Abstract
The aim of this prospective randomized double-blind clinical study is to examine the benefits of using olfactory ensheathing cells (OECs) combined with or without Schwann cells (SCs) in treating chronic complete spinal cord injuries (SCIs). This would offer patients a better alternative for neurological functional recovery. According to the initial design, 28 eligible participants with cervical chronic complete SCI were recruited and randomly allocated into four groups of seven participants each. The neurological assessments were to be performed according to the American Spinal Injury Association (ASIA) and International Association of Neurorestoratology (IANR) Functional Rating Scales, in combination with electrophysiological tests, for example, electromyography (EMG) and paraspinal somatosensory evoked potentials (PSSEPs). Here we have summarized the data from seven patients; three patients received an OEC intraspinal transplantation, one underwent SC implantation, and one received a combination of OECs and SCs. The remaining two patients were used as controls. The scores were evaluated independently by at least two neurologists in a blinded fashion for comparing the neurological functional changes during pre- and post-cell transplantation (6-month follow-up). All patients who received OECs, SCs alone, and a combination of them showed functional improvement. Mild fever occurred in one of the patients with OEC transplant that subsided after symptomatic treatments. All treated patients except one showed improvement in the electrophysiological tests. The functional improvement rate comprises 5/5 (100%) in the treated group, but 0/2 (0%) in the control group (p = 0.008). These preliminary findings show that transplanting OECs, SCs, or a combination of them is well tolerated and that they have beneficial effects in patients. Thus, further studies in larger patient cohorts are warranted to assess the benefits and risks of these intervention strategies. This manuscript is published as part of the IANR special issue of Cell Transplantation.
Keywords
Introduction
Recently, the treatment of spinal cord injury (SCI) has undergone novel changes in concept, though the disease still remains incurable (22,27,28). New research is largely being conducted in studies of clinical cell therapies [bone marrow (BM) or peripheral blood mononuclear cells (PBMNCs), olfactory ensheathing cells (OECs), and Schwann cells (SCs)] (9,19,31,37,46,54,63,66,71,75), growth factors [e.g., brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3)] (5,20,52,74), inhibitory molecules [e.g., NG2 (also known as chondroitin sulfate proteoglycan 4; CSPG4), myelin protein) (17,50,65), and combination therapies (e.g., cell grafts, neurotrophins, cotransplantation) (12,32,47,51,53).
OECs and SCs are closely related cell types with profound restoration-promoting properties (25,58,62,72). OECs may offer greater advantage as a cellular therapy for SCI by enhancing axonal regeneration and remyelination, providing trophic support for injured neurons and stimulating angiogenesis (13,33,36,40,48,49). Moreover, SC transplantation also results in significant functional improvement in animal models of SCI (21,68). This has become evident with several recent reports of combined OEC and SC therapies, showing improved outcomes in animal models of SCI (14,57,67).
We have already established the safe procedure of OEC transplantation in SCI patients (23,24,26,30,45,66,77), whereas other researchers have used SCs for restoring neurological function in patients with SCI (64,78). However, there is not enough data to understand the best clinical potential of OECs, SCs, or their combined use for therapeutic strategies. The purpose of this clinical trial was to examine and compare the benefits of using OECs with or without SCs in chronic complete cervical SCI cases. The data obtained from some of these patients are reported in this investigation.
Materials and Methods
Initial Design of This Study
This was a prospective randomized, double-blind clinical trial with an initial enrollment of 28 patients. The study commenced on May 15, 2006, and the observation duration was 12 months. The trial was approved by the ethical committee of the Beijing Xishan Hospital (Beijing Rehabilitation Hospital of Capital Medical University). All participants and their family members (legal representatives) gave written informed consent for this study. The patients did not pay for their inclusion or treatment in this study.
Inclusion Criteria
The inclusion criteria were that patients must be at least 12 months postinjury, male or female, aged ≤60 years. They had cervical (C4–C7) SCI as measured by the American Spinal Injury Association (ASIA) Impairment Scale grade of A (complete, no motor or sensory function present in the sacral segment). They were assessed 6 months before surgery and were able to communicate effectively and gave informed consent.
Exclusion Criteria
The exclusion criteria included several factors: any functional recovery demonstrated during the 6-month period leading to surgery, use of any experimental drug, or participation in any experimental treatment within 1.5 years during the study, and any medical condition that may interfere with the ASIA and International Association of Neurorestoratology Spinal Cord Injury Functional Rating Scale (IANR-SCIFRS) assessments (1,35). Exclusion criteria also included clinically significant neurological, cardiac, respiratory, hepatic, or renal disease and/or malignancy, hemophilia, or other bleeding abnormality as defined by platelet level lower than 100 × 109/L; activated partial thromboplastin time or international normalized ratio higher than the upper limit of normal range; baseline hematocrit lower than 0.25; any cognitive impairments, which may preclude accurate neurological assessments [e.g., traumatic head injury with Glasgow Coma Score (GCS) of less than or equal to 14], ankylosing spondylitis, or diabetes mellitus requiring insulin therapy. Further exclusion criteria included known immunodeficiency, including acquired immune deficiency syndromes, or use of immunosuppressive or cancer chemotherapeutic drugs, pregnancy or breastfeeding during the study, and any condition or situation likely to cause the patient to be unable or unwilling to participate in study procedures or participate in all scheduled study assessments, including follow-up through the 12th month of the study. Patients were also excluded if they had any preexisting condition likely to result in the patient's death within the next 12 months, nontraumatic causes of SCI (e.g., transverse myelitis, acute disc herniation), any neuroimaging evidence of compression to the spinal cord (e.g., by fractured vertebrae or disc), any neuroimaging evidence of transecting injury to the spinal cord (e.g., by stab wound), any infections or pressure sores at the site of surgery, any condition or situation likely to cause the patient to be unable to undergo rehabilitation procedures, and any other condition that, in the opinion of the investigator, would preclude the patient's participation in the study.
Researchers Involved
Researchers included attending neurosurgeons (in charge of surgical procedure administration), neurologists (in charge of gathering relevant medical information and neurological function assessments to be conducted 1 week prior to surgery, then 1 week, 3 weeks, and 6 months after surgery), a local neurologist, neurosurgeon, or orthopedic surgeon (in charge of gathering relevant medical information and neurological function assessments at 6 months and 1 week prior to coming to this center for surgery), statisticians (in charge of study design and statistical analyses), and laboratory technicians (in charge of cell culture).
Procedure and Assessment (Fig. 1)
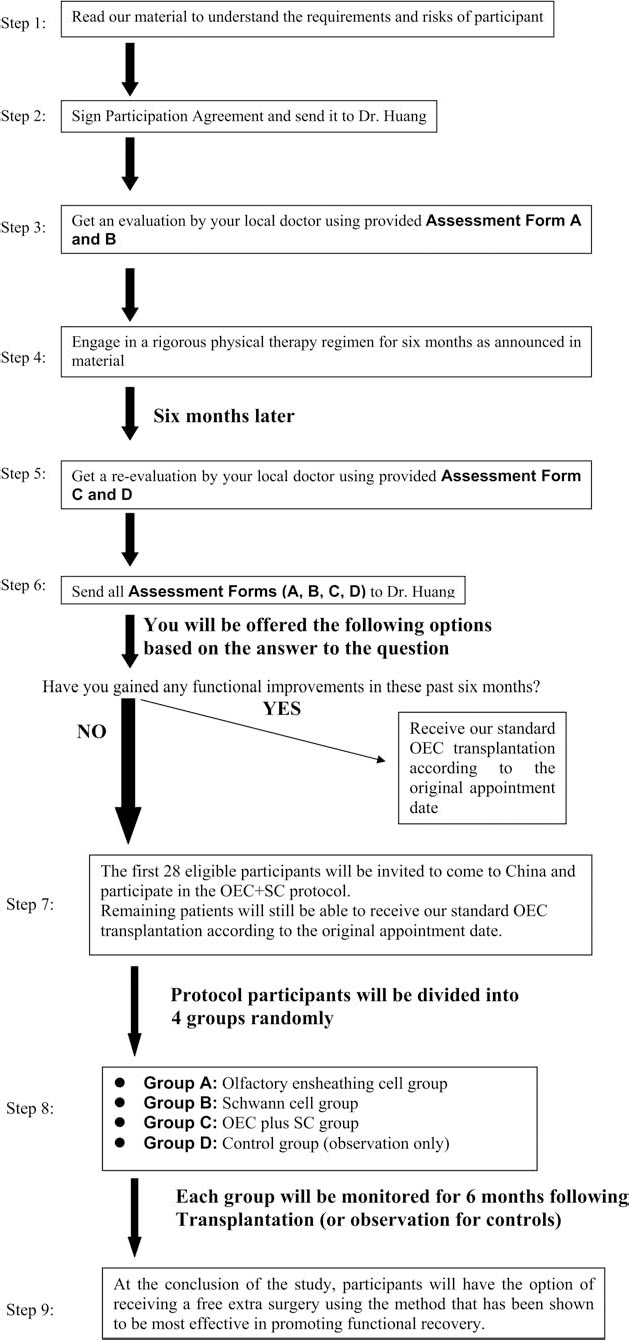
Flow diagram of procedure and assessment for this study.
Prospective study participants were evaluated 6 months before the scheduled surgery by undergoing a neurological assessment and magnetic resonance imaging (MRI) scan (1.5 Tesla, GE Medical Systems, Little Chalfont, UK). The local neurologist, neurosurgeon, orthopedic surgeon, or neurological rehabilitation therapist filled out two neurological assessment forms: (1) the ASIA scale (assessment form A) and (2) the IANR-SCIFRS (assessment form B).
The prospective participants then engaged in a rigorous physical therapy regimen for the next 6 months of training at least 6 h per day and 5 days per week.
The subsequent neurological assessments included (1) the ASIA scale (assessment form C) and (2) the IANR-SCIFRS (assessment form D). These were filled out approximately 1 week before coming to our center for the surgery, and all assessment forms, A, B, C, and D, described above were sent to this institute via e-mail or fax. If the physical therapy results showed any functional improvement during the observed 6-month period, the patients were excluded from the clinical trial.
Twenty-eight eligible participants were selected and randomly allocated into four groups of seven patients each. Eligible participants in three treated groups then came to our hospital to receive cell transplantation, whereas the control group just came for functional assessment.
All participants of the three experimental groups received treatment. Group A received transplantation of OECs only. Group B received transplantation of SCs only, and Group C received transplantation of OECs combined with SCs. Group D (control group) only got functional assessment.
Participants were given follow-up neurological assessments at 1 week pretransplantation, 3 weeks posttransplantation, and 6 months after the initial evaluation or surgery. The first two neurological assessments were performed during the participant's first trip to the center (1 week and 3 weeks), and the final postsurgery examination was performed during the participant's second trip to the center (6 months).
The neurological assessments were performed according to the ASIA and IANR-SCIFRS, electromyography (recruitment of maximal voluntary contraction; Keypoint-4, Skovlunde, Denmark), and evoked potential tests [para vertebrae somatosensory evoked potential (PVSEP), stimulation square wave, 3 Hz, 20–40 mA, 0.2 ms; Keypoint-4]. Participants in all three treated groups and the control group were instructed to undergo a rigorous physical therapy regimen for at least 6 months following the surgical transplantation in between the two visits.
The participants planned to stay in this hospital during their first trip for 30 days; the control group stayed at our center for 1 week. The control group (Group D) continuously engaged in a rigorous physical therapy regimen for the next 6 months of training for at least 6 h per day and 5 days per week. Another MRI was performed 6 months after the surgery (or observation–-control group).
Cell Culture
Tissue was obtained from fetuses that were aborted in accordance with guidelines issued by the Chinese Ministry of Health (91–006) (59).
OECs
Cells were isolated from aborted human fetal olfactory bulbs (unknown sex) with proper written informed consent from the donors. The OECs were cultured in Dulbecco's modified Eagle's medium/Ham's F12 (DMEM/F12; Hyclone, Logan, UT, USA) with 10% fetal bovine serum (FBS; Hyclone) and propagated for 14–21 days. They were characterized by immunostaining with antibodies (1:200) against the low-affinity nerve growth factor receptor, p75 (Sigma, St. Louis, MO, USA) and S100 (Sigma). The nuclei were stained with Hoechst 33342 (10 mg/ml; Sigma). Hoeschst-positive nuclei were counted as whole cells, and p75 or S100-positive cells were counted as OECs, from which was calculated the purification rate of OECs, which was about 94%. Cultured cells were detached with trypsin-ethylenediaminetetraacetic acid (EDTA) (Gibco, Grand Island, NY, USA) and resuspended in saline at 20,000 cells/μl (7,8). Cells taken from one fetus were used in one patient.
SCs
Cells were isolated from human fetal sciatic nerve (unknown sex) after the specimens were cut into slices, digested in trypsin (Invitrogen, Carlsbad, CA, USA), and made into a monoplast suspension. These cells were cultured in DMEM/F12 (Hyclone) with 15% FBS (Hyclone) and harvested in 7 days. Double-label immunofluorescence staining of S100 (1:200; Sigma) and Hoechst (Sigma) were performed. Hoechst-positive nuclei were counted as whole cells, and S100-positive cells were counted as SCs, from which was calculated the purification rate of SCs, which was around 95%. Cultured cells were detached with trypsin-EDTA and resuspended in saline at 20,000 cells/μl. The donors are the same as for OECs (73).
Cell Administration
The surgical procedures for intraspinal cord implantation were performed under general anesthesia. Small incisions in length of 2–3 cm with keyholes in the vertebral plate and a diameter of 1 cm were performed above and below the SCI site. Fifty microliters of saline containing about 1,000,000 cells [fetal OECs or fetal SCs or 50% OECs (500,000) and 50% SCs (500,000)] was injected into the spinal cord using a 4-gauge hypodermic syringe needle (Hanahao Medical Material Co., Ltd., Tianjin, China). One injection was performed in the dorsal mid-line of the spinal cord, above and below the lesion. The patients did not receive cyclosporine or any other immunosuppressive treatment.
Statistical Analysis
The data were expressed as either mean ± standard deviation (SD). Using SPSS 13.0 for Windows (IBM, Armonk, NY, USA), chi-square tests were performed for the comparisons of the entry state (age and gender) in controls and treated groups, though of course with n = 2 for the control group, any tests are meaningless. The improvement rates between the treated group and the controls were also compared by chi-square tests. Paired-sample t-tests were carried out for the comparisons of the scores prior to and after the cell therapy in the treated groups, and independent-sample t-tests were used for comparison of the scores between the treated group and controls. A value of p < 0.05 was considered statistically significant.
Results
Initially, 64 potential participants with cervical SCI were screened for this study according to the criteria, and 28 completed a 6-month rehabilitation program. However, only seven patients were able to be enrolled with short-term follow-up largely because of the limited resources. The patients were allocated into Groups A, B, C, and D randomly.
Given the very limited number of patients in each group, we compared only the data between the treated groups (i.e., groups A + B + C), and the control group (i.e., group D). The total number in the treatment groups (A + B + C) was five. Of them, patient numbers 1, 2, and 4 received the OEC intraspinal transplantation; patient No. 3 underwent the SC implantation, and patient No. 5 received the combination cell transplantation using OECs + SCs. There were two patients in the control group (D) (Table 1). At baseline, no significant differences were found based on sex (treated group vs. controls, p = 0.427), in the age of the patients (treated group vs. controls, p = 0.806), and initial scale scores at admission, that is, ASIA motor: 21.2 ± 2.8 versus 29.5 ± 21.9 (p = 0.372), light touch: 22.4 ± 7.9 versus 31.0 ± 21.2 (p = 0.425), pinprick: 16.6 ± 3.1 versus 26.5 ± 12.0 (p = 0.108), and the IANR score: 13.2 ± 4.7 versus 16.0 ± 5.7 (p = 0.523).
Demographic Characteristics of SCI Patients in This Clinical Trial

SCI, spinal cord injury; OECs, olfactory ensheathing cells; SCs, Schwann cells.
Among the five treated patients, only one patient in the OEC-alone group had a fever after surgery, but this was controlled following symptomatic treatment. There were no other side effects. The assessments of 1-week pretransplantation and 3-week posttransplantation were done within one admission, and the follow-up of 6 months was done in the next admission. There was no significant difference between the time point of 3 weeks and 6 months.
All patients (5/5) in the treated groups showed functional neurological improvements in one or more measurements (Table 2) and improvements on the electrophysiological test (except patient No. 4), while no changes were seen in the controls. The functional improvement rate of 5/5 (100%) was seen in the treated groups, whereas only 0/2 (0%) rating was seen in the control group [χ2(1) = 7.000, p = 0.008]. However, in comparing the improved scores between the treated group and controls, there were no significant differences: ASIA motor: 0.4 ± 0.5 versus 0.0 ± 0.0 (p = 0.374), light touch: 1.2 ± 1.3 versus 0.0 ± 0.0 (p = 0.273), pinprick: 2.6 ± 3.3 versus 0.0 ± 0.0 (p = 0.339), and the IANR score: 1.2 ± 1.1 versus 0.0 ± 0.0 (p = 0.203). Further comparison of the scores before and after cell surgery in the treated group were also not significant as follows: ASIA motor: 21.2 ± 2.8 → 21.6 ± 2.9 (p = 0.178), light touch: 22.4 ± 7.9 → 23.6 ± 8.4 (p = 0.109), pinprick: 16.6 ± 3.1 → 19.2 ± 3.8 (p = 0.152), and the IANR score: 13.2 ± 4.7 → 14.4 ± 5.5 (p = 0.070).
Neurological Functional Change Before and After Cell Transplantation Evaluated by the Same Independent Neurologist

Patient Nos. 6 and 7 are controls, and they were assessed at the admission and the discharge.
Case Reports
Patient 1
OECs were transplanted on April 24, 2007, and the patient was discharged on May 4, 2007. The functional improvements seen were in eating and drinking, grooming, transfers, and dressing. Electromyography (EMG) showed spontaneous activity and was increased in the part of the tested muscle. Recruitment of maximal voluntary contraction changed in the right extensor carpi radialis longus and left extensor digitorum communis muscles. Its pattern was improved and found to be more intense. PVSEPs showed the sensory level had moved down two segments on the left side (from T2 to T4).
Patient 2
OECs were transplanted on April 25, 2007, and the patient was discharged on May 14, 2007. The improvements included stronger muscles of his abdomen and recovery of sweating on trunk and lower extremities. EMG showed that the recruitment of maximal voluntary contraction changed in the right extensor carpi radialis longus muscle. Its pattern improved and became more intense compared with preoperative findings. There were a few low-amplitude motor unit potentials (MUPs) in the left extensor digitorum communis muscle after surgery, while no potential could be recorded before surgery.
Patient 3
SC transplant was performed on October 23, 2007, and the patient was discharged on November 15, 2007. There was an improvement in handwriting. The EMG showed that recruitment of maximal voluntary contraction was changed in both sides of the extensor carpi radialis longus muscles and the right biceps muscle. Its pattern was improved and more intense. Somatosensory evoked potentials (SEPs) were recorded from median nerves bilaterally after surgery, while no evoked potential could be recorded before surgery. PVSEPs showed that the sensory level moved down two segments bilaterally (from C7 to T2) after operation.
Patient 4
OEC transplant was performed on February 20, 2008, and the patient was discharged on March 12, 2008. The sweating pattern improved. The strength of elbow extensors increased from grade 2/5 to 3/5 on the right. Light touch was decreased at C5 on the right and disappeared below C8 bilaterally. Pinprick pain also disappeared at C5 and below C8 on the right, below C8 on the left.
Patient 5
OECs plus SCs were transplanted on May 21, 2008, and the patient was discharged on June 11, 2008. His arm movements became stronger. EMG showed recruitment of maximal voluntary contraction in the extensor carpi radialis longus muscles bilaterally. Its pattern improved and became more intense.
Discussion
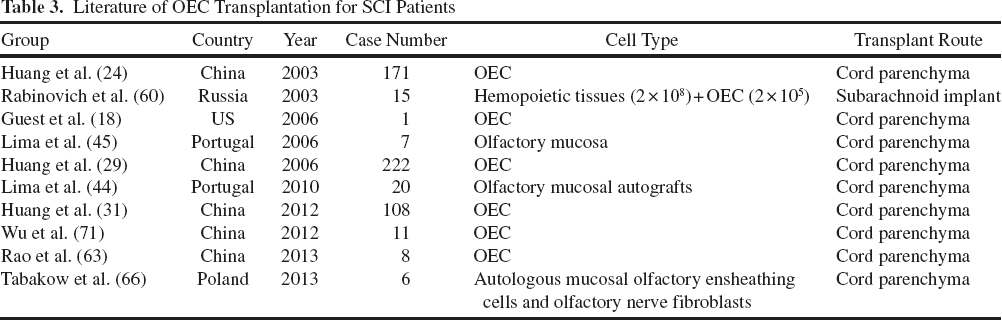
We initially observed the feasibility of cell transplants, including OECs, SCs, and OECs + SCs in SCI patients. Owing to the limited number of patients enrolled, we did not have comparative results among the OECs, SCs, and OECs + SCs groups. Since 2001, we and others have already established the clinical safety and efficacy of OEC transplant in SCI patients (Table 3). Further, application of SCs by Saberi and his coworkers demonstrated a 2-year follow-up of intraspinal transplantation without any problems (64). Feng's group also reported a 5-year observation in six patients after implanting autologous activated SCs (78).
Literature of OEC Transplantation for SCI Patients
Same Properties: OECs Versus SCs
Transplantation of OECs and SCs are considered as new promising options to promote axonal regrowth and remyelination after SCI in humans (70). OECs and SCs share a common developmental peripheral origin (11). In culture, SCs and OECs share a number of phenotypic properties, such as expression of p75 (39). There are only subtle immunological differences noted between SCs and OECs (4).
Individual Properties: OECs Versus SCs
OECs exhibit unique migratory, phagocytic, and myelinating properties. These features are not shared by SCs (15,69). Thus, OECs can migrate extensively in both gray and white matter and have the capability of moving into the delayed demyelinated lesion and remyelinated host axons (42).
N-cadherin, implicated in the adhesion of SCs to astrocytes, is differentially regulated in OECs and SCs (10).
Studies have also demonstrated that OECs integrate more extensively than SCs in astrocytic environments (41). In addition, bIII-tubulin mRNA and protein expression was high in OECs and low in SCs (55).
Do OECs Really have Advantages over SCs for CNS Repair?
Scar
The openings in the astroglial boundary at the lesion site may give OECs a potential advantage over SCs in promoting axonal growth through the astroglial scar. OECs injected 2.5 mm outside of a lesion could enter into the lesion in dorsal column crush (DCC) injury; however, SCs did not pass a region of increased astroglial immunoreactivity (2). In vitro, reactive astrocytes showed decreased chondroitin sulfate proteoglycans (CSPGs) in the presence of OECs (56). OECs could induce less of a host astrocyte response and CSPG expression than SCs following transplantation into adult central nervous system (CNS) white matter (41,61). Moreover, OECs and SCs exhibit a distinct interaction with meningeal cells (MCs), which are another major cellular component of the neural scar. OECs intermingle with MCs in cocultures, but SCs aggregate into well-defined cell clusters (14).
Remyelination
OEC and SC transplantation into dorsal columns (DCs) of adult rat spinal cords demyelinated by X-ray irradiation resulted in substantial SC-like patterns of remyelination to an equal degree (34).
Function Restoration
Although SCs and OECs behave differently in vivo in their interactions with astrocytes, as mentioned above, both cell types might produce similar improvements in motor function in contused rat spinal cords (43). In the acute phase of photochemically injured rats, both OEC- and SC-transplanted groups showed significant results in all the behavioral tests, though OEC-transplanted animals had higher motor-evoked potential (MEP) amplitudes and lower H-responses. They also had reduced astrocytic reactivity and proteoglycan expression than the SC group (16).
Regarding the speed and level of functional recovery, OECs implanted after 8 weeks into complete unilateral lesions of the rat corticospinal tract induced restoration of paw reaching function to 50% of normal, starting at 10 days after transplantation, whereas functional recovery in SC-treated rats was delayed until 30 days after transplantation and reached only 5–10% of the normal group (38).
Combination Transplantation: Possibility
Cao and her colleagues found that OECs can promote SC migration in the astrocytic environment by secreting nerve growth factor (NGF) (6). In chronic contusion injury in rats, transplanted OECs promote expansion of the cellular and axonal zones through stimulation of host SCs (76). Secreted protein acidic rich in cysteine (SPARC) was further identified as an OEC-derived matricellular protein that could indirectly enhance the ability of SCs to stimulate dorsal root ganglion outgrowth in vitro and enhance spinal cord repair (3). Thus, the transplanted OECs with the help of SCs might have more positive roles in the repair of SCI (38).
Summary
Taken together, our results suggest that the intraspinal transplantation of OECs, SCs, and OECs + SCs is feasible and well tolerated in patients. This combined treatment was beneficial for treated patients compared to the control group, as seen by functional assessment and electrophysiological tests. This suggests that our experimental design with combined treatment is a good strategy. However, due to the limitations of our study, the long-term observation of a large sample size is warranted in further clinical studies to examine, in detail, the benefits and risks of these cell-based interventional strategies in chronic complete SCI patients.
Footnotes
Acknowledgments
We thank all patients who took part in this trial. We thank Professor Hari Sharma from Uppsala University, Sweden, for his valuable comments and a critical review of this manuscript. We also thank our staff Xie Zixing, Jiang Chao, Liu Ruiwen, Jiang Zhao (neurosurgeons); Liu Qi (evaluator/neurologist); Wang Hongmei (cell center); Chen Tao, Wang Wei (data support); Zheng Lu and Wu Cui (assistants) for their excellent work. This work was supported by research funding (number: 06-A-01) of Beijing Hongtianji Neuroscience Academy. The authors declare no conflicts of interest.
