Abstract
We did a clinical trial to determine whether olfactory mucosa lamina propria (OLP) transplants promote regeneration and functional recovery in chronic human spinal cord injury (SCI). The trial randomized 12 subjects to OLP transplants (n = 8) or control sham surgery (n=4). The subjects received magnetic resonance imaging (MRI), electromyography (EMG), urodynamic study (UDS), American Spinal Injury Association impairment scale (AIS), and other functional assessments. OLP-transplanted subjects recovered more motor, sensory, and bladder function compared to sham-operated subjects. At 3 years after OLP transplant, one patient improved from AIS A to C and another recovered from AIS A to B, two recovered more than three segmental sensory levels, two had less spasticity, two had altered H-reflexes and SSEP, two regained bladder and anorectal sensation and had improved bladder compliance on UDS. OLP-treated patients had partial or complete tissue bridges at the injury site compared to cavitary gaps in sham-operated patients. The limited recovery suggests that OLP transplants alone do not have significant benefits but may provide a rationale for larger randomized trials or combination therapies.
Keywords
Introduction
The currently available clinical trials on spinal cord injury with cellular transplantation had limitations with respect to the study design, ethical issues, number of participants, and/or duration of follow-up. However, there are no reports on prospective, randomized, double-blind studies in clinical trials for olfactory tissue transplantation. Autologous olfactory ensheathing glial cells (OECs) have been used to promote spinal cord repair (39,46,47,51) and can be extracted in a relatively simple and reliable way from the olfactory mucosa (31,35,42). These cells have similar properties to Schwann cells and promote and guide axonal growth during development (15). They have the unique property of crossing from the peripheral nervous system (PNS) to the central nervous system (CNS) (19), and olfactory mucosa containing OECs are thought to be useful in neural repair (35-37,57). In addition, the growth factors secreted by OECs have been shown to support neuronal multiplication and differentiation (16,43,56). Studies have suggested that olfactory mucosal tissue transplantation has similar outcomes to the transplantation of purified populations of OECs (2,36). A few recent animal studies demonstrated that the transplantation of olfactory mucosa containing OECs could repair the injured spinal cord (26,35-37). Olfactory laminae propria transplants have also been shown to reduce posttraumatic cavity size, increase the sprouting of neurofilament- and serotonin-positive axons, and stimulate the autonomic nerve system (48).
Intraspinal OLP transplants have been used to restore intraspinal circuitry or to serve as a “bridge” for damaged axons or substrate for spinal axonal regeneration (31,35-37). Three of the few published human clinical trials used cultured fetal OEC transplantation (22-24,51), one was a phase I/II trial using cultured autologous OECs (40), and another was a pilot study that demonstrated the safety and feasibility of autologous olfactory mucosal transplant (32). The advantages of using autologous olfactory mucosal transplants as a source of olfactory ensheathing cells and other nerve growth factors to enhance neurological recovery after SCI have been reported (30), and the use of autologous transplant has been shown to reduce the complications such as rejection, overgrowth, disease transmission, and ethical issues (32).
The present study investigates the outcomes of autologous olfactory mucosal transplantation in chronic, complete spinal cord injuries following partial scar excision and adhesion release compared with similar patients without transplants. In this study, the olfactory lamina propria was prepared for transplantation by removing the epithelial layer that contains progenitor cells, to test the unique role of olfactory mucosa OECs in spinal cord regeneration and functional recovery.
Recently, clinical studies of OECs or olfactory mucosa (OM) transplantation in patients with SCI were reported (40,51). A phase I/II trial of autologous OEC transplantation in patients with SCI confirmed the safety of the procedure, but did not assess functional recovery (13,40). A phase I/II trial of whole-layer OM transplantation in human SCI has also been reported (32). However, these two trials were uncontrolled, nonblinded cases, which are different from our controlled, randomized, double-blind trial.
Materials and Methods
Participants
This RCT of autologous olfactory mucosa lamina propria transplantation for chronic spinal cord injury was approved by the Ethics Committee of the Second Teaching Hospital, The Wenzhou Medical College, according to Guidelines of the National Health Department of China [registration No.: wzyxy 2009 (034)]. It was also approved by the Chinese Clinical Trial Registry and monitored by our University Ethics Committee (Registration Number: ChiCTR-TRC-09000482, ChiCTR on following internet link: http://www.chictr.org/cn/proj/show.aspx?proj=917). This work is supported by the Wenzhou Medical University (clinical research funding: XNK05013), Zhejiang, China.
According to the protocol, patient selection criteria included a chronic prior paraplegic or tetraplegic SCI of more than 6 months. The inclusion criteria were that the patient had to be a grade A on the ASIA Impairment scale, age less than 50 years, presence of cervical or thoracic spinal cord lesion, absence of significant nasal and paranasal sinus pathology, and an absence of an additional serious medical problem, such as brain disease or psychological disorders. Exclusion criteria included substance abuse, spinal vertebral instability, major concurrent medical illness (e.g., carcinoma, autoimmune disease, diabetes mellitus), and an ASIA Impairment scale grade higher than A. Patients with major and current psychiatric illness who had a significant traumatic brain injury associated with their spinal cord injury were excluded. All patients were informed of the RCT, its potential risks, and signed a consent form in order to be assigned to one of the two groups: either to receive an autologous OLP transplant or to the control group (Fig. 1). During the 6 months of screening, candidates for this clinical trial were selected based on the patient's willingness to continue to accept treatment and fulfill follow-up, constant sensory and motor function loss with an ASIA score of A, and a definite cavitary cord lesion on MRI. Ultimately, 12 patients who met the criteria were enrolled in this clinical trial study (10 men and 2 women).

Flow chart for patient recruitment, assessment, and follow-up.
The mean age of the participants was 32.9 ± 8.9 years. Other demographic data, clinical, imaging/radiological characteristics of the participants, and the dates of operations are presented in Tables 1 and 2. The causes of SCI include falls from height in six cases, motor vehicle accident in four cases, and knife stabbing injury in two cases. The cord lesion size was not more than 5 cm on preoperative MRI measurements in longitudinal section images. Twelve patients were randomized into two groups according to the patient's registration number and the number from the random number table. Eight patients were randomized for OLP transplantation, and four were randomized for the control group. The RCT was started at 6 to 83 months after SCI.
Demographic and Clinical Features of the Patients

Patients 2, 8, 10, and 11 were randomly selected as control group; the rest of them were in the OLP transplantation group.
Parameters for Clinical, Electrophysiology, MRI, and Other Assessments

ASIA, American Spinal Injury Association; FIM, function independent measure; MAS, modified Ashworth scale; MRI, magnetic resonance imaging; EMG, electromyography; UDS, urodynamic study; ENTA, ear-nose-throat assessment.
Biopsy of Olfactory Tissue
Olfactory mucosa was taken from the patients' own nasal cavity by an ENT surgeon (B. Chen) using biopsy instrumentation through a nasal endoscope (STOZE Inc., Munich, Germany). Before this procedure, the location of olfactory mucosa was identified using cadaver dissection and immunohistochemistry studies of sectioned tissue until a reproducible biopsy specimen could be confidently harvested. Biopsies were taken from the nasal septum in the posterior superior region of the nasal cavity close to the cribriform plate, under visual guidance via a nasal endoscope. Three to four medium-sized (3 × 3 mm) biopsies of olfactory mucosa were collected via one naris of each patient. The tissue was excised through a nasal endoscope using ENT biopsy tools (STOZE Inc.) and placed immediately into a sterile container in cold normal saline (4°C). All subsequent procedures were performed under aseptic conditions. The olfactory tissue was rinsed repeatedly for 20 min with penicillin (Zhongnuo CSPC Pharmaceutical Co. Ltd., Shijiazhuang, China)/streptomycin (100 U/ml, 100 mg/ml, respectively) (Shandong Lukang Record Pharmaceutical Co. Ltd., Jining, China). However, if the patient had an allergy to penicillin, gentamycin 150 U/ml was used (Rui Pharmaceutical Co. Ltd., Zhejiang, Ruian, China). OLPs were carefully separated from the overlying epithelium under a dissection microscope (Zeiss S8, Kiev, Germany) using microsurgical scissors and forceps (Aesculap, B. Braun Melsungen AG, Tuttlingen, Germany), cut into pieces of 0.1-0.25 mm2, and kept in normal saline. A small amount of lamina propria tissue was sent for microbiology (culture bacteria/fungi/virus) and histopathological studies.
Surgical Technique
All patients had preoperative plain radiographs (Siemens Arcadis Orbic, Erlangen, Germany) and MRIs (Siemens Magnetom Avanto1.5T, Erlangen, Germany) to identify the level of the injury and to determine previous surgical decompression and internal fixation levels. The same surgeons (H. Z. Xu, H. Zhou, and J. Lu) performed all of the surgeries using general anesthesia with endotracheal intubation. The ENT surgeon first harvested the olfactory mucosa through a transnasal endoscopic approach with the patients in a supine position. After cleaning the nasal cavities with iodophors (Minsheng Pharmaceutical Co. Ltd., Hangzhou, China), a submucoperiosteal tunnel was created in the most posterior-superior region of the medial (septal) side of the olfactory groove, and sufficient tissue was biopsied to be used to fill the spinal cord cavity and for microbiology and immuno-histology studies. Nasal packing (IVALON) was tightly packed into the nasal cavity to avoid postoperative bleeding. For control patients, the same preparation procedures were performed as above, but olfactory mucosa was not taken. However, a full-thickness cut was made along the nasal mucosa until bleeding began, and then the nose was packed with nasal packing (IVALON). The patient did not know if he or she had OLP transplants.
Subsequently, the patient was placed on the operating table in a prone position. The head was stabilized using a Mayfield frame (Schaerer Medical USA, Inc., Cincinnati, OH, USA). All internal fixation implants were removed before the spinal cord decompression and scar tissue release. Under surgical microscope, the damaged spinal cord was exposed using a standard midline incision, posterior laminectomy, and opening of the dura mater. The damaged spinal cord was approached by a posterior midline myelotomy or scarlotomy. If there were minimal cavity lesions, the scar tissue of the lesion sites were partially excised to accommodate OLP transplants. The cavitary lesion in the cord was opened, and the scar tissues from the proximal and distal stumps were partially or minimally excised before the olfactory mucosa tissue was transplanted into the cavity. No normal cord tissues were handled during the operation. For controls, the same surgical procedures were performed without transplants. The meninges and the dura matter were then closed. The patient was then transferred to the surgical ward with standard spinal postoperative care.
Selection Criteria and Initial and Repeated Assessments
The ASIA score system (14,41) was used for preoperative and postoperative assessments to determine the differences between the ASIA neurological examination component scores (sensory pin prick, sensory light touch, motor upper and lower limbs) in OLP transplantation and control groups. ASIA scores were recorded at 12, 24, and 36 months after intervention and analyzed with Microsoft Excel 2003.
The severity of increasing spasticity/muscle tone was graded into five levels (0-4) according to the modified Ashworth scale (7).
The International Association of Neurorestoratology Spinal Cord Injury Functional Rating Scale (IANR-SCIFRS) was also used for preoperative and postoperative assessments, as it is another method that is popular among some researchers (12,21,25).
Each patient had a postoperative MRI at 12, 24, and 36 months to assess infection, hemorrhage/hematoma, cyst/syringomyelia, tumors, and increasing myelomalacia. With the exception of signal changes at the injury sites, the quantification of the cross-sectional area at the proximal and distal stumps for pre- and postintervention were performed using the PACS 1.5 image measurement programs (INFINITT PiviewSTAR PACS V3.113, Seoul, Korea).
Standard conventional electromyography (Keypoint; Dantec Electronics LTD, Skovlunde, Denmark), including H-reflexes (EMG), were performed for each group at each follow-up time point. SSEP was recorded pre- and postoperatively in the cortex of all patients following tibial nerve stimulation. H-reflex and SSEP were recorded by standard conventional electromyography (Keypoint; Dantec Electronics LTD) pre- and postoperatively.
A urodynamic study was performed preoperatively in all patients and at 12, 24, and 36 months postoperatively. All urodynamic studies were conducted in the same manner, with the same equipment, and by a single physician and his project partners. The equipment used in the present study was a Canada Laibo Rui UDS-94-R01-BT urodynamic analyzer. Before testing, the patients' bladders were emptied by catheterization using a 10F catheter for men and an 8F catheter for women. The patients were placed in the lithotomy or sitting position. The bladder was then filled with normal saline at a constant infusion rate of 50 ml/per second. The intra-abdominal pressure, intravesical pressure, and detrusor pressure were recorded throughout the voiding cycles. During the test, the patient was also asked to cough or apply abdominal strain to evaluate detrusor instability. The first desire to void was recorded as soon as perceived by the patient. The test then continued until the patient reached full bladder capacity, at which time the infusion stopped, and the patient was asked to void, and the voiding pressure was recorded. The bladder was then emptied to determine the postvoiding residual volume (PVR). Continuous blood pressure (B/P) monitoring was initiated and maintained throughout the study. The patients underwent urodynamic testing regardless of bladder management methods and were analyzed for maximum urinary flow rate (Qmax), residual urine volume (residual volume, RV), maximum bladder capacity (MCV), bladder compliance, A-G values, and detrusor external sphincter synergy.
The return of anal sphincter contraction was qualitatively assessed in a rectal digital examination (0-normal; 1-mild; 2-moderate; 3-severe). Bulbocavanous reflexes were examined in each follow-up visit. The severity of bulbocavanous reflexes was semiquantified as severe, moderate, mild, and normal.
Postoperative instruction for a home exercise program was given to all patients. All patients were interviewed carefully at each follow-up time point after the intervention and questioned specifically to identify any illness or need for hospitalization and/or any subjective neurological changes in power, sensation, continence, and other uncomfortable complaints. All patients were asked to complete a function independent measure (FIM) form (28) at 1-, 2-, and 3-year follow-up visits. ENT assessment (ENTA) for olfactory function was carried out preoperatively and at 6 and 12 months after the intervention.
Study Design and Patient Follow-Up
The present study was designed as a prospective, randomized, and double-blind trial. The surgical procedures were performed by the same surgeons (H. Zhou, H. Xu, J. Lu) for all 12 patients, but those surgeons were not involved in follow-up studies. The surgeons (S. Wang, W. Ni, S. Zhu) who were involved in patient follow-up did not know which surgical procedures had been performed. Olfactory function was assessed clinically, and nasal mucosa healing (tissue donor region) was evaluated through an endoscopic examination at the 6-month follow-up.
Statistical Analysis
The Mann-Whitney test and Kruskal-Wallis test were used to analyze the data (SPSS 17.0; IBM, Armonk, NY, USA). Values of p < 0.05 were considered to be significant.
Results
The first case in the present study was started on May 20, 2009 and the last case on December 2, 2009. The mean time between the injury and the operation was 32.16 ± 23.52 months (minimum, 6 months; maximum, 83 months). The spinal cord injury level was consistent with the level of the vertebral fracture. In our patients, it was easy to identify the normal tissue from the damaged nervous tissue under a surgical microscope after carefully reviewing preoperative plain radiographs, CT scans, and MRI images. No neurological deterioration, surgical wound infection, and nasal biopsy-related complications were found postoperatively. Although there was no significant difference in the assessment of patients' quality of life with the FIM score system, three cases in the OLP group greatly improved their daily activities. However, there was no significant difference in the FIM scores between the OLP transplant and control groups at the preoperative and postoperative assessments in different follow-up time points. Increased sweating was recorded in three cases in the OLP transplant group, but none in the controls. No bacteria or other microorganism growth was found in the OLP tissues that were prepared from patients' own olfactory mucosa for transplantation.
ASIA Scores
All 12 patients were evaluated using ASIA scores at the preoperative assessment and at 12, 24, and 36 months after surgery. The results from the ASIA neurological examination are presented in Figure 2A-C. Patient 3 with T3 paraplegia displayed an ASIA score change from A to C with improvement in motor function of the hip flexors (grade III), hip adduction (grade II), and hip abduction (grade II). Improvement of sensory function was recorded in left side lower limb to S4 and left side of the perineal region that was absent prior to OLP transplantation. Patient 7 with T7 paraplegia had a return of minor motor and sensory function at S4-S5 (right side perineal region sensory recovery) and minor improvement in abdominal motor function (visible muscle contraction) below the level of injury down to T10. There was no sensory alteration in the saddle area in the other six patients in the OLP group. There were six patients who had sensory light-touch improvement with OLP transplantation 36 months after surgery, but this was not reported in the control group (Fig. 2A1). However, there were no significant differences (p = 0.99) for light touch sensation between the two groups at different follow-up time points (Fig. 2A2). There were six patients who had improvement in ASIA scores for pinpricks (Fig. 2B1); however, there were no significant differences (p = 0.99) for pinprick sensation between the two groups at different follow-up time points (Fig. 2B2). Patient 8, who had quadriplegia, displayed little improvement in upper limb motor function, while other patients' upper limb motor functions were normal preoperatively (paraplegia). There was also one participant (patient 3) who had an ASIA motor score improvement from A to C after OLP transplantation (hip flex 3/5, hip abduction 2/5, and hip adduction 2/5). There were two cases whose abdominal reflexes were returned 3 years after OLP transplantation (patient 3 and 7), but this was none in controls (Fig. 2C1). However, there was no significant difference in the ASIA motor scores between the OLP transplant and control groups (Fig. 2C2).
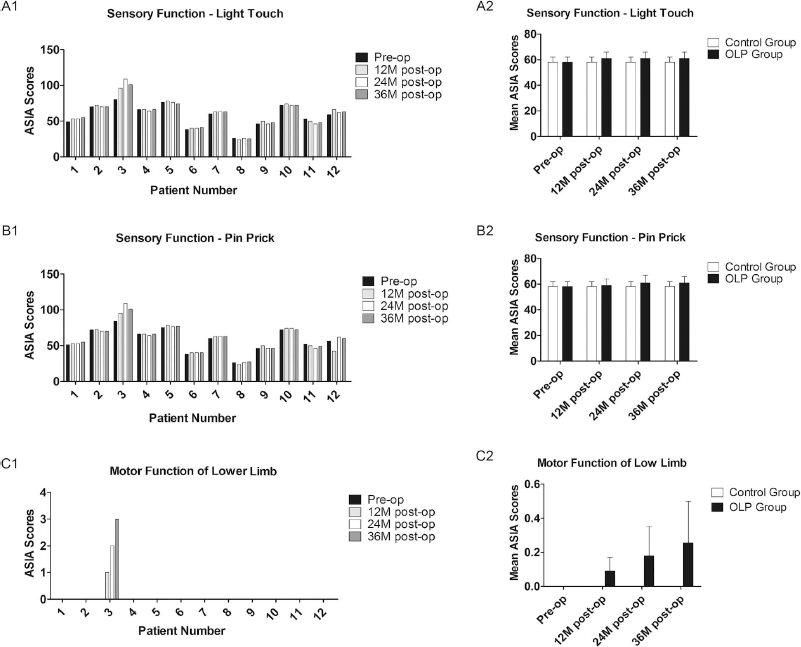
Bar graphs showing baseline (preoperatively), 12, 24, and 36 months postoperative ASIA scores for each patient. (A) The ASIA scores of light touch at scheduled follow-up. (A1) The bar graphs show the ASIA scores of light touch for each individual participant. There were five patients (patients 1, 3, 6, 7, 12) with sensory light touch improvement 12, 24, and 36 months after OLP transplantation but none in the control group (patients 2, 8, 10, 11). (A2) The bar graphs show the mean data of the ASIA scores of light touch at scheduled follow-up. (B) The ASIA scores of pinprick at scheduled follow-up. (B1) The bar graphs show the ASIA scores of pinprick for each individual participant. There were six patients (patients 1, 3, 5, 6, 7, 12) with sensory light-touch improvement 12, 24, and 36 months after OLP transplantation, but none in the control group (patients 2, 8, 10, 11). (B2) The bar graphs show the mean data of the ASIA scores of pinprick at scheduled follow-up. (C) The ASIA scores of motor function of lower limb at scheduled follow-up. (C1) The bar graphs show the ASIA scores of motor function of lower limb for each individual participant. There was one patient (patient 3) with motor function improvement 12, 24, and 36 months following OLP transplantation, but none in the control group (patients 2, 8, 10, 11). (C2) The bar graphs show the mean data of the ASIA scores of motor function of lower limb at scheduled follow-up.
In short, this study showed some sensory and motor recovery in the OLP transplant group compared with the controls, although there were no significant differences statistically between the two groups at different follow-up time points.
MAS
There was no significant reduction of spasticity in the OLP transplant group, which was similar to the control group. However, one case (patient 3) in the OLP transplant group had nearly normal muscle tone and a complete disappearance of spasticity at 12 months after surgery, and another patient in the OLP transplant group (patient 6) showed a slight reduction in spasticity 3 years after surgery. The reduction of spasticity and muscle tone was noticed by the patient and recorded by clinical examination in two cases in the OLP transplant group. Patient 3 showed normal muscle tone and with no spasticity 1 year following OLP transplantation, with the greatest improvement in motor and sensory functions in ASIA scores. In contrast, none of the control patients showed a reduction in spasticity at 1-, 2-, and 3-year follow-up visits. There was no statistically significant difference for modified Ashworth spasticity scores between the OLP transplant and the control groups at the different follow-up time points (p > 0.05).
IANR-SCIFRS
All 12 patients were evaluated preoperatively and 12, 24, and 36 months postoperatively for IANR-SCIFRS scores. The results from the IANR-SCIFRS scores are presented in Figure 3A and B. Patient 3 with T3 paraplegia was found to have a change in IANR-SCIFRS score from 20 to 28, patient 9 changed from 18 to 21, patient 10 changed from 26 to 27, patient 11 changed from 15 to 16, and patient 12 changed from 19 to 20. There was no statistically significant difference between the two groups (p = 0.50).

International Association of Neural Restoration Spinal Cord Injury Functional Rating Scale (SCI-FRS) for both OLP group and control preoperatively and 12, 24, and 36 months postoperatively. (A) The bar graphs show SCI-FRS scales for each participant. There were two patients (patients 3 and 9) with score improvement ≥ 3 after OLP transplantation but none in the control group. (B) The bar graphs show the mean scales of SCI-FRS for OLP and control group.
MRI
MRI was performed at 12 months after the transplantation and showed a partial or complete filling of the lesion site in all OLP transplant patients, but not the controls. The MRI showed that the grafted area had “peppers and/or cord-like signal” appearance (Figs. 4 and 5). In addition, there was no MRI evidence of neoplastic tissue overgrowth in any of the patients in this trial. Measurement of the cross-sectional areas of the proximal and distal stumps on preoperative and postoperative MRIs showed that the spinal cord stump atrophy was prevented in the OLP transplants group, but not in the controls (Fig. 6A1, A2; 6B1, B2). At the 36-month follow-up, there was no atrophy in the proximal [pre-operative cross-sectional area in 50th percentile (P50): 28.6 mm2, postoperative P50: 41.9 mm2, p = 0.02] and distal stumps (preoperative cross-sectional area P50: 28.2 mm2, postoperative P50: 39.3 mm2, p = 0.19) in the OLP transplant group, but attenuating cords were noticed in the proximal (preoperative cross-sectional area P50: 41.1 mm2, postoperative P50: 33.9 mm2, p = 0.97) and distal stumps (preoperative cross-sectional area P50: 28.7 mm2, postoperative P50: 27.2 mm2, p = 0.72) in the control. The statistical analysis did not reach significance between the OLP transplant and the control groups at different follow-up time points.

MRI images from different follow-up points. (A) Preoperative MRI images showed the posttraumatic syringomyelia cavity in thoracic spinal cord (arrow). (B) Twelve months after transplantation of OLP, the “pepper”-like signals visible in the cavity indicating viable transplants (arrow). (C) Twenty-four months after transplantation of OLP, reformation of the cavity in the cord and OLP transplants were migrating peripherally (arrow). (D) Thirty-six months after transplantation of OLP, enlargement of cavity in the cord was noticed, and clinically, this patient had limited sensory and motor function recovery in the final follow-up.
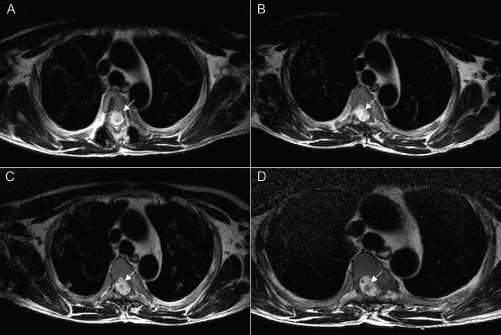
MRI images from different follow-up points. (A) Preoperative MRI showed a large posttraumatic cavity in the cord. (B) Twelve months post-OLP transplantation, “cord”-like MRI signals in the cavity. (C) Twenty-four months after transplantation of OLP, more “cord”-like signals in the cavity. (D) Thirty-six months after OLP transplantation, thick “cord”-like tissue signal in the previous cavity indicating spinal cord regeneration.

Bar graphs representing cross-sectional area of MRI measurements pre- and 12, 24, and 36 months postoperative ASIA scores. (A1) Bar graphs showed cross-sectional area of MRI of the proximal stumps for each individual participant. There was no atrophic tethering of the cord in the proximal stumps compared to controls at scheduled follow-up. (A2) Bar graphs show the mean data of cross-sectional area of MRI of the proximal stumps for OLP and control groups. (B1) Bar graphs show the cross-sectional MRI of the distal stump for each individual participant. There was no atrophic tethering of the cord in the distal stumps compared to controls at scheduled follow-up. (B2) Bar graphs show the cross-sectional area of MRI of the distal stumps for OLP and control groups.
SSEP and EMG Findings
There was no significant difference in semiquantified data for SSEP and H-reflex between two groups (p = 1.00 and p = 0.35, respectively). However, postoperative EMG changes were recorded in patient 3, who had voluntary control of skeletal muscles. This included abdominal muscles, hip flexors, and the large abductor and adductor muscles of the thigh, but most of their strength was less than 3 on the MRC grade. Pre- and postoperative SSEP were recorded and are presented in Figure 7. Bowel and bladder control for patient 3 was almost completely recovered, and voluntary anal sphincter contraction was also confirmed by rectal digital examination.
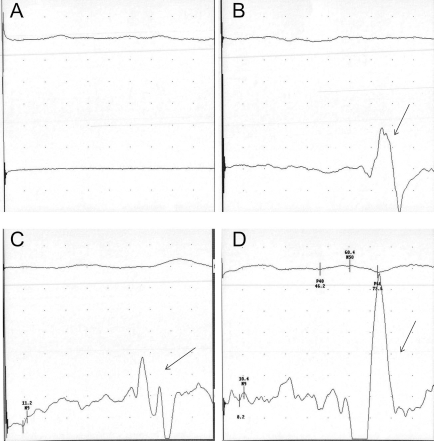
SSEP studies from different follow-up points. SSEP of patient 3, recorded pre- and postoperatively. There was no SSEP signal in the preoperative study (straight line after stimulation) (A). However, 12 months after the OLP transplantation, detection of the SSEP signal was recorded (arrow) (B). Twenty-four months postoperatively, SSEP signals were constantly present (arrow) (C). Thirty-six months postoperatively, the highest amplitude of SSEP was recorded (arrow) (D).
In patient 7, who had a knife stabbing transection injury of T7 cord, the H-reflex was suppressed 6 months after OLP transplantation (Fig. 8), even though he had no visible functional motor recovery.

H-reflexes from different follow-up points. High amplitude of H-reflex of patient 7 was recorded preoperatively (A). Twelve months post-OLP transplantation, the amplitude of H-reflex was significantly decreased compared with the preoperative recording (B). Twenty-four months postoperatively, the amplitude of H-reflex was maintained as in 12 months postoperatively (C). Thirty-six months postoperatively, the amplitude of H-reflex was consistently depressed (D).
Urodynamic Study
Urodynamic studies showed an improvement of bladder compliance in two cases (patients 3 and 7) in the OLP transplant group, but not in the controls (Fig. 9). Improvement of bladder function in the OLP transplant group at the 3-year follow-up included full and partial urination control, bladder sensation during urination, and reduced urinary frequency. Shortly after surgery (6 months), patient 3 reported a recovery of sensation when the bladder was full. Urodynamic studies revealed increased sensation, and desire to void was present when pressure was increased during bladder-filling cystometry. There were four patients who had residual urine volumes less than 50 ml 1 year after OLP transplantation, but only one in the control group. Patient 7 reported regaining sensation when the bladder was full, but postoperative urodynamic studies were inconclusive. Urinary tract infection occurred in two patients in the control group, and one in the OLP transplant group at the 1-year follow-up.

Urodynamic studies from different follow-up points. Patient 3 (A) and patient 7 (B) urodynamic studies showed most parameters were improved post-OLP transplantation. This included maximum cystometric volume (MCV); Abrams-Griffiths nomogram (AG); maximum flow rate (Qmax); residual urine volume (RUV); and bladder compliance (BC).
There was one patient whose preoperative bulbocavanous reflexes were reduced from severe to normal at the 3-year follow-up (patient 3), two patients from severe to mild, and two patients from severe to moderate in the OLP transplant group, but only one in the controls was reduced from severe to moderate. Although significant differences could not be found (p = 0.19), clinical improvement of bulbocavanous reflexes and anal tones were noticed in five patients in the OLP transplant group.
Adverse Events
Patient 10 had CSF leakage and watertight soft tissue and skin closure was performed 1 week following the surgery. CSF leakage was completely stopped 2 weeks after surgery. Patient 3, who had motor and sensory recovery at 1 year, developed hypersensitivity and an itching sensation of his scrotum, and superficial ulceration over the scrotum that had healed at the final follow-up. Posttraumatic syringomyelia was underestimated in patient 12, who had OLP transplantation, on preoperative MRI, while postoperative MRI showed an enlarged syrinx cavity that was similar to the preoperative size. There were two patients with OLP transplantation who had progressive postoperative enlargement of the syringomyelia cavity. Depression was observed in patient 2 (control) 1 year after surgery.
No adverse findings were observed with regard to increasing spasticity, autonomic dysreflexia, or temperature control disturbance. There were no detrimental postoperative nasal complaints. Olfaction was returned to normal in all patients within 6 months following surgery.
Discussion
The lack of axonal regeneration in the adult central nervous system has been attributed to a variety of factors, including a nonpermissive environment like the glial scar, the presence of inhibitory factors, and the lack of trophic support (35,36). Both animal experiments and clinical applications have demonstrated that olfactory ensheathing cell transplantation assists in spinal neurofunctional recovery (29,44,51,53,55). Several recent clinical trials have reported the potential therapeutic role of olfactory ensheathing cells for the repair of human chronic spinal cord injuries (13,17,31,32,40,47). In these clinical trials, ensheathing cells were cultured from either the olfactory lamina propria (40) or olfactory bulbs from the embryo (22,25) or direct transplantation of olfactory tissue from nasal olfactory mucosa (2,11,31,32). In human spinal cell or tissue transplantation, there are major ethical and technical problems associated with the use of embryonic tissue or tissue from olfactory bulbs of adult donors. A craniotomy procedure to remove the olfactory bulb itself may not be desirable and acceptable in humans. Furthermore, there may be difficulty in obtaining sufficient quantities of OECs from olfactory bulbs to produce an effective repair of the damaged spinal cord; this has restricted the use of this approach in humans. The olfactory lamina propria in the nose is an accessible source of OECs by performing a simple biopsy through the external nares. The OECs are taken from the anterior and posterior portion of the nasal cavity, to be used for autologous transplantation and provide a cellular bridge to join the gap between the damaged ends of the transected spinal cord. This approach avoids the need for an artificial material bridge (35,36). Peripherally derived OECs appear to have characteristics similar to those of olfactory bulbs in promoting axonal regeneration. It is well known that olfactory tissue shows a high level of nerve growth factor and neurotrophic receptor expression (10,33), and circumstantial evidence indicates that OECs secrete NGF (8,34). Therefore, OECs appear to provide a highly favorable substrate for axonal regeneration, partly because they secrete extracellular matrix molecules and neurotrophic factors (16,56). The current study also demonstrated that biopsy of lamina propria of the olfactory mucosa does not affect the sense of smell 6 and 12 months postbiopsy that is consistent with previous studies (3,6,18). Thus, the most distinct advantages of peripherally derived OECs are their accessibility from the patient's own olfactory mucosa in the nasal cavity, which can be transplanted in solid pieces as a “bridge” and rich source of OCEs, and eliminating problems of rejection or the need for immunosuppression. All of these advantages support a potential therapeutic role for peripherally derived OEC tissue implantation that was used in the present study for human chronic spinal cord injury (21,36-38). However, because Schwann cells from the trigeminal nerve are also innervating the olfactory mucosa (6) and p75 protein can be expressed by some nonmyelinating Schwann cells (4), we could not completely exclude a contribution of Schwann cells in our OLP tissue, although the epithelial layer of olfactory mucosa had been completely removed before OLP transplantation. The aim of the present RCT study was to determine whether grafts of the olfactory lamina propria from the patient's own nose could promote repair of chronic human spinal cord injury.
The current RCT using autologous OLP is designed as a randomized, controlled, double-blind prospective study that is different from previous studies (17,32,40). All 12 patients were pre- and postoperatively (12, 24, 36 months) evaluated using the ASIA score system. Patient 3 improved from an ASIA score of A to C and exhibited the best recovery in the OLP transplant group. The sensory level was lowered to L4 from preoperative level of T3 that was recorded at 4 weeks post-OLP transplantation. Motor recovery was noticed from 3 months post-transplantation, becoming more significant at 12 months and plateauing at 24 months. The mechanism of early recovery is thought to be related to surgery such as opening posttraumatic syrinx reducing pressure on adjacent residual neurons, in which transplants provide an awakening stimulus to dormant neurons for roles in functional recovery (24). It has been well known that OECs release BNDF and other NGFs (56) to induce the formation of new local synapses. In addition to their neuroprotective roles, they also promote axonal regeneration from intact fibers to connect to the residual neurons in the distal stump and form novel local neuronal circuits that may contribute to functional recovery. In addition, OECs are able to penetrate the scar tissues, which may facilitate and activate spinal circuits in more timely and effective manner. They may also activate novel circuits from regenerating fibers through scar tissue that activates local propriospinal neurons to form a novel ascending/descending pathway.
The postoperative MRI images in our study demonstrated that the transplants in the OLP group bridged the proximal and distal stumps, which may provide assistance in establishing the integrity of the local neuronal signal circuit and conducting neural signals for early motor or sensory recovery.
The scar that formed after SCI had an inhibitory effect on CNS axonal regeneration. The scar tissue that was partially resected in the present study is based on the capability of OECs to penetrate scar tissue and promote axonal regeneration in this inhibitory environment (35,36,45). In addition, partial excision of the scar tissue may minimize damage to the normal cord, such as postoperative sensory or functional deterioration or triggering myelomalacia changes. Functional deterioration was not detected in our patients either immediately after surgery or in the long-term follow-up (up to 36 months), likely due to careful surgical dissection and handling, which was focused at the damaged spinal cord tissues (cavity or scar) rather than on interfering with the normal spinal cord. Therefore, a partial resection of the scar tissue to accommodate the OLP graft tissue and to bridge the spinal cord above and below the injury site without harming the normal cord tissue is feasible and practical for accommodating and encouraging the grafts to integrate with the proximal and distal stumps.
MRI scans at 12 months after transplantation revealed partial or complete filling of the cystic cavity with neural tissue appearance signals at grafted areas. The obliteration of the cavities seems to indicate that the transplants had survived. At the 36-month follow-up, there was no atrophy of the proximal and distal stumps in the OLP transplant group, but attenuating cords were noticed in the proximal and distal stumps in the control, which is consistent with a previous report (20). The lack of atrophy of the proximal and distal stumps may be associated with the secretion of neurotrophic factors and neural signaling by olfactory tissues to maintain the structural integrity of the cords. Because of the small number of patients in the present study, statistical analysis did not reach significance between the two groups in proximal and distal stump cross-sectional area measurements (p=0.31 and p=0.17).
Four out of eight patients in the OLP transplant group showed some improvement in either motor or sensory function, although the statistical analysis was not significant. Nevertheless, the number of the patients enrolled in this RCT was relatively small; it seems unlikely that consistent recovery would be detected in patients with a chronic, severe SCI in the OLP transplant group. Most of the patients exhibited improvements within the first 12 to 24 months after surgery, and slowly increased their functional recovery, which plateaued 24 months after surgery. Overall, there appeared to be greater improvement in sensory function rather than motor function in the ASIA score assessment. Normalized bladder function in one patient and the return of bladder sensation in two of the eight patients at 12 months after OLP transplantation may reflect some type of long-term reorganization of long tracts in spinal cord. In one patient (patient 3), the return of voluntary anal sphincter contraction, as detected by the patient or clinical examination, occurred more than 6 months after OLP transplantation. There was no alteration in bladder function in the controls, which may indicate that a spontaneous return of bladder sensation or bowel control is not possible; therefore, partial bladder functional return may be related to the OLP transplantation. The urodynamic study demonstrated that bladder compliance had improved in three patients in the OLP group, but none in the controls. Therefore, the normal urinary function assessment in the urodynamic study may indirectly reflect the recovery of neurological function. From the patients' perspective, the limited changes in motor skills but any changes in bladder sensation and/or bowel function had the most significant impact on their daily lives. Of the eight patients in the OLP transplant group without bladder sensation at preoperative evaluation, three had regained the ability to sense bladder fullness (one of them regained nearly full bladder control).
Spasticity is a common problem after SCI and results from increased reflex activity that develops following the SCI and may contribute to pressure sores, the cause of joint contractures, preventing appropriate positioning, and may mask recovery of muscle function in rehabilitation. Reduction of spasticity and muscle tone in two cases in the OLP transplantation group in this study greatly facilitated the patients' transfer from the bed to the wheelchair and other daily activities.
Hyperreflexia is a well-known consequence of spinal injury in both animals and humans (9,53). Rate-sensitive depression in the normal cord primarily reflects presynaptic inhibitory mechanisms (34). Such inhibition is known to be under the influence of descending controls from the cortex and brainstem (1,27,49), and the reduction of rate-sensitive depression after cord injuries is considered to reflect the loss of these descending influences (52,53). The rate-sensitive reflex depression in H-reflexes in two patients with OLP transplants may be associated with descending motor pathways that were partially restored following autologous OLP transplantation.
However, although animal models of spinal cord injury are designed to develop new therapies (35,36), functional recovery will not necessarily translate into recovery for humans because of the many differences in anatomy and injury mechanisms between animals and humans. In our patients, olfactory tissue transplantation to bridge the proximal and distal stumps of a small cavity was easily achieved either from intraoperative findings or demonstrated on postoperative MRI images (Fig. 5). However, for large gaps, the OLP transplants cannot fully cover/bridge the length of lesions, which may be associated with poor functional recovery. The cause of postoperative enlargement of the syringomyelia cavity is unknown, but may be related to poor functional recovery outcomes (30).
Therefore, optimal outcomes may be related to age, the severity and level of the injury, the quality and quantity of transplants, surgical technique, and postoperative rehabilitation. The expectation of transplanting olfactory mucosal cells into spinal cord injury patients would be associated with clinically detectable effects, but it is less likely that patients would experience useful benefit in their everyday lives (5). Therefore, using OLP transplant as a sole treatment would be less likely to achieve clinical benefit for spinal cord injury patients. However, the effects of OLP transplantation on spinal cord function recovery in this RCT might provide some worthwhile insights if the olfactory mucosal tissue transplants are to be part of a multimodal intervention and is more likely to be developed as a future therapeutic strategy (50,54).
In some previous human studies, olfactory mucosal cell transplantation has been accompanied by rehabilitation therapies (12,13,31), which in itself may have a beneficial effect on outcomes of functional recovery and may thus confound the interpretation of the origin of the observed effects. In the current RCT study, physical exercises or rehabilitation programs had not been initiated after the patients were discharged from the hospital because of financial difficulties and other major social disadvantages.
Conclusions
This RCT study first demonstrated that transplantation of OLP affected motor functional recovery and improvement in ASIA sensory scores, bladder compliance, sensation and partial control, and daily life activities. Our study indicated that OLP therapy is promising and safe for chronic spinal cord injury, although a larger number of patients are required in a future clinical trial for final clinical implication. We believe that for clinical benefits of OEC transplants, a combination with other pharmacological agents is most likely to achieve significant axon regeneration and reestablish functionally useful connections across the injured spinal cord.
Footnotes
Acknowledgments
We thank Ms. Qing Yu for her assistance in the study design and statistic analysis, Bei-Lei Hu and Xun Wang for their assistance in EMG study, and Qian Wang for the urodynamic study, and appreciate Dr. Zhi-Kang Yu for his great help in MRI image data acquisition and analysis. The project (No: XNK05013) was sponsored by the Wenzhou Medical University, Zhejiang, China. The authors declare no conflicts of interest.
