Abstract
In this study, superparamagnetic iron oxide (SPIO) particle-labeled mesenchymal stromal cells (MSCs) were injected intra-articularly into osteoarthritic knee joints. Their fate and distribution were evaluated using magnetic resonance imaging (MRI) and macroscopic and histologic postmortem examination. Osteoarthritis was induced in 12 sheep by bilateral meniscectomy. After 6 weeks, one knee joint received 10 × 106 SPIO-labeled MSCs (Molday Ion Rhodamine B). Contralateral knees received a control injection of a) PBS, b) SPIO in PBS, c) 10 × 106 nonvital SPIO-labeled MSCs in PBS, or d) no injection. MR images were acquired immediately after injection and 1, 4, 8, and 12 weeks thereafter using a 0.5-T unit and a T2* sequence. Signal intensity of synovial fluid and synovial lining was assessed semiquantitatively using a scoring system. Viable SPIO-labeled MSCs produced a strong hypointense signal in the synovial fluid immediately after injection, but normal signal intensity of the synovial fluid was observed 1 week later. Synovial lining maintained its hypointensity throughout the study period. Nonvital SPIO-labeled MSCs induced hypointense signals of the synovial fluid; synovial lining appeared weak and inconsistently hypointense in the following weeks. Pure SPIO produced a strong hyperintense signal in the synovial fluid at the time of injection only. Histologically, in all knee joints receiving viable SPIO-labeled MSCs, SPIO particles were detected (Prussian blue) within the synovial lining, dorsal fat pad, and neomeniscus tissue, but not in osteochondral samples. Few SPIO particles were detected in joints injected with nonvital SPIO-labeled MSCs. Immunohistologically, no increased cell death (TUNEL) was observed in the area of detected SPIO particles, but we did observe potential chondrogenic cell differentiation (Safranin O or S100β). We conclude that viable SPIO-labeled MSCs remain detectable within the joint for 12 weeks and attach themselves to some but not all diseased joint structures.
Keywords
Introduction
Mesenchymal stromal cells (MSCs) are currently under investigation for the treatment of a variety of disorders due to their capacity for self-renewal, differentiation, and immunomodulation (12,28). Respective joint-related research is dominated by tissue engineering for chondral defects (38). However, cartilage defects with distinct margins are not a regular feature in osteoarthritis (OA), especially in early stages of the disease. Additionally, in OA, all tissues of the joint are involved in the disease process (20). A treatment approach that addresses the entire joint might therefore be desired over targeting a single tissue. Injecting MSC suspension into diseased joints may fulfill this criterion, as the current understanding is that MSCs exert their effect by trophic and immunomodulatory properties rather than by transformation into target cells (28). Previous studies on intra-articular MSC injection showed varying degrees of improvement. This was demonstrated in experimental animal studies (11,21,26,27), as well as in human (7,9,13,29,37) and animal patients (14,17). However, multiple patient and injection-related factors may influence the clinical outcome, and still, little is known about engraftment, fate, and contribution to healing of those intra-articularly applied MSCs.
Available in vivo cell tracing methods are via bioluminescence (16), fluorescence (27,30), or labeling with radionuclides (4,32) or superparamagnetic iron oxide particles (SPIO). The latter have the advantage of being radiation-free, in vivo longitudinal tracing added by concurrent three-dimensional assessment of the targeted tissue. A previous study tracing SPIO-labeled MSCs in OA joints of rabbits (21) demonstrated the feasibility of this method. MSC engraftment after systemic administration and homing is generally considered low (i.e., <1%) and transient in nature (24). Current challenges after any kind of labeled MSC delivery are cell quantification and characterization in the target tissue (23).
The data presented here are part of a larger project evaluating the effect and fate of intra-articularly applied MSCs in an ovine OA model. Data regarding the treatment effect of MSCs have been presented in part before (10) and will be the main content of a separate publication. Suitability of our selected SPIO product has been shown (22).
The objective of this study is the longitudinal tracking of intra-articularly applied, autologous, SPIO-labeled MSCs over a period of 12 weeks using MRI and their disposition and biological status evaluated postmortem at the conclusion of our study.
Our hypotheses for the current study are therefore (1) viable SPIO-labeled MSCs are retained within the joint after intra-articular injection in comparison to nonvital SPIO-labeled MSCs and pure SPIO markers detectable on MRI over a 12-week period, (2) viable SPIO-labeled MSCs attach themselves to diseased joint tissues detectable on MRI and postmortem evaluation, and (3) SPIO-labeled MSCs are still alive after 12 weeks, evaluated on a histologic and an immunohistologic level.
Materials and Methods
Induction of OA, Bone Marrow Aspiration, and Animal Management
All procedures were approved by the State Animal Care Committee (TVV 11/09; Landesdirektion Leipzig, Free State of Saxony, Germany). Figure 1 illustrates the study design: 12 purebred German Blackhead mutton ewes from a local breeder (Agrargenossenschaft Sonnewalde, Sonnewalde, Germany) aged 24-30 months and with an average body weight of 75.3 kg were used. Physical examination, complete blood count, and radiologic examination of both knee joints ensured normal health status and orthopedic soundness. For the OA induction by lateral meniscectomy, animals received 2.2 mg/kg flunixin IV (cp-pharma, Burgdorf, Germany) and 2 mg/kg ceftiofur DM (Pfizer, Berlin, Germany) and were placed routinely under general anesthesia. All surgical procedures were performed under aseptic conditions. After a 4-cm skin incision cranial to the lateral collateral ligament (CL) of the lateral femorotibial joint (FTJ), the joint cavity was entered dorsally and caudally to the CL, and approximately 90-100% of the lateral meniscus was removed. Fascia and skin were closed routinely, and the contralateral FTJ was operated in a similar manner. Subsequently, sheep were placed in sternal recumbency, and bone marrow was aspirated. Briefly, after aseptical preparation, the skin at the center of each iliac crest was incised at a length of 0.7 mm using a scalpel (No. 15; B. Braun, Tuttlingen, Germany). A sterile bone marrow aspiration needle (11 gauge; Angiotech, Gainesville, FL, USA) was inserted vertically and pushed with gentle to-and-fro motion into the medullary space. The obturator was removed, and 12.500 IU heparin (B. Braun, Melsungen, Germany) was injected into the medullary space. Subsequently, 20 ml of bone marrow was aspirated into a heparinized (10,000 IU heparin) syringe. The procedure was repeated at the other side, resulting in a total amount of 40 ml harvested bone marrow. Finally, the skin incisions were covered with iodine-PVP spray (Albrecht, Aulendorf, Germany).

Time bar displaying the study design (MRI, magnetic resonance imaging; OA, osteoarthritis; BM, bone marrow; MSC, mesenchymal stromal cell).
After surgery, sheep were administered 2.2 mg/kg flunixin orally and 2 mg/kg ceftiofur IM, both every 24 h for 3 days. Additional doses of flunixin were administered if needed, based on a standardized pain score. During the 7 days after surgery, animals were restricted to stall rest (3 × 3 m). After that, they were hand-walked daily 7 min for 3 weeks. At the beginning of the fourth week after surgery, sheep were turned out daily to graze on a small pasture area.
MSC Isolation, Expansion, and Labeling
Mononuclear cells (MNCs) were isolated by density gradient centrifugation within 8 h after sampling. For that, bone marrow was diluted 1:1 with phosphate-buffered saline (PBS; PAA, Pasching, Austria); 22.5 ml of the mixture was placed on 15 ml Ficoll PREMIUM (GE Healthcare, Uppsala, Sweden) and centrifuged (20 min, 1,000 x g, 20°C). The buffy coat containing MNCs was collected and washed twice with PBS. Cells were plated at a density of 1.5 × 105 cells/cm2 in T175 tissue culture flasks (Greiner Bio-One, Frickenhausen, Germany) in Dulbecco's modified Eagle medium (PAA) supplemented with 10% fetal bovine serum (FBS; Sigma-Aldrich, Steinheim, Germany), 1% penicillin/streptomycin (PAA), and 8.9 μg/ml ascorbic acid (Sigma-Aldrich). Cultures were kept at 37°C in a humidified atmosphere at 5% CO2. After 24 h, cells were washed with PBS, and culture medium was added again. Subsequently, culture medium was changed twice a week. MSCs were passaged at subconfluency. All cells of p1 were cryopreserved to prevent cell aging and senescence during the 6 weeks between bone marrow aspiration/OA induction and MSC injection. For cryopreservation, MSCs were harvested into 50-ml conical tubes (BD Biosciences, Erembodegem, Belgium) and centrifuged (5 min, 400 x g, 4°C). The supernatant was discarded, and cells were resuspended in freezing medium consisting of FBS and 10% dimethyl sulfoxide (Sigma-Aldrich). The suspension was transferred to cryotubes (Greiner Bio-One), which were placed in a freezing box (Nalge Nunc, Rochester, NY, USA) filled with isopropanol (Roth, Karlsruhe, Germany), allowing for a stepwise decrease in temperature at 1°C/min to −80°C. Cells were kept at −80°C for a maximal time frame of 3 weeks.
Ten days prior to injection, MSCs were thawed by placing the cryotube into a 37°C water bath. MSCs were then transferred to a 50-ml conical tube and washed twice with PBS. MSCs were further expanded until p3. Immediately before injection, approximately 10 × 106 MSCs (within two T175 tissue culture flasks) were labeled with Molday ION Rhodamine B (“Molday”; BioPal, Worcester, MA, USA). Briefly, Molday stock solution was added to the culture medium at a concentration of 50 μl/ml, and MSCs were incubated for 10 h under standard culture conditions. After labeling, the medium was removed, and cells were washed three times with PBS, and 10 × 106 labeled cells were harvested and then mixed with 10 × 106 unlabeled cells that had been cultured with normal culture medium without Molday. The mixture of both labeled and unlabeled cells were resuspended in 2 ml PBS and transferred to a 5-ml syringe (DE Healthcare Products, Gillingham, UK) for implantation.
For the contralateral control joint injection, four different setups were used; however, none of them contained viable MSCs. Specifically they were a) PBS (n = 4), b) pure liquid SPIO in PBS (n = 2), c) 10 × 106 nonvital SPIO-labeled MSCs (n=3), or d) no injection (n=3). The pure Molday sample was prepared by adding 25.5 pl Molday stock solution to 2 μl PBS and transferring it to a 5-ml syringe. The nonviable MSC sample was prepared as follows: 10 × 106 labeled MSCs were harvested and transferred to a 50-ml conical tube. Cells were centrifuged (5 min, 800 x g, 20°C), and supernatant was discarded. To devitalize the cells, they were incubated in 3 ml of 70% ethanol for 3 min at room temperature. Subsequently, cells were washed three times with 40 ml PBS, resuspended in 2 ml PBS, and transferred to a 5-ml syringe. Lack of cell viability was proven by trypan blue stain (Sigma-Aldrich). The time between sample preparation in the laboratory and joint injection did not exceed 30 min, and samples were kept at 4°C during that time.
Intra-articular Administration
Animals received a single dose of 2.2 mg/kg flunixin IV and 12 mg/kg penicillin/15.5 mg/kg streptomycin IM (Albrecht) and were placed under general anesthesia. The treatment knee was randomly selected, and the contralateral knee served as control. Intra-articular injection was performed under aseptic conditions with a 20-gauge needle (Terumo Europe, Leuven, Belgium). All suspensions to be injected were gently agitated within the syringe immediately before injection. Arthrocentesis of the lateral FTJ was always attempted first at the caudal recess. Correct intra-articular needle placement was ensured by synovial fluid aspiration. If unsuccessful, the femoropatellar joint (FPJ) was punctured dorsally instead, again ensured by positive synovial aspiration.
MR Image Acquisition and Evaluation
The protocol for the seven MR sessions per sheep was identical. Animals were placed under general anesthesia in lateral recumbency in the gantry with the evaluated leg uppermost. The imaged leg was extended caudally and slightly rotated outward and held in position with sandbags. The angle at the knee joint was approximately 90°-110°. A 0.5-T MRI unit (Gyroscan NT 0.5T; Philips HealthCare, Hamburg, Germany) and a flexible surface coil (Philips C3) were used. A T2*FFE sequence was used (field of view 160 mm, matrix scanned 256 × 256, matrix reconstructed 512 × 512, TE 41 ms, TR 27 ms, flip angle 25°, 1 mm slice width, no slice gap). Three orientations (i.e., sagittal, dorsal, transverse), and about 40 to 90 slices were obtained per scan. The orientations were planned in pilot images by the same MR technician. The sagittal image was planned in a transverse pilot view so that the distal condyle of the femur was split in half. The dorsal orientation was planned in a sagittal pilot view so that it was parallel to the long axis of the tibia. Finally, the transverse orientation was planned in a dorsal pilot view so that it was parallel to the tibial plateau and 90° to the cranial aspect of the long axis of the tibia.
All images were stored and evaluated en bloc in a blinded and randomized fashion (U.D.). Digital image software (eFilm 2.1.0.; Merge Healthcare, Milwaukee, USA) on double monitors (each 3.2 bit, 1,680 × 1,050 pixel; Fujitsu Technology Solutions GmbH, München, Germany) was used. The MRI scoring system for SPIO detection was developed based on our own observations gained during image acquisition (Table 1). The entire joint was always observed, that is, all intra- and extra-articular structures. Owing to natural hypointensity, bone and ligaments were excluded from the evaluation. Additionally, the soft tissue on the lateral joint aspect, including the newly formed tissue in the area of the lateral neomeniscus, contained multiple hypointense artifacts in all animals irrespective of the treatment allocation. Care was taken not to include this area in the scoring system. Therefore, only two structures in three locations were scored: The intensity of the synovial fluid and joint capsule of the lateral and medial FTJ caudal recess and FPJ proximal recess (suprapatellar pouch). All images of the three orientations were included in the grading, and the worst grade (i.e., the highest score) was given in each category if an abnormality was seen on any of the images. Synovial fluid and synovial lining score values of the three joint compartments were summarized. Thus, a combined score value for each structure was obtained. Consequently, a high combined score value indicated accumulation of SPIO particles.
Magnetic Resonance Image Evaluation Criteria for the Lateral Femorotibial Joint (FTJ), Medial FTJ, and Femoropatellar Joint (FPJ)

Postmortem Procedures and Sampling
All sheep were sacrificed (captive bolt stunning followed by exsanguination) 12 weeks after intra-articular administration, when knee joints were opened. The entire joint was assessed for presence of brown discolorations indicating SPIO presence. Special attention was paid to the cranial fat pad, synovial linings, and the reformed soft tissue structure in the area of the lateral meniscus (neomeniscus). When brown discoloration was visible, representative samples were obtained. In the absence of brown discolorations, standardized samples were obtained from the cranial fat pad, from the caudal synovial lining of the lateral FTJ, and from the cranial third of the reformed soft tissue structure in the area of the lateral meniscus. Additionally, samples of articular cartilage with underlying bone were harvested from the lateral and medial FTJ of areas with the most extensive OA lesion.
Histologic and Immunohistologic Evaluation
Soft tissue samples were fixed in 4% paraformaldehyde (Roth) for 72 h, and subsequently serial sections at 4 μm were obtained. Osteochondral samples were fixed in paraformaldehyde as well with ensuing decalcification in 20% Decal (Serva Electrophoresis, Heidelberg, Germany) for 6 weeks. Following paraffin embedment, slices of 5-μm thickness were obtained. All sections were stained with hematoxylin and eosin (H&E; Sigma-Aldrich) for cell and tissue morphology and Prussian blue (PB; BioPAL) for detecting iron (i.e., SPIO). If PB stain indicated iron particles (PB+) further sections were stained for TUNEL, indicating cell death (Roche, Mannheim, Germany), immunostained for chondrogenic marker S100β (rabbit; 1:600; Dako, Hamburg, Germany) (15), or histologically stained for Safranin O (Waldeck-Chroma, Münster, Germany), which marks proteoglycans as important extracellular matrix component (39). Both stainings were performed in order to show MSC chondrogenic differentiation or even chondrogenesis. TUNEL staining was performed according to the manufacturer's instructions. The positive control included DNAse I solution (300 U/ml) treatment for 10 min at room temperature to induce DNA strand break prior to labeling procedures. The immunohistological staining procedure for S100β was performed as described previously, including boiling of sections in citrate buffer (5). Incubation of sections with DAPI solution (200 ng/ml; Invitrogen, Darmstadt, Germany) for 30 min at room temperature stained cell nuclei and served as counterstain. For Safranin O staining, we followed the manufacturer's instructions and incubated sections for 10 min with Safranin O (0.1% w/v water), and unspecific dye was removed with aqua dest; cell nuclei were counterstained with Hemalaun (Waldeck-Chroma) as described.
All PB stained sections were semiquantitatively scored in a blinded fashion (H.J.) for the amount of (i.e., SPIO-labeled cells) (0=no PB stain; 1=few cells PB+; 2=moderate amount of PB+ cells; 3=large amount of PB+ cells) at 20x magnification (Nikon eclipse Ti-S; Nikon Instruments, Melville, USA). All samples with PB+ sections were additionally qualitatively analyzed for TUNEL-positive cells, S100β or Safranin O on adjacent serial sections (M.M.).
Statistical Analysis
None of the data were normally distributed (Shapiro-Wilk test). Therefore, group comparisons were performed using the Wilcoxon test. The control group always encompassed all four different approaches, and no separate comparisons were made between the treatment group and the individual four control setups due to the small number of cases. The absolute value was used for statistical analysis of negative score values. Significance was set at p < 0.05. Bonferroni correction was applied to adjust the p value. Data were expressed as median with interquartile range (IQR); however, calculating the IQR proved impossible in data sets containing few cases, and in this instance, only the median was presented. Statistical analysis was performed with IBM SPSS Statistics (version 22; IBM Corp., Armonk, NY, USA).
Results
Intra-Articular Administration
The lateral FTJ was successfully injected in 19/24 joints. Five times, including one bilateral case, the FPJ had to be injected instead.
MR Image Evaluation
Immediately after intra-articular injection, viable SPIO-labeled MSCs produced large hypointense signals in the joint cavity (Fig. 2). In 9/12 injected treatment joints, all three joint compartments appeared signal void or hypointense, whereas in the remaining three cases, at least one neighboring joint space was involved. Furthermore, the intensity of the synovial lining of injected treatment joints appearing in 8/12 cases decreased at that time. Within the group of control injections, this was observed only in one of the two joints injected with pure liquid SPIO in PBS. The difference in signal intensity between treated and control joints was significant for synovial fluid (6.5 IQR 3.0 vs. 0.5 IQR 1.75; p=0.005) and for synovial lining (2.0 IQR 3.5 vs. 0.0 IQR 0.8; p=0.007) (Fig. 3). Using control group d) no injection (n=3) as negative control only, significance was missed due to the small number of cases, even though a better demarcation between the two groups was seen on the graphic display (data not shown). Nonvital SPIO-labeled MSCs produced a moderate to strong hypointense signal within the synovial fluid as well as immediately after injection. Surprisingly, injection of pure SPIO (in PBS) produced a strong hyperintense signal within the synovial fluid of the injected joint compartment with few hypointense dots along the synovial lining (Fig. 4). PBS injection did not change the intensity of the synovial fluid in MR images.
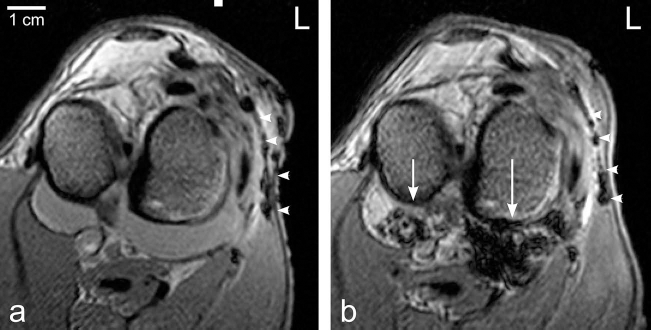
Transverse T2* magnetic resonance images of the left femorotibial joints (FTJ) in animal #7 (lateral is to the right) at time point 0 (a) immediately before injection and (b) after injecting 10 × 106 viable, SPIO-labeled MSCs into the lateral FTJ. Note the strong hypointense signal in the medial and more so in the lateral caudal joint space (arrows), and surgically induced hypointense artifacts lateral and dorsolateral of the FTJ (arrowheads).

Median, interquartile range (box), range (whisker), and outlier (°, *) of the combined synovial fluid (a) and synovial lining (b) MRI scores, that is, values of the three knee joint compartments were summarized; significant differences are denoted.

Transverse T2* magnetic resonance images of the right femorotibial joints (FTJ) in animal #7 (lateral is to the left) at time point 0 (a) immediately before injection and (b) after the injection of 25.5 μl Molday ION Rhodamine B stock solution in 2 ml PBS into the lateral FTJ. Note few hypointense artifacts in the lateral FTJ (arrowheads), but strong hyperintensity of the remaining synovial fluid in the medial, but more so in the lateral caudal joint space (arrows).
In week 1, the synovial fluid regained its physiological signal intensity in all joints, while the synovial lining still appeared significantly more hypointense in all treated joints compared to the control (4.0 IQR 2.0 vs. 0.0 IQR 1.8; p = 0.005). This difference in the joint capsule remained significant for weeks 4 and 8 (both p = 0.002) but became insignificant in week 12 (p = 0.049; Bonferroni-adjusted significance level was p < 0.008). Thus, the hypointensity of the synovial lining in the treated joints did not change until week 12 (i.e., the end of the study) (Fig. 5). Nonvital SPIO-labeled MSCs and, to a far lesser degree, pure SPIO inconsistently produced a very weak hypointense signal within the synovial lining.

Transverse T2* magnetic resonance images of the left femorotibial joints (FTJ) in animal #10 (lateral is to the right) at time point 0 (immediately before injection), 0+ (immediately after injecting 10 × 106 viable, SPIO-labeled MSCs into the lateral FTJ), and 1, 4, 8, and 12 weeks later. Note the persistence of a hypointense signal within the synovial membrane of the caudal recess of the lateral FTJ throughout the entire study period (arrowheads).
Macroscopic Postmortem Evaluation
Macroscopically, brown discolorations were detected in multiple locations in the majority of the joints treated with viable SPIO-labeled MSCs as well as in one case within the synovial lining of the control group c), which had received nonvital-labeled MSCs (Table 2). Figure 6 depicts representative focal brown discoloration of joint structures due to SPIO particles.

Macroscopic images of the left knee in animal #11. (a) View of the tibial plateau (lateral is to the left); note the discoloration due to SPIO particles located in the caudolateral synovial lining (arrowheads) and craniomedial fat pad (arrows). (b) Close-up view of the caudolateral synovial lining showing the brown discoloration (lateral is to the right). (c) Transected newly formed meniscoid tissue; note the discoloration within the tissue matrix (arrowhead).
Results of Macroscopic Evaluation of Tissue Samples With Respect to Brown Discolorations Within the Tissue Indicating Presence of Iron (SPIO)
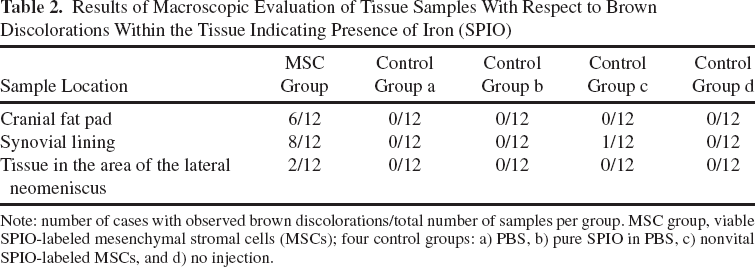
Note: number of cases with observed brown discolorations/total number of samples per group. MSC group, viable SPIO-labeled mesenchymal stromal cells (MSCs); four control groups: a) PBS, b) pure SPIO in PBS, c) nonvital SPIO-labeled MSCs, and d) no injection.
Histologic and Immunohistologic Evaluation
SPIO particles were detected histologically in all knee joints that received intra-articular injection of viable SPIO-labeled MSCs (Table 3). Specifically, PB+ cells were identified in 9/12 cranial fat pad samples, in 9/12 caudal synovial lining samples, and in 8/12 samples of the newly formed meniscoid tissue. Few SPIO particles were detected in 2/3 fat pad and neomeniscus tissue samples in the control group c) only, where nonvital MSCs had been injected. Contrary to the MSC treatment group, where SPIO particles were intracellular, the detected SPIO particles in control group c) were observed to be within an accumulation of cell debris. Histological evaluation of osteochondral samples revealed the absence of iron, that is, SPIO particles in all samples irrespective of the treatment mode.
Results of Semiquantitative Histologic Evaluation [Median (Interquartile Range)] of Tissue Samples for Amount of Iron (SPIO) in Prussian Blue (PB) Staining

0 = no PB stain; 1 = few PB-positive (PB+) cells; 2 = moderate amount of PB+ cells; 3 = large amount of PB+ cells. MSC group, viable SPIO-labeled mesenchymal stromal cells (MSC); four control groups: a) PBS, b) pure SPIO in PBS, c) nonvital SPIO-labeled MSCs, and d) no injection.
Subsequently, serial sections of PB+ images were analyzed for potential chondrogenic differentiation or chondrogenesis and viability status (Fig. 7). Adjacent to PB+ cells, the cartilaginous extracellular matrix was identified by red-stained proteoglycans using Safranin O (Fig. 7B′, red histologic staining). Also, intracellular chondrogenic markers, such as S100β (Fig. 7C, red immunofluorescence staining), were observed in the area of PB+ cells.
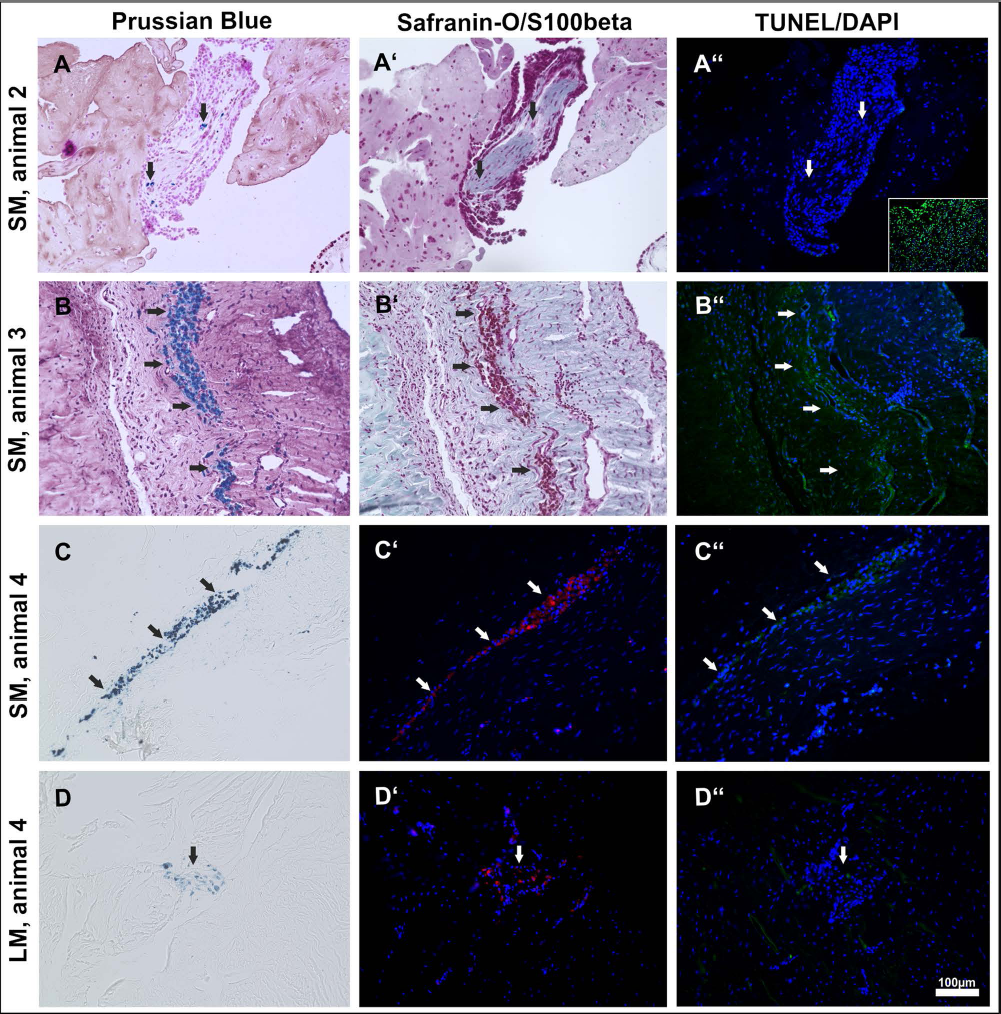
Serial histologic and immunohistologic images of synovial membrane (SM) or lateral meniscoid tissue (LM) of joints treated with viable, SPIO-labeled MSCs (treatment group). (A-D) Prussian blue-positive (PB+) cells (arrows) are shown in blue in H&E-counterstained sections (A, B) and single PB-stained sections (C, D). (A′, B′) Colocalization of Safranin O (A′, B′) and S100β/ DAPI. (C′, D′) Positive areas may indicate chondrogenic differentiation (arrows point to the same tissue area as shown in A-D and indicate positively stained cells in red). (A″-D″) In comparison to untreated joints no increased cell death was identified by TUNEL stain (i.e., no increased green nuclei stain); DAPI indicates all cell nuclei (blue); the inset in image A″ shows a DNAse-treated section and served as positive control. White arrows point to the same tissue area as shown in A-D and A′-D′, respectively.
However, due to the fact that we compared serial sections for separate stainings, a cell-to-cell allocation was difficult, particularly in sections with low numbers of PB+ cells (Fig. 7A′, D′). Immune cell infiltrates were detected based on their cell morphology in some H&E-stained sections in the area of PB+ cells (Fig. 7B). Importantly, no increase in TUNEL-positive cells was identified in the area of the transplanted cells (Fig. 7A″-D″).
Discussion
On MRI, viable SPIO-labeled MSCs produced a strong hypointense signal in the joint space immediately after injection. One week later, this strong signal (i.e., the labeled cells) had disappeared from the synovial fluid, and a strong hypointense signal was observed within the synovial lining instead, presumably due to cell attachment. This appearance remained almost unchanged throughout the follow-up period, potentially indicating that viable MSCs were still locally present. This is supported by the lack of persistence of hypointense signals in the synovial lining of joints injected with nonvital SPIO-labeled MSCs or pure SPIO. The presence of viable labeled MSCs over 12 weeks is in agreement with comparably designed studies; specifically, labeled MSCs were monitored after intra-articular injection in a rabbit model (19), or labeled MSCs were monitored in a cartilage defect model in rats (21). In contrast to our study, other authors (19) observed a persistence of the hypointense signal throughout the observation period of 12 weeks in their control group, consisting of mitomycin pretreated (i.e., according to the author apoptotic) SPIO-labeled MSCs.
An important question relates to the accuracy of MRI as a detection method. Various digital evaluation systems, including determination of the relaxation rate R2*, number of hypointense voxels in a user-defined region of interest (ROI), and the calculation of a signal-to-noise ratio (SNR) are described (1,3,19). In our study, the size and intensity of signal void areas were subjectively evaluated and scored. This allows differentiation from surgically induced hypointense artifacts and naturally hypointense structures (like cortical bone), but with the downside of being potentially inaccurate. To decrease the chance of false-positive assessment, we excluded the surgical access region (i.e., the entire lateral joint aspect), including neomeniscus formation, from the evaluation. We were also not able to accurately assess the potential presence of SPIO markers on the joint surface or even bone. The cartilage appeared as a very thin (<1 mm) structure and might therefore be susceptible to volume artifact (overlap with surrounding structures). Bone, especially subchondral cortical bone, exhibits a hypointense signal. Additionally, the subchondral bone underwent osteoarthritic changes, like sclerosis, thus further increasing hypointensity and therefore the differentiation difficulties to SPIO signals. This contrasts to previous studies (21), though in this report, no statements about surgically induced or other artifacts and their discrimination to SPIO markers were made. We also observed instant distribution of SPIO particles in all three knee joint compartments after injection irrespective of the administration site, thus reinforcing previous findings of free communication of those three cavities in sheep (36). This further impedes computerized evaluation systems because they rely on clearly defined and consistent boundaries, which are impossible to draw in a complex three-dimensional structure like the three compartments of the knee joint.
Using SPIO products for cell labeling and cell tracking raises some questions. While the hypointense MRI signal can be reliably related to SPIO particles, deducing engraftment, viability, type, or differentiation status of labeled and injected cells may be arguable. One may postulate that MSCs will not survive after injection, and hypointense signals indicate SPIO within nonvital cells. Alternatively, SPIO particles could be released from vital cells into the surroundings and detected as free iron by MRI. Finally, fragments of SPIO carrying nonvital MSCs, or even free iron particles could be phagocytized by immune cells, which are in turn visualized via MRI. We tried to address these issues in our study design and in the choice of histologic and immunohistologic methods. The inclusion of control group b) injection of pure SPIO particles and control group c) injection of nonvital MSCs allows differentiation to our treatment group. On a histologic level, the combination of H&E, PB, and TUNEL clearly indicates the presence of viable cells with intracellular SPIO particles within the synovial lining, neomeniscus, and fat tissue after injection of labeled MSCs. There was no increased amount of dead cells based on TUNEL stain in the respective area. However, we did not detect any labeled cell in the osteochondral samples. This is in partial agreement with a previous study (21), where no cells were found within the cartilage, but they observed cells in the subchondral bone at 12 weeks.
The following question addresses the phenotype of the detected local cells, that is, as to whether they are MSCs or differentiated progeny cells. At present, there are no comprehensive methods for characterizing implanted cells, and as it was pointed out in a previous study (23), there is a “big unmet need in the field of MSC trafficking.” Most of the studies assessing this topic are related to homing ability of systemically (intravenous, intra-arterial) injected MSCs. Maintained stemness, specifically proliferation capacity of locally engrafted MSCs, was proven by using serial transplantation (25). In our study, chondrogenic activity was detected histologically by S100β or Safranin O staining, which might indicate that injected MSCs differentiated to the requested, that is, the chondrogenic cell type as these markers are not present in undifferentiated MSCs. Expression of cell surface markers of the targeted tissue following systemic MSC infusion has been described in a muscle dystrophy model (8), damaged intestinal epithelial cells model (40), and after homing in healthy bone marrow (31). In contrast to that, studies evaluating the fate of SPIO-labeled MSCs after intramyocardial injection revealed the presence of iron-containing CD68+ cells (34) or ED1+ cells (2), that is, macrophages, in positive PB areas. Both authors concluded that MRI overestimates MSC survival after local administration but noted a treatment effect.
This leads to the question as to how MSCs may contribute to joint healing as a positive effect of intra-articularly injected MSCs has been described in clinical (14,37) and experimental settings (11,21,26,27). It has been suggested that not the differentiations of the MSCs into the targeted cells of the host tissue, but rather secreted bioactive factors are responsible for the effects seen after MSC administration in various tissues (6). MSCs are described as suppressing the local immune system, inhibiting fibrosis (scar tissue), inhibiting apoptosis, enhancing angiogenesis, and stimulating mitosis and differentiation of local MSCs into repair tissue.
Another aspect is the exact identity of the applied MSCs. From studies evaluating homing ability of MSCs, it is known that several factors influence cell character, namely, MSC culture conditions, timing, and mode of delivery (23). Subsets of MSCs express different surface markers (18) and may therefore differ in behavior and fate. We used standard culture conditions for isolation, proliferation, and cryopreservation, and cells of passage 3 were always used for injection, though we did not characterize surface markers of our cells. Additionally, SPIO particles influence chondrogenic differentiation (22), and therefore 10 × 106 nonlabeled MSCs were additionally injected. In our study, we monitored only labeled cells. The in vivo fate of nonlabeled cells is unknown, and it is unclear how much SPIO labeling may influence cell fate.
A further question arises as to how much coinjected substances may influence cell engraftment and fate. We refrained from any additives on purpose; however, this hampers direct comparison to previous studies. In clinical studies, hyaluronic acid is the most frequently used compound (14,37). A comparable tracking study also using SPIO particles and MRI added chitosan and glycerophosphate gel (21).
Measuring the degree of engraftment is another recognized unsolved problem (23). This starts with the type of sampling, which may lead to false-negative errors. We obtained four standardized samples: areas where brown discoloration was most frequently seen (caudal joint capsule, cranial fat pad), and research target tissue (neomeniscus, cartilage defect). Additional niches of cell engraftment might have been missed. Comparative quantification of radionuclide-labeled MSCs was achieved by measuring radioactivity of the target tissue after systemic injection (4) or local delivery (32,33,35). We semiquantitatively scored the PB histologic samples for the amount of iron (SPIO-labeled cells) (0=no PB stain; 1 = few cells PB+; 2=moderate amount of PB+ cells; 3 = large amount of PB+ cells) to be able to estimate the number of local cells.
Several questions arose from our results, and further studies are needed to improve techniques for assessing MSC engraftment and MSC efficacy, that is, by modifying cell surface molecules by chemical or genetic coating in order to improve selective adherence to particular organs or tissues (23,24).
In conclusion, we accept our first hypothesis that, based on MRI evaluation, viable SPIO-labeled MSCs are retained intra-articularly for up to 12 weeks in contrast to nonviable cells or pure SPIO markers. Our second hypothesis can only be partially accepted since SPIO-labeled cells were verified within the synovial membrane and the neomeniscus, but none were found in the osteochondral tissue. Our third hypothesis can also be accepted in part: on histologic and immunohistologic evaluation, we were able to show viable, SPIO-containing cells with potential chondrogenic phenotype.
Footnotes
Acknowledgments
The authors would like to thank Mrs. Ines Merseburger for her excellent work in acquiring the MR images and Prof. Dr. Johannes Seeger and Mrs. Gabriele Lindner for their support during histologic processing. We would also like to thank Mr. Miguel Espina, Dr. Christin Veit, and Mrs. Doreen Stein for taking such good care of our research sheep. Thanks also go to Ms. Sabine Wilhelm, Wuerzburg, for her technical assistance. The work presented in this article was made possible by funding from the German Federal Ministry of Education and Research (BMBF, PtJ-Bio, 0313909/0315883). Results of this project have been published previously as an oral presentation at the World Conference on Regenerative Medicine (WCRM), Leipzig, Germany, in November 2011. The authors declare no conflict of interest.
