Abstract
Cyclin D2 knockout mice show decreased levels of endogenous dentate neurogenesis. We investigated whether transplanted dentate progenitor cells from wild-type mice respond in vivo to an enriched environment and whether they improve deficient dentate neurogenesis through a neurotrophic effect. Adult cyclin D2 knockout mice were transplanted with passaged adult progenitor cells and kept in an enriched environment or under standard housing conditions in isolation. After 1 week, animals living in an enriched environment underwent water maze testing. Progenitor cells grown on a laminin/poly-D-lysine monolayer expressed Sox2 and nestin and could be differentiated in vitro into neurons and astrocytes. After transplantation into the dentate gyrus, cells preferentially survived along the laminin-rich ependymal lining of the basal cistern or basal membrane of capillaries. A subpopulation of transplanted cells migrated into the interstitial space of the hippocampus and was not associated with laminin. Environmental enrichment led to a significant increase in the survival of transplanted progenitor cells on laminin in the dentate gyrus after 2 weeks. However, animals did not show an enhanced performance in the Morris water maze, and transplantation failed to exert a neurotrophic effect on endogenous neurogenesis after 2 weeks. However, a major limitation of the study is the short-term period of investigation, which may have been insufficient to capture functional effects. In conclusion, initial survival of transplanted neural progenitor cells was dependent on the presence of laminin and was significantly enhanced by environmental enrichment. Further studies are needed to address whether an enriched environment continues to promote graft survival over longer periods of time.
Keywords
Introduction
Dentate neurogenesis has been implicated in pattern separation, the ability to differentiate between similar memories in space and time (40). Dentate neurogenesis in humans and rodents declines with age (23,27), and so does the ability to perform pattern separation (45,54). The cyclin D2 knockout (KO) mouse exhibits decreased levels of endogenous neurogenesis (3) and may therefore serve as a model of the neurogenesis-deficient aging dentate gyrus. While spatial navigation in the water maze environment, including platform reversal learning, is not affected in this mouse model (17), mice with decreased endogenous neurogenesis have shown worse performances in long-term memory acquisition of spatial memory (16,18).
Environmental enrichment and running promote the generation of progenitor cells in the granule cell layer of the dentate gyrus (22,24,47), also in the senescent dentate gyrus (23), as early as 1 week after enrichment (30). Cell restoration techniques aim to replenish the declining pool of dentate progenitor cells during aging to maintain spatial navigation memory. Transplantation success has been variable and seems to be dependent on graft cell origin and culture history (11). While transplantation of enteric-derived or whole brain-derived neural progenitor cells into the hippocampus of irradiated mice failed to restore spatial memory and caused disruption of the granule cell layer (36,41), transplantation of subventricular zone cells from postnatal rats into the lesioned hippocampus was shown to restore spatial memory in the probe trial (13). Improved spatial memory was also observed in several other hippocampal transplantation experiments, which utilized various cell lines (1,39,53). Other investigators have reported a positive influence of precursor cell transplantation on endogenous neurogenesis (5,14,19,21,31,38,46).
Since adult-derived neural progenitor cells have shown poor neuronal differentiation and functional integration even after prolonged periods of time in vivo (6,35,41), several investigators have come to the conclusion that observed effects are due to neurotropism and not due to differentiation and functional integration (6,10,53).
We chose a short study period of 2 weeks to investigate whether transplanted adult neural progenitor cells have an early neurotrophic effect on endogenous neurogenesis and performance in spatial navigation. Furthermore, we asked whether survival of transplanted adult dentate progenitor cells is influenced by environmental enrichment in a mouse model of endogenously decreased dentate neurogenesis.
Materials and Methods
Mouse Model
The study was approved by the ethics committee of the Technische Universität Dresden and by the responsible authority, Landesdirektion Dresden (TVA-2011-7-2). Cyclin D2 KO mice (44) were obtained after rederivation by embryo transfer and backcrossing into the C57BL/6J background for more than six generations. Embryos were taken from the Ccnd2 colony at the Nencki Institute (Warsaw, Poland). Rederived mice were group housed at the animal facility of the Medical Faculty of the Technical University Dresden with 12 h light/dark cycles and food ad libitum. Cyclin D2 KO mice were verified by polymerase chain reaction genotyping [cyclin D2(G): 5'-CCAGATTTCAGCTGCTTCTG-3’; cyclin D2(D): 5'-GCTGGCCTCCAATTCTAATC-3'; cyclin D2(N): 5'-CT AGTGAGACGTGCTACTTC-3’].
Progenitor Cell Harvest and Proliferation
Eight adult (8-week-old) β-actin-green fluorescent protein (GFP) female mice (kindly supplied by Masahito Ikawa, Institute for Microbial Diseases, Osaka University) were euthanized with isoflurane (Sigma-Aldrich, Seelze, Germany), and the dentate gyri were microscopically removed. The tissue was enzymatically digested using the Neural Tissue Dissociation Kit (Miltenyi, Bergisch-Gladbach, Germany) according to the manufacturer's instructions. Following a final wash in Hank's buffered saline solution (PAA; GE Healthcare, Little Chalfont, UK), the pellet was resuspended in 1 ml of proliferation medium containing 20 ng/ml fibroblast growth factor 2 (FGF-2; Pepro Tech, Hamburg, Germany) and epidermal growth factor (EGF; Pepro Tech) and filtered through a 40-μm cell sieve (Falcon; BD Biosciences, Heidelberg, Germany).
Cells were plated in two wells of a 24-well-plate (Falcon; BD Biosciences Discovery Labware) coated with poly-D-lysine (10 mg/ml; Sigma-Aldrich) and laminin (10 mg/ml; Roche, Grenzach, Germany). The first passage was performed after 19 days, and cells were plated in a poly-D-lysine- and laminin-coated T25 flask (TPP, Trasadingen, Switzerland). Cells were passaged again on days 26, 30, 33, and 36. After 39 days, cells were detached with Accutase (PAA Laboratories, Cölbe, Germany), spun for 2 min at 2,000 × g, resuspended in proliferation medium with 10% dimethyl sulfoxide (DMSO; Sigma-Aldrich), and snap frozen at a concentration of 1 million cells per ml.
Cell Culture Analysis
Stocked cells were thawed at 37°C with 1 ml proliferation medium. Subsequently, the volume was slowly brought up to 10 ml with proliferation medium. Cells were centrifuged at 1,000 × g for 2 min and resuspended in 1 ml proliferation medium. The viability and concentration of cells was determined in the Neubauer chamber (Carl Roth, Karlsruhe, Germany) with trypan blue (Sigma-Aldrich) staining. Cells were seeded in poly-D-lysine- and laminin-coated 24-well plates (Falcon; BD Biosciences Discovery Labware) and grown for 3 days in proliferation medium consisting of neurobasal medium (Gibco, Life Technologies, Grand Island, NY, USA), supplemented with 2% B27 (Invitrogen, Life Technologies, Grand Island, NY, USA), 1x GlutaMAX (Life Technologies), and 50 U/ml penicillin/streptomycin (Life Technologies). As growth factors, 20 ng/ml EGF and 20 ng/ml FGF-2 were added for proliferation conditions.
Cells under proliferation conditions were fixed with 4% paraformaldehyde (PFA; Sigma-Aldrich) in 0.1 M phosphate-buffered saline (PBS; local production) at room temperature for 20 min. After washing with PBS, cells were incubated in blocking solution [10% normal donkey serum (NDS; Sigma-Aldrich) in 0.1 M PBS containing 0.2% Triton X-100 (Sigma-Aldrich)] for 60 min at room temperature. The cells were then incubated in fresh blocking solution containing 3% NDS and monoclonal mouse nestin antibody (1:200; BD Biosciences), polyclonal rabbit Sox2 antibody (1:500; Chemicon, Schwalbach am Taunus, Germany) or monoclonal mouse GFP antibody (1:400; Molecular Probes, Grand Island, NY, USA) overnight at 4°C. The cells were washed three times with PBS and incubated in fresh blocking solution with donkey anti-mouse Cy3 (1:500; Jackson ImmunoResearch, Suffolk, UK), donkey anti-rabbit Cy3 (1:500; Jackson ImmunoResearch), or donkey anti-mouse DyLight 488 (1:500; Jackson ImmunoResearch) and 4',6-diamidino-2-phenylindole (DAPI; 1:5,000; Sigma-Aldrich), respectively.
For the differentiation assay after eight passages, cells were grown after standard proliferation conditions for 4 days with 10 ng/ml FGF-2, 2% fetal bovine serum (FBS; Invitrogen), and 1 μM retinoic acid (Sigma-Aldrich), followed by 7 days of 1% FBS, 1 μM retinoic acid and complete withdrawal of growth factors. The differentiated adherent cultures were fixed with 4% PFA in 0.1 M PBS at room temperature for 10 min. After washing with PBS, the cells were incubated in blocking solution (10% NDS in 0.1 M PBS containing 0.2% Triton X-100) for 60 min at room temperature. The cells were then incubated in fresh blocking solution containing mouse monoclonal βIII-tubulin antibody (1:1,000; Promega, Mannheim, Germany) or polyclonal rabbit glial fibrillary acidic protein (GFAP; 1:2,000; Dakocytomation, Gross Lindow, Germany) overnight at 4°C. The cells were washed three times with PBS and incubated in fresh blocking solution containing donkey anti-mouse Cy3 antibody (1:250; Jackson ImmunoResearch) or Cy3 anti-rabbit antibody (1:250; Dianova, Hamburg, Germany), and DAPI (1:5,000; Sigma-Aldrich) for 30 min at room temperature, washed with PBS, and mounted with Aqua-Poly/Mount (Polysciences, Warrington, PA, USA). Images were taken with an Axiocam MRm camera (Zeiss, Jena, Germany) and A1 confocal microscope (Nikon, Tokyo, Japan).
Primary neurospheres from the dentate gyrus were differentiated in proliferation medium without growth factors for 5 days, seeded on coverslips (Engelbrecht, Edermünde, Germany) coated with poly-D-lysine and laminin, and fixed with 4% PFA at room temperature for 20 min (50). The stains for βIII-tubulin and GFAP were performed with the same protocol that was used for passaged neural progenitor cells (see above). For O4 immunohistochemistry, cells were washed in PBS, blocked with 10% normal goat serum (Sigma-Aldrich) in 0.1 M PBS for 60 min at room temperature, and incubated overnight in 3% fresh blocking solution and mouse anti-O4 antibody (1:100; R&D Systems, Wiesbaden, Germany) at 4°C. Cells were washed with PBS and incubated with Alexa Fluor 555 goat anti-mouse IgM (1:1,000; Molecular Probes) for 30 min at room temperature.
Cell Transplantation
Stocked cells were thawed at 37°C with 1 ml proliferation medium. Subsequently, the volume was slowly brought up to 10 ml with proliferation medium. Cells were centrifuged at 1,000 × g for 2 min and resuspended in 1 ml proliferation medium. The viability and concentration of cells was determined in the Neubauer chamber (Carl Roth) with trypan blue (Sigma-Aldrich) staining. Cells were seeded in poly-D-lysine- and laminin-coated T25 flasks at a concentration of approximately 250,000 cells per flask. Cells were grown for 5 days either in proliferation medium or differentiation medium containing 100 ng/ml brain-derived neurotrophic factor (BDNF; Peprotech), 1 μM retinoic acid (Sigma-Aldrich), 5 ng/ml FGF-2, and 1% FBS.
After 5 days in vitro, cells were detached with 500 μl Accutase for 15 min and spun at 300 × g for 2 min. The supernatant was removed. Cells were resuspended in a total volume of 10 μl. Graft concentrations were determined with trypan blue staining in the Neubauer chamber. For the sham group, cells were shredded by centrifugation through a biopolymer column (QiaShredder; Qiagen, Hilden, Germany).
Forty cyclin D2 KO mice were anesthetized with an intraperitoneal injection of 100 mg/kg ketamine (Pharma Partner, Hamburg, Germany) and 10 mg/kg xylazine (Pharma Partner) and positioned in a Just for mice® stereotactic frame (Stoelting, Wood Dale, IL, USA). Animals were injected bilaterally with 1 μl of cell suspension at anterior posterior (AP): −1.58 mm, lateral: ±1.15 mm, and z: −2, as well as AP: −2.2, lateral: ± 1.25, and z: −2.2. Infusion rate was 0.2 μl per minute, and the needle was withdrawn 0.2 mm after each minute to achieve a greater coverage area of the graft in the z plane. These coordinates correspond to the ventral dentate gyrus. One group of animals (n = 10) received a sham injection of shredded cells, one group of animals received cells pretreated for 5 days with BNDF (n=20), and one group of animals was transplanted with cells that were grown under proliferation conditions for 5 days (n = 10).
Behavioral Groups and Water Maze
Animals of one group (n = 10) were housed in isolation under standard housing conditions, while all other animals (n=30) were in an enriched environment and group housing for 2 weeks. Animals housed in an enriched environment underwent water maze testing in the second week.
Animals were given 120 s to find the hidden platform in the Morris water maze (Weinert Wassertechnik GmbH, Dresden, Germany) paradigm. The platform stayed in the same location for all trial days. Mice were tested three times each day to find the hidden platform for the first 2 days and six times each day to find the hidden platform on the last 2 days. Mice were guided to the platform if they were not able to reach it within 120 s. Latency to reach the platform, number of platform crossings, and swim speed were recorded. Tracking files were analyzed with Ethovision 10 software (Noldus, Leesburg, VA, USA) and by repeated measure analysis of variance (ANOVA) followed by Tukey's post hoc comparisons using SPSS statistical software package (IBM, Armonk, New York).
Immunohistochemistry
Two weeks after transplantation, mice underwent transcardiac perfusion with 4% PFA. Whole mouse brains were resected, stored in 4% PFA overnight, cryoprotected with 30% sucrose (Sigma-Aldrich), and sectioned at 40-μm width. Sections were washed with PBS and blocked with 10% NGS (EMD Millipore, Darmstadt, Germany), in 0.1 M PBS with 0.2% Triton X-100 for 1-2 h at room temperature. Sections were subsequently incubated overnight at 4°C with primary antibodies for GFP (1:2,000; polyclonal chicken; Abcam, Cambridge, UK), nestin (1:200; monoclonal rat; EMD Millipore), microtubule-associated protein 2 (MAP2A + 2B; 1:200; monoclonal mouse; Sigma-Aldrich), neuronal nuclei (NeuN; 1:200; monoclonal mouse; EMD Millipore), neuron-glial antigen 2 (NG2; 1:250; monoclonal mouse; EMD Millipore), laminin (1:400; polyclonal rabbit; Sigma-Aldrich), GFAP (1:800; polyclonal rabbit; Dakocytomation), myelin basic protein (MBP; 1:200; monoclonal rat; EMD Millipore), platelet-derived growth factor receptor β (PDGFR-β; 1:100; polyclonal rabbit; Santa Cruz Biotechnology, Santa Cruz, CA, USA), vimentin (1:100; mouse monoclonal; Santa Cruz Biotechnology), ionized calcium adaptor molecule 1 (Iba-1; 1:250; polyclonal rabbit; Wako Chemicals, Neuss, Germany), and doublecortin (1:250; polyclonal goat; Santa Cruz Biotechnology) in 3% NGS in 0.1 M PBS with 0.2% Triton X-100. Sections were washed three times in PBS and then incubated for 1 h with Alexa Fluor 488 AffiniPure donkey anti-chicken IgG, Cy3 AffiniPure donkey anti-rabbit IgG, Cy3 AffiniPure donkey anti-mouse IgG, Cy3 AffiniPure donkey anti-rat IgG, or Cy3 AffiniPure donkey anti-goat IgG (1:250; Jackson ImmunoResearch). Cells were washed three times, mounted on 1% gelatin-coated SuperFrost slides (Fisherbrand, Waltham, MA, USA), and coverslipped with Fluoromount GT (Electron Microscopy Sciences, Munich, Germany).
Cell Survival Analysis
Cell survival of transplanted progenitor cells was analyzed in every third 40-μm coronal section spanning the ventral dentate gyrus. The ventral dentate gyrus was defined as the area between the anterior border of the dentate and the habenular commissure. GFP-positive transplanted cells were counted in the cortex, corpus callosum, thalamus, subventricular zones of the third and lateral ventricle, ependymal layer of the basal cistern, hippocampal fimbria, non-dentate hippocampus, and dentate gyrus (molecular layer, granule cell layer, subgranular zone, and hilus). The examiner was blinded to the experimental groups during counting. Animals were randomly chosen for analysis. GFP-positive cells were counted in 13 sections, ranging from bregma −0.70 mm and −2.30 mm according to The Mouse Brain in Stereotaxic Coordinates atlas. At least 200 cells were counted for each brain except for one. The cell counts were multiplied by three to obtain total counts, and the total counts were divided by number of cells injected and multiplied by 100 to obtain the percentage of cell survival. Transplanted cell survival in mice living in an enriched environment (n = 11) was compared to mice living isolated in standard housing conditions (n = 5) using an unpaired t-test with Welch's correction.
Quantification of GFP Expression
ImageJ (National Institutes of Health, Bethesda, MD, USA) was used to convert images to eight-bit grayscale. ImageJ was calibrated using an eight-bit grayscale density wedge. Using the circle tool, optical densities were measured for each cell. Approximately 19-24 cells were sampled from the group of cells present around the basal cistern and the cells present in the hilus.
Results
Passaged Progenitor Cells Are Neural Progenitors and Differentiate In Vitro After Removal of Growth Factors
Primary neurospheres from the dentate gyrus could be differentiated into βIII-tubulin-positive neurons, GFAP-positive astrocytes (Fig. 1A), and O4-positive oligodendrocytes (Fig. 1B). After six passages and 39 days in vitro, 100% of cells continued to express GFP (Fig. 1C). Under proliferation conditions, all cells expressed markers of neural progenitors, such as nestin and sex-determining region Y-box 2 (Sox2) (Fig. 1C-E). Using a protocol of sequential growth factor withdrawal with 2% FBS and 1 μM retinoic acid supplementation, progenitor cells could be differentiated into βIII-tubulin-positive neurons and GFAP-positive astrocytes (Fig. 1F, G). No oligodendrocytic differentiation was observed with this protocol. Cells retained their efficiency to differentiate into neurons and astrocytes after eight passages. Primary dentate gyrus neurospheres yielded 8.23 ± 7.97% βIII-tubulin-positive neurons, 80 ± 15.25% GFAP-positive astrocytes, and 1.71 ± 1.03% O4-positive oligodendrocytes (Fig. 1H), whereas 8.5 ± 9.66% of passage 8 neural progenitor cells differentiated into βIII-tubulin-positive neurons, and 67 ± 15.15% differentiated into GFAP-positive astrocytes with FBS supplementation (Fig. 1I)
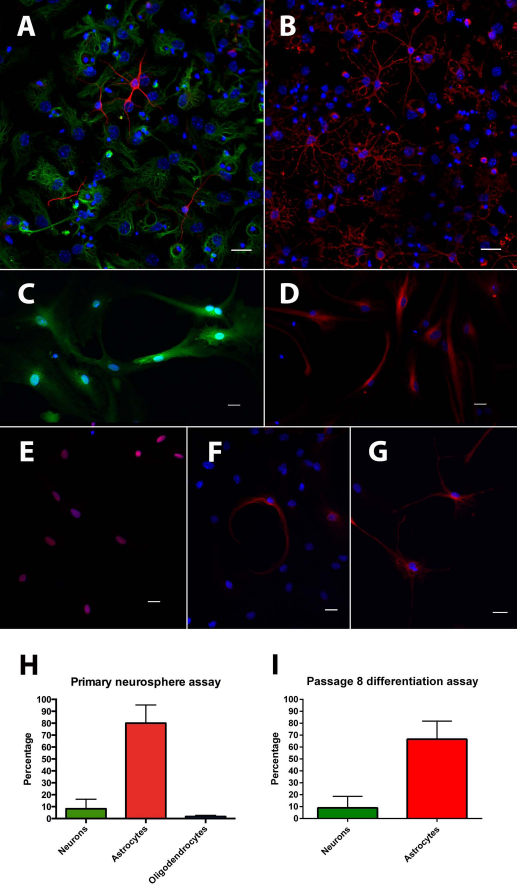
In vitro characterization of dentate neural progenitor cells. Primary neurospheres from the dentate gyrus can be differentiated into βIII-tubulin-positive neurons (red, A), GFAP-positive astrocytes (green, A), and O4-positive oligodendrocytes (red, B); DAPI (blue). All adherent monolayer cells expressed GFP (green, C) after six passages in vitro; DAPI (blue). Under proliferation conditions, cells expressed markers of neural progenitor cells, such as nestin (red, D) and Sox2 (red, E); DAPI (blue). After 10 days of differentiation with gradual withdrawal of growth factors and addition of 2% fetal bovine serum and retinoic acid, cells could be differentiated into βIII-tubulin-positive neurons (red, F) and GFAP-positive astrocytes (red, G). Primary dentate neurospheres differentiated into βIII-tubulin-positive neurons (8.2%), GFAP-positive astrocytes (80%), and O4-positive oligodendrocytes (1.7%, H). Neural progenitors retained their capacity to differentiate into βIII-tubulin-positive neurons (8.5%) and GFAP-positive astrocytes (67%) if the differentiation medium was supplemented with 2% FBS and 1 μm retinoic acid (I). Scale bars: 20 μm.
Transplanted Cells Differentiate In Vivo Into GFAP-Positive Astrocytes
Analysis of transplanted cells revealed that a small portion of grafted cells differentiated into GFAP-positive astrocytes 2 weeks after grafting (Fig. 2A, B). Transplanted cells did not differentiate in vivo into MAP2a+b- or NeuN-positive neurons (Fig. 2C, D) or myelin basic proteinpositive oligodendrocytes (Fig. 2E) in this time period. Also, no evidence for expression of the pericyte markers NG2 (Fig. 2F) and PDGFR-β (Fig. 2G) was found. Transplanted cells, which had retained their GFP expression, were also not found to express nestin (Fig. 2H). Some transplanted cells were observed in the subgranular zone of the dentate gyrus. These cells exhibited a morphology and orientation similar to endogenous dentate progenitor cells (Fig. 2I); however, they were not doublecortin positive.

In vivo differentiation analysis. Some transplanted cells on the ependymal lining of the basal cistern expressed GFAP (red, A and B). Transplanted cells did not express the neuronal markers MAP2a + b (red, C) or NeuN (red, D), or the oligodendrocytic marker MBP (red, E). They did not stain for the pericyte markers NG2 (red, F) and PDGFR-β (red, G), or for a marker of undifferentiated progenitor cells, nestin (red, H). Some cells showed a similar morphology to endogenous doublecortin-positive progenitor cells but did not express doublecortin (red, I). No cell fusion of transplanted cells with microglia was observed (Iba-1, red, J). Transplanted cells also did not express the fibroblast marker vimentin (red, K). DAPI: blue; scale bars: 20 μm.
The Transplant Graft Does Not Fuse with Host Microglia
The transplant graft elicited an inflammatory response along the ependymal lining of the basal cistern marked by recruitment of endogenous microglia to this area.
We stained for the microglial marker Iba-1 to confirm that observed transplant cell migrations were not due to phagocytosis of transplanted cells by endogenous microglia. No fusion of endogenous microglia with transplant cells was observed with Iba-1 (Fig. 2J). The graft was also not contaminated with fibroblasts, which was verified by staining for vimentin (Fig. 2K).
Laminin Is Mandatory for the Initial Survival of Transplanted Progenitor Cells In Vivo
None of the progenitor cells within the transplant core in the dentate gyrus survived after 2 weeks. Instead, progenitor cells survived along the ependymal lining of the basal cistern covering the molecular layer of the dentate gyrus. Transplanted cells seemed to migrate horizontally along the ependymal lining of the basal cistern into the fimbria of the hippocampus laterally and medially into the roof of the third ventricle. Some cells were detached from the ependymal lining of the basal cistern and seemed to migrate radially into the hippocampus and dentate gyrus.
Laminin is constitutively expressed in the ependymal layer of the basal cistern, which coats the molecular layer of the dentate gyrus, in the wild-type and cyclin D2 KO mouse (Fig. 3A). The ependymal layer of the basal cistern provides for a dense laminin scaffold for progenitor cells to settle on and survive after transplantation (Fig. 3B). Transplanted cells seemed to migrate along this scaffold horizontally (Fig. 3C). Laminin is not only expressed on the ependymal layer of the basal cistern, but also along dentate capillaries, which are in continuity with the basal cistern (Fig. 3D). Some cells appeared to migrate along the laminin-rich basal membrane of blood vessels into the hippocampus and dentate gyrus (Fig. 3E). Other transplanted cells were observed along cortical capillaries (Fig. 3F). All surviving transplant cells with strong GFP expression were associated with the basal membrane of blood vessels within the hippocampus or cortex.

Laminin is important for in vivo survival and migration of neural progenitor cells. Laminin is constitutively expressed in the ependymal layer of the basal cistern (red, A, scale bar: 200 μm). Transplanted neural progenitor cells show a predilection for settling on this layer (GFP, green; laminin, red; B and C, scale bar: 20 μm). The laminin scaffold of the ependymal layer of the basal cistern is connected with the laminin-rich basal membrane of dentate capillaries (red, D, scale bar: 100 μm). Transplanted neural progenitor cells appear to migrate from the ependymal layer of the basal cistern along the laminin-rich basal membrane of dentate capillaries into the dentate gyrus (GFP, green; laminin, red; E, scale bar: 20 μm). Transplanted cells also show a close association with the laminin-rich basal membrane of capillaries in the cortex (GFP, green; laminin, red; F, scale bar: 20 μm). A fraction of cells leaves the laminin scaffold of the ependymal lining of the basal cistern and seems to migrate into the interstitial space of the hippocampus (G, scale bar: 50 μm; H, scale bar: 20 †im). These cells show a markedly reduced expression of GFP (I). Some of these cells in the hippocampus express the astrocytic marker GFAP (J-L, scale bar: 50 μm).
A Subpopulation of Transplanted Cells with Weaker GFP Expression Is Not Associated with Laminin
A subpopulation of cells with weaker GFP expression was identified that was not associated with laminin. These cells seemed to have detached from the ependymal lining of the basal cistern and then migrate radially into the hippocampus (Fig. 3G, H). Optical density analysis showed that cells that had detached from the laminin scaffold exhibited a significantly weaker expression of GFP (Fig. 3I). Some of these cells were GFAP-expressing astrocytes (Fig. 3J-L).
Environmental Enrichment Promotes Survival of Transplanted Progenitor Cells in the Dentate Gyrus
Overall in vivo survival of cells was 11.82 ± 11.29% after 2 weeks. Pretreatment in vitro with BDNF had no significant effect on overall survival of transplanted cells or survival in any of the evaluated areas.
While enrichment had no effect on overall survival of transplanted cells (Fig. 4A) or survival in the ependymal lining of the basal cistern (Fig. 4B), enrichment significantly increased survival of transplanted cells in the dentate gyrus (Fig. 4C). With enrichment, 0.43 ± 0.17% of transplanted cells survived in the dentate gyrus, whereas without enrichment only 0.03 ± 0.01% cells survived in this location (p < 0.05). No significant difference was found with enrichment in the non-dentate hippocampus; 0.33 ± 0.14% of transplanted cells survived here with enrichment versus 0.04 ± 0.04% without enrichment (p=0.0832). There was no significant difference in survival in the fimbria of the hippocampus (Fig. 4E), corpus callosum, thalamus, or subventricular zones of the third and lateral ventricles (data not shown). A significant difference was found in the cortex (Fig. 3F), where 0.74 ± 0.25% of cells survived with enrichment, and 0.1 ± 0.03% survived without enrichment (p < 0.05).

Environmental enrichment leads to increased survival of transplanted neural progenitor cells in the dentate gyrus and cortex. No significant difference was seen in the total survival or survival along the ependymal layer of the basal cistern where most transplanted cells settled (A, B). Environmental enrichment significantly increased survival of transplanted neural progenitor cells in the dentate gyrus (C). No significant difference was also seen in survival in the non-dentate hippocampus and the fimbria of the hippocampus (D, E). Survival was also significantly increased in the cortex (F).
Transplantation of Neural Progenitor Cells Does Not Enhance Endogenous Neurogenesis or Performance in the Morris Water Maze After 2 Weeks
Cyclin D2 KO mice transplanted with neural progenitor cells did not show improved performance in the Morris water maze compared to sham surgery (Fig. 5A, B). Analysis of latency showed that there was no significant difference between groups [F(2, 21) = 1.00, p=0.39], and no group × day interaction, [F(6, 63) = 1.196, p = 0.32]. However, there was a significant day effect reflecting the fact that animals reduced their latency to find the escape platform over the course of the 4 days of training [F(3, 63) = 10.44, p = 0.0001].

Transplanted dentate progenitor cells do not enhance spatial memory or endogenous dentate neurogenesis in the first 2 weeks. Transplantation of progenitor cells pretreated for 5 days with differentiation medium (BDNF) or proliferation medium (PM) did not result in a decreased latency to the platform (A) compared to sham surgery. Swim speeds remained constant among groups (B). Transplantation of progenitor cells (C; doublecortin, red; DAPI, blue) versus sham surgery (D; doublecortin, red; DAPI, blue) did not result in a significant difference in endogenous dentate neurogenesis (E). Scale bars: 20 μm.
Progenitor cell transplantations were not observed to significantly enhance endogenous neurogenesis in the neurogenesis-deficient dentate gyrus of cyclin D2 KO animals. Stereological analysis of transplanted animals showed 108.3 ± 49.87 doublecortin-positive neural progenitor cells per ventral dentate gyrus compared to 244.3 ± 177.9 doublecortin-positive progenitor cells in the sham surgery group (Fig. 5C-E).
Discussion
Despite the knowledge that neural progenitor cells in the dentate gyrus proliferate in the setting of an enriched environment, little is known about the behavior of transplanted neural progenitor cells in an enriched environment. In this study, we show that transplanted neural progenitors show increased survival in the dentate gyrus if animals are living in an enriched environment.
The mechanism for the increased survival of transplanted progenitor cells in an enriched environment may lie in the association between transplanted cells and the endogenous capillary network. Palmer et al. have shown the importance of the vascular stem cell niche for proliferation and maturation of dentate progenitor cells (37). Mice with narrower capillaries in the dentate gyrus show decreased cell proliferation and survival of newborn neurons (12). Other investigators have implicated vascular endothelial growth factor (VEGF) as a coregulator of neurogenesis (8,29,42).
We observed that transplanted progenitor cells initially only survived on the laminin-rich scaffold of the ependymal lining of the basal cistern or the basal membrane of blood vessels that contain laminin. The mouse brain contains laminin in the ependymal lining of the basal cistern and in the basal membrane of blood vessels (see Fig. 3A-F). None of the cells of the transplant graft at the target location within the dentate gyrus survived after 2 weeks. Laminin coating of collagen scaffolds has been shown to be important for neural progenitor cell proliferation and infiltration in a dose-dependent manner (25,32). Laminin-based extracellular matrix significantly enhanced neural progenitor cell migration, survival, or acute neuronal differentiation (2,4,20,28,34,52). Laminin-derived polypeptides in a collagen hydrogel have been used to enhance survival of transplanted neural stem cells in the rat brain (33). One of the shortcomings of our study is that our protocol may have selected progenitor cells that can only survive initially on laminin in vivo, since the cells had been grown for 39 days in laminin-coated flasks. However, even when cells were grown as neurospheres for 5 days before transplantation, they still showed a similar strong affinity to laminin after transplantation in vivo (data not shown). In addition, we have observed that primary sorted neural progenitors also show an affinity to the laminin-rich ependymal lining of the basal cistern after transplantation (51).
An enriched environment is expected to activate the neural network of the dentate gyrus by increased spatial navigation of the animal. Functional magnetic resonance imaging studies have shown an increased hemodynamic response of hippocampal regions that are active (43,49). Since transplanted progenitor cells initially only survive close to the vascular network in the dentate gyrus, it is possible that an increased delivery of glucose and oxygen in the setting of an enriched environment results in better nutritional support and survival conditions for these cells.
Most of the transplanted cells initially only survived along the laminin-rich ependymal lining of the basal cistern. From this area, cells seemed to migrate horizontally along the ependymal lining of the basal cistern or radially along capillaries into the dentate gyrus or non-dentate hippocampus. Some cells survived along capillaries in the cortex. Therefore, it is possible that an enriched environment and increased perfusion of the dentate gyrus promoted migration of transplanted progenitors from the laminin-rich ependymal lining of the basal cistern along capillaries into the dentate gyrus.
We observed poor survival of transplanted cells in the corpus callosum and the CA1 area of the hippocampus. The poor survival of transplanted cells in the corpus callosum may be due to the lower density of capillaries in white matter tracts (26). The CA1 region has also been found to have a lower capillary density than other regions of the hippocampus (9). The capillary density may be the reason why cells in the cortex and dentate gyrus showed the strongest response to an enriched environment.
Two populations of transplanted cells could be distinguished: Cells with strong GFP expression that were always associated with laminin and cells with lower levels of GFP expression that had detached from the laminin scaffold and migrated into the interstitial space of the hippocampus. The laminin receptor interacts with β-actin and is critical for cell migration (48). Thus, the strong GFP expression of transplanted progenitor cells associated with laminin may be due to the activation of the laminin receptor. The further cells had migrated away from the laminin scaffold of the ependymal lining of the basal cistern or the basal membrane of capillaries, the less they expressed β-actin. GFAP-positive progenitor cells migrated radially into the hippocampus and did not show a preference for a particular location or anatomical structure. The strong association of surviving progenitor cells with the capillary network raises the question whether transplanted cells are pericytes, which are characterized by expression of NG2 (7,55). However, no expression of the pericytic marker NG2 or PDGFR-β was observed in transplanted progenitor cells.
We chose a short study period of 2 weeks to investigate whether transplanted cells exert an early neurotrophic effect on the dentate gyrus. We did not pursue a study with a longer time interval since earlier research has shown that in contrast to fetal-derived cells, adult-derived neural progenitors have poor in vivo neuronal differentiation potential, observed in studies spanning 6 or more weeks (35,41). Even though cells migrated into the dentate gyrus, unlike previously published data (38), they did not provide trophic support or stimulation of the depleted pool of endogenous neural progenitor cells and did not enhance performance of animals in the water maze test. Although treadmill exercise has been shown to enhance spatial memory of scopolamine-induced amnesia rats in the water maze after 2 weeks (15), transplanted adult-derived progenitor cells in our study did not have a neurotrophic effect on mice endogenously deficient in dentate neurogenesis. Nevertheless, a major limitation of the study is the short-term period of investigation since functional effects of the graft may not have been fully captured.
In conclusion, we show that environmental enrichment and laminin promote survival of transplanted neural progenitor cells in the dentate gyrus after 2 weeks in vivo. Further studies with a longer time interval will be necessary to observe whether transplanted cells show the same positive survival response to environmental enrichment in vivo.
Footnotes
Acknowledgments
This work was supported by the 2011 William P. Van Wagenen Fellowship Award from the American Association of Neurological Surgeons (B.W.), a Marie Curie International Incoming Fellowship (T.L.W.), and basic institutional funding (B.W. and G.K). We thank Anne Karasinsky for care and maintenance of all animals used in this study and Nick Wheat, Rahil Ghiasvand, Emily Doisy, Alexander Garthe, Daniela Lasse, and Klaus Fabel for technical support. The authors declare no conflicts of interest.
