Abstract
Schwann cells (SCs) are the main glial cells of the peripheral nervous system, which can promote neural regeneration. Grafting of autologous SCs is one of the well-established and commonly performed procedures for peripheral nerve repair. With the aim to improve the clinical condition of patients with spinal cord injury (SCI), a program of grafting autologous activated Schwann cells (AASCs), as well as a series of appropriate neurorehabilitation programs, was employed to achieve the best therapeutic effects. We selected six patients who had a history of SCI before transplantation. At first, AASCs were obtained by prior ligation of sural nerve and subsequently isolated, cultured, and purified in vitro. Then the patients accepted an operation of laminectomy and cell transplantation, and no severe adverse event was observed in any of these patients. Motor and sensitive improvements were evaluated by means of American Spinal Injury Association (ASIA) grading and Functional Independence Measure (FIM); bladder and urethral function were determined by clinical and urodynamic examination; somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) were used to further confirm the functional recovery following transplantation. The patients were followed up for more than 5 years. All of the patients showed some signs of improvement in autonomic, motor, and sensory function. So we concluded that AASC transplantation might be feasible, safe, and effective to promote neurorestoration of SCI patients.
Keywords
Introduction
Fracture/dislocation of the spine often leads to laceration of the spinal cord and a loss of axonal connection as well as motor and sensory function. Studies over the past two decades have shown that axonal growth after spinal cord injury (SCI) can occur once provided with the appropriate microenvironment. In that case, tissue transplantation or peripheral nerve grafting is usually taken to repair damaged or diseased regions of the central nervous system (CNS). However, donor shortage and immunological and ethical problems may appear at the same time.
During Wallerian degeneration, Schwann cell (SC) responses initiate, and several growth factors as well as cytokines that facilitate regeneration of injured peripheral axons (4,16,17) are synthesized and secreted. Then activated SCs produce a high level of growth factors including brain-derived neurotrophic factor (BDNF), glial-derived neurotrophic factor (GDNF), ciliary neurotrophic factor (CTF), nerve growth factor (NGF), neurotrophin-3 (NT-3) (12,22,28), insulin-like growth factor (IGF-1), and erythropoietin (13,14,19). On the other hand, SCs can be easily obtained from a piece of peripheral nerve of a patient with SCI, cultured in vitro (18), and transplanted into the patient's SCI area. So transplantation of the autologous transplantation of SCs is feasible without immunological and ethical problems.
In the past experimental research, we compared three different grafting routes of autologous activated Schwann cells (AASCs) (intravenous, intrathecal, and intraspinal cord), which demonstrated that direct transplantation of AASCs directly into the spinal cord may be one of the most promising transplantation approaches to injured spinal cord (2). Bachelin et al.'s experiment in monkeys also indicated that SCs can promote functional and anatomical repair after its transplantation in the lesioned spinal cord (1).
We selected six patients with a history of SCI to observe the clinical result of AASC transplantation.
Materials and Methods
Participants and Preparation before Operation
General information of the six patients is shown in Table 1. Clinical evaluation, conventional laboratory examination, and imaging (X-rays, CT, and MRI) were taken in our treatment plan. Before the treatment, all the patients signed a written informed consent, which had been reviewed and approved by Institutional Ethics Committee and notarized by a lawyer.
General Information of the Six Patients

ASIA, American Spinal Injury Association.
One week before the activation of SCs, we used American Spinal Injury Association (ASIA) score, Functional Independence Measure (FIM), and magnetic resonance imaging (MRI) to evaluate preoperative function of these patients. Then sural nerve ligation in one lower extremity was performed 1 week before the operation in order to activate the SCs (Figs. 1–4).

Isolation of sural nerve before the operation.
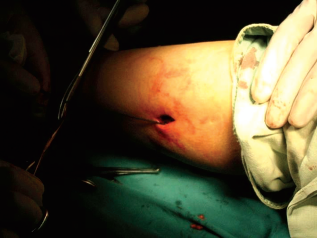
Free sural nerves.

Removal of the sural nerve.

Digestion of the sural nerve and isolation and culture of Schwann cells.
AASC Culture and Purification
One week after ligation, the sural nerve was dissected out, the epineurium and connective tissue were removed under a microscope, and then the nerve was cut into 2-to 3-mm fragments and dissociated with 0.25% trypsin for 15 min at 37°C. After that, the tissue blocks were put in six-well plates in DMEM medium containing 10% fetal bovine serum. Cells were incubated at 37°C in 5% CO2. Medium was changed every 2–3 days and the cells were purified by differential adhesion. After five passages, the purity of AASCs was identified by S-100 immunostaining.
Operation and Functional Exercises
Under general anesthesia with the patients positioned prone, the patients were treated with surgical decompression and stabilization of the spine (anterior or posterior). Careful dissection was also performed to open the dura and expose the cord. For these cases, 200 μl AASC suspension (in cerebrospinal fluid and autoserum in a 2:1 ratio; the cell concentration was 2–3 × 104/μl) was injected into the adjacent area of the injured site with a microinjector (EndoBionics, CA). The injection was distributed in six to seven locations on each side of the spinal cord and after each injection the syringe injector would be indwelled for 1–2 min in the spinal cord and then the needle was withdrawn slowly. The dura was closed anatomically with nonabsorbable sutures. Vital signs of the patients were carefully monitored, and they were given the corresponding symptomatic supportive treatment after operation. All operations were done by the same group of surgeons and operational procedures went on as usual.
Three days after the operation, gradual functional exercises began. Mobility activities, aerobic resistance, strength, coordination, recreation, relaxation, and wheelchair skills were combined; FIM motor score (FIM MS), ASIA motor, and sensory index total score were calculated after operation; somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) were used to further confirm the functional recovery following transplantation; urodynamic tests involving functional and dynamic assessment of the lower urinary tract were also taken to assess detrusor and bladder outlet function.
Results
Cell Identification
Three days postisolation of cells, the AASCs began to grow out from the rim of the tissue lumps with radiant shape (Fig. 5). Another 5 days later, the cells proliferated and covered the whole bottom of the six-well plates. The outline of the cells was clear and bright and some of the AASCs presented with a whirlpool shape. After purification with the method of differential adhesion, the number of fibroblasts decreased evidently and the morphology of the AASCs appeared more unified (Fig. 6). After five passages, AASCs exhibited stable expression of S-100 antigen, and the purity of AASCs reached 98% (Fig. 7).

At 3 days postisolation of cells, activated autologous schwann cells (AASCs) crept out of the rim of the tissue lumps with radiant shape.

After purification with the method of differential adhesion, the morphology of AASCs appeared more unified.
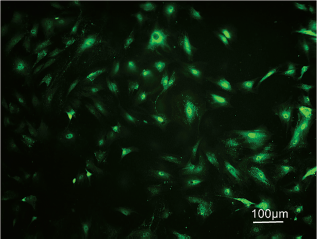
AASCs expressed S-100 antigen; the cell cytoplasm was stained with green color.
Functional Improvement
Because there were only six patients, we did not perform any further statistical analysis. The follow-up showed that the six patients' motor and sensory functions were improved to some extent. Three to 10 weeks after transplantation, a sensory and motor recovery pattern, which descended along the trunk and limbs from proximal to distal endings, took place on the patients and the ASIA scale and FIM motor score increased in all patients (Table 2). Apart from that, the improvement of autonomic function (skin nutrition improvement, reduced spasticity, increased bladder volume, decreased residual urine volume, smooth intestinal defecation) was earlier than sensory and motor function, which showed a gradual improvement with time. Then after a period of rehabilitation exercises, the quality of life was also improved.
Follow-up of the Patients

FIM, Functional Independence Measure.
Urodynamic testing also revealed that the maximum detrusor pressure, the Valsalva leak point pressure, the detrusor voiding pressure, postvoid residual urine volume, and maximal and average flow rates improved significantly.
At the same time, latency period and wave amplitude of SSEPs and MEPs were increased at different times after AASC transplantation (Figs. 8 and 9).

Somatosensory evoked potentials (SSEPs) 10 weeks after AASC transplantation in the fourth case.

SSEP 7 years after AASC transplantation in the fourth case.
Variation of MRI
These six patients were also studied with MRI before and after transplantation. Any development of glial tumor, tumor-like mass, new hemorrhage, edema, expanding cyst, new cyst formation, infection, or disruption of neural structure were not found by MRI at transplantation site. However, the MRI indicated that the volume of myelomalacia and cystic degeneration became small after AASC transplantation (Figs. 10–13).
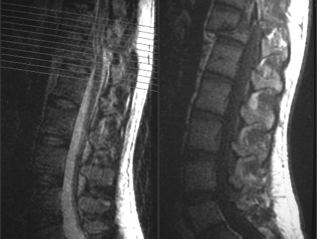
Preoperative magnetic resonance imaging (MRI) the fourth patient with spinal cord injury.

Postoperative MRI in the same patient with spinal cord injury.

MRI in the patient with spinal cord injury 2 years after AASC transplantation.
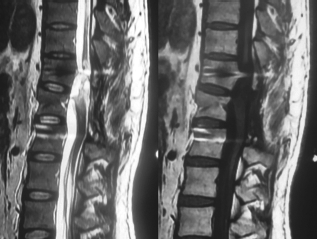
MRI in the patient with spinal cord injury 7 years after AASC transplantation.
Discussion
The clinical pathophysiological protocol of SCI is a complex and devastating condition that combines hemorrhage, ischemia, and/or edema. It develops and results in necrosis with tissue loss, glial scar, and necrotic cyst formation (27). SCI affects all cell types in the spinal cord and causes marked pathological changes, including early neuron degeneration, activation of astroglial and microglial cells, and secondary demyelination (3,11).
Experimental work indicates that cell transplantation approaches can facilitate axonal regeneration, remyelination, neuroprotection, and possible neovascularization (8,9,23,32). Lesions of peripheral nerves initiate a sequence of cellular changes in their distal of the injury site including axonal degeneration, myelin breakdown, and differentiation and proliferation of SCs (24,29,31). These activated SCs produce growth factors and extracellular matrix components that aid in fostering this regeneration, and they myelinate and ensheath axons (10).
Our experimental results also indicate that a preinjured peripheral nerve graft is useful in stimulating chronically injured descending nerves to overcome a local inhibitory environment. The resulting sprouting and growth in the injury site of the spinal cord is associated with a significant improvement in locomotor function (2,10).
Saberi and coworkers explored autologous SC transplantation to treat SCI patients. They concluded that autologous SC transplantation was generally safe for the selected number of SCI patients but it did not prove beneficial effects after 1 year of follow-up (26). Here, we tested the validity of transplanting AASCs into the injury site of six SCI patients. We ligated unilateral sural nerve and cut only a piece of it without any anesthesia because each patient selected was paralyzed in his lower extremity. Then the patients accepted an operation that had been preceded for many years. The only different thing was to open the dura and grafting AASCs to the injured site of the spinal cord. However, no immunological problems happened and no severe adverse event was observed in any of the six patients. Apart from that, all patients had an increase of ASIA scale and FIM motor score, which may be due to the following reasons.
Firstly, although the precise mechanisms of the functional recovery after SC transplantation are not fully understood, several mechanisms have been proposed: 1) secreting numerous neurotrophic factors (NTs) as BDNF, GDNF, NT-3 and -4/5, NGF (12,22,28); 2) an increase in cell surface adhesion molecular synthesis (15); 3) production of a basement membrane that consists of extracellular matrix proteins (5,25); 4) formulating “conduits” or “frames” that are capable of aiding axon growth or sprouting after transplantation (30). All these may promote axonal regeneration, axonal sparing, sprouting, and plasticity associated with novel polysynaptic pathways, recruitment of endogenous SCs, and remyelination.
Secondly, after traumatic SCI, areas of glial scar tissue and cystic degeneration create a structural barrier to regeneration and consequently recovery of function. During the operation before grafting AASCs, we performed decompression anteriorly or posteriorly, opening the dura and draining necrotic liquids, which can produce the possibility of regeneration of injured spinal cord.
Thirdly, after AASC transplantation, all the fractures or dislocations were reduced, internally fixed, and fused with bone segments (Figs. 14 and 15). This facilitated the postoperation care of the patients and helped them to adapt to daily life. After a progressive training program, the patients' ability and coordination were well improved.

Preoperative X-Ray in the fourth patient with spinal cord injury.

Postoperative X-Ray in the fourth patient with spinal cord injury.
FIM motor score assesses disability by reporting on motor skills through 13 subscales, including self-care, sphincter control, mobility transfer, and locomotion. All scores evaluating changes in the physical capacity (i.e., the weights lifted, exercise repetitions, wheelchair skills) showed improvement to some extent (21).
At present, the ASIA standards are internationally accepted standards for neurological classification of SCI. ASIA motor and sensory index total score was also calculated. SSEPs and MEPs may be of additional value to clinical examination and functional scores (6,7). The follow-up showed that the six patients' motor and sensory function was improved to some extent and the improvement was partly attributed to their functional training.
One of the limitations of this study is that there was no control group with rehabilitation alone to separate the effects of rehabilitation and AASCs and rehabilitation. However, two of the six patients had about half a year or more of rehabilitation before the AASC transplantation with no change in their ASIA grade. This suggests that rehabilitation alone was not sufficient for these patients with SCI.
The combination treatment of cell transplantation and rehabilitation is suspected to benefit the SCI patients through a safe pathway (20). Further research is necessary to identify this finding.
Conclusion
After more than 5 years of follow-up, we concluded that the treatment of AASC transplantation approach associated with a specific neurorehabilitation scheme might be feasible, safe, and effective to promote neurorestoration of SCI patients, although the clinical pathophysiology of SCI is so complex that further studies are under way to certify these findings. Many questions and uncertainties still exist.
We still have good reasons to believe that SCI patients should accept some sort of treatment such as an operation if it is permitted in the early stage after injury. And we should provide an opportunity to patients for the recovery of neurological function rather than wait until the chronic stage of injury.
Footnotes
Acknowledgments
This work was sponsored by Wu Jie-Ping medical fund of Ministry of Health (TJB-2004-1), the National Natural Science Foundation of China (30872603, 81070982,81171714), the Applied Basic Research Programs of Science and Technology Commission Foundation of Tianjin, China (07JCYBJC10200), the Key Technology Foundation of Tianjin Health Bureau (07KG2), and the Research Foundation of Tianjin Health Bureau (09kz104). The authors declare no conflicts of interest.
