated cells.
Effect of ginger extract on the histological changes in inflammatory joints in CIA mice
To investigate the effect of the ginger extract on arthritic knee joint damage due to CIA, we observed histological changes through H&E and safranin-O staining. Upon H&E staining, the knee joints of CIA mice displayed partial cartilage destruction with inflammatory cell infiltration into the joint capacity and notable synovial hyperplasia (Figure 6(A)). These pathological events were substantially reduced by treatment with 100 and 200 mg/kg ginger extract. Moreover, safranin-O staining revealed that the ginger extract markedly reduced cartilage loss compared to the CIA group (Figure 6(B)). This result indicates that the ginger extract exerts a protective effect on joint inflammation and bone destruction in RA.

Effect of ginger extract on the histological changes in the knee joints of CIA mice. The joint tissues were isolated from all mice, from which paraffin-embedded slides were prepared, followed by staining with (A) H&E and (B) safranin-O. A representative result from at least three independent experiments is presented (original magnification = ×200).
HPLC analysis
Four compounds (vanillylacetone, 6-gingerol, shogaol, and 1,4-cineol) in the ginger water extract were identified through the comparison of retention times of authentic standards analyzed by HPLC. The content of each compound was calculated by comparison of peak areas of sample (Figure 7(A)) and authentic standards (Figure 7(B)). The equations of calibration curves for vanillylacetone, 6-gingerol, and shogaol were y(peak area) = 4117 × (concentration, mg/mL) − 4.4, y = 5733 × −1.3, and y = 5967 × +9.7, respectively. The correlation coefficients of three calibration curves were higher than 0.9998 through the concentration range of 0.005–0.3 mg/mL. The concentrations of vanillylacetone, 6-gingerol, and shogaol in the extract were 0.11%, 0.70%, and 0.24%, respectively. The relative standard deviation of the analysis was less than 4.5% for all marker compounds with three times of analysis.

HPLC pattern analysis of the ginger extract: (A) ginger extract and (B) each standard compound. Peaks: (1) vanillylacetone, (2) 6-gingerol, (3) shagaol, and (4) 1,4-cineol.
Discussion
In Western medicine nosology, RA is an autoimmune disease characterized by chronic inflammation due to synovial hyperplasia that progresses with massive irreversible bone and cartilage destruction. 1 Thus, the development of RA follows a complex chain of autoimmune events involving activation of immune cells such as T cells, B cells, macrophages, and fibroblasts and orchestrates the interaction of pro-inflammatory cytokines. In the pathophysiology of RA, activated CD4+ T cells are allowed to penetrate into extracellular fluid by passing through the endothelial wall in inflamed tissues and releasing pro-inflammatory Th1 cytokines, such as TNF-α, IL-2, IFN-γ, IL-17, and RANKL. 24 Therefore, the therapeutic approach for RA is focused on relieving the pain and symptoms of RA with anti-inflammatory drugs, such as NSAIDs.
In the theories of Traditional Korean Medicine (TKM) and Traditional Chinese Medicine (TCM), RA is a complicated, chronic, and intractable process, described under the category of arthromyodynia due to weakness of nutrient qi/defense qi and weakness of the viscera and bowels resulting from insufficient healthy qi. 25 Arthritic patients cannot resist external pathogens such as wind, dampness and heat, and phlegm. Their static blood can debilitate long-term conditions. Therefore, RA treatment is based on the pathogenesis determined through syndrome differentiation into an excess pattern of the acute phase and a deficient pattern of the chronic phase and involves acupuncture and the prescription of various herbs. Ginger is an essential herb for the treatment of chronic arthritis in TKM and TCM due to its pharmacological characteristics of dispelling wind, eliminating dampness, dissipating cold, clearing heat, activating blood circulation to dispel static blood, and tonifying the liver, kidney, and spleen.25,26
The therapeutic benefits of ginger for arthritis include good analgesic and anti-inflammatory effects. Ginger extract reduces the release of inflammatory mediators, prostaglandin E2 (PGE2), nitric oxide (NO), and inflammatory cytokines (TNF-α, IL-1β, and IL-6) in macrophages and synovial cells. Ginger extract in the ranges of 100–500 mg/kg also suppresses LPS-induced acute systemic inflammation and adjuvant-induced chronic inflammation in animal models and improves the knee symptoms of OA patients.13,16,17 Based on these previous findings, we investigated the anti-arthritic effects of ginger extract at 100 and 200 mg/kg in CIA mice and in the synovial fibroblasts of RA patients, with a focus on regulating Th1 and Th17 cytokines and MMPs.
The Th1-type cytokines tend to product the pro-inflammatory responses responsible for killing intracellular parasiters and for perpetuating autoimmune responses. IFN-γ is the main Th1 cytokine. 19 In RA, autoreactive T-cell responses, including autoantibody production, are biased toward a Th1-type response, and the exploration of therapeutic strategies for inhibiting IFN-γ, the main Th1-type cytokine has begun. 27 IFN-γ induces initial inflammation in RA, and its net effect may depend on the phase of disease and the location, such as the joint versus the spleen or lymph node. The Th2-type cytokines include IL-4, IL-5, and IL-13, which are associated with the promotion of IgE and eosinophilic responses in allergic diseases. 19 IL-4 is a main Th2-type cytokine and a positive regulator of inflammatory arthritis. 20 IL-4 is variable consequence in arthritis models. Some CIA models have indicated that arthritis is reduced in IL-4-deficient mice, 28 but other studies indicate that IL-4 deficiency induces more severe arthritis. 29 The actual roles of various Th1- and Th2-type cytokines in the induction of RA remain elusive. However, in our study, ginger extract significantly decreased IFN-γ and IL-4 levels in the sera of CIA mice and IL-1β-stimulated synovial fibroblasts. These results indicate that ginger extract can prevent tissue damage due to the excessive pro-inflammatory responses in RA by regulation of the Th1/Th2 cytokines. Similar to our results, it has been reported that ginger extract prevent the Th2-mediated immune response in airway inflammation or allergic rhinitis of mice through inhibition of IFN-γ, IL-4, and IL-5 production.30,31
IL-17 is a pro-inflammatory cytokine that is primarily produced by Th17 cells and is involved in all stages of RA as an important contributor to preventing or controlling autoimmunity.19,20 In fact, RA patients commonly exhibit increased IL-17 levels in their serum and synovial fluid compared to normal controls. 32 In this study, ginger extract was shown to significantly decrease the level of IL-17 in not only in the serum of CIA mice but also in their spleen and paw tissues. Ginger extract also inhibited IL-1β-induced production in human synovial fibroblasts. These results indicate that ginger can prevent joint inflammation partly through blocking the production of IL-17.
Synovial fibroblasts are responsible for the synthesis of extracellular matrix proteins including MMPs, collagen, fibronectin, and hyaluronic acid as well as other molecules that facilitate the lubrication and function of cartilage surfaces, but these cells also act as a major source of inflammatory mediators. MMPs are a family of zinc-dependent enzymes that degrade all components of the extracellular matrix synthesized in synovial joints by synovial cells and chondrocytes. 33 MMP-1, MMP-13 (collagenases), and MMP-3 (stromelysin-1) are activated by pro-inflammatory cytokines, degrade proteoglycans in articular cartilage, activate procollagenase, and have been proposed as markers of joint damage in RA. 34 In this study, ginger extract was found to significantly inhibit the expression of MMP-1, MMP-3, and MMP-13 in paw tissues of CIA mice and IL-1β-activated human synovial fibroblasts. These results indicate that ginger extract protects bone and cartilage from MMP-induced degradation in RA.
We detected vanillylacetone, 6-gingerol, shogaol, and 1,4-cineol as main compounds in the ginger extract via HPLC analysis. The obtained HPLC pattern was consistent with previous reports. 18 6-shogaol and 6-gingerol are main bioactive compounds in ginger with multiple known beneficial effects, including analgesic and anti-inflammatory properties.7,16,17 Recently, the phenolic compounds including 6-gingerol, 6-shogaol, and zingerone in the rhizome of ginger have been found to possess various pharmacological and physiological activities with anti-oxidant, anti-inflammation, and anti-cancer. In relation to arthritic diseases, it has been reported to the anti-catabolic and anti-inflammatory properties of 6-gingerol and 6-shogaol in streptococcal cell-wall-induced arthritis of rats, 35 in complete Freund’s adjuvant-induced chronic inflammation of rats, 36 and in chondrocytes. 37 6-gingerol is known as a responsible compound for the downregulation of the Th2-type cytokines-mediated allergic response. 31 Our analysis also suggested that these compounds support the anti-arthritic effect of ginger, but their regulatory effects on Th1/Th17 imbalance and MMP release require further research.
In conclusion, ginger extract exerts an anti-arthritic effect through inhibiting the production of the Th1 inflammatory cytokine IFN-γ and the Th17 cytokine IL-17, as well as the expression of MMP-1, MMP-3, and MMP-13 in CIA mice and IL-1β-activated synovial cells. These results suggest that ginger is a potential natural source for the development of RA drugs.
Footnotes
Author contribution
J.H.H. and H.W.J. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
