Abstract
The mechanisms underlying the functional improvement after injection of multipotent mesenchymal stromal cells (MSCs) in infarcted hearts remain incompletely understood. The aim of this study was to investigate if soluble factors secreted by MSCs promote cardioprotection. For this purpose, conditioned medium (CM) was obtained after three passages from MSC cultures submitted to 72 h of conditioning in serum-free DMEM under normoxia (NCM) or hypoxia (HCM) conditions. CM was concentrated 25-fold before use (NCM-25X, concentrated normoxia conditioned medium; HCM-25X, concentrated hypoxia conditioned medium). The in vitro cardioprotection was evaluated in neonatal ventricular cardiomyocytes by quantifying apoptosis after 24 h of serum deprivation associated with hypoxia (1% O2) in the absence or presence of NCM and HCM (nonconcentrated and 25-fold concentrated). The in vivo cardioprotection of HCM was tested in a model of myocardial infarction (MI) induced in Wistar male rats by permanent left coronary occlusion. Intramyocardial injection of HCM-25X (n = 14) or nonconditioned DMEM (n = 16) was performed 3 h after coronary occlusion and cardiac function was evaluated 19—21 days after medium injection. Cardiac function was evaluated by electro- and echocardiogram, left ventricular catheterization, and treadmill test. The in vitro results showed that HCM was able to decrease cardiomyocyte necrosis. The in vivo results showed that HCM-25X administered 3 h after AMI was able to promote a significant reduction (35%) in left ventricular end-diastolic pressure and improvement of cardiac contractility (15%) and relaxation (12%). These results suggest that soluble factors released in vitro by MSCs are able to promote cardioprotection in vitro and improve cardiac function in vivo.
Keywords
Introduction
Multipotent mesenchymal stromal cells (MSCs) are plastic-adherent cells with heterogeneous morphology obtained from different tissues, including bone marrow (12,15). Several studies have shown that these cells are capable of differentiation, in vitro, into various cell types such as adipocytes, osteocytes, chrondocytes, and, in some instances, cardiomyocytes (17,24,39). Given the large amount of in vitro data regarding the plasticity of various bone marrow-derived cell populations, it is tempting to conclude that cell therapy exerts its dominant effects through differentiation. Based on this principle, cell therapy with MSCs after myocardial infarction (MI) was first proposed aiming to regenerate the heart by replacing lost tissue (21,30,45).
However, there is increasing evidence showing that differentiation of bone-marrow derived cells into host tissue cells and/or even cell fusion (2,49) do not occur in vivo at a frequency high enough to explain functional improvement observed after cell therapy (5,7,16). Furthermore survival of MSCs and engraftment after cell injection seems to be minimal despite functional improvement, which suggests that beneficial indirect effects may manifest earlier and independent of engraftment (8,38,54,59). Thus, other mechanisms are likely to contribute to the benefits seen after therapy using MSCs, such as the paracrine release of soluble factors by these cells (22,23). One of the studies aimed at directly evaluating this hypothesis in infarcted hearts was conducted using MSCs overexpressing Akt1 (Akt-MSCs) (13). These authors showed that conditioned medium obtained from Akt-MSCs was able to promote cardioprotection in cardiomyocytes subject to hypoxia in vitro and improved cardiac function evaluated in a Langendorf preparation.
An antiapoptotic paracrine effect has been suggested as a mechanism for the improvement of heart function after cell therapy with MSCs in infarcted hearts (7,13,51). It is known that after MI, necrosis is the prevailing form of damage produced by coronary artery occlusion, but apoptotic cell death also contributes to the loss of myocytes in the ischemic heart (20) and the remote area of the infarct (6,11,42). Although the relevance of cell death in the pathophysiology of MI has been reported by several studies in animal models (20,29,37) and in humans (1,28,35,42), cytoprotective therapy and its clinical relevance are still limited (18,26,46). Studies focused on investigating antiapoptotic and antinecrotic effects of soluble factors released from MSCs and their relevance for recovery of cardiac function after MI are still scarce (13,44,55).
Therefore, the aim of the present study was, first, to investigate if soluble factors released by MSCs in culture under normoxia and hypoxia conditions are able to promote cardioprotection in vitro by antiapoptotic and/ or antinecrotic effect. In addition, we conducted experiments in vivo using hypoxic MSC-conditioned medium in animals infarcted by left anterior descendent coronary artery ligation.
Materials and Methods
Animals
All procedures were performed in accordance with US National Institutes of Health guidelines as attested to by the competent institutional board.
Wistar rats (200—250 g) were housed at controlled temperature (23°C) with daily exposure to a 12-h light/ dark cycle (lights on 08:00—20:00 h) and free access to both water and standard rat chow.
Multipotent Mesenchymal Stromal Cell Culture
The isolation and primary culture of MSCs was performed as described by Caplan (9). Briefly, total rat bone marrow was harvested by flushing the tibias and femurs with Dulbecco's modified Eagle medium (DMEM, Gibco-Invitrogen, Carlsbad, CA, USA) and the cell suspension centrifuged in Ficoll gradient at 400 x g for 30 min (Histopaque 1.083 g/ml, 1:1, Sigma-Aldrich, St. Louis, MO, USA). Mononuclear cells were collected from histopaque medium interface. Cells were washed in buffered saline solution (BSS) three times, counted in a hemocytometer, and checked for viability using 0.4% trypan blue. Cells were then plated at a density of 1.2 × 106 cells/cm2 and maintained at 37°C in a 5% CO2 incubator for 1 week, during which medium was changed at least twice, washing away all floating hematopoietic cells. Culture medium used was DMEM supplemented with 10% fetal bovine serum (Gibco-Invitrogen, Carlsbad, CA, USA), 2 mM l-glutamine (Sigma-Aldrich), and antibiotics (100 U/ml penicillin G and 100 μg/ml streptomycin, Gibco). At approximately 80—90% confluence, adherent cells were detached from the culture flasks with 0.25% trypsin-EDTA (Sigma-Aldrich) and replated at a density of 1.2 × 104 cells/cm2. These cells were further propagated until passage 3 for collecting conditioned medium. These cells, purified by adherence to plastic culture flasks, were demonstrated to be positive for CD29 and CD90 surface markers and negative for CD34, CD45, and CD11b and, additionally, capable of differentiating into osteoblasts and adipocytes (10). Together, these findings are in accordance to criteria for MSC definition established by International Society for Cellular Therapy (15).
Collection of Conditioned Medium
After the third replating (1.2 × 104 cells/cm2) had reached approximately 70—80% confluence, the culture was gently rinsed three times with BSS and the standard medium was replaced by serum-free DMEM. Cells were then incubated under normoxia or hypoxia condition (O2 1% and CO2 5%) at 37°C in a CO2/O2 incubator (Thermoforma model 3130). After 72 h of conditioning, conditioned medium (CM) obtained under normoxia (NCM) or hypoxia (HCM) condition was centrifuged and filtered through a 0.22-μm filtration device. After each medium collection, the cells were detached by tripsin-EDTA 0.25% and counted in hematocytometer. Approximately 15 ml of CM was obtained by conditioning 106 cells. In addition, cell viability was tested after both normoxia and hypoxia conditions. NCM and HCM were concentrated 25-fold by ultrafiltration using a centriprep® device (YM-10 Millipore) equipped with a 10 kDa cut-off membrane. Nonconditioned serum-free culture medium (DMEM) was submitted to the same procedure and used as control.
Cardiomyocyte Culture and Experimental Conditions
In vitro effect of CM was tested on ventricular myocytes isolated from the hearts of neonatal Wistar rats (1—2 days after birth). Neonatal cardiomyocytes were obtained according to Suadicani et al. (47). Briefly, hearts from neonatal rats were removed, minced, and serially digested by dissociative solution (NaCl 136.7 mM; KCl 2.68 mM; Na2HPO4 0.352 mM; NaHCO3 11.9 mM; dextrose 11 mM) containing pancreatin (1.25mg/ ml; sigma) and bovine serum albumin (3 mg/ml; Sigma). Each successive digestion lasted for 5 min at 37°C with gentle stirring and the cells obtained were suspended in growth medium containing 10% fetal bovine serum (FBS) for inhibition of proteolytic enzymes. Elimination of non-muscle cells was achieved by preplating twice for 1 h each. After that, the cells were counted and seeded in 24-well plates at a density of 1.5 × 105 cells/well and cultured in DMEM high glucose containing 10% FBS at 37°C and 5% of CO2. Experimental in vitro conditions were established 48—72 h after plating. Then cultures were washed with BSS and cardiomyocyte apoptosis was induced by serum deprivation (S-) associated to hypoxia (H+) at 1% O2 using a CO2/O2 incubator for 24 h in the absence (cell death induction; S-/H+) or presence of CM (NCM or HCM) at two different dilutions: nonconcentrated and 25-fold concentrated. For the control normoxic condition (control, S+/H-), we used DMEM with 10% FBS and the culture was submitted to 24 h of 21% O2 and 5% CO2. Three independent experiments were made in triplicate for each experimental condition.
Cardiomyocyte Apoptosis/Necrosis Assay
Apoptosis and necrosis were identified by means of double fluorescence staining with Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis detection kit (Sigma). Briefly, after 24 h under experimental conditions, cardiomyocytes from each well were separately collected after rinsing with Ca2+ and Mg2+-free saline solution three times. The remaining attached cardiomyocytes were removed by adding 500 μl/well of trypsin-EDTA (0.25%) without rinsing for 3 min. After that, trypsin was inactivated by adding 2 ml of DMEM supplemented with 10% FBS and the cells were centrifuged at 250 x g for 5 min. For quantification of cardiomyocyte death, the pellet was resuspended in 500 μl of binding buffer (HEPES 10 mM; NaCl 140 mM; CaCl2 2.5 mM; dextrose 11 mM) and incubated in the dark for 10 min with 5 μl of Annexin V-FITC and 10 μl of PI at room temperature. Dual color analysis was performed using a FACScalibur flow cytometer (Becton Dickinson) by counting 20,000 cells (FL1-H filter for Annexin V-FITC and FL3-H filter for PI). This combination detects cells in apoptosis (Annexin V positive, PI negative), necrosis (Annexin V positive, PI positive), and viable cells (Annexin V negative, PI negative) (52).
In Vivo Study of Effect of Conditioned Medium
In vivo cardioprotective effect of CM was tested in a model of acute myocardial infarction (AMI) induced in Wistar male rats weighing 200—250 g.
Myocardial Infarction
MI was induced by left coronary artery ligation as previously described (34). Briefly, rats were anesthetized with halothane (Merck, Deutschland) and a small thoracotomy was made on the left side between the fifth and sixth ribs. After exposing the heart, the descending branches of the left coronary artery were permanently occluded with a 5.0 silk suture, and then the chest was closed. Sham-operated animals underwent the same procedure but without the coronary ligation.
Experimental Protocol
The aim of these experiments was to investigate if soluble factors released in vitro by MSC under hypoxia would improve cardiac function in AMI. For this purpose, 3 h after coronary occlusion, animals were reanesthetized and electrocardiography (ECG) and echocardiography (ECHO) were performed to confirm infarction. Immediately after that, rats were randomly selected and the chest was reopened for intramyocardial single injection of 100 μl of CM previously obtained under hypoxia and concentrated 25-fold (HCM-25X; n = 14) or of nonconditioned serum-free culture medium (DMEM, control; n = 16). Injection was made at the border of MI. Cardiac function was evaluated 19—21 days later. Sham animals (SHAM; n = 9) were used as controls.
Functional Analyses
Electrocardiogram
The animals were anesthetized with ketamine and xylazine (50 and 5 mg/kg, respectively, IP) and placed in the supine position. Electrodes were connected to surgical needles and inserted subcutaneously in order to record the classical six limb leads (L1, L2, L3, aVR, aVL, and aVF) as previously described (41). âQRS > 90° and presence of Q wave in L1 were used as evidence of infarction, as previously shown (27).
Echocardiogram
Echocardiographic examination was performed blinded by the same echocardiographist, following the American Society of Echocardiography (ASE) recommendations. We used an echocardiographic color system (Megas/Esaote) equipped with a 10-MHz electronic-phased-array transducer. Short-axis two-dimensional views of the left ventricle (LV) were taken at the level of the papillary muscles. In order to estimate LV global function, we calculated LV diastolic and systolic area (LVDA, LVSA, respectively) to determine fractional shortening area: FSA (%) = [(LVDA − LVSA)/ LVDA] x 100.
Treadmill Test
Nineteen days after MI animals were weighted and had their cardiac function analyzed under physiological stress (exercise). The test was performed according to an already established protocol (53). We evaluated the maximal time of endurance running of the animals. In brief, after an adaptation period of 3 days running at 17 cm/s for 5 min, the animals started to run in a treadmill with a constant slope of 10° and velocity of 17 cm/s. Each 2 min, the velocity was increased by 2 cm/s until exhaustion. Stainless steel grids provided an electrical shock to keep the animals running. Exhaustion was determined whenever the animal stayed on the steel grids for more than 15 s despite the shocks.
Left Ventricular Pressure Measurements in Unanesthetized Rats
Twenty-one days after MI, animals were anesthetized with ketamine/xylazine and the right carotid artery was cannulated with a PE10 catheter. One extremity of the catheter was inserted into the left ventricle and the opposite end was tunneled back and exposed between the scapulae. With the animal free to walk about in its cage 24 h after this procedure, the catheter was connected to a pressure transducer (MLT0380/D, ADInstruments) coupled to a Power Lab400 acquisition system (ADInstruments) and LV pressure was measured in awake nonanesthetized animals according to protocol established per our group in infarcted rats (53). After an adaptation period of 30 min, LV pressure acquisition started. The left ventricular parameters evaluated were: left ventricular end diastolic pressure (LVEDP), left ventricular end systolic pressure (LVESP), left ventricular developed pressure [(LVDP) = LVESP − LVEDP], cardiac index of left ventricular contractility (dPdT+: rate of pressure rise), and index of left ventricular relaxation (dPdT-: rate of pressure decay). All values were measured using Chart 4 software (PowerLab, ADInstruments).
Histological Procedures
After all cardiac function analysis, the animals were sacrificed and had their hearts excised for histological analysis. After arrest in diastole by KCl (30 mM) perfusion, the hearts were fixed by immersion in 4% paraformaldehyde in phosphate buffer. Ventricles were sectioned in four slices from apex to base (A, B, C, and D, respectively). Infarct size determination was performed by computer-assisted planimetry (ImageJ version 1.27z, National Institute of Health, USA) as the mean percent value of the circumference occupied with the scar tissue on each section, after staining with Picrosirius (sirius red) for collagen scar evaluation.
Statistical Analysis
Values are mean ± SEM. For the in vitro study we used one-way ANOVA followed by Dunnett post hoc test. For the in vivo study, ECHO and AQRS parameters (baseline and after injection) were evaluated by two-way ANOVA followed by Bonferroni post hoc test. The ECG QRSi at 21 days, exercise time, and left ventricular parameters between DMEM and HCM-25X were analyzed by nonpaired t-test. Values were considered significant when p < 0.05.
Results
In Vitro Effect of Conditioned Medium Obtained Under Normoxia
The putative cytoprotective effect of CM was tested first in vitro on cardiomyocytes submitted to serum deprivation associated to hypoxia (S-/H+). Nonconcentrated and 25-fold concentrated NCM and HCM were used. As expected (Fig. 1A), apoptosis (Annexin-V+/PI-) was higher under hypoxia (S-/H+: 16 ± 4%) than normoxia (S+/H-: 5 ± 1%) condition. Nonconcentrated NCM was not able to prevent cardiomyocyte apoptosis (NCM: 18 ± 6%) even when 25-fold concentrated. Although with the concentrated medium there was tendency to reduce apoptosis, this did not reach statistical significance (NCM-25X: 10 ± 5%).
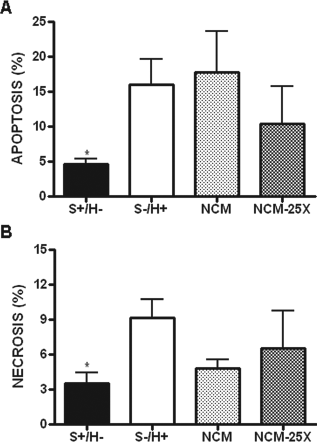
In vitro effect of conditioned media obtained under normoxia (NCM). Apoptosis/necrosis percentage quantified by flow cytometry after Annexin V/propidium iodide (PI) staining in cardiomyocytes submitted for 24 h to normoxia with serum (S+/H-), or to hypoxia and serum deprivation (S-/H+) or to hypoxia with different concentrations of NCM (NCM: nonconcentrated normoxic CM and NCM-25X: 25-fold concentrated normoxic CM). (A) Apoptosis percentage characterized by Annexin-V+/PI- staining. (B) Necrosis percentage characterized by Annexin V+/PI+ staining. *p < 0.05 versus S-/H+.
The percentage of necrosis (Annexin-V+/PI+) (Fig. 1B) was again higher at hypoxia (9 ± 2%) than normoxia (4 ± 1%). Under this condition, neither NCM nor NCM-25X was able to reduce percentage of cardiomyocyte necrosis (NCM: 5 ± 1%; NCM-25X: 7 ± 3%).
In Vitro Effect of Conditioned Medium Obtained Under Hypoxia
We postulated that CM obtained under hypoxia (HCM) would better mimic soluble factors release by MSC injected at the border zone in vivo after a myocardial infarction. After 24 h of hypoxia (Fig. 2A), we observed that independent of the HCM concentration used, cardiomyocyte apoptosis had a tendency to decrease but did not reach statistical significance when compared to hypoxia and serum deprivation (HCM: 12 ± 4%; HCM-25X: 13 ± 6% vs. 17 ± 4% under S-/H+). However, necrosis (Fig. 2B) was significantly attenuated by HCM 25-fold concentrated (HCM-25X: 4 ± 1% vs. 9 ± 2% under S-/H+; p < 0.05).

In vitro effect of CM obtained under hypoxia (HCM). Apoptosis/necrosis percentage quantified by flow cytometry after Annexin V/PI staining in cardiomyocytes submitted for 24 h to normoxia with serum (S+/H-), or to hypoxia and serum deprivation (S-/H+) or to hypoxia with different concentrations of HCM (HCM: nonconcentrated hypoxic CM and HCM-25X: 25-fold concentrated hypoxic CM). (A) Apoptosis percentage characterized by Annexin-V+/PI- staining. (B) Necrosis percentage characterized by Annexin-V-/PI+ staining. *p < 0.05 versus S-/H+.
In Vivo Effect of Concentrated Hypoxia Conditioned Medium
ECG Parameters
Since only HCM-25X was able to significantly decrease the necrosis process in our in vitro experiments, we decided to test only this CM in our in vivo experiments. All animals included in the infarcted groups presented Q wave in L1 3 h after coronary ligation. Intramyocardial injection of DMEM or HCM-25X was not able to reverse rightward deviation, but 21 days after MI QRSi was approximately 28% lower in DMEM-injected animals (0.92 ± 0.07 mV) than in HCM-25X (1.18 ± 0.05 mV, p < 0.01), which suggests a larger mass of depolarizing myocytes and/or better electrical conductivity in the infarcted hearts that received HCM-25X.
ECHO
Three hours after MI, all infarcted groups presented similar decreased FSA values (DMEM: 40 ± 2%; HCM-25X: 35 ± 3%; p > 0.05) when compared to Sham (65 ± 4%) (Fig. 3). At 21 days, FSA was maintained in Sham animals while in the DMEM control group additional reduction of approximately 22% was observed (31 ± 2% vs. 40 ± 2% at 3 h; p < 0.01). HCM-25X injection diminished this reduction in FSA (31 ± 3% vs. 35 ± 3% at 3 h; p > 0.05). However, FSA at 21 days in HCM-25X was not different from the DMEM group.

In vivo effect of CM 25-fold concentrated obtained under hypoxia (HCM-25X) on echocardiographic (ECHO) parameter. Fractional shortening area (FSA) calculated from LVEDA (left ventricular end diastolic area) and LVESA (left ventricular end systolic area). ECHO was performed at 3 h (preinjection; black bars) and 21 days after intramyocardial injection (white bars). Groups: DMEM (n = 16) and HCM-25X (n = 14). Dashed line indicates FSA mean value in the Sham group (n = 9). *p < 0.05 versus DMEM at 3 h.
Exercise Test
All infarcted groups presented lower exercise capacity, shown as total exercise time (Fig. 4), compared to sham animal (Sham: 22.5 ± 1.1 min). Exercise capacity was unchanged after medium injection and no difference was observed between infarcted groups (DMEM: 14.47 ± 0.7 min; HCM-25X: 14.92 ± 1.3 min). Therefore, intramyocardial injection of HCM was not able to improve cardiac function under physiological stress induced by exercise.

In vivo effect of CM 25-fold concentrated obtained under hypoxia (HCM-25X) on total exercise time. Total exercise time obtained 20 days after intramyocardial injection. Groups: DMEM (n = 16) and HCM-25X (n = 14). Dashed line indicates mean value of total exercise time in Sham group (n = 9).
Left Ventricular Pressure Measurements in Unanesthetized Rats
The left ventricular pressure (LVP) parameters in unanesthetized rats measured 21 days after medium injection are shown in Figure 5. All infarcted groups showed increases in LVEDP and reductions in LVSP, LVDP, and contractility (DPDT+) and relaxation (DPDT-) when compared to Sham animals (LVEDP: 4 ± 1 mmHg; LVSP: 133 ± 5 mmHg; LVDP: 130 ± 7 mmHg; DPDT+: 8119 ± 353 mmHg/s; DPDT-: −6200 ± 263 mmHg/s). The HCM-25X group presented levels of LVEDP (Fig. 5A) about 35% lower than those measured for the DMEM group (HCM-25X: 13 ± 2 mmHg vs. DMEM: 20 ± 2 mmHg, p < 0.05). In addition, HCM-25X injection increased cardiac contractility (Fig. 5C) by approximately 15% (HCM-25X: 6169 ± 347 mmHg/s vs. DMEM: 5231 ± 266 mmHg, p < 0.05) and improved cardiac relaxation (Fig. 5D) by about 12% when compared to DMEM administration (HCM-25X: −4549 ± 223 mmHg/s vs. DMEM: −4000 ± 133 mmHg/s, p < 0.05). However, LVEDP, DPDT+, and DPDT- in HCM-25X were still different when compared to Sham. HR was slightly higher in HCM-25X than in the DMEM group (352 ± 10 bpm vs. 317 ± 13 bpm in DMEM, p < 0.05). LVSP (HCM-25X: 110 ± 3 mmHg; DMEM: 109 ± 2 mmHg, p > 0.05) and LVDP (HCM-25X: 97 ± 4 mmHg; DMEM: 88 ± 3 mmHg, p > 0.05) were not different between infarcted groups.

In vivo effect of CM 25-fold concentrated obtained under hypoxia (HCM-25X) on left ventricular parameters. (A) Left ventricular end-diastolic pressure (LVEDP), (B) heart rate (HR), (C) cardiac index of left ventricular contractility (dP/dT+), and (D) cardiac index of left ventricular relaxation (dP/dT-) were obtained 21 days after intramyocardial injection of DMEM (n = 13) or HCM-25X (n = 13). Dashed line indicates mean value of respective LV parameters in Sham group (n = 6). *p < 0.05 versus DMEM.
Infarct Size
Figure 6 summarizes the results of infarct size. Left coronary artery ligation induced large infarcts (40% in average). Intramyocardial injection of HCM-25X was not able to reduce infarct size when administered 3 h after MI in comparison to the DMEM group.
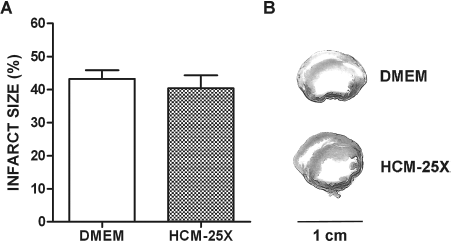
Infarct size as percentage of left ventricular perimeter. (A) Infarct size measured 21 days after myocardial infarction. (B) Representative slices of infarcted hearts from the DMEM (n = 16) and HCM-25X (n = 14) groups stained by Picrosirius red for collagen scar evaluation.
Discussion
A number of studies suggest a paracrine effect in which MSCs secrete angiogenic (22,23) and/or antiapoptotic factors (13,55) capable of protecting tissues from ischemic damage. Mangi et al. (25) showed that intramyocardial injection of MSCs genetically modified to overexpress Akt protein (MSC-Akt) improved cardiac function 72 h after cell implantation. Because the functional improvement was observed after a short period of time, the authors suggested that cytoprotective factors released by MSCs were responsible for attenuating myocyte loss and thus promoting improved function. This same group later showed that injection of MSC-Akt conditioned medium only was sufficient to promote similar improvements in cardiac function and that cell incorporation in native tissue was not necessary (13).
In the present study, we opted to obtain CM from MSCs not genetically modified but exposed to conditions similar to those encountered in the postinfarct myocardium (i.e., under hypoxic conditions). It is known that hypoxia increases the secretion of growth and angiogenic factors, and cytokines such as fibroblast growth factor (FGF), vascular endothelial growth factor (VEGF), angiopoietin 1 (Ang-1), platelet-derived growth factor (PDGF), interleukin-1 (IL-1) and IL-6 by MSCs in culture (23). The conditioning time of 72 h was chosen based on the literature (22,23) and on experiments using CM on chicken embryo sympathetic neurons, a throphic sensor model (40), where CM obtained after 72 h of conditioning was able to prevent neuronal death and induce neuritogenesis, an effect absent using 24 and 48 h of conditioning (unpublished data).
The putative cardioprotector effect of CM was then investigated in vitro in rat neonatal cardiomyocytes submitted to hypoxia for 24 h. Preliminary results using ethidium bromide staining in permeabilized cardiomyocytes followed by flow cytometry quantification of cell hypodiploid content showed that CM obtained under normoxia reduced cardiomyocyte apoptosis induced by serum deprivation for 24 h (data not shown). In subsequent experiments, we opted to use Annexin V/PI staining and flow cytometric quantification aiming to discriminate apoptosis from necrosis. In vitro results showed that CM obtained either under normoxia or hypoxia conditions was not able to significantly reduce the percentage of cardiomyocytes in apoptosis induced by hypoxia and serum deprivation. On the other hand, HCM-25X diminished the percentage of cardiomyocytes in necrosis. Spontaneously beating cardiomyocytes were visualized under experimental conditions with NCM and HCM, in contrast to that observed with control DMEM in which spontaneous contractions were always absent. These results suggest that soluble factors released by MSCs under hypoxia protect cardiomyocyte from necrosis.
Based on our in vitro results and considering that necrosis is the main cell death mechanism after MI, we aimed to investigate in vivo if a single intramyocardial injection of CM obtained under hypoxia would be able to improve cardiac function when injected 3 h after MI. Hypoxic medium for in vivo experiments was chosen, aiming to better mimic the ischemic microenvironment and to test the hypothesis of a paracrine mechanism. It is known that hypoxia can induce upregulation of various factors related to angiogenesis and survival, and even lead to expression of cytoprotective factors not normally expressed under normoxia (13,23).
We showed that a single intramyocardial injection of HCM-25X was not able to improve cardiac performance but prevented additional reduction in cardiac function evaluated by ECHO by means of FSA. In addition, ECG studies showed that although QRS right axis deviation was not prevented by CM injection, QRS index at 21 days was 28% higher in the HCM-25X than in the DMEM group. In awake nonanesthetized rats, a single intramyocardial injection of HCM-25X promotes significant reduction (~35%) in LVEDP and improvement in cardiac contractility (~15%) and relaxation (~12%) compared to control DMEM injection. Improvement in cardiac contractility and relaxation per se may be responsible for decreased LVEDP observed in the HCM-25X group. In addition, we can speculate that this reduction in LVEDP and improvement in relaxation may also be related to an antifibrotic effect of CM (5,58) in areas remote to infarction or even remodeling effect on scar tissue (57), increasing cardiac compliance and promoting improvement in diastolic function (33,44). It is known that reduction in LVEDP promotes attenuation of wall stress in infarcted hearts (19), preventing additional cell death in remote areas to infarction along time (32).
It could be argued that the increased heart rate observed in the HCM group was responsible for the lower LVEDP and for better cardiac contractility and relaxation. However, the relationship between heart rate and contractility seems to be complex in rat heart, differing from other mammals (31,48,50). In fact, Nalivaiko et al. (31), using a rat heart—brain stem preparation, showed that the contractility falls if the pacing rate rises within the physiological range (200—500 beats min−1), but it also tends to fall during substantial drops of heart rate. This force—frequency relationship seems to be related to Ca2+ dynamics. These same authors showed that LVEDP increases with augmentation in heart rate due to reduction in relaxation time. Together, these observations exclude the fact that variations in heart rate in our study, which was kept within rat physiological range, are responsible for alteration on LV parameters in the HCM-25X group.
Our in vivo results are at odds with Gnecchi et al. (13), who showed no improvement in cardiac function with CM obtained under hypoxia from nonmodified MSCs. These authors only demonstrated improvement in cardiac function when CM was obtained from MSCs genetically modified to overexpress Akt. Here, we demonstrated that although modest, CM obtained under hypoxia from nonmodified MSCs can prevent additional reduction in cardiac function with time, improve cardiac contractility and relaxation, and decrease LVEDP. Conditioning time of 12 h and/or the window for myocardial injection (30 min after MI) besides different methodological approaches for functional analysis chosen by these authors may explain the difference in results. In addition, functional improvement using non-genetically modified MSCs has been reported in several studies in the literature, suggesting that factors secreted by these cells promote cardioprotection (3,7,16,45).
In relation to infarct magnitude, the results of our study showed that CM obtained under hypoxia was not able to alter infarct size. However, functional improvement has been recorded after cell therapy with MSCs even without changes in infarct size (7,16). In addition, we cannot rule out the putative hypertrophic effect of CM on the remaining myocardium. Indeed, Xu et al. (56) demonstrated that MSCs promote cardiomyocyte hypertrophy through hypoxia-induced paracrine mechanisms.
Since we did not measure capillary density, one cannot rule out the possibility of functional improvement also being due to a paracrine angiogenic effect, since many of the MSCs secreted factors are known to participate in the angiogenesis process. However, functional improvement after therapy with MSCs described by some authors seems to occur more rapidly than the time required for angiogenesis (13,25).
CM could also be promoting proliferation and/or activation of native cardiac stem cells (4,14), thus stimulating an endogenous repair mechanism. Still another possible explanation for the cardiac improvement observed in our study can be related to cardiac nerve sprouting. Indeed, Pak et al. (36) observed that intramyocardial injection of MSCs induced sympathetic nerve sprouting in infarcted swine hearts. Moreover, it is important to emphasize that experiments in our laboratory showed that CM obtained from MSCs was able to prevent cell death and induced neuritogenesis in a culture of sympathetic neurons obtained from chicken embryos. So putatively, sympathetic hyperinnervation could be responsible for cardiac functional improvement observed in the HCM-25X group.
Our results show a modest improvement in cardiac function when compared to results from direct MSC injections, which on average can promote increases of about 30—60% in contractility and relaxation, respectively (7,14,16). This suggests that the presence of MSCs in the tissue amplifies the cardiac functional improvement in infarcted rat hearts. The additional benefit given by the presence of cells can be related to different causes. First is the continuous secretion of factors while MSCs are alive, which contrasts with a single injection of conditioned medium as performed in our study. Second, we cannot exclude differences in secretory pattern when these cells are injected into the receptor tissue, whose microenvironment is not perfectly simulated by the in vitro hypoxic conditions. In addition, human cardiac explants have been shown to promote proliferation and differentiation of MSCs into a cardiac-like phenotype (43). Therefore, future studies will be important to elucidate the nature and importance of soluble factors released by MSCs on cardiac function of infarcted hearts. Maybe, once these factors and the intracellular pathways involved in their cardioprotective action have been dissected out, a therapy based only on soluble factors derived from MSCs could be used therapeutically.
In conclusion, the present study showed that MSCs under hypoxia secrete soluble factors capable of promoting cardioprotection in vitro. For in vivo experiments, HCM-25X promotes a modest improvement in cardiac function when compared to the much larger effect of MSC therapy.
Footnotes
Acknowledgments
This work was supported by Decit, CNPq, CAPES, FAPERJ, and NIH (R01 HL073732-08). The authors declare no conflict of interest.
