Abstract
Oligodendrocyte (OL) replacement can be a promising strategy for spinal cord injury (SCI) repair. However, the poor posttransplantation survival and inhibitory properties to axonal regeneration are two major challenges that limit their use as donor cells for repair of CNS injuries. Therefore, strategies aimed at enhancing the survival of grafted oligodendrocytes as well as reducing their inhibitory properties, such as the use of more permissive oligodendrocyte progenitor cells (OPCs), also called glial restricted precursor cells (GRPs), should be highly prioritized. Schwann cell (SC) transplantation is a promising translational strategy to promote axonal regeneration after CNS injuries, partly due to their expression and secretion of multiple growth-promoting factors. Whether grafted SCs have any effect on the biological properties of grafted GRPs remains unclear. Here we report that either SCs or SC-conditioned medium (SCM) promoted the survival, proliferation, and migration of GRPs in vitro. When GRPs and SCs were cografted into the normal or injured spinal cord, robust survival, proliferation, and migration of grafted GRPs were observed. Importantly, grafted GRPs differentiated into mature oligodendrocytes and formed new myelin on axons caudal to the injury. Finally, cografts of GRPs and SCs promoted recovery of function following SCI. We conclude that cotransplantation of GRPs and SCs, the only two kinds of myelin-forming cells in the nervous system, act complementarily and synergistically to promote greater anatomical and functional recovery after SCI than when either cell type is used alone.
Keywords
Introduction
Spinal cord injury (SCI) results in the destruction of descending motor and ascending sensory pathways leading to permanent damage with limited prospects for spontaneous repair (23,54,55). The failure of injured axons to regenerate results from a combination of factors including the lack of permissive growth substrates at lesion sites, the presence of inhibitory molecules that impair axonal outgrowth (17,45,58), the lack of neurotrophic support (7,24,30,49,52), and the death of oligodendrocytes that induces demyelination of surviving axons (13,29,48,50). Since cavity formation is evident following SCI, cell transplantation into the lesion site is essential to provide permissive substrates for axonal extension through and beyond it. Different cell types, including neural stem cells (NSCs) (41), Schwann cells (SCs) (36,51), olfactory ensheathing glial cells (OEGs) (16,31), and marrow stroma cells (MSCs) (14,39) have been used as donor cells for transplantation after SCI with variable degrees of success.
Theoretically, oligodendrocytes (OLs) should be a strong candidate for cell-based therapy because they are central nervous system (CNS)-derived myelin-forming cells. These cells are CNS environmentally compatible and form multiple myelin segments. A single oligodendrocyte can myelinate up to 50 axon segments and thus is highly efficient (5). However, ample evidence suggests that mature OLs and their myelin are nonpermissive substrates for neurite outgrowth in vitro (44) and axonal regeneration in vivo (43). We reason that, although mature OLs are inhibitory to neurite outgrowth, their progenitors glial restricted precursor cells (GRPs), also called oligodendrocyte progenitor cells (OPCs), may not be. Our previous study showed that GRPs or OPCs are significantly more permissive than mature OLs (32), indicating that GRP transplantation could be an attractive strategy not only for remyelination but also for regeneration of damaged axons following SCI. However, the poor survival of grafted GRPs remains a major challenge.
In contrast to GRPs, SCs, a type of peripheral nervous system (PNS)-derived myelin-forming cells, are strong promoters of axonal regeneration. Mounting evidence suggests that SCs play a central role in the repair of both CNS and PNS injuries due to their abundant production of growth-promoting factors and cell adhesion molecules (11,36,55). Our previous study showed that transplanted SCs successfully bridged the injured spinal cord and promoted axonal regeneration across the lesion gap following SCI (51,53,56). However, SCs do not normally reside in the CNS and form only one myelin segment on a single axon (5), making them less efficient in myelin formation than their CNS counterparts, the OLs.
To date, both GRPs (9,22,25) and SCs (38,51,53,56) have been used individually as donor cells for transplantation after SCI with variable degrees of success. However, cotransplantation of the two cell types has not been examined. In the present study, we examined whether SCs have any effect on the biological properties of GRPs including their survival, proliferation, migration, differentiation, and myelination in vitro. We further investigated whether cotransplanted SCs promote better survival, migration, and myelination of grafted GRPs and, collectively, better recovery of function in a clinically relevant model of contusive SCI.
Materials and Methods
Animals
All animal care and surgical interventions were undertaken in strict accordance with the Public Health Service policies on humane care and the use of laboratory animals and with the approval of the Indiana University Institutional Animal Care and Use Committee and Institutional Biosafety Committee.
Isolation and Culture of Embryonic GRPs
GRPs were immunopanned on embryonic day (E) 14 from wild (for in vitro experiments) or green fluorescent protein (GFP)-transgenic (for cell transplantation) Sprague–Dawley (SD) (Harlan, Indianapolis, IN, USA) rat spinal cords using an anti-A2B5 IgM mAb (1:1, a kind gift from Dr. Scott R. Whittemore, University of Louisville, KY, USA) and a protocol modified from previous studies (9,32,33). Immunopanned GRPs were plated onto fibronectin (BD Biosciences, Bedford, MA, USA)-coated 10 × 12-cm culture dishes (Becton Dickinson, Franklin Lakes, NJ, USA), and the growth medium was added and changed every other day. The GRP growth medium (GRPM) contained Dulbecco's modified Eagle's medium (DMEM)/Ham's F12 (DMEM/F12; Invitrogen, Carlsbad, CA, USA), 1 × N2 and 1 × B27 supplements (Invitrogen), fibroblast growth factor 2 (FGF-2) (20 ng/ ml; Chemicon, Temecula, CA, USA), and platelet-derived growth factor-AA (PDGF-AA; Chemicon) (10 ng/ml). After 5–7 days, the cells were passaged and the purity of GRPs was examined. The GRP preparations were highly pure with >95% expressing A2B5. In all experiments, only passage 2 (P2) cells were used.
Isolation and Culture of SCs
The methods for SC isolation, purification, and amplification were developed by Morrissey et al. (34) and described in our previous publications (52,53). Briefly, sciatic nerves were obtained from adult rats anesthetized with by an intraperitoneal (IP) injection of ketamine (87.7 mg/kg; Ben Venue Labs, Bedord, OH, USA) and xylazine (12.3 mg/kg; Butler, Columbus, OH, USA) under aseptic conditions. After epi-neurium and connective tissue were removed, the nerves were cut into 1-mm2 explants. The explants were placed in 35-mm Corning tissue culture dishes (Baxter, Stone Mountain, GA, USA) with low levels of DMEM supplemented with 10% fetal bovine serum (FBS; Invitrogen). When the outgrowth of migratory cells (predominantly fibroblasts) reached a near-confluent monolayer around the explants (7 days), the explants were transferred to new culture dishes with fresh medium. After four to six such passages (4–6 weeks), the cells that emerged from the explants were primarily SCs. The explants were then transferred to a 35-mm dish containing 1.25 U/ml dispase (Boehringer Mannheim Biochemicals, Indianapolis, IN, USA), 0.05% collagenase (Worthington Biochemicals Corp., Freehold, NJ, USA), and 15% FBS in DMEM for incubation overnight at 37°C in 5% CO2. On the following day, the explants were dissociated and the cells were plated onto poly-l-lysine (100 μg/ml, Sigma, St. Louis, MO, USA)-coated 100-mm dishes (Becton Dickinson) in DMEM/10% FBS. Later, the cultures were refed with the same medium supplemented with 20 μg/ml pituitary extract (BTI, Stoughton, MA, USA) and 2 μM forskolin (Sigma) for dividing. When the SCs reached confluence (≍ 4–5 × 106 cells/dish), they were rinsed in Ca2+- and Mg2+-free Hank's balanced salt solution (CMF-HBSS; Gibco, Gaithersburg, MD, USA) and briefly treated with 0.05% trypsin (Gibco) and 0.02% ethylenediaminetetraacetic acid (EDTA; Gibco) in CMF-HBSS. Cells were washed twice in DMEM/10% FBS and passed into new dishes at a density of 2 × 106 cells/100-mm dish. The purity of the SCs was quantified according to the methods described previously (54). All SCs used in this study were P2 cells with a purity of >98%.
Preparation of SC-Conditioned Medium (SCM)
To prepare SCM, purified P2 SCs were grown on a poly-l-lysine-coated 75-mm2 flask (Corning/Costa, Cambridge, MA, USA) in DMEM/10% FBS for 1–2 days. When SCs obtained good growth state and reached 80% confluence, the cultures were washed with DMEM/F12 medium and then were cultured in 12 ml DMEM/F12 with 1 × N2 and 1 × B27 supplements. After 3 days, SCM was removed from the cultures and immediately passed through a membrane filter (0.2 μm pore diameter, Millipore, Billerica, MA, USA) to remove cells and debris. The SCM was stored at −80°C and diluted to 20% or 50% with DMEM/F12 before use.
GRP Survival Assay
To determine the effect of SCM on the survival/death of GRPs, the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) was performed using the In Situ Cell Death Detection Kit (TMR red; Roche, Mannheim, Germany) according to the manufacturer's manual. Briefly, GRPs were plated onto fibronectin-coated coverslips and cultured in different mediums including control (DMEM/F12 with 1 × N2 and 1 × B27 supplements), GRPM, 20% SCM, and 50% SCM. After 3 days, cells were incubated with mouse anti-A2B5 IgM mAb (1:1, a kind gift from Dr. Scott R. Whittemore, University of Louisville) for 40 min at 4°C to label live GRPs. Then cells were rinsed and fixed with 4% paraformaldehyde (PFA, Sigma) in PBS (pH 7.4; Invitrogen) for 15 min at room temperature and rinsed twice in PBS. After blocking with 10% donkey serum (Jackson Immunoresearch Lab, West Grove, PA, USA)-0.3% Triton X-100 (Sigma)/PBS, cells were then incubated with fluorescein isothiocyanate (FITC)-conjugated donkey anti-mouse IgM (1:200; Jackson ImmunoResearch Lab). For TUNEL, cells were then permeabilized with 0.1% Triton X-100 in freshly prepared 0.1% sodium citrate (Sigma) for 2 min on ice. After being rinsed twice in PBS, the coverslips were incubated with TUNEL reaction mixture for 1 h at 37°C. The coverslips were rinsed with PBS and mounted with Gel/Mount aqueous mounting media (Biomeda, Foster City, CA, USA) containing Hoechst 33342 (Sigma). The TUNEL-positive (red) cells in GRPs (green) were counted under the BX60 Olympus fluorescence microscope (Center Valley, PA, USA) and the percentage of TUNEL+A2B5+ cells out of total A2B5+ cells was calculated. At least five randomly selected fields in each coverslip with a total of more than 500 A2B5+ cells were counted for each culture. Each experiment included four cultures and the experiments were independently repeated for five times for final quantification and statistical analysis.
BrdU Incorporation Assay
To assess the effect of SCM/SCs on the proliferation of the cultured GRPs, GRPs or the mixture of GRPs and SCs (1:1) were plated onto fibronectin-coated coverslips and cultured in different media including control (DMEM with 1 × N2 and 1 × B27 supplements), GRPM, 20% SCM, and 50% SCM. After incubation with BrdU (5-bromo-2′-deoxyuridine, 10 μM; Sigma) for 16 h, cells were incubated with mouse anti-A2B5 IgM mAb for 40 min at 4°C to label live GRPs. Then cells were rinsed and fixed with 4% PFA in PBS (pH 7.4) for 15 min at room temperature and then rinsed twice in PBS. For BrdU and A2B5 immunofluorescence double labeling, a BrdU incorporation assay was used. Briefly, the fixed cells were treated with 1 N HCl for 40 min at 37°C to denature the DNA prior to the use of primary rabbit anti-BrdU antibody (1:100; Sigma) and mouse anti-A2B5 IgM mAb (1:1) overnight at 4°C and then secondary antibody (rhodamine-conjugated donkey anti-rabbit IgG; 1:200; Sigma; FITC-conjugated donkey anti-mouse IgM; 1:200; Jackson ImmunoResearch Lab) at room temperature for 2 h. The cells were examined and photographed using an Olympus BX60 microscope and the percentage of Brdu+A2B5+ cells out of the total A2B5+ cells was calculated. At least five randomly selected fields in each coverslip with a total of more than 500 A2B5+ cells were counted for each culture. Each experiment included four cultures, and the experiments were independently repeated for five times for final quantification and statistic assay.
To assess GRP proliferation after transplantation in normal spinal cord, 3 days before 1 and 4 weeks posttransplantation time point termination, corresponding randomly selected rats (n = 5/group) received 10 IP injections of BrdU (50 mg/kg/injection; three injections per day for 3 days and one injection at the last day) and were killed 2 h after the last injection. The animals were perfused, spinal cords were collected, and tissue sections were prepared as described in the following. One set of serial sections were randomly selected for BrdU incorporation assay. Similarly, the fixed tissue sections were treated with 1 N HCl for 40 min at 37°C to denature the DNA prior to the use of primary rabbit anti-BrdU antibody (1:100; Sigma) 4°C overnight and then secondary antibody (rhodamineconjugated donkey anti-rabbit IgG; 1:200; Sigma) at room temperature for 2 h in the second day. The BrdU+ cells were examined and photographed using an Olympus BX60 microscope, and the percentage of BrdU+ GFP+ cells in GFP+ cells was calculated. At least five randomly selected fields with a total of more than 800 A2B5+ cells were counted in each section. The results of all sections from the selected set were averaged.
Inverted Coverslip Migration Assay
The inverted migration assay was based on a method described previously (18). Briefly, GRPs were plated onto the coverslip fragments (about 2 mm2) precoated with fibronectin. After the cells were allowed to attach for 16–18 h, the coverslips were washed with culture medium three times to remove any loose cells and then cells were pretreated with aphidicolin (12 mg/ml; Sigma) for 30 min. To determine the effect of SCM on GRP migration, the coverslip fragments were inverted with cells facing downward onto 12-well plates coated with fibronectin and cells were cultured in different media. To assess the effect of SCs on GRP migration directly, the coverslip fragments were inverted with GRPs facing downward onto a monolayer of SCs or NIH3T3 fibroblast cell line (control; ATCC, Manassas, VA, USA) in 12-well plates. Cells were cultured for 3 days to allow cell migration and then fixed. Cells were immunostained with mouse anti-A2B5 IgM mAb as mentioned above, and nuclei were labeled with Hoechst 33342. The distance of A2B5-positive GRPs migrated away from the edge of the fragment was analyzed. The maximum distance of the leading cells was measured in each experiment, and the number of migrated cells occupying successive 0.1 × 0.1-mm2 areas from the edge of the coverslip was counted. All experiments were carried out four times independently (n = 4) in the presence of aphidicolin, a mitotic inhibitor, in order to be certain that movement away from the coverslip was due to migration alone with no contribution from proliferation.
GRP + SC Transplantation Into the Normal Spinal Cord
To examine whether grafted SCs had an effect on the survival, proliferation, and migration of GRPs in vivo, we cografted GRPs and SCs into normal, unlesioned spinal cords (GRP + SC cografted group, n = 25); GRP-grafted group was set as the control (n = 25). To trace the fate of transplanted GRPs, GRPs were isolated from GFP-expressing transgenic rats (GFP-GRPs). To trace the fate of grafted SCs, SCs were incubated in 10 μM BrdU overnight prior to transplantation. GRPs (1 × 105/μl) or a mixture of GRPs and SCs (1 × 105 each/μl) were collected by centrifugation and resuspended in PDGF- and FGF-2-containing GRP medium. For cell transplantation, adult female SD rats (160–180 g in body weight) were anesthetized using ketamine (87.7 mg/kg; Ben Venue Labs, Bedford, OH, USA) and xylazine (12.3 mg/kg; Butler, Columbus, OH, USA) to receive a dorsal laminectomy at the T10 level. Two injections of cell suspension (1 μl/ injection/site) were made bilaterally into the normal spinal cord at a depth of 1.2 and 0.6 mm laterally from the midline through a glass micropipette (World Precision Instruments, Sarasota, FL, USA) with an outer diameter of 50–70 μl and the tip sharp-beveled to 30°–50° at a rate of 0.5 μl/min, as described previously (9). The animals were perfused at different time points after transplantation (1 day, 3 days, 1 week, 2 weeks, and 4 weeks; n = 5 at each time point), and the spinal cords were collected for further analysis.
GRP + SC Transplantation Into the Contused Spinal Cord
Contusive SCI was induced using a weight-drop device developed at New York University (NYU) (20). Briefly, after anesthesia, adult female SD rats (160–180 g in body weight) received a dorsal laminectomy at the T10 level, and the dorsal surface of the spinal cord was subjected to a weight-drop impact, using a 10-g weight (2.5 mm diameter) dropped at a height of 12.5 mm (28). At the 9th day post-SCI, cell transplantation was performed. To initiate and enhance OL lineage differentiation, GFP-GRPs were incubated with triiodothyronine (T3, 50 ng/ml; Sigma) for 3 h prior to transplantation. Cells were collected and resuspended according to the methods described above. A total of 40 rats were randomly assigned into five groups that received injections of (1) control (GRP medium), (2) GFP-GRPs (2 × 105) + GRPs (2 × 105; isolated from wild-type SD rats), (3) SCs (4 × 105), and (4) GFP-GRPs (2 × 105) + SCs (2 × 105). For cell transplantation, animals were reanesthetized and the laminectomy site was reexposed. A total of five injections were made into the spinal cord: one at the lesion epicenter (2 μl of cell suspension at a concentration of 105 cells/μl), two at 2 mm rostral to the epicenter bilaterally, and two at 2 mm caudal to the epicenter bilaterally at a depth of 1.2 and 0.6 mm laterally from the midline (0.5 μl/injection/side). Thus, a total of 4 × 105 cells were grafted into each injured spinal cord. The animals received a daily subcutaneous injection of cyclosporine A (10 mg/ kg; Sigma) beginning at 3 days prior to the transplantation and continued until the 7th week after transplantation when the rats were scarified. During this period, behavior tests including Basso, Beattie, Bresnahan (BBB) locomotor rating scale, grid walking and foot printing analysis were performed at indicated time points.
Tissue Preparation and Immunohistochemistry
At the indicated time points postinjury or posttransplantation, rats were anesthetized with 60 mg/kg pentobarbital and perfused transcardially with 4% PFA in 0.01 M PBS, pH 7.4. The spinal cord segments containing the grafts were removed, postfixed in the same perfusion fixative for overnight at 4°C, and cryoprotected in 30% sucrose buffer (Sigma) for another 5–7 days. A 2-cm length of the spinal cord centered at the injection site or the injury epicenter was dissected out. Then the cords were embedded in tissue-embedding medium (HistoPrep; Fisher Scientific, Pittsburgh, PA, USA) on dry ice. For uninjured and cellgrafted spinal cords, cords were cut longitudinally (5 sets) at 20 μm, and for injured and cell-grafted spinal cords, half of the cords were sectioned transversely (25 sets) and half of the cords were cut longitudinally (5 sets) at 20 μm on a cryostat and mounted onto gelatin-subbed slides (Fisher Scientific, Fairlawn, NJ, USA) and stored at −70°C.
For immunohistochemistry staining, the frozen slides were air dried at room temperature for 10 min and washed with PBS for 10 min. Then they were blocked with 10% donkey serum in Tris-buffered saline (TBS) containing 0.3% Triton X-100 (TBST) for 1 h at room temperature (RT), and the primary antibody was applied in the same blocking solution overnight at 4°C. The following primary antibodies were used: polyclonal chicken anti-GFP (1:500; Chemicon), mouse anti-adenomatous polyposis coli (CC1, 1:100; Calbiochem, La Jolla, CA, USA) for oligodendrocytes, mouse anti-glial fibrillary acidic protein (GFAP, 1:200; Sigma) for astrocytes, and mouse anti-phosphorylated heavy neurofilament (SMI31; 1:1,000; Chemicon) for axons, mouse anti-active caspase 3 antibody (1:1,000; Sigma) for cell apoptosis detection, and mouse anti-myelin basic protein (MBP) antibody (1:200; Chemicon) for myelin detection. The slides were washed in PBS three times and incubated with FITC or rhodamineconjugated goat anti-mouse IgM or IgG (Jackson Immu no-Research Lab; all 1:200) for 1 h at 37°C. The slides were washed three times with PBS and coverslipped with Gel/Mount aqueous mounting media containing Hoechst 33342 to counterstain the nuclei. The images were taken with an Olympus BX60 microscope or Zeiss 510 laser confocal microscope (Thornwood, NY, USA). In all experiments, primary antibody omission controls were used to confirm the specificity of the immunofluorescence labeling.
Cell Counting on Tissue Sections
For cell survival assay, we quantified all the GFP+ cells within 2-cm length of the spinal cord centered at the injection site in all sagittal sections in GFP-GRP or GFP-GRP + SC groups grafted into the normal spinal cord. One of the 25 sets of transverse sections spaced at 500-μm intervals in groups GRP (2 × 105 GRP + 2 × 105 GFP-GFP) or GFP-GRP + SC (2 × 105 GFP-GRP + 2 × 105 SC) were grafted into contused spinal cord was selected, immuno-labeled with appropriate cell markers as CC1 and GFAP, and the CC1+ GFP+ cells, GFAP+ GFP+ cells, and GFP+ cells were counted respectively using Image J software (NIH, Bethesda, MD, USA) to calculate the differentiation fate of grafted cells in different areas of the injured spinal cord.
In Vivo Cell Migration Analysis
To analyze the migration ability of grafted cells, we checked all the longitude sections of all animals with cell transplantation under Olympus BX60 microscope. We measured the most distal distance (mm) of the GFP+ cells from the injection site in each uninjured animal (n = 5) and then calculated the averaged maximum migration distance of each group. In the injured group, we measured the longest distal distance of the GFP+ cells from the injection sites that were 2 mm caudal or rostral to the epicenter and then calculated the average maximum migration distance of each group (n = 5).
Serotonergic Fiber Counts
5-Hydroxytryptamine (5-HT; serotonin) fiber counts were modified according to a previously published method (15). Briefly, three transverse sections, at about 6 mm caudal to the lesion epicenter, were randomly selected and were immunolabeled with anti-5-HT antibodies (1:5,000; DiaSorin, Stillwater, MN, USA). All 5-HT immunopositive fibers within the ventral gray matter, at a 20× view field (27,000 μm2), were counted and averaged for each animal. Statistical analysis, discussed below, was performed to compare the 5-HT fiber counts among different groups.
Histological Analyses
Seven weeks after SCI, animals were sacrificed and two sets of transverse sections, spaced 500 μm apart, were stained with Luxol fast blue (LFB; Sigma) for white matter sparing analysis (n = 5/group) and stained with cresyl violet-eosin (CVE; Sigma) for lesion volume assessments (n = 5/group), respectively, as described previously (10). Briefly, white matter sparing was defined as tissue showing normal myelin appearance and density, the lesion epicenter was defined as the section containing the least amount of spared white matter. The areas of whole transverse section and the myelinated white matter at the lesion epicenter were outlined and quantified following LFB staining, and the percentage of the area of white matter sparing to the area of the whole section was calculated. After CVE staining, the total and cross-sectional area of the spinal cord and the lesion boundary were measured using a Neurolucida System (MicroBrightField Inc., Colchester, VT, USA) attached to an Olympus BX60 microscope. The total volume of the lesion area (which included areas of cavitation and degeneration) was calculated by summing their individual subvolumes. Individual subvolumes of the lesion area were calculated by multiplying the cross-sectional area (A) × D, where D represents the distance between sections (500 μm). The percentage total volume of the injured area was calculated by dividing the total volume of the lesion area by the total volume of corresponding length of the spinal cord.
Behavioral Assessments
The BBB open-field locomotor rating scale (4) was used to assess hindlimb locomotor recovery including joint movements, stepping ability, coordination, and trunk stability. The tests were performed by two examiners who are blinded to theanimals' treatments at 2 days preinjury, 1 day post injury, 7 days postinjury, and 1, 2, 3, 4, 5, 6 weeks after cell transplantation for all 40 rats assigned to four groups (control, GFP-GRP + GRP, SC, and GFP-GRP + SC). Animals with a minimum BBB score of 12 were further analyzed and included in the footprint and grid-walk analyses at 6 weeks after cell transplantation.
For footprint analyses, the hindlimbs of the rats were dipped in blue dyes (nontoxic), and the animals walked on a paper-covered narrow runway (100 cm length and 7 cm width). To perform the measurements, the first and last 15 cm of the prints were excluded to avoid analyzing the beginning and end of the limb movements. The angle of rotation (AOR) was assessed by measuring the angle between two lines connecting the third toe and the stride line at the center of the paw pad. The base of support was assessed by measuring the distance between the central pads on the hind paws, respectively. The trail was repeated three times, and the results were averaged for each rat.
Grid-walk analysis was performed to assess the deficits in the descending motor functions (6) in a manner complimentary to the footprint. The rats were allowed to traverse over a wire mesh grid (2.5 × 2.5-cm2 grid spaces; The Home Depot, Indianapolis, IN, USA). Animals must walk for at least 30 s on this device up to 3-min test period during which the total number of hindlimb movement and the number of footfalls of the hindlimb were counted, respectively, by two examiners blind to the experiment. One footfall was defined when the hindlimb paw protruded entirely through the grid with all toes and heel extended below the wire surface. Data are expressed as the percentage of footfalls over the total number of hindlimb movement. For each session, the average number of paw falls of each rat was taken from three trials.
Statistical Analysis
Data were presented as mean±standard deviation (SD). One-way ANOVA with Tukey's honestly significant different (HSD) or Fisher's least significant different (LSD) post hoc tests were used to determine statistical significance. A value of p < 0.05 was considered statistically significant. Data were analyzed with appropriate software (SPSS 14.0; SPSS, Chicago, IL, USA).
Results
SCM and SCs Reduced Apoptosis, Promoted Proliferation, and Enhanced Migration of GRPs In Vitro
To determine whether SCs affect the growth property of GRPs, we examined the effect of SCM on survival of GRPs in vitro (Fig. 1). Our result showed that in the presence of SCM (Fig. 1C, D), GRP cell death, identified by TUNEL, was significantly reduced as compared to the control medium (DMEM/F12 with 1 × N2 and 1 × B27 supplements) (Fig. 1A). Only 10.31±1.81% (n = 5) and 5.32±1.01% (n = 5) TUNEL+ cells were detected when cultured in 20% and 50% SCM, respectively (Fig. 1E). Next, we examined whether SCs affect proliferation of GRPs using a BrdU incorporation assay. We counted the number of BrdU+ GRPs cultured in the control medium (Fig. 2A), GRPM (Fig. 2B), 20% SCM (Fig. 2C), 50% SCM (Fig. 2D), and cultured directly with SCs (Fig. 2E). We found that the percentage of BrdU+ GRPs was significantly increased in A2B5+ cells cultured in SCM of both 20% (79.18±5.98%, p < 0.001, n = 5) (Fig. 2C, F) and 50% (89.02±4.76%, p < 0.001, n = 5) (Fig. 2D, F) or cultured directly with SCs (88.20±3.29%, p < 0.001, n = 5) (Fig. 2E, F) as compared to the control group (15.24±5.76%, n = 5) (Fig. 2A, F). The percentage of BrdU incorporation in GRPs cultured in 50% SCM or cultured with SCs was not statistically significant (p = 0.46, n = 5) compared with those cultured in GRPM containing PDGF-AA/FGF-2 (85.49±4.01%, n = 5) (Fig. 2F). These results strongly suggest that SCs and their secreted growth factors are strong promoters of GRP proliferation. Finally, we examined the effect of SCM or SCs on migration of GRPs in vitro using an inverted coverslip migration assay. As shown in Figure 3, SCM at both 20% (Fig. 3B) and 50% (Fig. 3C) and GRPM (Fig. 3D) all significantly promoted GRP migration, indicated by the maximum migration distance of GRPs (Fig. 3G) and the total number of GRPs that migrated away from their original sites (Fig. 3H) as compared to the control (Fig. 3A, G). The SCM at 50% promoted an equal GRP migration to GRPM (p = 0.27, n = 5). When GRP migration was examined on SCs or 3T3 cell monolayer, significantly longer distances of GRP migration were found when placed on the SC monolayer (417.32±22.41 μm, n = 5) (Fig. 3F) than on the 3T3 cell monolayer (16.35±5.40 μm, p < 0.001, n = 5) (Fig. 3E), indicating that induction of GRP migration is cell specific. Figure3G and H show the average maximum migration distances of GRPs and the number of migrated GRPs under different culture conditions, respectively. It is clear that SCs provide the most permissive substrate for the migration of GRPs as compared with all other experimental conditions.

SCM reduces apoptosis of cultured GRPs in vitro. (A–D) Representative photomicrographs show patterns of terminal deoxynucleotidyl transferase-mediated dUTP nickend labeling (TUNEL) (red) counterstained with A2B5 staining (green) in glial restricted progenitors (GRPs) cultured in (A) control [Dulbecco's modified Eagle's medium (DMEM)/F12 with 1 × N2 and 1 × B27 supplements], (B) GRP proliferation medium (GRPM) containing growth factors platelet-derived growth factor (PDGF)-AA and fibroblast growth factor (FGF)-2, (C) 20% Schwann cell-conditioned media (SCM), and (D) 50% SCM. (E) Quantification of TUNEL+ cells shows that SCM at both concentrations significantly reduced apoptosis of GRPs. Their effects are similar to GRPM. Data shown are mean±SD (n = 5). **p < 0.01, ***p < 0.001. Scale bar: 50 μm.

SCM and SCs promote proliferation of cultured GRPs in vitro. (A–E) GRPs were cultured for 3 days in (A) control, (B) GRPM, (C) 20% SCM, (D) 50% SCM, or (E) with SCs. Cells were incubated with bromodeoxyuridine (BrdU; 10 μM) for 16 h before cell fixation. Immunofluorescent double labeling of BrdU (red) and A2B5 (green) was performed, and all nuclei were stained with Hoechst 33342 (blue). (F) Quantitative measures demonstrate that treatments of GRPs with SCM, SCs, as well as GRPM, resulted in a significant increase of BrdU+ cells in A2B5+ GRPs. Data presented are mean±SD (n = 5). ***p < 0.001, compared to the control. Scale bar: 50 μm.
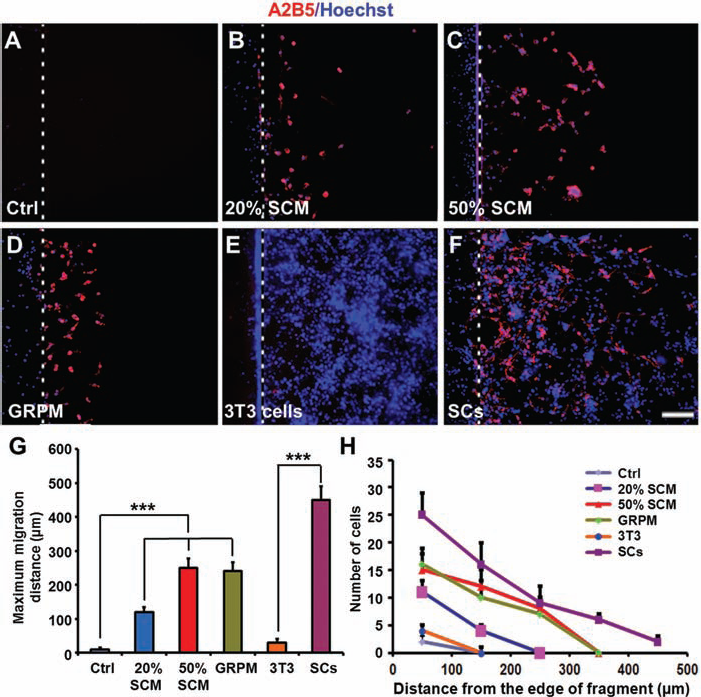
SCM and SCs promote migration of cultured GRPs in an inverted coverslip migration assay. (A–F) GRPs were plated on coverslip fragments and cultured for 1 day, and then the fragments were inverted facing downward onto poly-l-lysine (PLL) (A–D), monolayer of NIH3T3 fibroblast cell line (E), or SCs (F) in 12-well plates. Culture media used were DMEM/F12 with 1 × N2 and 1 × B27 supplements (A, E, and F), 20% SCM (B), 50% SCM (C), and GRPM (D). Cells were cultured for another 3 days and then were immunostained with A2B5 antibody (red). All cell nuclei were labeled with Hoechst 33343 (blue). (G) Average maximum distance of GRPs traveled from the edge of the inverted fragment in different culture conditions (vs. control or 3T3). (H) Plot of the number of migrated cells occupying successive 0.1 × 0.1-mm2 areas from the edge of an inverted fragment of different culture conditions. Data shown are mean±SD (n = 5). ***p < 0.001. Scale bar: 100 μm.
SCs Promoted Survival, Proliferation, and Migration of Cografted GRPs in the Normal Adult Spinal Cord
Since we demonstrated that both SCM and SCs had strong effects on GRP survival (reduction of apoptosis), proliferation, and migration in vitro, our next step was to examine whether SCs had similar effects on GRPs when they were cografted into the adult normal spinal cord. We examined the effect of SCs on biological properties of GRPs by comparing rats receiving GRPs alone (n = 25) with those receiving SCs and GRPs (n = 25) at 1 day, 3 days, 1 week, 2 weeks, and 4 weeks posttransplantation. Our results showed that cografting of SCs and GRPs significantly enhanced the number of grafted GFP+ GRPs in the spinal cord as compared to the GRP single-grafted group at all time points except at 1 day after cell transplantation (Fig. 4A, B). Moreover, the maximum migratory distance of the grafted GRPs was significantly increased in the SC and GRP cografted group as compared to the GRP singlegrafted group at all five time points (Fig. 4A, C). Since the increased numbers of grafted GRPs could be a result of proliferation or increased survival, we performed BrdU incorporation (Fig. 5A–M) and activated caspase 3 staining (Fig. 5N–R) to determine the proliferation and death/survival of grafted GRPs. More BrdU+ GFP+ cells in GFP+ cells (25.37±3.45% vs. 5.20±1.345 at 1 week, p < 0.001, n = 5; 6.83±1.72% vs. 2.70±0.90% at 4 weeks, p < 0.05, n = 5) (Fig. 5M) and less caspase 3+ GFP+ cells in GFP+ cells (7.63±2.00% vs. 31.15±3.13% at 1 week, p < 0.001, n = 5; 8.13±2.15% vs. 14.52±2.87% at 4 weeks, p < 0.05, n = 5) (Fig. 5R) were found in the SC and GRP cografted group as compared to the GRP single-grafted group. These data not only confirmed the in vitro results but also provided first evidence that cografted SCs have a remarkable effect on the survival, proliferation, and migration of grafted GRPs in the normal adult spinal cord.

Survival and migration of grafted GFP+ GRPs in the normal adult spinal cord. (A) Representative longitudinal sections show the survival and migration of grafted green fluorescent protein positive (GFP+) GRPs in the normal spinal cord at different time points posttransplantation. Note that substantial migration of GFP+ GRPs occurred only in the group that received GRP + SC cograft as compared to the lack of GFP+ GRP migration in the GRP alone graft. (B) Quantification of the number of GFP+ GRPs in the normal spinal cord at different time points between GRP single and GRP + SC cografts. The number of GFP+ GRPs was determined using Image J software to count all GFP+ GRPs in a series of 20-μm-thick sections. (C) Comparison on average maximum migration distances of grafted GFP+ GRPs in the normal spinal cord between GRP single and GRP + SC cografted groups. Data shown are mean±SD (n = 5). *p < 0.05, ** p < 0.01. Scale bar: 100 μm.

Proliferation and apoptosis of grafted GRPs in the normal spinal cord after GRP single or GRP + SC cografting, assessed by BrdU incorporation and activated caspase 3 staining, respectively. (A–L) Representative photomicrographs showed that BrdU+ cells (red) were colocalized with GFP+ GRPs (green) in either GRP single or GRP + SC cografted animals at 1 or 4 weeks post-grafting (arrows). (M) Quantification data showed that significantly more BrdU+ GFP+ double-labeled cells were found in the GRP + SC cografted group than in the GRP single-grafted group. (N–Q) Representative photomicrographs show that caspase 3+ cells (red) were colocalized with GFP+ GRPs (green) in either GRP single or GRP + SC cografted animals at 1 or 4 weeks postgrafting. (R) Quantification data showed that significantly less caspase 3+ GFP+ double-labeled cells were found in the GRP + SC cografted group than that in the GRP single-grafted group. Data shown are mean±SD (n = 5). *p < 0.05, ***p < 0.001. Scale bar: 25 μm.
SCs Promoted the Survival, Proliferation, and Migration of Cografted GRPs in the Adult Contused Spinal Cord
To examine the feasibility of cografting SCs and GRPs as a potential therapy for SCI, we transplanted spinal cordderived GFP+ GRPs into the injured spinal cord of rats at 9 days after SCI. This time point provides a clinically relevant time window for therapeutic interventions. We used a widely accepted model of contusive SCI, that is, the New York University Impactor (20), to produce a moderate contusive SCI at the T10 level. We first examined the survival and migration of transplanted GRPs within the injured spinal cord (Fig. 6). Similar to the results observed in the normal spinal cord, cografted SCs promoted a remarkable increase in the number (Fig. 6A) and the maximum migration distance of grafted GRPs from the injection sites (Fig. 6C) at 7 weeks after transplantation. In the SC-GRP cografted group, numerous GFP+ GRPs survived within and migrated away from the injured epicenter. Importantly, many GFP+ GRP-derived cells were in close association with axons that were either survived or regenerated at the injury epicenter (Fig. 6A). On the contrary, very few GFP+ cells survived and migrated for considerable shorter distances in the GRP single-grafted group (Fig. 6B). BrdU and activated caspase 3 assays at both 2 and 7 weeks showed that cotransplantation of SCs and GRPs remarkably promoted proliferation and survival of grafted GRPs in the contused spinal cords (data not shown), indicating that the increased number of GFP+ cells was likely due to both enhanced proliferation and survival. Finally, we did not observe any signs of tumorogenesis macroscopically or microscopically within the spinal cord after transplantation.

Survival and migration of grafted GFP+ GRPs in the spinal cord following contusive SCI. Representative longitudinal sections show that more GFP+ GRPs survived and migrated for longer distances from the injury epicenter (*) in the GRP + SC cografted group (A) as compared to the GRP single-grafted group (B) at 7 weeks after transplantation. Notably, numerous GFP+ GRPs survived at the injury epicenter, and many of them were associated with axons either spared or regenerated (*) in the GRP + SC cografted group (A). (C) Comparison on average maximum migration distances of grafted GFP+ GRPs rostrally and caudally from the site of injection in the injured spinal cord between GRP single and GRP + SC cografted groups. Data shown are mean±SD (n = 5). ***p < 0.001. Scale bar: 500 μm.
Differentiation of Grafted GRPs in the Injured Spinal Cord
We observed that, after SC-GRP or GRP transplantation, GFP+ GRPs integrated well into the injured spinal cord (Figs. 6 and 7), these cells differentiated into either oligodendrocytes or astrocytes, identified by an oligodendrocyte marker CC1 (Fig. 7A–G) and an astrocyte marker GFAP (Fig. 7I–O). Quantification data showed that about 40% of cells differentiated along an oligodendroglial lineage (Fig. 7H) and over 50% of cells differentiated into GFAP-positive astrocytes (Fig. 7P) at 7 weeks posttransplantation. We also compared the differentiation results of grafted GFP+ GRPs cells in SC-GRP and GRP transplantation groups in white matter and in gray matter and found no significant difference between the two groups in both areas (all p > 0.05, n = 5). These findings suggest that cografted SCs did not affect differentiation of grafted GRPs toward an oligodendrocyte lineage, although it promotes proliferation and survival of the grafted GRPs.

Differentiation of grafted GFP+ GRPs in the injured spinal cord. (A–G, I–O) Representative images showed that grafted GFP+ GRPs (arrows in A, D, I, L) differentiated into CC1+ (arrows in B, E, J, M) oligodendrocytes (arrows in C, F) and GFAP+ (arrows in A, D, I, L) astrocytes (arrows in K, N) in the contused spinal cord at 7 weeks posttransplantation in the GRP single or GRP + SC cografted group. (G, O) High-magnification confocal single plane images of boxed areas in (F) and (N) showing GFP+ cells expressed CC1 (G) and GFAP (O). (H, P) Quantification data showed that no statistically significant difference was found in the percentage of CC1+ oligodendrocytes (H) and GFAP+ astrocytes (P) between the two grafted groups or between the gray and white matter. Data shown are mean±SD (n = 5). Scale bar: 25 μm.
SC-GRP Cografting Enhanced Serotonergic Innervation Caudal to the Injury
Since almost all serotonergic axons, immunoreactive for 5-HT, in the spinal cord descend from supraspinal raphe nuclei, we examined 5-HT immunoreactivity (IR) caudal to the lesion as one measure of axon sparing and/or regeneration. We found that the number of 5-HT-IR axons in the gray matter at 6 mm caudal to the lesion epicenter in SC-GRP cografted group was twofold as many as those that received either GRP or SC single transplantation (p < 0.05, n = 5) (Fig. 8).

Quantification of 5-HT+ fibers in the ventral horn caudal to the injury. (A–D) Representative images show serotonin (5-hydroxytryptamine; 5-HT+) fibers in the ventral horn of spinal cord at 6 mm caudal to the lesion epicenter in the control (A), GRP (B), SC (C), or GRP + SC (D) grafted group. (E) Quantification data showed that the number of 5-HT+ fibers in the ventral horn was significantly higher in the GRP + SC cograft group than in all other groups at 7 weeks posttransplantation. Data shown are mean±SD (n = 5). *p < 0.05. Scale bar: 50 μm.
SC-GRP Cografting Enhanced Myelination
To investigate whether SC-GRP cografting has an effect on myelin preservation and/or remyelination, LFB staining was performed to examine the extent of residual myelin at the injury epicenter at 7 weeks after transplantation. As shown in Figure 9A–E, single transplantation of GRPs or SCs significantly enhanced the area of residual myelin at the injury epicenter as compared to the control group. Importantly, the area of residual myelin was further increased in the SC-GRP cografted group as compared to those received GRPs alone (p < 0.05, n = 5), SCs alone (p < 0.05, n = 5), or the control (p < 0.01, n = 5).
To further determine the ability of newly generated oligodendrocytes to form new myelin on spared or regenerated axons of the spinal cord, we performed double or triple labeling to identify grafted GRPs (GFP+), axons (NF+), and myelin (MBP+). GFP expression in the grafted GRPs was robust in both cell bodies and processes, so we were able to examine the close proximity of donor processes (GFP+) wrapping around host axons (NF+). Confocal images clearly displayed that numerous GFP+ OL processes wrapped around NF+ axons within the SC-GRP grafted region at the injury epicenter (Fig. 9F–G). Triple labeling showed that GRP-derived OL (GFP+) formed new myelin (MBP+) on axons (NF+) that either spared or regenerated at the lesion sites (Fig. 9H–J).

Myelination and tissue sparing of grafted GFP+ GRPs in the spinal cord following contusive SCI. (A–D) Representative images of the sections stained with Luxol fast blue showed the extent of myelin at the injury epicenter at the 7th week after transplantation. (E) Quantitative data of percent spared myelin area showed that cografting of GRPs and SCs resulted in a significant increase in spared myelin area as compared to GRP or SC single grafted group as well as the control group receiving no cell transplantation (*p < 0.05, **p < 0.01, n = 5). (F) Anti-GFP (green) and anti-neurofilament (NF, red) double-labeling at the site of GRP + SC grafting showed that numerous highly branched GFP+ oligodendrocytes (OLs), derived from grafted GRPs, were wrapping around NF+ axons. (G) A high-magnification single-plane image of the boxed area in F shows the close relationship between NF+ axons and GFP+ OLs. The dark area between the OL process and axon likely represents an unstained myelin sheath. (H–J) A confocal single plane image clear shows the formation of new myelin (blue, MBP+) on axons (red, NF+) by differentiated OLs, derived from grafted GRPs (green, GFP+). (K–N) Representative images of the sections stained with cresyl violet-eosin showed the extent of tissue sparing at the lesion epicenter at the 7th week after transplantation. (O) Quantitative data of relative lesion volume showed that cografting of GRPs and SCs significantly decreased the lesion volume as compared to GRP or SC single grafted group as well as the control group receiving no cell transplantation (*p < 0.5, **p < 0.01, n = 5). Scale bar: 200 μm (in A–D, K–N).
SC-GRP Cografting Enhanced Tissue Sparing
To assess the possibility that SC-GRP cotransplantation induced tissue sparing, we examined the gross morphology of the injured spinal cord and the extent of tissue sparing/loss relative to the corresponding total spinal cord volume in animals sacrificed at 7 weeks posttransplantation. The results showed that the lesion volume was significantly decreased in SC-GRP cografted animals when compared with those grafted with GRPs alone (p < 0.01, n = 5), SCs alone (p < 0.01, n = 5), or the control (p < 0.01, n = 5) (Fig. 9K–O), indicating that SC-GRP cografting has a greater neuroprotective effect than when a single cell type, either SCs or GRPs, is used.
SC-GRP Cografting Promoted Recovery of Function After SCI
To determine whether SC-GRP cografting could improve recovery of function after SCI, we assessed functional recovery using three independent behavioral tasks: BBB, grid-walking, and footprint analysis. All behavioral assessments were undertaken by observers blinded to the experimental groups. The BBB test was performed to evaluate the hindlimb locomotor function in an open field. One day after SCI, all injured rats were paraplegic with no observable hindlimb movement (BBB = 0) (Fig. 10A). Injured rats were allowed to recover until 1 week after injury (2 days before transplantation) and showed a mean BBB score of 6.1±0.5 (n = 40). We assigned these rats to four groups randomly and checked the BBB score distribution to make sure that there was no significant difference of BBB score between groups (Fig. 10B). In the first 3 weeks after transplantation, we continued to assess the BBB score and no significant difference was found between these groups (Fig. 10A). However, with continued recovery, rats received SC-GRP cografting showed a significant improvement in the BBB score as compared to the GRP or SC single-grafted group or the control group at 4–6 weeks postgrafting (Fig. 10A). The SC-GRP cografted group had a mean BBB score of 13.2±0.6 (n = 10) as compared to the GRPs alone (11.8±0.5, p < 0.01, n = 10), SCs alone (12.0±0.4, p < 0.05, n = 10), or the control group (11.5±0.5, p < 0.01, n = 10) at 6 weeks posttransplantation (Fig. 10A).
Grid-walk analysis was performed to examine the deficits in descending fine motor control after SCI (6). Neither GRP nor SC transplantation alone showed a significant difference in grid-walking performance when compared with the control group. However, SC-GRP cografting exhibited a notable improvement in grid-walking performance as compared to any other groups. As is shown in Figure 10C, the SC-GRP cografted group exhibited significantly lower footfall errors at 6 weeks after transplantation (41.00±5.20%, n = 10) as compared to the GRP (52.03±7.08%, p < 0.05, n = 7) or SC (51.06±6.45%, p < 0.05, n = 8) single-grafted group, or the control group (52.52±6.90%, p < 0.05, n = 5).
The footprint patterns of transplanted and control rats were analyzed for angle of rotation and base of support at 6 weeks after transplantation. As is shown in Figure 10D, the SC-GRP cografted group displayed a significant reduction (15.90±1.92°) of hindlimb angle of rotation as compared to the GRP group (20.48±1.96°, p < 0.05, n = 7) or SC single-transplanted (20.20±3.75°, p < 0.05, n = 8), or the control group (22.56±2.49°,p < 0.05, n = 5). Moreover, the SC-GRP cografted group demonstrated a significantly reduced base of support (2.84±0.42 cm, n = 10) as compared to the GRP (3.56±0.37 cm, p < 0.05, n = 7) or SC (3.52±0.23 cm, p < 0.05, n = 7) single grafted group or the control group (4.10±0.62 cm, p < 0.05, n = 7). No statistically significant difference was found in the foot print analysis between graft of the GRP, SC, or control group (Fig. 10E).

GRP + SC cografting significantly promoted locomotor recovery after SCI. (A) All animals (n = 40) scored 21 at 1 day preinjury, 0 at 1 day after injury, and an average of 6.1 at 1 week pretransplantation. The animals were randomly assigned to four groups (n = 10/group) for cell grafting. GRP + SC cografting significantly improved Basso, Beattie, Bresnahan (BBB) locomotor score at 4–6 weeks posttransplantation compared with all other groups. (B) The bar graph of BBB scores showed that there were no significant differences between groups at 2 days before cell grafting (1 week postinjury) and GRP + SC cografting significantly improved BBB score at 6 weeks posttransplantation. The distribution of BBB scores was plotted for individual rats (dots) for each of the groups at the first week post injury and sixth week posttransplantation. (C) GRP + SC cografting (n = 10) also showed percentage reduced footfall errors in hindlimb placements in the grid-walk analysis at 6 weeks posttransplantation compared to the control (p < 0.05, n = 5), GRP-grafted (p < 0.05, n = 7), or SC-grafted (p < 0.05, n = 8) group. (D, E) Footprint analysis revealed that GRP + SC cografting significantly reduced the hindlimb angle of rotation and the distance of base of support between the hind paws at 6 weeks posttransplantation compared to the control (p < 0.05, n = 5), GRP-grafted (p < 0.05, n = 7), or SC-grafted (p < 0.05, n = 8) group. (A–E) Data shown are mean±SD. *p < 0.05, **p < 0.01.
Discussion
Oligodendrocytes replacement can be a promising strategy for SCI repair. However, there exist two major challenges, that is, poor posttransplantation survival and inhibitory properties to axonal regeneration, which limit their use as donor cells for the repair of SCI. Thus, strategies aimed at enhancing the survival of grafted OPCs or GRPs as well as reducing their inhibitory properties should be highly prioritized. SCs are myelin-forming cells in the PNS and have been repetitively shown to promote axon regeneration after being grafted into the injured CNS (2,37,51,53,55,56). It has been well established that SCs express and secrete many growth factors including glial cell line-derived neurotrophic factor (GDNF) (12,46), nerve growth factor (NGF) (3,57), brain-derived neurotrophic factor (BDNF) (1,57), neurotrophin-3 (NT-3) (8), and fibroblast growth factor (FGF) (35). These secreted factors may be required for the growth and differentiation of OL lineage cells. Hence, it is important to determine whether SCs or their secreted factors in SCM have any effect on the survival, proliferation, differentiation, and migration of GRPs or OLs in vitro and in vivo. If so, we can further reason that cografting SCs and GRPs into the injured spinal cord may promote similar biological properties of GRPs/OLs and, therefore, enhance greater axonal regeneration and remyelination. As a final result, cografting GRPs and SCs may promote functional recovery after SCI.
To validate these hypotheses, we first examined whether SCs affect growth properties of GRPs in vitro. We showed that both SCs and SCM significantly protected GRPs from apoptosis and promoted the survival, proliferation, and migration of cultured GRPs isolated from the embryonic spinal cord and maintained the purity of GRPs. More importantly, when a mixture of SCs and GRPs was transplanted into the normal rat spinal cord, cografted SCs significantly increased the number and migratory distance of the grafted GRPs. BrdU incorporation and activated caspase 3 staining assays showed that the marked increase of GRPs attributed to both the proliferation and survival of GRPs, promoted by cografted SCs. These in vivo results are highly consistent with the in vitro results, strongly suggesting that SCs might have constructed a favorable microenvironment to stimulate or support the survival, proliferation, and migration of grafted GRPs. SCM is sufficient to enhance these biological properties of GRPs in vitro in the absence of SCs themselves, meaning that the SC-derived factors that affect GRPs appear to be diffusible molecules.
The remarkable effect of SCs on the proliferation, cell survival, migration, and differentiation properties of GRPs strongly suggests that cograft of GRPs and SCs may be a novel strategy for SCI repair. It should be noted that the microenvironment after SCI is totally different from that in the normal spinal cord, which could be hostile for the grafted GRPs. For example, injury-induced cytokines produced by inflammatory cells such as macrophages and reactive microglia initiate a cascade of cellular responses (26,27,40) that could be detrimental to the grafted GRPs. We predicted that cografted SCs might have a similar beneficial effect on the biological properties of GRPs in the injured spinal cord as they did in the normal cord. Indeed, SCs also decreased the apoptosis of GRPs and promoted proliferation and migration of GRPs after they were cografted into the injured spinal cord. Importantly, many grafted GRPs survived in the injury epicenter and were in close association with axons that were either spared or regenerated. These results were consistent with our previous report that GRPs or OPCs expressing Nogo-A but not MAG are significantly more permissive than mature oligodendrocytes expressing both myelin inhibitors in vitro (32). A previous study demonstrated that about 15% of grafted GRPs and 30% of grafted GRPs that express multineurotrophins differentiated into APC+ oligodendrocytes, which formed myelin; many of the grafted GRPs also differentiated into GFAP+ astrocytes, and some other GRPs remained undifferentiated in injured spinal cord 6 weeks posttransplantation (9). In the present study, we characterized the fate of grafted GRPs and found similarly that these GRPs differentiated into CC1+ oligodendrocytes, which remyelinated axons as well as GFAP+ astrocytes at the site of injury. Some GRPs remained undifferentiated in both GRP-only and GRP + SC grafted groups. Quantitative analyses revealed that about 50% of grafted cells differentiated into astrocytes, whereas 40% differentiated into oligodendrocytes in both groups. Higher OL lineage differentiation in the present study than the previous study may be, in part, due to the induction of GRPs with T3 for 3 h prior to transplantation in our study, and to the difference in end point examinations (6 weeks vs. 7 weeks), different cellular markers used (APC vs. CC1), different SCI models applied [Infinite Horizon (IH) vs. NYU], and different injection sites (4 sites vs. 5 sites) between the two studies.
SCI inevitably induces neuronal death and axonal degeneration, which contribute to functional deficits after the injury (27,47). Descending serotonergic system plays important roles in mediating motor, sensory, and autonomic functions (21). Initiation of hindlimb movement is influenced by serotonergic activation of motor output via a central pattern generator (19,42) that organizes automated execution of hindlimb movements. To determine whether cografted GRPs and SCs play a role in axonal protection after SCI, we examined serotonergic innervation in the gray matter caudal to the injury. We demonstrated that a significantly greater number of 5-HT-positive fibers were found in SC-GRP cografted animals when compared with cells that were either transplanted alone or the control. Moreover, quantitation of lesion volume and myelin sparing at the lesion epicenter also showed enhanced tissue and myelin sparing in the SC-GRP cografted group. Triple staining of GFP, NF, and MBP further confirmed that grafted GRPs had differentiated into mature OLs that formed new myelin on axons, which were either spared or regenerated in a model of contusive SCI. Together, these results indicate that SC-GRP cografting contributes to axonal protection and myelination by cografted GRPs on spared or regenerated axons following SCI and thus helps to explain the subsequent functional improvement.
An important observation of the present study is that locomotor function was significantly improved only in the group that received the SC-GRP cograft after SCI. Animals received GRPs or SCs alone after SCI demonstrated no significant improvement in locomotor recovery. Previous study showed similar functional effects following GRP transplantation alone (9), although the recovery of transcranial magnetic motor-evoked potential responses and hindlimb locomotor function (BBB) was significantly enhanced in the GRP-grafted animals that express D15A [with both BDNF and neurotrophin 3 (NT3) activities] (9). While substantial myelination has also been observed in the latter two groups in this study, such myelination might not be enough for significant behavioral improvement. In other words, the lack of functional recovery in the latter groups could also be caused by the fact that the behavioral tests that we chose might not be sensitive enough to detect these differences. The functional recovery observed in the SC-GRP cografted group is consistent with enhanced survival, proliferation, migration, and myelination of grafted GRPs observed in this group. Thus, we suggest that cografted SCs exhibited a powerful and beneficial effect on grafted GRPs and, together with SCs, they promoted axonal protection, tissue and myelin sparing, and new myelin formation, resulting in improved locomotor recovery.
In conclusion, our results show that SCs promoted robust survival, proliferation, and migration of grafted GRPs in vitro and in vivo. The increased number of grafted GRPs and their differentiation into functional OLs, together with cografted SCs, may positively modify the detrimental microenvironment of the injured spinal cord and improve functional neurological recovery. These results strongly support the translation of SC-GRP cografting as a potential therapeutic strategy for the treatment of SCI.
Footnotes
Acknowledgments
We would like to thank Ms. Terren Green for her critical reading of the manuscript. This work was supported by National Institutes of Health (NINDS NS36350, NS52290, NS50243, NS59622), Indiana Spinal Cord and Brain Injury Board (91910, 91913), the Mari Hulman George Endowment, and the National Natural Science Foundation of China (81171465). The authors declare no conflicts of interest.
