Abstract
Deep brain stimulation (DBS) is used to treat a variety of neurological disorders including Parkinson's disease. In this study, we explored the effects of striatal stimulation (SS) in a rat model of chronic-phase ischemic stroke. The stimulation electrode was implanted into the ischemic penumbra at 1 month after middle cerebral artery occlusion (MCAO) and thereafter continuously delivered SS over a period of 1 week. Rats were evaluated behaviorally coupled with neuroradiological assessment of the infarct volumes using magnetic resonance imaging (MRI) at pre- and post-SS. The rats with SS showed significant behavioral recovery in the spontaneous activity and limb placement test compared to those without SS. MRI visualized that SS also significantly reduced the infarct volumes compared to that at pre-SS or without SS. Immunohistochemical analyses revealed a robust neurogenic response in rats that received SS characterized by a stream of proliferating cells from the subventricular zone migrating to and subsequently differentiating into neurons in the ischemic penumbra, which exhibited a significant GDNF upregulation. In tandem with this SS-mediated neurogenesis, enhanced angiogenesis was also recognized as revealed by a significant increase in VEGF levels in the penumbra. These results provide evidence that SS affords neurorestoration at the chronic phase of stroke by stimulating endogenous neurogenesis and angiogenesis.
Keywords
Introduction
Cerebral ischemia is a major cause of death and disability around the world. To date, tissue plasminogen activator is the only effective treatment, but its narrow 3-h therapeutic window limits the benefits to 2% of ischemic stroke patients (63). Cell therapy using stem cells advances the possibility of treating stroke beyond its acute stage (4,57). However, among the many logistical and ethical issues inherent with stem cell use, the rapid deterioration of the stroke brain also hinders the success of restorative cell therapy in the chronic phase of the disease. In particular, once the blood supply to the cerebral tissue stops, primary cell death ensues with the release of various cytokines compounding the cascade of secondary neurodegeneration (16,24). Accordingly, the acute stage of cerebral ischemia remains the most appealing target for cell therapy (14,73). In the clinic, however, the population of patients who will be rescued is undoubtedly huge if therapies are found effective in the chronic stage of cerebral ischemia. An alternative cell-based therapeutic approach is to rescue host endogenous stem cells rather than transplanting exogenous stem cells. Indeed, neurotrophic factor treatment, such as glial cell line-derived neurotrophic factor (GDNF) for Parkinson's disease, has reached clinical trials but with mixed results (29,52). The potential of neurotrophic factor therapy has been extended in experimental stroke models, but for it to be effective the treatment needs to be initiated proximal to the onset of brain injury (26,68), indicating again a limited clinical application of this technique. Thus, the rationale for the present study was to find a therapeutically effective method in brain repair targeting endogenous stem cells at the chronic stage of the disease in order to enhance the translation of such experimental treatment into the clinic.
Deep brain stimulation (DBS) of the subthalamic nucleus and globus pallidus pars interna (GPi) has been shown as a potent treatment for Parkinson's disease patients with consequent dramatic amelioration of symptoms through the suppression of abnormal discharge or transmission from affected neurons (3,27,61). In addition to Parkinson's disease, electrical stimulation has been used for other diseases of the central nervous system, such as GPi-DBS for severe cervical dystonia (25), electroconvulsive therapy, vagal nerve stimulation, and DBS for treatment-resistant depression (23), bilateral thalamic DBS for Tourette syndrome (33), and bilateral central thalamic-DBS for the severely disabled patients of the chronic stage of head trauma (50). Recently, our laboratory demonstrated that epidural electrical stimulation exerted strong therapeutic effects on middle cerebral artery occlusion (MCAO) model of rats in the acute phase (2). Additionally, electrical stimulation has been employed to achieve the three-dimensional organization of cultured muscle cells (54) or increase endorphin secretion from genetically modified Neuro-2a cells (55). Altogether these studies suggest various applications of electrical stimulation in regenerative medicine.
We hypothesized that striatal stimulation (SS) could restore function in the stroke brain by stimulating endogenous stem cells, which remained nascent, but viable, after the ischemic insult. In this study, we explored the behavioral, neuroradiological, and immunohistochemical effects of intraparenchymal electrical stimulation directed to the ischemic penumbra of rats in the chronic stage of middle cerebral artery occlusion (MCAO) model.
Materials and Methods
We used adult male Wistar rats following guidelines approved by the institutional animal care and use committee of Okayama University. Rats were housed two per cage in a temperature- and humidity-controlled room that was maintained on a 12-h light/dark cycle. All animals were given free access to food and water.
Experiment 1: Comparison of Infarct Volumes Measured by MRI, TTC Staining, and NeuN Staining (Fig. 1, n = 30)

Experimental protocol and the accuracy of MRI in delineating infarct volumes. (A) The protocols for experiment 1, 2, and 3 are shown. BT: behavioral tests, IH: immunohistochemical investigation, WB: Western blotting. (B) MRI revealed significant reduction of infarct volumes at day 7, compared to those at day 1. Data are shown as the mean infarct volumes + SE. *p < 0.05 versus infarct volumes measured at day 1. (C) There was strong correlation of infarct volumes taken by MRI with those evaluated by TTC and NeuN staining at day 1, 7, 14, 21, and 28. Data are shown as the mean infarct areas at the coronal level 0.3 mm posterior to bregma + SE. (D) Representative images of MRI (D1), TTC staining (D2), and NeuN staining (D3) at 2 weeks after MCAO are shown.
In this study, MRI was used to assess the infarct volumes over time. At first we confirmed whether or not the neuroradiological evaluation using MRI might be feasible. At 1, 7, 14, 21, and 28 days after MCAO, rats (n = 6 in each time point) were euthanized after MRI investigation with subsequent 2,3,5-triphenyltetrazolium chloride (TTC) staining and NeuN staining.
Experiment 2: SS in the Normal Brain (Fig. 1, n = 24)
The effects of SS in the normal brain without MCAO were examined using adult male Wistar rats (200–250 g, Charles River). Rats in the SS group (n = 12) received continuous 1-week electrical stimulation in the striatum [1.0 mm anterior and 3.5 mm lateral, i.e. the right side to bregma with the depth of 4.0 mm to the brain surface; Paxinos and Watson (43)]. The stimulation condition for the frequency and current were preset at 2 Hz and 100 μA, respectively, which were revealed as optimal based on preliminary experiments (2). Rats in the control group (n = 12) were given a “sham” operation using the same electrode without connection to the generator and consequently no current generated into the striatum. The rats were euthanized at 1 month after the initiation of SS for immunohistochemical investigations and protein assay using Western blotting.
Experiment 3: SS in the Ischemic Brain (Fig. 1, n = 24)
The stimulation electrode was implanted into the ischemic penumbra (using coordinates as in Experiment 1, but further confirmed and corrected by MRI examination before SS) at 1 month after MCAO with subsequent continuous stimulation delivered to the electrode for 1 week. Limb placement and cylinder tests were performed at 30 and 60 days after MCAO (pre- and post-SS). MRI examination was performed in stroke rats at day 28 and 44 under general anesthesia (30 mg/kg pentobarbital, IP). Thereafter, rats were euthanized at 60 days after MCAO for immunohistochemical investigations.
Electrical Stimulation
Electrical stimulation was performed as described in our previous study (2). Rats were fixed to a stereotaxic apparatus (Narishige, Japan) under general anesthesia using pentobarbital (30 mg/kg, IP) and the electrodes were implanted as described above. The electrodes were bipolar twisted stainless steel wires 200 μm in diameter. The electrode was connected to an implantable pulse generator unit (ISE1000SA, Unimec Co., Ltd., Japan), which was embedded in the skull with dental cement (15). Rats were continuously stimulated for 1 week after the implantation by implantable remote pulse generator system command equipment (ISE1010C, Unimec Co., Ltd.) and electronic stimulator (SEN-7203, Nihon Kohde, Japan). After 1 week of stimulation (day 31–37) the electrode was removed (day 42) and its placement later confirmed by MRI (see below).
BrdU Injection
Bromodeoxyuridine (BrdU, 50 mg/kg, IP) was injected intraperitoneally at 48 h before electrical stimulation with subsequent injection every 12 h for 1 week (Fig. 2).

Enhanced neuronal differentiation induced by SS in the SVZ of normal rats. Cell counts of green-colored BrdU and red-colored Dcx double-positive cells in the SVZ of normal rats revealed SS significantly increased cell proliferation and migration compared to rats without SS. (A–C) The SVZ of rats with SS; (C) merged images of (A) and (B). (D, E) The SVZ of rats without SS. (A, D) BrdU staining, (B, E) Dcx staining. (F) Quantitative analyses revealed significant differences of the number of BrdU/Dcx double-positive cells. Scale bar: 120 μm.
Western Blotting
We evaluated whether neurogenic and angiogenic factors were altered by SS. For protein assay, brains from normal rats with or without SS were quickly removed after decapitation using an overdose of pentobarbital (200 mg/kg, IP) at 1 week and 1 month after the initiation of SS. Brains were sliced at the thickness of 2 mm and each two brain tissues of the cortex and striatum near the electrode (Fig. 3) were punched out with Harris Uni-Core (2-mm-hole, Ted Pella, Inc., CA). Brain tissues were then suspended in the lysis buffer (CelLytic™MT, Sigma) and centrifuged at 15,000 rpm for 10 min at 4°C twice to remove cell debris, nuclei, and large particulates and obtain the supernatant containing cytosolic protein fraction. Two volumes of 4× NuPage LDS sample buffer (Invitrogen Life Technologies, CA) were added and boiled at 95°C for 4 min. Samples were subjected to electrophoresis in a 7.5% SDS-polyacrylamide gel and transferred electrophoretically to a Hybond-P pure nitrocellulose membrane (Amersham Pharmacia Biotech, Buckinghamshire, UK). The membranes were probed at room temperature for 12 h with rabbit anti-GDNF (1:500, #sc-328, Santa Cruz, CA), mouse anti-VEGF antibody (1:1000, #555036, BD Pharmingen, NJ), or chicken anti-β-actin (1:2000, ab13822, Abcam, MA) as primary antibodies, followed by exposure to peroxidase-conjugated secondary antibodies (1:2000, anti-mouse IgG; 1:2000, anti-rabbit IgG, Amersham Pharmacia Biotech; 1:2000, anti-chicken IgY) for 1 h. Signal development was achieved using an ECL plus Detection kit (Amersham Biosciences, UK) (76). The relative densities and areas of the bands were analyzed with Image J software, the numerical integration value (density × area) was calculated, and finally corrected using the value of β-actin (38).

Upregulation of VEGF and GDNF in normal brain induced by SS. (A) The schematic diagram shows the region punched out for Western blotting and the area evaluated as ischemic penumbra for immunohistochemical investigations. Black boxes: cortex, white boxs: striatum. (B) Western blots revealed that VEGF levels in the cortex and striatum of nonischemic rats with SS were significantly upregulated compared to nonischemic rats without SS. GDNF was also upregulated in the striatum of rats with SS, compared to those without SS, but GDNF level in the cortex did not significantly differ between the two groups. (C) The semiquantitative analyses of the Western blots revealed that VEGF levels in the cortex and striatum as well as GDNF level in the striatum were significantly upregulated.
MCAO Procedure
MCAO was carried out according to the intraluminal suture method described elsewhere (20,22). The rats were anesthetized (1.0% halothane in 70% N2O and 30% O2 using a face mask) and a 4-0 monofilament nylon suture with a silicone-coated tip (Xantopren® L blue & ACTIVATOR Universal Liquid; Heraeus Kulzer GmbH & Co. KG, Hanau, Germany) was inserted through an arteriotomy in the right common carotid artery. The blood supply to the MCA was occluded for 75 min, while the rats were allowed to move freely in the cage, and consequently the MCA reopened with animals under anesthesia as above.
Behavioral Tests (Limb Placement and Cylinder Tests)
We performed limb placement and cylinder tests for behavioral evaluation at pre- and post-MCAO (day 30 and 60). The limb placement test (LPT), composed of 8 trials, was used to assess the degree of hemiparesis, as previously described (20). The rats were evaluated for their ability to place their limbs on the top or edge of a countertop. For each trial, animals received a score of 0, 1, or 2 (0: unable to place limbs; 1: able to place limbs, but partially and/or taking time—i.e., over 2 s; 2: able to place limbs immediately and correctly). Rats with an LPT score of 6 and below at 30 min after reperfusion were used for further experiments as they were appropriate for the ischemic model.
The cylinder test was used to assess the degree of forepaw asymmetry. Rats were placed in a transparent cylinder (diameter: 20 cm, height: 30 cm) for 3 min with the number of forepaw contacts to the cylinder wall counted (49,73). The score of cylinder test in this study was calculated as a contralateral bias; that is, [(the number of contacts with the contralateral limb) – (the number of contacts with the ipsilateral limb)/(the number of total contacts) × 100] (47).
Magnetic Resonance Imaging
At pre- and post-SS periods (day 28 and 44 after MCAO), rats were anesthetized with 30 mg/kg of pentobarbital (IP) prior to MRI examinations using a clinical imaging system at 3.0T (Signa EXCITE 3.0T HD; General Electric, Yokogawa, Japan). A 3-in. microscopy coil was used for reception of the signal. Continuous, coronal T2-weighted images (T2-WI) were obtained using a spin-echo technique with the following parameters: repetition time (TR)/echo time (TE) 4000/85 ms; acquisition matrix 320 × 256; field of view (FOV) 10 cm; number of excitations (NEX) 3. A total of 9 sections were obtained from the pole of the frontal lobe to the most caudal portion of the cerebellum, with a section thickness of 2 mm and 0.2 mm intersection gap. To avoid overestimation of the infarct area due to brain edema in the acute phase and underestimation due to the brain atrophy in the chronic phase, a “corrected infarct area” (CIA) was calculated as follows, according to Neumann-Haefelin et al. (37): CIA = LT – (RT – RI), where LT and RT are the area of the left and right hemisphere, and RI is the high intensity area of T2-WI in square millimeters. Infarct volumes (IV) were calculated using the formula, IV = ∫ [CIA (mm2) × thickness (2 mm)].
TTC Staining
For TTC staining, rats were euthanized under deep anesthesia using pentobarbital before saline perfusion and brains were quickly removed at 3 days after MCAO for TTC staining. Thereafter, six serial coronal sections of 2 mm thickness were prepared. Brain slices were incubated in a 0.2% solution of TTC (Kanto Chemistry Co) in phosphate-buffered saline (PBS) at 37°C for 30 min and fixed by immersion in 4% buffered formaldehyde solution. The normal area of brain was stained dark red based on intact mitochondrial function, whereas infarct area remained unstained. Each brain slice was scanned by a color flatbed scanner, and infarct area (the level at 0.3 mm posterior to bregma) was measured using a computerized image analysis (Image J; National Institutes of Health, Bethesda, MD).
Immunohistochemistry
Rats were euthanized with an overdose of pentobarbital at 1 and 2 months after MCAO for experiments 1 and 2, respectively. The rats were perfused transcardially with 200 ml of cold PBS and 200 ml of 4% paraformaldehyde in PBS. Brains were removed and postfixed in the same fixative overnight at 4°C with the subsequent replacement with 30% sucrose in PBS for 72 h. The brains were coronally sectioned at a thickness of 14 μm. Sections were washed three times for 5 min in PBS. For laminin, Iba-1, glial fibrillary acidic protein (GFAP), doublecortin (Dcx), microtubule-associated protein 2ab (MAP2ab), and NeuN staining, sections were incubated overnight at 4°C with each primary antibody and washed three times in PBS. Then sections were incubated with corresponding Cy3-conjugated secondary antibodies (1:1000, Jackson ImmunoResearch Lab, PA). For BrdU detection, sections were pretreated with 2 N HCl for 30 min at 37°C and washed three times for 5 min in PBS. Sections were then incubated for 36 h at 4°C in primary antibody to BrdU, diluted in PBS containing 10% normal horse serum, washed with PBS, and consequently incubated with biotinylated antibodies for 8 h at room temperature followed by incubation with streptavidin-FITC secondary antibodies (1:500) for 1 h at room temperature (1:800, Jackson ImmunoResearch Lab). Sections were then processed for other immunofluorescent labeling for double staining with BrdU. Finally, sections were washed three times for 5 min each, washed in distilled water, and coverslipped with Gelmount (Biomedia Corp., CA). Control studies included exclusion of primary antibody substituted with 10% normal horse serum in PBS. No immunoreactivity was observed in these controls.
The following primary antibodies (final dilution and source) were used for immunohistochemical investigations: mouse anti-NeuN (1:100, Chemicon), rabbit anti-MAP2ab (mature neuron marker, 1:200, Chemicon), mouse anti-GFAP (1:50, Sigma-Aldrich), rabbit anti-GFAP (astrocyte marker, 1:500, Dako Denmark AS), rabbit anti-Iba1 (1:150, Wako Pure Chemical Industries, Osaka, Japan), rabbit anti-Dcx (neuroblast marker, 1:100, sc-8066, Santa Cruz Biotechnology, Santa Cruz, CA), and rat anti-BrdU (1:100, Oxford Biotechnology).
Morphological Analyses
Cell survival, migration, and differentiation in the subventricular zone (SVZ) and ischemic penumbra were examined as well as angiogenesis and microglial activation using a Zeiss LSM510 confocal microscope (Oberkochen, Germany). Specifically, six coronal sections at every 336 μm corresponding to the striatum (AP −2.0 to +2.0 mm from bregma) were collected from each rat and the number of BrdU-positive cells was counted. In the middle two sections collected from the SS group, many BrdU-positive cells were detected near the needle tract and striatum. The number of BrdU-positive cells of the middle two sections residing in the SVZ, 100, 100–200, 200–400 μm from the SVZ was counted in each six HPFs and the averages were used for the statistical analyses. In order to explore the differentiation of BrdU-positive proliferating cells, GFAP, Dcx, or MAP2ab staining was performed with BrdU staining. Six representative regions of interest were randomly selected per each section using six sections per each rat (using a 20× objective lens on a Zeiss LSM510 confocal microscope). In order to examine microglial activation and angiogenesis in the ischemic penumbra, the areas occupied by the Iba1- and laminin-immunopositive structures were calculated with binary images using Image J software, with six representative regions of interest per each section (36). The area of Iba1- and laminin-immunopositive structures was expressed as the percentages relative to the area of captured images. For the analyses of ischemic penumbra, each two areas of the cortex and striatum were used, as shown in Figure 3.
Statistical Analysis
Almost all data are presented as the mean ± SE. Data were evaluated statistically using ANOVA, followed by a post hoc Scheff's F-test. Evaluation of Iba-1 staining was analyzed using Mann-Whitney U-test with the data presentation as the median [Q1]. Statistical significance was preset at p < 0.05.
Results
Experiment 1: Comparison of Infarct Volumes Measured by MRI, TTC Staining, and NeuN Staining
At 1, 7, 14, 21, and 28 days after MCAO, TTC and NeuN staining was performed and infarct volumes were measured, respectively. Infarct volumes at day 1 evaluated by MRI were significantly larger than those at day 7, 14, 21, and 28 (Fig. 1). There were no significant differences of infarct volumes at day 7, 14, 21, and 28. In order to demonstrate the accuracy of infarct volumes by MRI, infarct areas evaluated by MRI, TTC staining, and NeuN staining were compared at day 1, 7, 14, 21, and 28 (Fig. 1). There was a tight correlation across these parameters at all time points; thus, we used MRI evaluation to assess infarct volumes over time.
Experiment 2: SS in the Normal Brain
We examined the effects of 1-week SS in the normal brain of adult male Wistar rats. Rats were euthanized at 1 month after the initiation of SS. Immunohistochemical investigations revealed that the number of BrdU and Dcx double-positive cells in the SVZ of rats receiving SS significantly increased (38 ± 9.8 cells/high power field: HPF, p < 0.05) (Fig. 2), compared to that of the control rats (13 ± 3.6 cells/HPF), although the number of BrdU-positive cells did not significantly differ between the two groups (SS: 93 ± 12.5, control: 86.3 ± 7.5 cells/HPF, p > 0.05).
Western blotting revealed that vascular endothelial growth factor (VEGF) levels were significantly upregulated both in the cortex (122 ± 2.3%) and striatum (134 ± 2.5%) of rats receiving SS compared to those without SS (cortex: 103 ± 7.3%, striatum: 105 ± 3.8%; p's < 0.05) at 1 month after the initiation of SS. Similarly, GDNF levels were significantly higher in the striatum of SS-treated rats than controls (SS: 137 ± 1.4%, control: 122 ± 0.4%, p < 0.05) (Fig. 3), but not in the cortex (SS: 126 ± 3.2%, control: 120 ± 7.7%, p > 0.05). Upregulation of both VEGF and GDNF expression was significantly higher at 1 week after SS initiation, just after SS for 1 week (VEGF in the cortex: 164 ± 5.8% and striatum: 185 ± 4.3%; GDNF in the cortex: 148 ± 4.4% and striatum: 166 ± 8.2%).
Laminin staining revealed that angiogenesis in the ischemic penumbra both in the cortex and striatum of rats with SS was significantly enhanced (20 ± 0.96 and 28.8 ± 2.8%, p's < 0.05) (Fig. 4) compared to those without SS (13 ± 0.77% and 18.1 ± 1.1%).

Enhanced angiogenesis induced by SS in the cortex and striatum. Angiogenesis was enhanced by SS in both cortex (C) and striatum (D) compared to that of rats without SS (A: cortex, B: striatum). Scale bar: 60 μm. (E) The bar graph demonstrates that the percentages of laminin-positive structures relative to the area of ROI in the ischemic striatum with SS significantly increased those without SS.
Experiment 3: SS in the Ischemic Brain
We next evaluated the effects of SS on MCAO stroke rats in the chronic stage. SS, continuously delivered over a week, was performed at 1 month after MCAO with animals subjected to behavioral and neuroradiological investigations, and thereafter at 2 months after MCAO, rats were euthanized for immunohistochemical evaluations.
Behavioral Amelioration by SS
As shown in Figure 5, MCAO produced significant behavioral impairments as revealed by LPT scores corresponding to a deficient limb use in both groups of stroke animals prior to SS treatment (control: 8.2 ± 0.5, SS: 8.0 ± 0.4 at 1 month after MCAO). Following the SS treatment, the LPT score at 2 months after MCAO significantly improved (11.4 ± 0.4, p < 0.05, Mann-Whitney U-test) compared to the control group (8.8 ± 0.3).
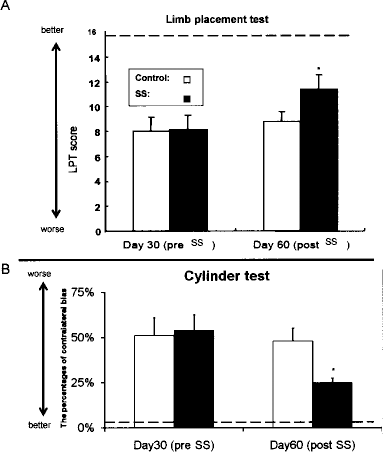
Behavioral amelioration by SS in ischemic stroke rats at the chronic stage. (A) Limb placement test revealed that the LPT score was improved by SS compared to controls. Data are shown as mean values ± SE. *p < 0.05 versus rats in the control group. (B) Cylinder test revealed that the ratio of the contralateral bias (post-/pre-SS) of rats in SS group was significantly better than the control group. Data are shown as ratio of post-SS contralateral bias relative to that of preSS ± SE. *p < 0.05 versus rats in the control group. Dotted line demonstrates the averages of normal rats from our historical data.
The ratio of contralateral bias in cylinder test of rats receiving SS relative to the value at pre-SS was significantly ameliorated (0.46 ± 0.05, p < 0.05), compared to that in the control group (0.94 ± 0.14), although there was no significant difference of the contralateral bias at pre-SS in the two groups (control: 51 ± 9.8%, SS: 54 ± 8.5%). The results indicated that SS afforded significant therapeutic effects on hemiparesis and forepaw asymmetry induced by MCAO.
Infarct Volumes Seasured by MRI
At 28 days after MCAO (pre-SS), infarct volumes were almost the same in the control and SS group (26.9 ± 1.9% and 23.9 ± 2.0% relative to the corrected hemispheric area). However, at 44 days after MCAO (post-SS), MRI demonstrated that infarct volumes of rats receiving SS (14.6 ± 1.9%) were significantly reduced compared to those of rats without SS (24.0 ± 1.5%, p < 0.05, Mann-Whitney U-test) (Fig. 6), indicating that SS induced cellular recovery from cerebral ischemia.

Reduced infarct volumes in rats receiving SS measured by MRI. (A) Representative MRI signals from rats with SS at 28 and 44 days after MCAO demonstrated that infarct volumes appear smaller than those that did not receive SS. Scale bar: 3.0 mm. (B) Quantitative analyses revealed that percentages of the corrected infarct volumes of rats receiving SS are significantly reduced at 44 days after MCAO. Data are shown as percentages of the infarct volumes relative to the corrected hemispheric area ± SE. *p < 0.05 versus rats in the control group.
Proliferation, Migration, and Differentiation of Newly Developed Cells in the SVZ
We postulated that the reduction in cerebral infarction was mediated by endogenous neurogenesis involving neural progenitor cells proliferating in the SVZ and subsequently migrating toward the ischemic penumbra. In the SVZ, there were no significant differences in the number of BrdU/Dcx double-positive cells both in the SS and control group (14 ± 1.2 and 16.3 ±1.4 cells/HPF) (Fig. 7). In contrast, the number of BrdU- and BrdU/Dcx-positive cells located at 200–400 μm from the SVZ, toward the ischemic penumbra, was significantly higher in the SS group than in the control group (BrdU: 16.8 ± 1.3 and 8.5 ± 1.3 cells/HPF, p < 0.05; BrdU/Dcx: 8.8 ± 2.1 and 5.3 ± 1.1 cells/HPF). Moreover, the number of BrdU-, BrdU/Dcx,- and BrdU/MAP2ab-positive cells in the ischemic penumbra was significantly increased in the SS group (31.3 ± 2.0, 9.4 ± 1.3, and 19.8 ± 1.6 cells/HPF) compared to the control group (14 ± 1.9, 3.2 ± 0.7, and 6.3 ± 0.9 cells/HPF).

Enhanced migration of the neural progenitor cells toward ischemic penumbra following SS treatment. BrdU (A, D) and Dcx (B, E) staining in the SVZ revealed an apparent increase in the number of double-positive cells in the SS-treated group compared to the control group (F). The merged image shows double positive cells (C). (A–C) SS group, (D, E) control group. Scale bar: 120 μm. Quantitative analyses revealed that the number of double-positive cells in 200–400 μm from the SVZ of rats in SS group is significantly higher than the control group. Data are shown as mean cell numbers ± SE. *p < 0.05 versus rats in the control group.
Next, the differentiation of the migrating cells in the ischemic penumbra was examined by double staining of BrdU with MAP2ab (mature neuron marker), GFAP (astrocyte marker), and DcX (neuroblast marker). As shown in Figure 8, many BrdU-positive cells in the SS group expressed MAP2ab, but there were few BrdU/GFAP and BrdU/Dcx double-positive cells, which did not significantly differ from the control group.

Enhanced differentiation of the neural progenitor cells to mature neurons in the ischemic penumbra after SS treatment. BrdU/MAP2ab. (A) Control, (B) SS, (C) higher magnification of the SS group. (D) BrdU/Dcx-positive and (E) BrdU/GFAP-positive cells in the ischemic penumbra are shown. The number of BrdU-positive and BrdU/MAP2ab double-positive cells increased in the SS group compared to the control group, but no other detectable differences could recognized in other phenotypic marker expression. (F) Quantitative analyses revealed significant increase of BrdU-positive cells in the ischemic penumbra of the SS group compared to the control group. Data are shown as mean number ± SE. *p < 0.05 versus rats in the control group.
Iba1 Staining
To evaluate brain inflammation inherent after stroke, microglial proliferation was examined by Iba1 staining (Fig. 9). Morphological examination revealed many amoeboid phagocytic and spherical fully activated microglial cells in the ischemic penumbra of the control group, but only found sporadically in SS group. The area occupied by the Iba1-immunopositive structures in SS group was significantly smaller [9.3% (median: 7.9%), p < 0.05] (Fig. 9) than the area in the control group [45.5% (median: 37.0%)]. The SS group significantly inhibited microglial activation, indicating that anti-inflammatory effects of SS could have abrogated the ischemic brain and promoted a conducive microenvironment for the newly developed neurons.

Suppressed microglial activation in the ischemic penumbra with SS treatment. (A, B) Iba-1-stained microglial cells in the ischemic penumbra appear suppressed by SS (B) compared to control group (A). Scale bar: 120 μm. (C) Quantitative analyses revealed significant reduction in the percentages of Iba-1-positive structures relative to the analyzed area in the SS group compared to the control group. Data are shown as mean percentages ± SE. *p < 0.05 versus rats in the control group.
Discussion
In this study, we showed that SS exerted neurorestorative effects in rats exposed to transient MCAO in the chronic stage. The SS-mediated neurestoration, in the advanced phase of the disease and in the absence of exogenous cell transplantation, implicates that dormant endogenous stem cells still persist in the normally considered “fixed” stroke brain, which can be stimulated to achieve neural repair. Here, we demonstrate that SS prompted a surge in the number of neural progenitor cells migrating from the SVZ toward the ischemic penumbra, which subsequently differentiated into neurons, suggesting that the therapeutic effects of SS on cerebral ischemia was at least partially mediated by neuronal replacement. These findings further advance the concept of endogenous reparative mechanism, but also extend the therapeutic window for such modality up to late disease progression, which is normally limited to the acute phase of brain injury. SS might activate mechanisms of proliferation, chemoattraction, and neuronal differentiation for newly formed cells, thereby enhancing the potency of self-regeneration by the stroke brain. These neurogenic proliferation, migration, and differentiation effects of SS were accompanied by an increase in neurogenic and angiogenic growth factors in the ischemic striatum, which was also rendered as a less microglial activated injury site, altogether harnessing a favorable host microenvironment for the recently recruited endogenous neural progenitor cells.
Electrical Stimulation and Ischemic Injury
The use of electrical stimulation in ischemic injury has been previously shown to result in functional recovery, including its application to the affected cortex (5), sympathoexcitatory dorsal periaqueductal gray mater (11), spinal cord (48), contralateral forepaw (6), muscle of posterior limbs (8), or epidural stimulation (1).
The optimal duration for electrical stimulation to produce therapeutic benefits remains to be determined. The present 1-week stimulation was based on a previous study demonstrating that a week-long electrical stimulation to the cortex significantly increased the action potentials of the damaged neurons and reduced the required current to elicit motion with subsequent amelioration of skilled movements in stroke rats (58). Additionally, our previous study demonstrated neuroprotective effects through antiapoptotic, angiogenic, and anti-inflammatory effects at least partly because of enhanced expression level of neurotrophic factors (2).
The demonstration of beneficial effects of electrical stimulation at the chronic stage of cerebral ischemia is deemed clinically relevant. Preclinical studies of SS targeting this advanced phase of stroke are scarce, but have been previously explored using nonhuman primates. In combination with rehabilitation therapy, cortical stimulation of motor area at a few months after the insult significantly ameliorated behavioral functions characterized by the generation of new hand representations in peri-infarct motor cortex (44). The development of the newly formed motor cortex suggests the robust effects of electrical stimulation on neural circuitry reorganization, while advancing the utility of cortical mapping in detecting brain plasticity after injury and therapeutic intervention.
The frequency and the stimulated site of electrical stimulation are equally important variables in improving functional outcome. Our study employed the intraparenchymal delivery of 2 Hz stimulation compared to many studies (1,3,61) using 100 Hz, which is designed to suppress, rather than stimulate, the neuronal activity. The present low-frequency stimulation enhanced neuronal activity as revealed by increased endogenous cell proliferation in the SVZ, followed by migration and differentiation of these newly formed cells into neurons. Moreover, such direct intraparenchymal stimulation and the extreme low frequency exerted anti-inflammatory effects. The observed anti-inflammatory effects might represent a novel cellular pathway for low-frequency electrical stimulation to exert neural repair. Additionally, the concept of electrical stimulation (i.e., electroconvulsive seizure treatment therefore high frequency) promoting neurogenesis has recently gained support (51,69).
Trophic Factor and Deep Brain Stimulation
Various trophic factors and growth factors have been implicated as potent therapeutic agents for stroke, including VEGF (72), GDNF (66), brain-derived neurotrophic factor (BDNF) (28), or placenta growth factor (PlGF) (32). These therapeutic molecules are delivered either as sole exogenously infused compound, secreted from transplanted cells (28) or encapsulated cells (72), or genetically engineered via viral vectors (66) and have been demonstrated as neuroprotective in cerebral ischemia as well as other diseases of the central nervous system, such as Parkinson's disease, Huntington's disease, traumatic brain injury, among others (18,34,75). The growth factors GDNF and VEGF have been closely associated with endogenous neurogenesis and angiogenesis (31,67), with enriched environment and/or exercise and lack thereof altering both GDNF and VEGF expression levels in the brain accompanied by neurogenic and angiogenic cellular responses (74,77). In the present study, GDNF and VEGF were upregulated by SS in the normal brain, suggesting that these growth factors partially mediated the neurorestorative effects of SS in stroke rats, in agreement with previous reports demonstrating that electrical stimulation enhanced neurogenesis and angiogenesis (12,17,51,69).
Neural Stem/Progenitor Cells: The Proliferation, Migration, and Differentiation
Neural stem/progenitor cells have the capacity of self-renewal and the potential to differentiate into multiple lineages, including neurons and glial cells. Endogenous cell proliferation in neurogenic sites, such as SVZ, after experimental stroke injury has been well documented (7,19,60,64). Accumulating evidence has also revealed novel pathways underlying the migration of newly formed cells from the SVZ to the ischemic penumbra with the overarching hypothesis that chemical signals released from the injured blood vessels attract cells to hone towards the stroke site (39,46,78). Upon reaching the ischemic area, there appears to be a tendency for the newly recruited cells to preferentially differentiate into neurons (41,59,62,65,71). The enhanced cell proliferation and migration properties of endogenous stem cells after stroke are recapitulated by electrical stimulation in that increased neurogenesis in the rat dentate gyrus occurred following kindling or electroconvulsive seizures (42,53), and in an explant embryonic cell culture model, neuronal stem/progenitor cells migrated toward the cathode after electrical stimulation (30). Of further interest, although GDNF and VEGF are established growth factors for neurogenesis and angiogenesis, as mentioned above, they are also able to act as cell migratory cues (9,40,56), suggesting that upregulation of GDNF and VEGF within the ischemic penumbra after SS might have contributed to both neuronal migration and differentiation.
Anti-inflammatory Effects of SS
Reduced infarct volumes after therapeutic intervention in stroke is usually associated with suppressed microglial activation (13), but some studies argue that amelioration of cerebral infarcts does not necessarily equate to an attenuated microglial proliferation (70). That might be due to the two-sided effects of the inflammatory cells to the damaged tissue, where the microenvironment vertiginously alters over time after ischemic insult. Indeed, examination of tissues adhered to the electrodes used for DBS in Parkinson's disease patients revealed no suppression of microglial activation (35). The discrepant results in microglial activation following electrical stimulation may be due to the disease pathophysiology (Parkinson's disease vs. cerebral ischemia), duration of the electrical stimulation (several years vs. 1 week), or species (human vs. rat). Controversy also surrounds the effects of growth factors on inflammation in that while GDNF suppressed microglial activation (18,21), VEGF was observed to exacerbate microglial proliferation (10). In the end, a homeostatic balance in microglial activation and suppression by a cocktail of counteracting trophic factors primed by electrical stimulation may translate into a therapeutic benefit. In addition to directly reducing cerebral infarcts, the reduction in microglial activation by SS in the present study might have fostered a healthy niche for endogenous cells because an inflamed brain after stroke adversely affects cell proliferation and migration (45).
Conclusions
Cell replacement in cerebral ischemia remains a challenging treatment approach, especially in the chronic stage of the disease. The present study demonstrates that SS abrogated stroke behavioral and histological deficits in ischemic rats following a long delay between injury and treatment initiation. Here, intraparenchymal electrical stimulation at low frequency over 1 week at 1 month after brain insult in rats enhanced proliferation of neural stem/progenitor cells in SVZ, then facilitated their migration to and eventual neuronal differentiation in the ischemic penumbra. Furthermore, the normally inflamed stroke brain was restored into a tolerable niche for the newly recruited cells to proliferate, migrate, and differentiate, thereby allowing an unprecedented endogenous repair process to assert functional recovery in chronic-phase stroke.
Footnotes
Acknowledgment
This work was supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
