Abstract
The current study aimed to determine the safety profile of intra-articular-injected allogeneic adipose-derived mesenchymal stem cells (ADSCs) GXCPC1 in subjects with knee osteoarthritis (OA) and its preliminary efficacy outcome. The 3 + 3 phase I study was designed with two dose-escalation cohorts: low dose (6.7 × 106 GXCPC1, N = 5) and high dose (4 × 107 GXCPC1, N = 6). The primary endpoint was safety, which was evaluated by recording adverse events throughout the trial; the secondary endpoints included total, pain, stiffness, and function subscales of the Western Ontario and McMaster Universities Arthritis Index (WOMAC), Visual Analogue Scale (VAS) for pain, and 12-Item Short Form (SF-12) health survey questionnaire. The GXCPC1 treatment was found to be safe after 1 year of follow-up with no treatment-related severe adverse events observed. When compared to baseline, subjects in both the low- and high-dose cohorts demonstrated improving trends in pain and knee function after receiving GXCPC1 treatment. Generally, the net change in pain (95% confidence interval (CI) = −7.773 to −2.561t at 12 weeks compared to baseline) and knee function (95% CI = −24.297 to −10.036t at 12 weeks compared to baseline) was better in subjects receiving high-dose GXCPC1. Although this study included a limited number of subjects without a placebo arm, it showed that the intra-articular injection of ADSCs was safe and well-tolerated in subjects with therapeutic alternatives to treat knee OA. However, a larger scale study with an appropriate control would be necessary for clinical efficacy in the following study.
Keywords
Introduction
Osteoarthritis (OA) is a common chronic degenerative joint disease that affects elderly and obese population1,2. The prevalence of knee OA in females is significantly higher than that in males 3 and around the age of 75 years was 16%–17%, of which 10%–12% for men and 18%–20% for women 4 . The most common clinical manifestations of OA are pain, swelling, stiffness, function limitation, limited mobility, and causes to decrease health-related quality of life (HRQOL). Current treatments for knee OA are mostly symptomatic, focusing on pain reduction, inflammation control, and joint function maintenance, including paracetamol (acetaminophen), nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, and hyaluronic acid (HA). However, the aforementioned treatments can only alleviate and the progression of OA is continuing to aggravate 5 .
Mesenchymal stem cells (MSCs) have multilineage differentiation potential, immunosuppressive properties, and low immunogenicity and are extensively available 6 . Among the others, adipose-derived stem cells (ADSCs) represent a promising source for their abundance, accessibility, and less invasive collection procedure 7 . ADSCs have been proposed as an appealing treatment for knee OA8–17. Many preclinical studies have confirmed intra-articular injection of ADSCs reduced synovitis, osteophyte formation, and cartilage degeneration8,9. Furthermore, clinical studies showed that the autologous ADSCs improved knee function and relieved pain without causing adverse events11–17. A recent study found that using the autologous ADSCs in patients resulted in significant improvements in pain and life quality for up to 12 months when compared to the baseline 16 . While most studies have examined the efficacy and safety of the autologous ADSCs, the allogenic ADSCs have the advantage of application accessibility19–22. Only a few clinical trials for the allogenic ADSCs treatment in patients with knee OA were investigated19–22. In addition, different procedures or methodologies of adipose tissue derivation are very important in the application of orthopedics23,24. Different harvest and isolation methods will affect the cell viability and functional phenotype of ADSCs 24 . Until now, only a few clinical orthopedics-related applications with high-quality and high-concentration cells that meet regulatory requirements have been studied 24 . Therefore, we conducted a phase I clinical trial in this study to evaluate the safety, tolerability, and efficacy of injecting the allogenic ADSCs that meet regulatory requirements in knee OA patients’ knee.
Materials & Methods
GXCPC1 Preparation
The ADSCs in GXCPC1 were obtained from a trial in Hualien Tzu Chi Hospital, Taiwan (IRB number IRB-106-88-A). The inclusion/exclusion criteria for the adipose tissue donors followed the guideline for donor eligibility by Taiwan Food and Drug Administration and were tested negative for human immunodeficiency virus-1/2, hepatitis B virus, hepatitis C virus, and syphilis infections as well as screened for high risks for transmissible spongiform encephalopathies and Creutzfeldt-Jakob disease. The preparation process of GXCPC1 was modified from reference25–27. To put it briefly, the adipose tissues of eligible donors were collected via liposuction and immediately transferred to a Good Tissue Practice (GTP) lab (Gwo Xi Stem Cell Applied Technology Co., Ltd.). Adipose tissues were dissociated with collagenase and the resultant dispersed cells were collected and cultured at 37°C in a carbon dioxide incubator for the primary culture. GXCPC1-related quality testing was executed by a third-party certification laboratory (accredited in respect of laboratory by Taiwan Accreditation Foundation, Accreditation Criteria: ISO/IEC 17025, Accreditation Number: 2800). In-process and safety testing of GXCPC1 for microbiological assessments (including mycoplasma, sterility, and endotoxin tests) were employed to assure safety. The immunophenotypes (human clusters of differentiation CD34, CD45, CD90 and CD105), the viability, and tri-lineage differentiation properties were also evaluated. The quality tests such as sterility, mycoplasma, identity, purity, potency, and stability were conducted to meet regulatory requirements. GXCPC1 is a turbid white cell suspension containing 6.7 × 106 or 4 × 107 ADSCs in 3 ml of normal saline and packed in sterile glass bottles.
Trial Design
This trial (protocol number GXCPC1-01) was conducted at Taipei Veterans General Hospital in Taiwan. This trial was registered on clinicaltrials.gov (NCT03943576) and was approved by the Institutional Review Board (IRB number 2018-10-005C) in accordance with the Declaration of Helsinki.
The purpose of this 3 + 3 dose-escalation phase I study was to assess the safety, tolerability, and efficacy of allogeneic ADSCs (GXCPC1) in the treatment of knee OA. Two dose levels were tested: 6.7 × 106 (low dose) and 4 × 107 (high dose). Eligible subjects were assigned to one of the two doses according to the chronological order of eligibility confirmation.
A subject was considered evaluable if this subject contributed to the assessment of tolerability. Tolerability assessment contributes when (1) an eligible subject received GXCPC1 injection and experienced dose-limiting toxicity (DLT), or (2) an eligible subject received a successful GXCPC1 injection and no DLT was observed until week 8. The DLT was defined as any adverse event that is equal to or greater than grade 3 (CTCAE v.5.0) and was causally related to GXCPC1.
Subjects
Before enrolling, all subjects provided written informed consent to participate in the study. Adults aged 40 to 80 years old with Kellgren-Lawrence grading II-IV and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score range 7 to 17 in the target knee were enrolled in the study 28 . The study excluded subjects who had previous surgery for articular fracture, ligament reconstruction, meniscal reconstruction, or knee arthroplasty on the target knee. Subjects who had intra-articular intervention 12 weeks before the screening were also excluded, as were those who had received a systemic or local immunosuppressive agent, an anti-inflammatory drug, steroid, analgesics, opioid, or duloxetine for knee OA 1 week before screening. Acetaminophen and NSAID act as pain relievers when subjects were in need. Subjects should, however, stop taking pain relievers at least 24 h before the pain evaluation.
One of the eligible subject’s knees that met the criteria was chosen as the target knee for efficacy evaluation. Each subject received a single intra-articular injection at the target knee on day 1, followed by scheduled visits on weeks 1, 4, 8, 12, 24, 36, and 48 (Fig. 1A).
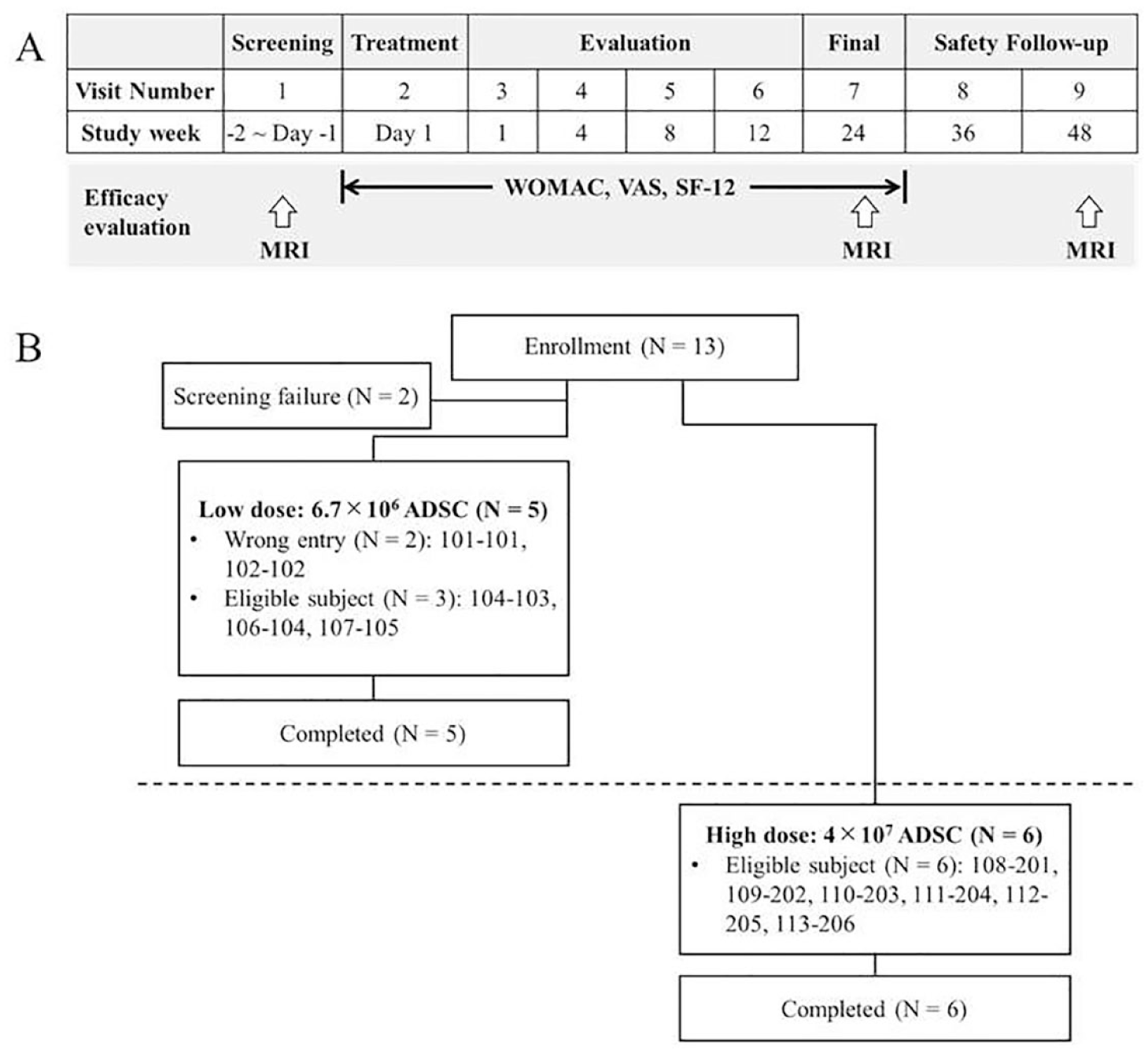
Study schedule and schema of study subject disposition. (A) Study schedule. (B) Schema of study subject disposition. ADSC: adipose-derived mesenchymal stem cells; SF-12: 12-item Short Form; VAS: Visual Analogue Scale; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
Outcome Measures
The primary endpoint was the incidence of adverse events (AEs) and serious adverse events (SAEs) up to 48 weeks after administration. The changes in laboratory examination results from baseline to post-treatment visits were included in the safety assessments. Secondary endpoints included net changes in WOMAC total and sub-scores (pain, stiffness, and physical function), Visual Analogue Scale (VAS) for pain, and 12-item Short Form (SF-12) from baseline to post-treatment visits.
Clinical Laboratory Evaluation
Laboratory assessments, including hematology and biochemistry, were performed at the screening, week 1, week 8, week 24, and week 48 visits. The measured items were listed as follows: for hematology: white blood cells, neutrophils, lymphocytes, monocytes, eosinophils, basophils, hemoglobin, hematocrit, platelet, and red blood cells; for biochemistry: aspartate aminotransferase, alanine aminotransferase, serum creatinine, blood urea nitrogen, total bilirubin, and albumin.
Statistical Analysis
For statistical analysis, the intend-to-treat (ITT) population was used, which included all subjects who had ever received GXCPC1. For the WOMAC, VAS for pain, and SF-12, one-sample t test (denoted as t) was used to assess the net change from baseline to post-treatment visits within group. If the net changes were not normally distributed, Wilcoxon rank-sum test (denoted as w) would be used to analyze the difference. All within-group comparisons were performed educing a 95% confidence interval for difference. Statistical Analysis Software (SAS) version 9.4 was used for all the statistical analyses.
Results
Subjects’ Disposition and Baseline Characteristics
A total of 13 subjects were screened in the study; two subjects were screen failures, resulting in five subjects in the low-dose cohort and six subjects in the high-dose cohort (Fig. 1B). Two subjects, who had WOMAC pain scores of 4 and 2, respectively, were mistakenly received GXCPC1 at low dose. However, the two subjects still completed the trial for long-term safety monitoring and efficacy evaluation.
The demographic and baseline characteristics of the ITT population are shown in Table 1. In the ITT population, the mean (SD) age of all subjects was 66.2 (5.89) years in the low-dose cohort and 64.5 (5.32) years in the high-dose cohort. All subjects had body weight index (BMI) < 35 kg/m2 and the mean (SD) BMI of all subjects was 26.6 (4.68) kg/m2 in the low-dose cohort and 29.3 (2.82) in the high-dose cohort. In both cohorts, the subjects were all females with knee OA grades II to IV. The average duration of knee OA since diagnosis was 7.07 years in the low-dose cohort and 4.31 in the high-dose cohort.
Summary of Demographics Data in ITT Population.
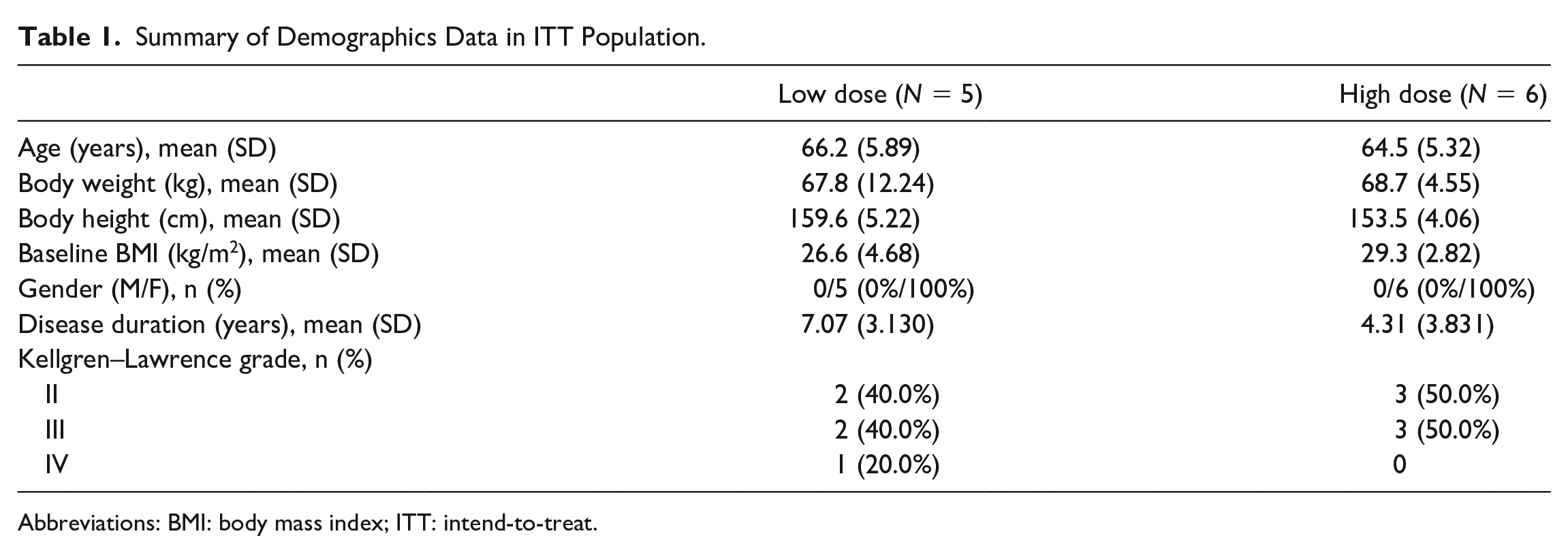
Abbreviations: BMI: body mass index; ITT: intend-to-treat.
Safety
No subjects in the trial experience DLT. AEs were reported by all subjects in both cohorts (Tables 2 and 3). Three (60.0%) subjects in the low-dose cohort and one (16.7%) subject in the high-dose cohort experienced grade ≥ 3 AEs. Treatment-related AEs were reported by two (40.0%) subjects in the low-dose cohort and six (100.0%) subjects in the high-dose cohort, but none of them were severe. Two (40.0%) subjects in the low-dose cohort and one (16.7%) subject in the high-dose cohort had SAEs that necessitated or prolonged hospitalization. All SAEs were considered unrelated to treatment. Moreover, no suspected unexpected serious adverse reaction (SUSAR) or death was reported in this study. Table 4 showed that the most common treatment-related AEs were injection site pain in both cohorts.
Summary of Adverse Events in ITT Population.
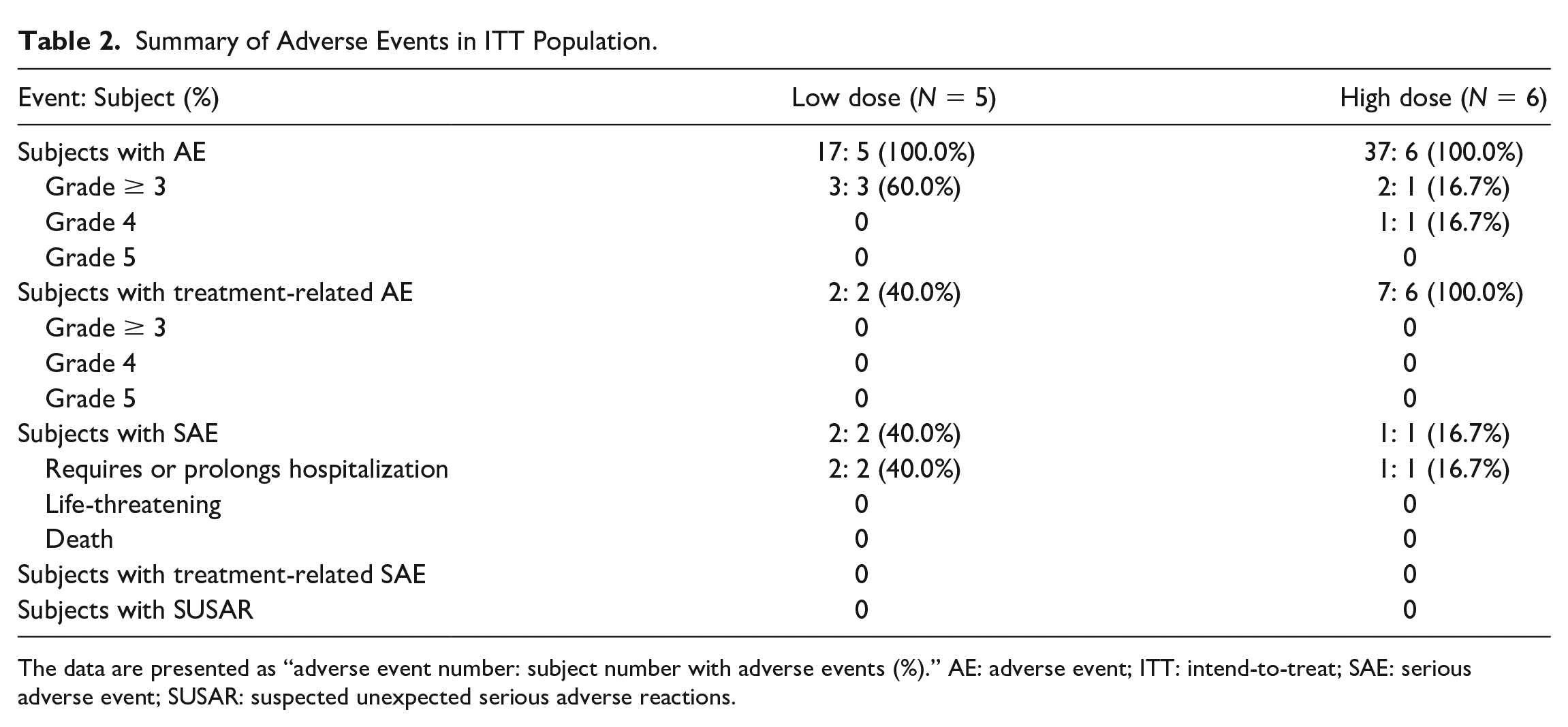
The data are presented as “adverse event number: subject number with adverse events (%).” AE: adverse event; ITT: intend-to-treat; SAE: serious adverse event; SUSAR: suspected unexpected serious adverse reactions.
Summary of Adverse Events by System Organ Class and Preferred Term in ITT Population.
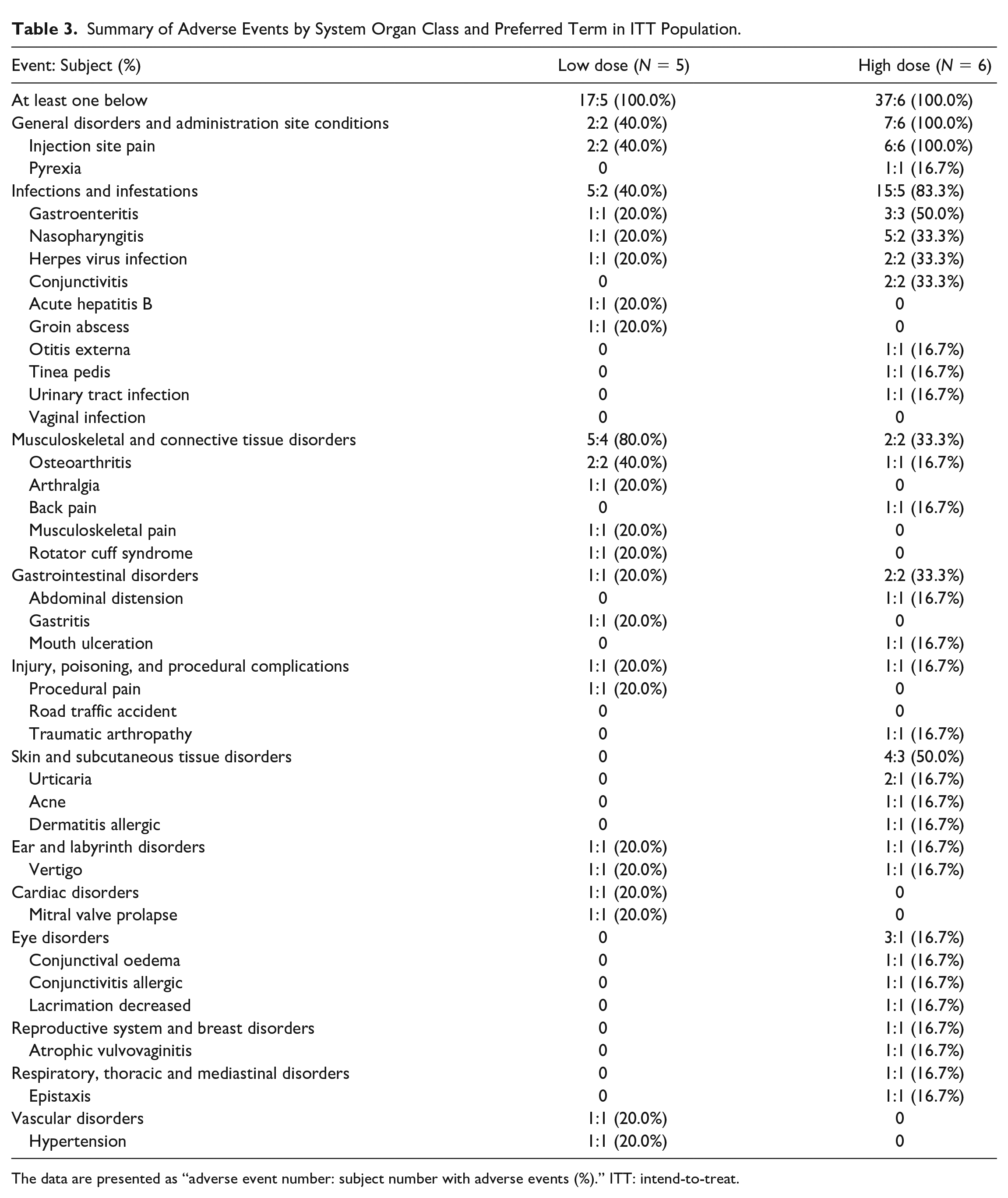
The data are presented as “adverse event number: subject number with adverse events (%).” ITT: intend-to-treat.
Summary of Treatment-Related AEs in ITT Population.
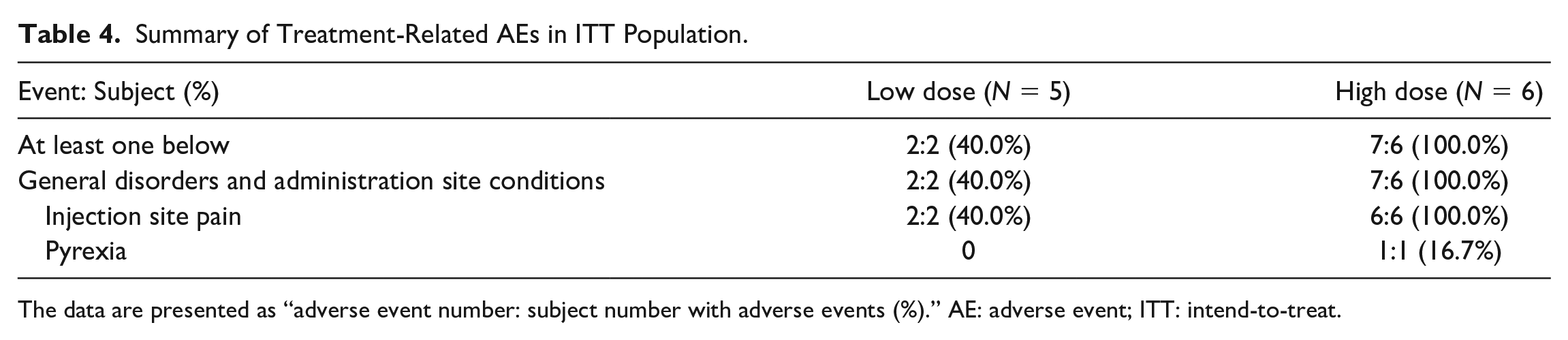
The data are presented as “adverse event number: subject number with adverse events (%).” AE: adverse event; ITT: intend-to-treat.
All subjects had either normal or abnormal without clinically significant (NCS) values in hematology and biochemistry parameters. No clinically significant values were found throughout the study period in both cohorts.
Efficacy
In both cohorts, there was a decreasing trend in WOMAC total and sub-scores (pain, stiffness, and physical function) in the net change from baseline to post-treatment visit (Fig. 2). Over time, the high-dose cohort had greater reductions, which represented the greater improvement, in WOMAC total and sub-scores than the low-dose cohort. The net change from baseline to post-treatment in high-dose cohort showed significant differences in WOMAC total, pain, and physical function scores at 1, 4, 8, and 12 weeks compared to baseline (Fig. 2A, B, and D). There were also significant differences in WOMAC stiffness scores from baseline to 1 and 8 weeks after administering a high dose of GXCPC1 (Fig. 2C). However, the low-dose cohort had no significant difference in WOMAC total, pain, stiffness, and physical function sub-scores.
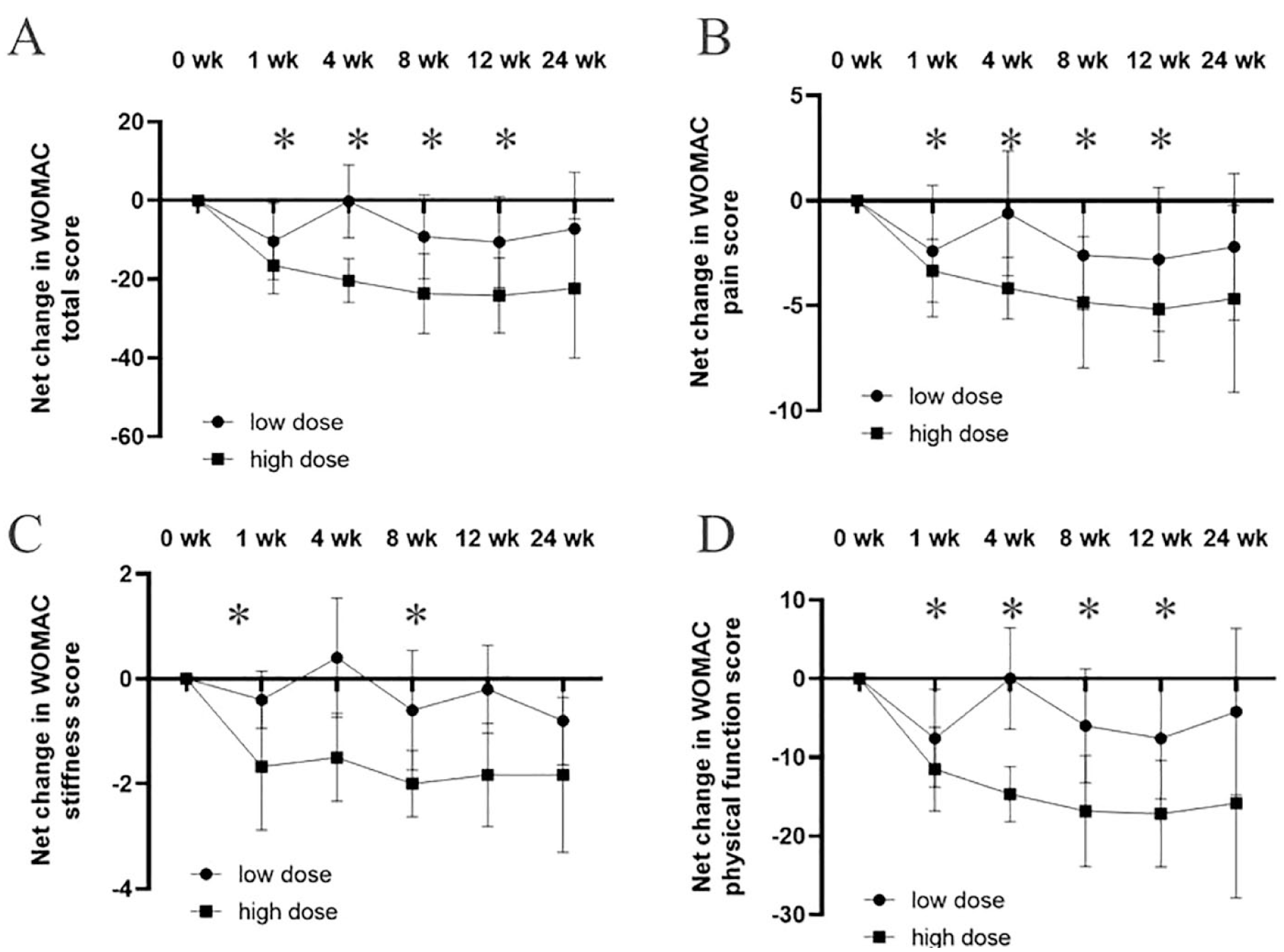
Net changes in WOMAC from baseline to 24 weeks after intra-articular injection of GXCPC1. (A) WOMAC total score, (B) WOMAC pain score, (C) WOMAC stiffness score, and (D) WOMAC physical function score. The data are presented by mean and error bars indicate standard deviation. WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index. *: Sign of significant difference in the high-dose cohort.
Consistent with these results, both cohorts had a decreasing trend in the VAS for pain in the net change from baseline to post-treatment visit (Fig. 3). A significant difference in VAS for pain was observed in the low-dose cohort at 12 and 24 weeks. A significant decrease in VAS for pain was observed in the high-dose cohort at 4, 8, and 12 weeks.
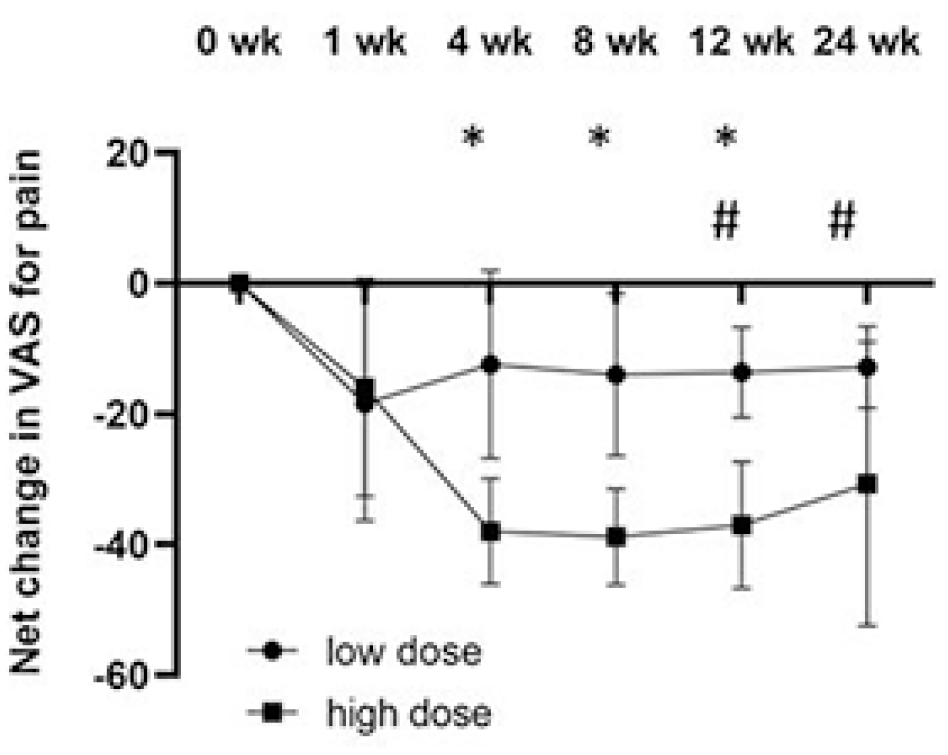
Net changes in VAS for pain from baseline to 24 weeks after intra-articular injection of GXCPC1. The data are presented as mean and error bars indicate standard deviation. VAS: Visual Analogue Scale. *, #: Sign of significant difference in the high-dose cohort and low-dose cohort, respectively.
The net change in SF-12 physical component summary (PCS) from baseline to post-treatment visit had an increasing trend in both cohorts (Fig. 4A). An increase in SF-12 PCS in the high-dose cohort was observed from 1 week to 24 weeks after GXCPC1 administration. On the contrary, we found no statistical differences in SF-12 MCS (mental component summary) over time in either cohort (Fig. 4B).
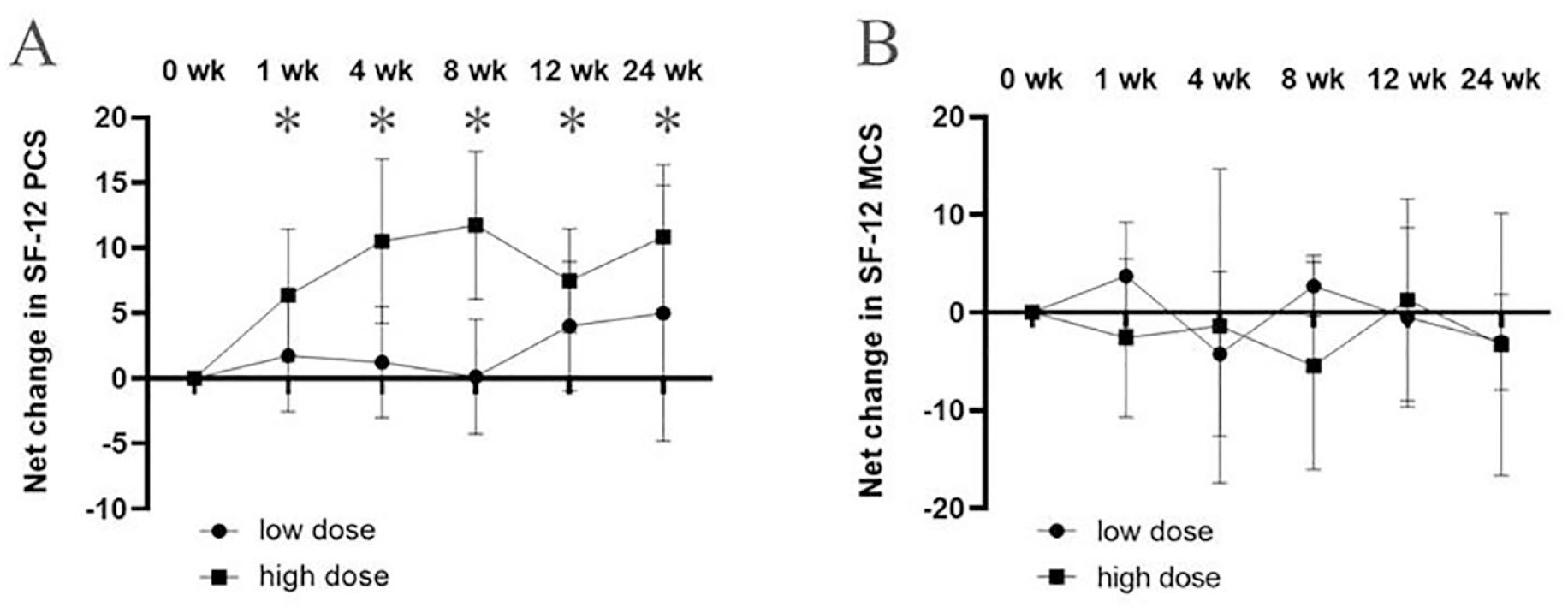
Changes in SF-12 during 24 weeks after intra-articular injection of GXCPC1. (A) PCS. (B) MCS. The data are presented as mean and error bars indicate standard deviation. MCS: mental component summary; PCS: physical component summary. *: Sign of significant difference in the high-dose cohort.
Discussion
This study preliminarily confirmed that intra-articular injection of GXCPC1 was safe and well-tolerated in subjects with knee OA. Based on a phase I clinical trial, this study focused on treatment-related adverse events in subjects. We observed that the most common complication related to the allogeneic ADSCs was injection site pain in 40% subjects in the low-dose cohort and 100% subjects in the high-dose cohort. All the treatment-related adverse events were graded as mild or moderate and they all soon subsided. These transient side effects were also observed in the other reported clinical trials19–22. Perhaps, physiological responses in the joint cavity or anti-histone H2B antibody production were resulted after intra-articular injections with ADSCs5,29. Moreover, the allogeneic ADSCs had already been proven to be safe in numerous clinical trials19–22. A systematic review of literature discovered only four SAEs in 844 intra-articular MSCs procedures, including one infection, one pulmonary embolus, and two unrelated events to the intervention 30 . In our study, no severe adverse events related to the GXCPC1 were reported over the 48-week study period. Overall, this study proved that GXCPC1 for knee OA treatment was safe and well-tolerated.
These results of WOMAC scores, VAS score, and SF-12 implied that better results might be achieved with an intra-articular injection at the dose of 4 × 107 GXCPC1. The choice of optimal cell numbers for intra-articular injection was challenging and varied across studies. One clinical study used higher cell doses of up to 1 × 108 ADSCs, and the study found that at least 1 × 108 ADSCs per injection was required to reduce cartilage defects through regeneration 16 . Chen et al. reported pain significantly decreased using 1.6 × 107 or 3.2 × 107 or 6.4 × 107 ADSCs; especially, 3.2 × 107 ADSCs had the best effect on pain relief among them, but no cartilage regeneration was observed 20 . Lu et al. indicated two intra-articular injections of 1.0 × 107, 2.0 × 107, and 5.0 × 107 ADSCs, and only 5.0 × 107 ADSCs significantly decreased VAS at week 12 and 48, while no significantly decreased WOMAC at week 24 and 48. Interestingly, there was no significant trend toward increased cartilage in 1.0 × 107 ADSCs 22 . Some reported that the high MSCs injection (1 × 107) generated free bodies of scar tissue in the rat joint which may cause considerable dysfunction of the joints, whereas others reported that sufficient cell density is required for rabbit cartilage production8,11. In our study, significant differences in WOMAC pain score were reported from baseline to week 1, 4, 8, and 12 while the differences in VAS were found at week 4, 8, and 12 in the high-dose cohort. Only VAS at week 12 and 24 showed significant differences from baseline to post-treatment in the low-dose cohort. According to the results of safety and preliminary clinical efficacy, high-dose cohort conferred an improvement in pain and functional scores.
The preliminary clinical efficacy results confirmed that GXCPC1 could improve HRQOL in OA patients according to SF-12 results. The SF-12 is a self-reported questionnaire to assess the impact of HRQOL on disease status. Previous studies used SF-12 to assess HRQOL in OA patients31–34. This trial intended to enroll both genders but subjects were all female. The result showed that the population of OA was predominantly female. According to gender differences in the 911 elderly Taiwanese individuals with knee OA, significant differences in SF-12 PCS were reported while no statistical differences in SF-12 MCS were observed in Taiwanese females with knee OA. On the contrary, significant differences in SF-12 MCS were found while no statistical differences in PCS were observed in Taiwanese males with knee OA 35 . This report indicated the presentation and natural history of condition may be different in male and female groups of Taiwanese. This result aligns with our study. In our study, an increase in SF-12 PCS in the high-dose cohort was observed from 1 week to 24 weeks after GXCPC1 administration. On the contrary, we found no statistical differences in SF-12 MCS over time in either cohort (Fig. 4B). By contrast, neither SF-12 PCS nor MCS found significant improvement after bone marrow MSCs treatment in knee OA patients’ knee 33 . Another study indicated a significant reduction was observed in SF-36 at 6 and 12 months in knee OA patients’ knee 17 . Therefore, the preliminary clinical efficacy results confirmed that GXCPC1 could improve HRQOL in OA patients. However, due to the small size and gender of clinical trials, more scientific evidence is needed to confirm the therapeutic effect of GXCPC1.
Robert et al. indicated that the types of performed procedures and the choice and processing adipose tissue derivates are high heterogeneity 24 . Bi et al. also reported that different processing methods of adipose tissue should be explicitly explained with application in orthopaedic 23 . GXCPC1, an expanded ADSCs product, is dissociated with enzymes and the resultant dispersed cells were collected and cultured according to our published methods. Flow cytometry immunophenotyping analysis revealed that GXCPC1 was positive (≥95%) for the surface markers CD90, and CD105, but essentially negative (≤2%) for CD34 and CD45 (data not shown). Moreover, GXCPC1 that meets regulatory requirements was shown to be safe, well-tolerated, and efficient in knee OA patients’ knee in our study. However, the main limitation of this study was the small number of subjects (11). In addition, it was not randomized and did not include a control arm (placebo). However, this study showed that intra-articular injection of ADSCs was safe and well-tolerated in subjects with knee OA. Nonetheless, a larger scale study with an appropriate control would be necessary for clinical efficacy in the following study.
Moreover, many studies have reported that ADSCs treatment can improve pain, swelling, stiffness, and function of the knee in patients with KL grading I-III, and even KL grading IV. Both Chahal et al. 14 and Kuwasawa et al. 36 showed that MSCs improved pain, stiffness, symptoms, and quality of life relative to baseline in patients with late-stage KL knee OA. In addition, some patients with KL grading IV are unwilling to undergo arthroplasty. Therefore, we would like to know whether GXCPC1 can improve symptoms in patients with late-stage KL knee OA or not. In this study, we recruited one patient with KL grading IV and this patient with KL grading IV in this study was special. According to her X-ray, this patient’s knee was diagnosed as KL grading IV. However, the WOMAC pain score and VAS of baseline were 8 and 14, respectively. Perhaps, this patient seems relatively insensitive to pain. Therefore, all pain questionnaire scores are not high. Even though all pain questionnaire scores are not high, after GXCPC1 treatment, the improvement of WOMAC total score and VAS from baseline to post-treatment was observed (3 and 14, respectively) at 24 weeks. This patient completely improved painful symptoms from the VAS questionnaire score after GXCPC1 treatment. Therefore, GXCPC1 has therapeutic potential for OA patients with KL grading IV from these results. In summary, GXCPC1 therapy was found to be safe and effective for treating knee OA subjects. GXCPC1 groups at low or high doses demonstrated a safety profile with a long duration of the pain relief. GXCPC1 administration is low risk, and these findings support the continued development of GXCPC1.
Footnotes
Acknowledgements
We appreciate Gwo Xi Stem Cell Applied Technology Co., Ltd for the sponsorship. We also like to express appreciation to all the subjects and the medical professionals for their participation in the study.
Authors’ Note
This study was done in Taipei Veterans General Hospital, Taiwan.
Author Contributions
C.-F.C., S.-W.T., P.-K.W., C.-M.C., W.-M.C. and H.-T.H.W. contributed to the research design and wrote the manuscript; Y.-S.F. drafted and revised the work; Y.-C.C. was responsible for the research execution, data collection, and data analysis. C.-H.L. contributed to the research design, data curation, validation, project administration, investigation, supervision, writing-review and editing. P.-C.L. contributed to the conceptualization, resources, supervision, project administration, and funding acquisition. C.-L.C., Y.-C.K., and C.-H.C. contributed to the validation. M.-H.C. contributed to the conceptualization and funding acquisition. All authors have read and approved the final manuscript.
Availability of Data and Materials
The data sets used/or analyzed are available by the corresponding author on reasonable request.
Clinical Trial Notation
Allogeneic Adipose Tissue-Derived Mesenchymal Stem Cells (GXCPC1) for Knee Osteoarthritis (NCT03943576).
Ethical Approval
Ethical aspects of the study were approved by the Research Ethics Committee of Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (IRB No: IRB106-88-A) and the Institutional Review Board of Taipei Veterans General Hospital (IRB number 2018-10-005C).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
All the patients provided informed consent for inclusion in the study.
Research Ethics and Patient Consent
All procedures involving human subjects were conducted in accordance with policies adopted by the Research Ethics Committee of Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (IRB No: IRB106-88-A) and the Institutional Review Board of Taipei Veterans General Hospital (IRB number 2018-10-005C).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Gwo Xi Stem Cell Applied Technology Co., Ltd., Hsinchu, Taiwan.
