Abstract
Glial scar formation is the major impedance to axonal regrowth after spinal cord injury (SCI), and scar-modulating treatments have become a leading therapeutic goal for SCI treatment. In this study, human neural stem cells (NSCs) encoding interferon-β (INF-β) gene were administered intravenously to mice 1 week after SCI. Animals receiving NSCs encoding IFN-β exhibited significant neurobehavioral improvement, electrophysiological recovery, suppressed glial scar formation, and preservation of nerve fibers in lesioned spinal cord. Systemic evaluation of SCI gliosis lesion site with lesion-specific microdissection, genome-wide microarray, and MetaCore pathway analysis identified upregulation of toll-like receptor 4 (TLR4) in SCI gliosis lesion site, and this led us to focus on TLR4 signaling in reactive astrocytes. Examination of primary astrocytes from TLR4 knockout mice, and in vivo inhibition of TLR4, revealed that the effect of IFN-β on the suppression of glial scar formation in SCI requires TLR4 stimulation. These results suggest that IFN-β delivery via intravenous injection of NSCs following SCI inhibits glial scar formation in spinal cord through stimulation of TLR4 signaling.
Keywords
Introduction
Spinal cord injury (SCI) is a devastating clinical condition that results in permanent disability due to very limited regenerative capability of the adult human spinal cord. One major impediment to axonal regeneration in SCI is glial scar formation, a process mainly directed by reactive astrocytes (43). Normally, quiescent astrocytes in adults respond vigorously to injury; during the acute phase of injury, some of these responses have beneficial effects, such as isolating the injury site and minimizing the area of inflammation and cellular degeneration. Some astrocyte populations may even support axonal regrowth (6); however, astrocytes eventually become hypertrophied and proliferative, upregulate the expression of glial fibrillary acidic protein (GFAP), and form a dense network of glial processes around the injury site (11). Therefore, scar-modulating treatments have become a leading therapeutic goal for the treatment of SCI (9,38).
We have previously examined the ability of liposomemediated interferon-β (IFN-β) gene delivery to inhibit the formation of glial scar tissue in a SCI mouse model (12) and found that the IFN-β administration induced functional and structural recovery in injured spinal cord including regrowth of corticospinal tract (CST) axons.
Recently, there has been a great deal of interest in the potential use of stem cells in SCI treatment because of their abilities to self-renew, migrate, and differentiate into all types of neural cells (18). Neural stem cells (NSCs), in particular, are characterized by the capability to home in and deliver therapeutic genes (41), and constitute a promising source for cell replacement therapy (22,25,45).
In the present study, we attempted to attain neuronal regrowth via intravenous transplantation of human NSCs encoding genes for cytosine deaminase (CD) and IFN-β (F3.CD.IFN). We investigated whether human NSCs transduced with IFN-β gene can inhibit glial scar formation and improve spinal function after SCI, while the CD gene, a therapeutic suicide gene that converts nontoxic prodrug 5-fluorocytosine (5-FC) into toxic 5-fluorouracil (5-FU), provides a safe guard to allow removal of cells in cases of undesirable proliferation. Further, in order to clarify the function of IFN-β, we attempted to identify profile changes in SCI gliosis lesion site by using gliosis site-specific microdissection, genome-wide microarray, and MetaCore pathway analysis. This systematic data processing revealed the upregulation of toll-like receptor 4 (TLR4), and we subsequently focused on the functional role of TLR4 signaling cascades in reactive astrocytes. We here verify that the SCI-induced proliferation of reactive astrocytes in lesion is suppressed by the ligation of TLR4 in the presence of IFN-β.
Materials and Methods
Neural Stem Cells
The HB1.F3 (F3) human NSC line was generated from human fetal telencephalon and immortalized by transfection with a retroviral vector encoding the v-myc myelocytomatosis viral oncogene homolog (v-myc), as described previously (28). It has been confirmed that this human NSC line is capable of self-renewal and has multipotent capacity to differentiate into neuronal or glial cell lineages both in vivo and in vitro (23,28). The F3 cell line was infected with a replication-incompetent retroviral vector encoding β-galactosidase (lacZ) and puromycinresistance genes. The cell line was subsequently designated as F3.LacZ. In this study, the clonal F3.CD. IFN-β line (F3.CD.IFN) was derived from parental F3.CD cells, as previously described (1,12). F3.LacZ and F3.CD.IFN cells were cultured in Dulbecco's modified Eagle's medium (DMEM) with high glucose (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Life Technologies, Grand Island, NY, USA), 100 U/ml penicillin, and 100 μg/ml streptomycin (both from Sigma-Aldrich, St. Louis, MO, USA).
In Vitro Assay of Astrocyte Suppression by F3.CD.IFN Cells
The suppressive effects of F3.LacZ and F3.CD.IFN cells on the growth of primary-cultured astrocytes were quantified in the presence or absence of 5-FC (Sigma-Aldrich). Primary astrocyte cultures were prepared from the cerebral hemispheres of fetal BALB-c nude mice (SLC, Shizuoka, Japan). The hemispheres were cleared of the meninges and choroid plexus and digested with 0.1% trypsin (Invitrogen) in phosphate-buffered saline (PBS; Sigma-Aldrich) for 30 min at 37°C, followed by dissociation into single cells by repeated pipetting. A suspension containing 2 × 105 cells was seeded into 35-mm poly-l-lysine-coated Petri dishes (BD Biosciences, San Jose, CA, USA), and the majority of cells (90–95%) were confirmed to be astrocytes based on immunocytochemical analysis for GFAP expression. Astrocytes were cultured on Petri dishes for 1 week, after which, F3.NSCs were added to the astrocyte cultures. Astrocytes were cocultured with either F3.LacZ or F3.CD.IFN at various ratios of astrocytes to F3 cells (1:0, 20:1, and 40:1). F3 cells were prelabeled by incubating for 20 min in culture medium containing the dye Cell Tracker™ DiI (Invitrogen), which emits 570-nm fluorescence. For experiments using F3.CD.IFN cells treated with 5-FC, 5-FC was added to the conditioned medium at a final concentration of 500 mg/ml following 24 h of culture, and the culture was maintained for 24 h. Cells were subsequently immunostained with anti-GFAP antibody (DAKO, Glostrup, Denmark) and Alexa 488-labeled IgG (Molecular Probes, Eugene, OR, USA). The cultures were analyzed using an Olympus FV5-PSU confocal laser microscope (Olympus, Tokyo, Japan), and the total numbers of GFAP-positive cells were counted. Each experiment was performed in triplicate. The suppressive effects of F3.LacZ and F3.CD.IFN on the growth of primary-cultured TLR4-deficient astrocytes were also quantified. Primary TLR4-deficient astrocyte cultures were prepared from the cerebral hemispheres of fetal TLR4 knockout mice (Oriental Bio, Kyoto Japan). A suspension containing 2 × 105 cells was seeded onto 35-mm poly-l-lysine-coated Petri dishes. The majority of cells (90–95%) were immunocytochemically confirmed to be astrocytes and cultured on Petri dishes for 1 week, after which, F3.NSCs were added to the astrocyte cultures. Astrocytes were cocultured with either F3.LacZ or F3.CD.IFN cells at various ratios of astrocytes to F3 cells (1:0, 20:1, and 40:1). F3 cells were prelabeled by the Cell Tracker CM-DiI, and the culture was maintained for 2 days. Cells were subsequently immunostained with anti-GFAP antibody followed by Alexa 488-labeled IgG, and the total numbers of GFAP-positive cells were counted. Each experiment was performed in triplicate.
Spinal Transection Procedure
Adult female Balb-c nude mice (8–12 weeks old; SLC) were used in this study. All experiments were performed in accordance with the ethical guidelines of the Nagoya University Institutional Animal Care and Use Committee. The mice were anesthetized with 1.5% halothane and maintained on 1.25% halothane (Takeda Pharmaceutical, Osaka, Japan) in an oxygen–nitrous oxide gas mixture. Laminectomy was performed at vertebral level T9–T10. The dura was opened, and the dorsal half of the spinal cord was transected to a depth of 1 mm with a pair of extrafine microscissors (Kent Scientific Corporation, Torrington, CT, USA). In the sham group, laminectomy was conducted without the accompanying SCI. Following spinal transection, the overlying muscle and skin were sutured. The mice were placed on soft bedding on a warming blanket held at 37°C for 1 h after surgery. One week later, the motor function of each animal was evaluated according to the Basso mouse scale (BMS), as described below. Only animals with BMS scores below 4 out of 9 points were used. These were divided randomly into four treatment groups: (1) PBS group, SCI with intravenous (IV) administration of 100 μl PBS; (2) F3.LacZ group, SCI with IV administration of F3.LacZ (2 × 106 cells in 100 μl of PBS); (3) F3.CD.IFN group, SCI with IV administration of F3.CD.IFN (2 × 106 cells in 100 μl of PBS); and (4) F3.CD.IFN + 5-FC group, SCI with IV administration of F3.CD.IFN (2 × 106 cells in 100 μl of PBS) and, beginning 2 days later, an intraperitoneal (IP) injection of 5-FC daily (900 mg/kg) for 10 consecutive days. In order to elucidate the mechanism by which IFN-β elicits effects following SCI, we used the same spinal transection procedure and assigned the mice to two treatment groups 1 week later: (1) F3.LacZ group, SCI with IV administration of F3.LacZ (2 × 106 cells in 100 μl of PBS); (2) F3.CD.IFN group, SCI with IV administration of F3.CD.IFN (2 × 106 cells in 100 μl of PBS). In addition, the F3.CD.IFN group was randomly divided into two further groups 1 week after the initial group assignment: (2-a) F3.CD.IFN group, (2-b) F3.CD.IFN + OxPAPC group, SCI with IV administration of F3.CD.IFN (2 × 106 cells in 100 μl of PBS), and 7 days later, an IP injection of 50 μg of OxPAPC (TLR4 Inhibitor; InvivoGen, San Diego, CA, USA). Finally, we compared the functional recovery in F3.LacZ, F3.CD. IFN, and F3.CD.IFN + OxPAPC groups.
Laser-Captured Microdissection (LCM) and Microarray
On day 10 after surgery, five animals from the laminectomy only and plain SCI groups were deeply anesthetized with barbiturate overdose and intracardially perfused with PBS. Their spinal cords were removed and immediately frozen in Tissue-Tek OCT medium (Sakura Finetek, Tokyo, Japan). The spinal cords were sectioned in the sagittal plane onto uncoated slides. A PixCell II LCM instrument (Arcturus, Mountain View, CA, USA) was used to dissect the injury site, and RNA was extracted from the microdissected samples using the PicoPure RNA Isolation Kit (Arcturus), according to the manufacturer's instructions. Total RNA was pooled from the five animals from each group and amplified and labeled using the Amino Allyl MessageAmp aRNA Kit (Ambion, Austin, TX, USA). Briefly, after reverse transcription (2 mg total RNA/sample), double-stranded cDNA was transcribed in vitro using the amino allyl cRNA. The RNA was amplified twice, and the purified and concentrated cRNA (5 mg) was coupled with either Cy3 or Cy5 dyes (GE Healthcare, Wauwatosa, WI, USA). The dye-labeled aRNA was purified from uncoupled dye using Micro Bio-Spin P-30 Tris chromatography columns (Bio-Rad, Hercules, CA, USA) and Microcon YM-30 centrifugal filter devices (Millipore, Billerica, MA, USA). The cRNA was fragmented in a fragmentation buffer [40 mmol/L Tris acetate (pH 8.1), 100 mmol/L potassium acetate, and 30 mol/L magnesium acetate; Life Technologies] at 94°C for 15 min and purified with Microcon YM-10 (Millipore). An oligonucleotide-based mouse DNA microarray, AceGene (mouse Oligo Chip 30K; DNA Chip Research, Yokohama, Japan) was preblocked with 1% bovine serum albumin (BSA; Sigma-Aldrich) solution. The fragmented cRNA was added to the microarray in hybridization solution and subsequently hybridized at 42°C for 16 h. The arrays were then washed, scanned at a pixel size of 10 mm, gridded, and analyzed (GenePix 4000B; Axon Instruments, Union City, CA, USA). The background was subtracted, and the medium sum intensity (CH1 and CH2) of < 100 absorbance units was excluded. Data were normalized by the trimmed mean at 10% to account for the differences in the amounts of labeled RNA or labeling efficiencies.
Pathway Analysis
We next attempted to identify the biological pathways involving novel gene networks that are activated in reactive astrocytes after SCI. For this purpose, we analyzed the genes whose expressions were upregulated in the lesion by at least a factor of 2.0, relative to their respective expressions in the normal spinal cord. The functional mapping tool MetaCore (GeneGO, St Joseph, MI, USA) was used in this analysis. MetaCore is a Web-based computational platform designed primarily for the analysis of high-throughput experimental data in the context of mouse regulatory networks and pathways. It includes a curated database of protein interactions, metabolism, and bioactive compounds. For a network of a particular size, MetaCore can be used to calculate statistical significance based on the probability of the network's assembly from a random set of nodes (genes) that is of the same size as the input list (p value).
In Vivo Migration of DiI-Labeled NSCs
F3.LacZ cells were prelabeled by incubating for 20 min in culture medium containing Cell Tracker CM-DiI. We evaluated whether DiI-labeled NSCs could migrate to the SCI site in adult mice after IV transplantation. Seven days after the induction of transection injury, the injured animals were injected via the tail vein with 2 × 106 DiI-labeled F3.LacZ NSCs in 100 μl of PBS. The control injured animals were injected with PBS alone. Seven days after NSC transplantation, the animals were euthanized and transcardially perfused with 4% paraformaldehyde (PFA; Tokyo Chemical Industry, Tokyo, Japan). Subsequently, we injected F3.LacZ cells without labeling into the injured animals. Seven days later, the mice were deeply anesthetized with an overdose of barbiturate and intracardially perfused with PBS followed by 4% PFA in PBS. The lesioned spinal cord region was removed and postfixed overnight in the same fixative. The fixed tissues were embedded in paraffin, and 3-μm sections were prepared for immunohistochemistry. Immunohistochemical staining was performed as per the manufacturer's instructions. Anti-mouse β-galactosidase antibody (Sigma-Aldrich; dilution 1/10,000) was applied to the samples.
RT-PCR for IFN-β and TLR2 and TLR4 Expression at the SCI Site
The total RNA extracted from the harvested tissues was subjected to DNase (Invitrogen) treatment prior to reverse transcription using a Transcriptor First-Strand cDNA Synthesis Kit (Roche, Mannheim, Germany), according to the manufacturer's protocol. PCR amplification was performed using GoTaq DNA polymerase (Promega, Madison, WI, USA) with the following primers: human IFN-β, 5′-GCCGCATTGACCATCTATGAGA-3′ (sense) and 5′-GAGATCTTCAGTTTCGGAGGTAAC-3′ (antisense); mouse TLR2, 5′-ACAGCTACCTGTGTGAC TCTCCGCC-3′ (sense) and 5′-GGTCTTGGTGTTCATT ATCTTGCGC-3′ (antisense); mouse TLR4, 5′-ACCTGGC TGGTTTACACGTC-3′ (sense) and 5′-CAGGCTGTTTG TTCCCAAAT-3′ (antisense); mouse β-actin, 5′-GACATG GAGAAGATCTGGCACCACA-3′ (sense) and 5′-ATCT CCTGGTCGAAGTGTAGAGCAA-3′ (antisense); human and mouse glyceraldehyde 3-phosphate dehydrogenase (GAPDH; product length; 454 bp), GACCACAGTCCAT CGCATCA (sense) and GTCCGCCACCCTGTTGCTGT (antisense).
Immunohistochemistry
The mice were deeply anesthetized with an overdose of barbiturate and intracardially perfused with PBS followed by 4% PFA in PBS. The lesioned spinal cord region was removed and postfixed overnight in the same fixative. The fixed tissues were embedded in paraffin, and 3-μm sections were prepared for immunohistochemistry. Immunohistochemical staining was performed as per the manufacturer's instructions. Briefly, sections were preblocked with the blocking reagent (DAKO REAL Peroxidase-Blocking Solution, DAKO), and antigen retrieval was performed for GFAP. After sections were stained with rabbit anti-GFAP (DAKO), mouse anti-neurofilament (NF) (Nichirei, Tokyo, Japan), and a polymer reagent (Chemomate Envision kit/HRP, DAKO), color was developed with diaminobenzidine (DAB). Finally, sections were counterstained with hematoxylin. The corresponding tissue sections were routinely stained by hematoxylin and eosin (Sigma-Aldrich) for morphological evaluation. Five areas of immunostained sections were randomly selected, and the color intensity was measured using NIH image software (Bethesda, MD, USA) as reported (15). Background intensity was subtracted from the mean intensity of the images, and the means were used for statistical analysis.
Basso Mouse Scale for Locomotion
The recovery of hindlimb motor function in seven mice in each treatment group was measured using the BMS for locomotion, as previously described (2). The behavior of each animal was videotaped for 5 min, and two investigators provided a score (on a scale of 0–9) for each hindlimb at 1, 2, 3, 4, 6, and 8 weeks after injury. The scores from both hindlimbs were averaged to obtain a single value per animal for each time point.
Inclined Plane Test
We constructed an inclined plane as previously described (37) and tested the ability of each animal to maintain its position on the incline at 1, 2, 3, 4, 6, and 8 weeks after injury. The maximum inclination at which a mouse could maintain itself for 5 s was recorded; this value was considered to represent the functional ability of the mouse. In practice, the angle was either increased or decreased by 5° intervals until the mouse could maintain its position on the inclined plane for 5 s without falling.
Electrophysiology
To measure signal conduction in mouse motor pathways following SCI, transcranial electrical motor evoked potentials (MEPs) were measured at 4 and 8 weeks after injury. To record MEPs, mice were anesthetized with 1.5% halothane and maintained in 1.0% halothane in oxygen. All recordings were performed using standard clinical electromyographical analysis with a 3000-Hz hi-cut filter and a 30-Hz low-cut filter (Nihon Kohden, Tokyo, Japan). Electrical stimulation (1 Hz, 15 mA) was delivered over the cranium at an area 1 mm lateral to the bregma. Evoked responses were recorded with needle electrodes from the contralateral femoral muscle. Response amplitudes were measured from base to peak.
In Vivo MRI
To investigate the status of injured neural tissue at the SCI site in a noninvasive manner, magnetic resonance imaging (MRI) was performed using 7.0-T MRI (superconducting magnet: Kobelco and Jastec, Japan; console: Avance-I system, Bruker BioSpin, Ettlingen, Germany), with a volume coil for transmission (Bruker BioSpin), and two-channel phased array coil for reception (Rapid Biomedical, Rimpar, Germany) dedicated to examining spinal cord lesions in live mice. Diffusion tensor imaging (DTI) and T2-weighted (T2WI) MRI were conducted 4 weeks after injury in mice assigned to the PBS, F3.LacZ and F3.CD.IFN groups. DTI data sets were acquired with a respiratory-gated gradient-echo echo-planar diffusion-weighted pulse sequence, based on the Stejskal-Tanner diffusion preparation (42). Scanning parameters of the DTI were as follows: repetition time (TR), 3000 ms; echo time (TE), 34 ms; flip angle, 90°; field-of-view (FOV), 25.6 × 12.8 mm; data matrix, 128 × 128; reconstructed image resolution, 200 × 100 μm 2; slice thickness, 1.0 mm; b-value, between 0.68 and 1000 s/mm2; motion-probing gradient (MPG) orientations, 30 axes and 5 B0 images; number of averaging (NA), 1; number of echo planar imaging (EPI) segment, 4; fat saturation pulse, on. T2WIs were acquired through rapid acquisition with relaxation enhancement (RARE) sequence as follows: TR, 4200 ms; TE, 12 ms; effective TE, 36 ms; FOV, 25.6 × 12.8 mm; data matrix, 256 × 128; reconstructed image resolution, 100 × 100 μm2; slice thickness, 1.0 mm; fare factor, 8; NA, 8; fat saturation pulse, on. Mice were placed under anesthesia with a mixture of 1.5% isoflurane, 40% oxygen, and 58% air delivered via tracheal intubation prior to T2WI and DTI analysis. Vital signs (respiration, heart rate, and body temperature) of the anesthetized animals were monitored using an MRI-compatible monitoring and gating system (Model 1025; SA Instruments Inc., Stony Brook, NY, USA). Respiratory-gated acquisition was used to increase the image quality by minimizing breathing-related image artifacts whenever necessary. To reduce motion artifacts, animals were immobilized on an acrylic bed, and a specially designed head positioner (Rapid Biomedical) for gated imaging was attached to each animal's front thorax. Diffusion tensor analysis was performed using ParaVision with Jive software (Bruker BioSpin). Three eigenvectors (e1, e2, e3) associated with the largest eigenvalue (λ1) were visualized as a color map (left–right, red; posterior–anterior, green; superior– inferior, blue) (35).
Statistical Analysis
The statistical significance of observed differences was determined by ANOVA (StatView; SAS Institute, Cary, NC, USA), and Bonferroni's correction was employed for multiple comparisons. All reported p-values are two-tailed; a value of p < 0.05 was considered statistically significant.
Results
Molecular Signature of Reactive Astrocytes Following SCI
A laser-captured microdissection technique was used to procure specific areas from each heterogeneously damaged tissue structure. In order to analyze the molecular signaling of reactive astrocytes following SCI, the sites of lesions that were expected to form a glial scar were captured, and a microarray was used to profile the expression of 30,000 genes on day 10 after dorsal hemisection of the spinal cord at the T9–T10 level. We evaluated gene expression as either significantly upregulated or significantly downregulated by using the fold-level criteria (greater than twofold change: the significance of upregulation was set at >2 and that of downregulation at <0.5). Data analysis identified 488 significantly upregulated genes and 226 downregulated genes in several different categories. Only 1.6% of the probe set corresponding to individual genes was expressed at a level >2.0-fold after SCI. After the microarray data were imported into MetaCore, functional gene networks were generated to integrate reactions and interactions around the identified genes of interest. Significantly activated pathways are displayed in Figure 1A (p < 0.05). In this study, we focused on TLRs that may be related to IFN-β as reverse transcriptase-polymerase chain reaction (RT-PCR), and quantitative RT-PCR revealed that TLR4 was markedly upregulated in the lesioned astrocytes (Fig. 1B).

Significantly activated pathways and TLR's following spinal cord injury. (A) Significantly activated pathways at glial scar following spinal cord injury. (B) Toll-like receptor 4 (TLR4) is markedly upregulated in the lesioned astrocytes. Cdc42, cell division cycle 42; GTPase, guanosine triphosphatase, TGF, transforming growth factor; IL-1, interleukin-1; pAS, primary cultured astrocytes; nSC, normal spinal cord; SCI-glia, glial scar in spinal cord injury.
F3 Cells Migrate to SCI Lesion Sites in Mice
We evaluated the efficient migration of intravenously injected F3 NSCs tagged with DiI or lacZ to SCI sites in mice (Fig. 2A). It was revealed that a large number of DiI-labeled F3 cells migrated to the lesion site (Fig. 2B), and immunohistochemical staining with anti-β-galactosidase (β-gal) antibody demonstrated that β-gal-positive cells were also extensively distributed in the lesioned area (Fig. 2C, D). All NSCs were found to be located in or around the injured parenchyma, indicating that there was specificity of migration. Conversely, in the control injured animals injected with PBS alone, no β-gal-positive cells were observed in the spinal cord (Fig. 2E, F). IFN-β expression was also detected at the SCI site of the animals injected with 2 × 106 F3.CD.IFN by RT-PCR (Fig. 2G).

F3 human neural stem cells migrate to mouse spinal cord injury (SCI) lesion site. (A) A hematoxylin and eosin-stained section shows that a large number of transplanted neural stem cells infiltrated in the lesioned site. (B) Fluorescence microscopy reveals a large number of DiI-labeled F3 cells at the lesion site. (C) Immunohistochemical staining with anti-b-galactosidase (b-gal) antibody demonstrates that b-gal-positive cells are extensively distributed in the lesioned area of a mouse injected with F3 cells expressing b-gal (F3.LacZ). (D) Higher magnification of (C). (E) A control injured animal injected with PBS alone; no b-gal-positive cells are observed in the spinal cord. (F) Higher magnification of (E). (G) Interferon (IFN)-b mRNA expression is detected at the SCI site of the animals injected with cytosine deaminase and IFN-b-expressing (F3.CD.IFN-b) neural stem cells by RT-PCR. F3.CD.IFN, positive control; SCI/F3.CD.IFN, SCI site of an animal injected with F3.CD.IFN-b cells; SCI, SCI site of an animal injected with PBS only.
F3.CD.IFN Cells Inhibit Growth of Primary Astrocytes, But Not TLR4-Deficient Astrocytes
To quantify the inhibitory effect of F3.CD.IFN on astrocyte growth, DiI-labeled F3 cells were cocultured with primary normal astrocytes in different ratios. As shown in Figure 3A and B, the number of astrocytes significantly decreased with a reduction in the astrocyte/F3 ratio in the F3.CD.IFN group. When they were cocultured with F3.CD.IFN with added 5-FC (F3.CD.IFN + 5-FC), there was no growth inhibitory effect on cultured astrocytes. To evaluate whether TLR4 is necessary for IFN-β to exert this inhibitory effect on astrocyte growth, DiI-labeled NSCs were also cocultured with astrocytes derived from TLR4-deficient mouse in 40:1 ratio. As shown in Figure 3C and D, the number of TLR4-deficient astrocytes did not change. Thus, the inhibitory effect by the F3.CD.IFN cells was not exerted in the absence of TLR4.

F3.CD.IFN-β cells inhibit growth of primary normal astrocytes. Astrocytes were cocultured with either F3.LacZ or F3.CD. IFN-β cells (F3.CD.IFN) at various ratios of astrocytes to F3 cells (1:0, 20:1, and 40:1). (A) Immunofluorescence images of primary astrocytes alone or coculture (astrocyte:F3 ratio, 40:1) of astrocytes and F3.LacZ, F3.CD.IFN, or F3.CD.IFN + 5-fluorocytosine (5FC), respectively. Green cells and red cells indicate glial fibrillary acidic protein (GFAP)-positive astrocytes and DiI-labeled F3 cells, respectively. (B) Subsequently, cells were immunostained with anti-GFAP antibody, and the total numbers of GFAP-positive cells were counted. The number of astrocytes decreased significantly in F3.CD.IFN group, *p < 0.05. (C) To evaluate whether TLR4 is necessary for IFN-β to exert this inhibitory effect on astrocyte growth, DiI-labeled NSCs were also cocultured with primary astrocytes derived from TLR4-deficient mouse in 40:1 ratio. (D) The number of TLR4-deficient astrocytes did not change.
Intravenous Administration of F3.CD.IFN-β NSCs Spares Host Neural Tissue and Enhances Axonal Regeneration, and TLR4 Blockade Cancels Regeneration
To determine the effects of the IV administration of F3.CD.IFN cells on neuronal regeneration and the inhibition of glial scar formation, the expression of GFAP and NF at the injury site was immunohistochemically evaluated. SCI was shown to result in a severe loss of neurons at the injury site, as well as the extensive growth of reactive astrocytes forming a glial scar. Hematoxylin and eosin (H&E) staining revealed severe destruction of the dorsal half of the spinal cord above the central canal (Fig. 4A), and the formation of a granular scar was clearly detected at the injury site. Immunohistochemical staining for GFAP showed hypertrophy of GFAP-positive reactive astrocytes even in the area ventral to the central canal (Fig. 4B). The intensity of GFAP staining was significantly decreased in the F3.CD.IFN-treated group compared to that observed in the other groups. GFAP density was calculated quantitatively using NIH imaging (mean±SEM): sham group, 17±0.5; PBS, 38±2.9; F3.LacZ, 35±2.3; F3.CD.IFN, 28±1.2; F3.CD.IFN + 5FC, 35±1.1 (Fig. 4C). In the PBS and F3.LacZ groups, NF-stained transverse sections demonstrated significant loss of neural fibers extending to the ventral side (Fig. 4D), while only partial preservation of neural axons was detected in the F3.CD. IFN + 5FC group. Compared to these findings, sections from the F3.CD.IFN group revealed the dramatic preservation of fiber number and alignment, which was confirmed by the quantification of NF density: sham, 31±0.4; PBS, 21±1.5; F3.LacZ, 21±1.3; F3.CD.IFN, 30±1.8; F3.CD.IFN + 5FC, 24±3.5) (Fig. 4E). Next, the expression of GFAP and NF at the injury site was compared in mice treated with F3.CD.IFN in the presence or absence of the TLR4 inhibitor OxPAPC. While the dorsal half of the spinal cord was destroyed equally in every group, the anterior half of the spinal cord was found to be markedly preserved in the F3.CD.IFN group when compared to the F3.CD.IFN + OxPAPC group (Fig. 5A). The intensity of GFAP staining was also significantly decreased in the F3.CD.IFN group compared to that observed in the F3.CD.
IFN + OxPAPC and F3.LacZ groups (Fig. 5B). NF-stained transverse sections also demonstrated a significant loss of neural fibers in the F3.CD.IFN + OxPAPC and F3.LacZ groups, while neural fiber was dramatically preserved in sections from the F3.CD.IFN group (Fig. 5C).

IV administration of F3.CD.IFN-β neural stem cells spares host neural tissue and enhanced axonal regeneration. (A) Hematoxylin and eosin-stained sections of spinal cord injury sites, showing severe destruction of the dorsal half of the spinal cord above the central canal (arrow) and the formation of a granular scar at the injury site. (B) GFAP immunohistochemical staining, showing hypertrophy of GFAP-positive reactive astrocytes even in the area ventral to the central canal. The intensity of GFAP staining is markedly decreased in the F3.CD.IFN group compared to that in the other groups. (C) GFAP density was calculated quantitatively using NIH imaging, *p < 0.05. (D) Neurofilament (NF) immunohistochemical staining. In the PBS and F3.LacZ groups, NF-stained transverse sections demonstrate significant loss of neural fibers. In the F3.CD.IFN + 5FC group, only partial preservation of neural axons is detected. Sections from the F3.CD.IFN group reveal dramatic preservation of fiber number and alignment. (E) Quantification of NF density. The intensity of NF staining is markedly increased in the F3.CD.IFN group compared to that in the other groups, *p < 0.05
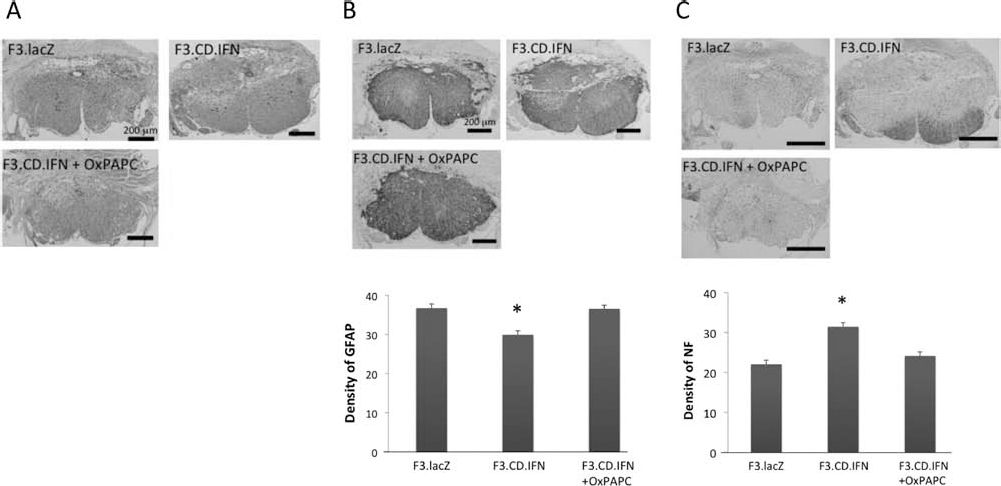
The expression of GFAP and NF at the injury site was compared in mice treated with F3.CD.IFN in the presence or absence of the TLR4 inhibitor OxPAPC. While the dorsal half of the spinal cord was destroyed equally in every group, the anterior half of the spinal cord was found to be markedly preserved in the F3.CD.IFN group when compared to the F3.CD.IFN + OxPAPC group (A). The intensity of GFAP staining was also significantly decreased in the F3.CD.IFN group compared to that observed in the F3.CD.IFN + OxPAPC andF3.LacZ groups (B). NF-stained transverse sections also demonstrated a significant loss of neural fibers in the F3.CD.IFN + OxPAPC and F3.LacZ groups, while neural fiber was dramatically preserved in sections from the F3.CD.IFN group (C), *p < 0.05.
Behavioral Studies
To determine whether F3.CD.IFN administration improved motor function after SCI, we assessed functional recovery using two independent behavioral tests: the BMS and the inclined plane test. Animals were evaluated for locomotor recovery at 1, 2, 3, 4, 6, and 8 weeks after injury.
One week after SCI, on the day of PBS or NSC administration, all injured animals exhibited a mean BMS score of <3, indicating extensive ankle movement without stepping ability. Animals with SCI receiving only PBS (PBS group) or F3.LacZ.NSCs (F3.LacZ group) spontaneously recovered to a BMS score of 3–4 (plantar placing of the paw or occasional plantar stepping) by 4 weeks after injury, and further recovery was limited. Animals treated with F3.CD.IFN followed by 5-FC administration (F3.CD.IFN + 5-FC group) had slightly better outcomes. In contrast, mice injected with F3.CD.IFN in the absence of 5-FC (F3.CD.IFN group) showed significant improvement in BMS scores compared to the other three groups, whereby the F3.CD.IFN group exhibited a mean BMS of 4.9±0.5 at 4 weeks, and the PBS, F3.LacZ, and F3.CD.IFN + 5-FC groups attained mean scores of 3.7±0.4, 3.6±0.5, and 4.0±0.5, respectively. Differences between the mean BMS scores of the F3.CD.IFN group and the other groups were statistically significant 4 weeks after injury and thereafter (p < 0.05) (Fig. 6A).
The inclined plane test was performed to examine additional post-SCI locomotor function. There was significant recovery of performance 2 weeks following the injury in the F3.CD.IFN group, whereas the performance of the other three groups remained severely impaired (p < 0.05). None of the groups recovered to preinjury levels of performance (Fig. 6B).
Finally, the improvements in the BMS scores and inclined plane test results that were observed in the F3.CD. IFN group were shown to be inhibited by the OxPAPC TLR4 inhibitor (Fig. 6C, D).

Behavioral studies. (A) Basso mouse scale (BMS) for locomotion. We measured the recovery of hindlimb motor function in seven mice in each treatment group. Animals were evaluated for locomotor recovery at 1, 2, 3, 4, 6, and 8 weeks after the injury. F3.CD. IFN group showed a significant improvement in BMS scores compared to the other three groups. Differences between the mean BMS of the F3.CD.IFN group and of the other groups were statistically significant 4 weeks after injury and thereafter (*p < 0.05). (B) Inclined plane test. It demonstrates significant recovery of performance 2 weeks following the injury in the F3.CD.IFN group, whereas performance of the other three groups remained severely impaired (*p < 0.05). (C, D) The improvements in the BMS scores and inclined plane test results that were observed in the F3.CD.IFN group were shown to be inhibited by the OxPAPC TLR4 inhibitor. As a result, the F3.CD. IFN group made a significant recovery compared to the other two groups 4 weeks after the injury in the BMS score. Similarly, the inclined plane test demonstrated marked improvement in the F3.CD.IFN group 8 weeks after the injury compared to the other two groups.
Electrophysiology
The mean motor nerve evoked-action potentials (MEP) amplitude observed in the sham operation group was approximately 600 μV at both 4 and 8 weeks after injury. This amplitude was reduced to a range of approximately 150–200 μV in the PBS, F3.LacZ, and F3.CD.LacZ + 5-FC groups at 4 and 8 weeks, while animals in the F3.CD.IFN group showed significantly better recovery, to approximately 300 μV at both 4 and 8 weeks after injury (p < 0.05) (Fig. 7A).
However, OxPAPC administration in the F3.CD.IFN group significantly worsened the mean MEP amplitude to 178 μV and 228 μV at 4 and 8 weeks, respectively (p < 0.05) (Fig. 7B).

Electrophysiology. (A) To measure signal conduction in motor pathways after SCI, transcranial electrical motor nerve evoked-action potentials (MEPs) were measured at 4 and 8 weeks after injury. Animals in the F3.CD.IFN group showed significantly better recovery at both 4 and 8 weeks after injury (*p < 0.05). (B) OxPAPC administration in the F3.CD.IFN group significantly worsened the mean MEP amplitude to 178 μV and 228 μV at 4 and 8 weeks, respectively (*p < 0.05).
Assessment of Regenerative Sprouting of CST Axons by 7-T MRI
Four weeks after dorsal hemisection, in vivo proton DTI was performed for noninvasive evaluation of regenerative axonal sprouting (Fig. 8). We visualized the diffusion tensor of the thoracic spinal cord. The eigenvectors associated with the tract were depicted as colors according to their orientation: blue illustrates a longitudinal (superior–inferior) orientation. The continuity of this longitudinal tract demonstrated functional regeneration of the axons. DTI was performed 4 weeks after the injury in mice that received PBS, F3.LacZ, or F3.CD.IFN. In the PBS and F3.LacZ groups, the injury site and its surrounding area displayed high signal intensity in T2-weighted sagittal sections, indicating spinal cord edema or necrotic tissue alteration, whereas this high intensity was not detected in the F3.CD.IFN group. The DTI of the injured spinal cord in the PBS and F3.LacZ groups revealed discontinuity of longitudinal fibers, which were mostly visible in the F3.CD.IFN group.
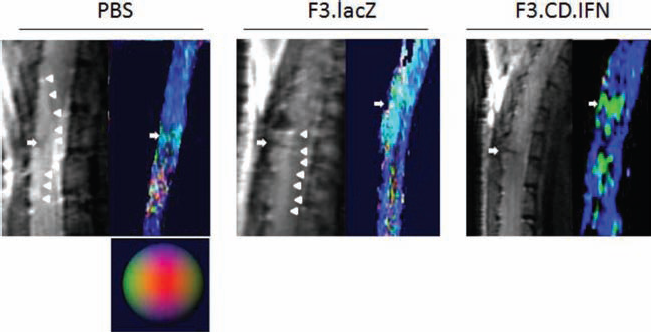
Assessment of regenerative sprouting of CST axons by 7-T MRI. Four weeks after dorsal hemisection. Magnetic resonance imaging [MRI; diffusion tensor imaging (DTI)] was performed in mice receiving PBS, F3.LacZ, or F3.CD.IFN. Diffusion tensor of the thoracic spinal cord was visualized. The eigenvectors associated with the tract were depicted as colors according to its orientation; blue illustrates a longitudinal (superior–inferior) orientation. Continuity of this longitudinal tract demonstrated functional regeneration of the axons. In the PBS and F3.LacZ groups, the injury site (arrow) and its surrounding area display high intensity in T2-weighted sagittal sections (arrowheads), indicating spinal cord edema or necrotic tissue alteration, whereas high intensity is not detected in the F3.CD.IFN group. DTI of the injured spinal cord in the PBS and F3.LacZ groups reveals discontinuity of longitudinal fibers, while these fibers were mostly visible in the F3.CD.IFN group. CST, corticospinal tract.
Discussion
In the present study, we transplanted intravenously human NSCs transduced with IFN-β gene in SCI mice, and the animals exhibited extensive suppression of glial scar formation and preservation and/or regeneration of nerve fibers in the lesioned spinal cord. Significant neurobehavioral and electrophysiological recovery was attained, as measured by the BMS for locomotion, inclined plane test, and transcranial MEPs. Evaluation with 7.0-T MRI also confirmed axonal regeneration. Systematic evaluation of spinal cord 10 days after SCI with gliosis-specific microdissection, genome-wide microarray, and MetaCore pathway analysis revealed the upregulation of TLR4 in the spinal cord lesion site. We therefore focused on the functional role of TLR4 signaling in reactive astrocytes, as there is a strong relationship between TLR4 and IFN-β in glia (8). TLR4 in the presence of IFN-β was shown to suppress the SCI-induced proliferation of reactive astrocytes within lesion site. Furthermore, as mice that were administered with IFN-β-expressing NSCs and TLR4 inhibitor simultaneously gained less functional recovery than mice in which only IFN-β-expressing NSCs were injected, we have verified that TLR4 plays an extremely important role in the effects of IFN-β during astrocytic gliosis. These results suggest that IFN-β delivery by IV injection of genetically engineered NSCs is capable of inhibiting glial scar formation after SCI and promotes functional recovery in the presence of TLR4.
Benefit of the Utilization of NSCs in the Treatment of SCI
Three different delivery methods can be considered for SCI cell therapy: intralesional, intrathecal, and IV. A cardinal feature of NSCs is their exceptional migratory ability, which has led to their emergence as a therapeutic paradigm in various animal models of neurodegeneration, stroke, and brain tumors (24,26,29,47). Previously, we and others have demonstrated that the transplantation of genetically engineered F3 human NSCs into sites of SCI lesions induces functional improvements in rat SCI models (10,18,19,27). We have also demonstrated that there is glial and neuronal differentiation of NSCs, indicating that IV NSC delivery activates the microenvironment of the injured spinal cord to stimulate self-regeneration (44). Thus, IV injection of human NSCs is a promising tool for replacing depleted cells or for delivering therapeutic genes in the treatment of SCI, with the potential to be used in clinical applications. We have also demonstrated that intravenously injected F3 human NSCs migrate preferentially to the SCI site (44). As a novel strategy for SCI treatment, we combined these two studiesroles of reactive astrocytes and examined whether functional recovery and axonal regeneration could be achieved through the intravenous administration of IFN-β-expressing NSCs. F3 NSC is a clonally isolated, multipotent human neural stem cell line that has the ability to self-renew, stably express genes, and differentiate into neurons, astrocytes, and oligodendrocytes (23). F3 NSCs have previously been used in a clinical trial for gene therapy in glioma patients (a pilot feasibility study of oral 5-FC and genetically modified neural stem cells expressing E. Coli CD for treatment of recurrent high-grade glioma—City of Hope National Medical Center, Duarte, CA, USA, June 2010; NCT01172964.) Inhibition of glial scar formation is a promising approach for the treatment of SCI. Recent studies have focused on the multifaceted roles of astrocytes in response to SCI.
It has previously been demonstrated that selective depletion or ablation of a subpopulation of reactive astrocytes causes widespread tissue disruption and pronounced cellular degeneration (6). Another study has reported that the emergence and migration of reactive astrocytes are beneficial in limiting inflammatory cell infiltration in the subacute phase of injury, before glial scar completion (34), while others have indicated that the elimination of reactive astrocytes is beneficial for SCI recovery at the subacute stage (38). Considering the above-described contradictory roles of reactive astrocytes after SCI, an ideal treatment strategy may involve inhibiting the aberrant development of hypertrophic reactive astrocytes without eliminating normal quiescent astrocytes, thus, preventing a cascading wave of uncontrolled tissue damage. Based on our findings, the timing of NSC transplantation is extremely important. On the basis of our previous studies regarding glial scar formation and effective NSC migration (44), we have determined that the optimal timing of IV NSC injection is 7 days after injury.
Critical Role of TLR4 in the Effect of NSCs Expressing IFN-β
We have previously been successful at restoring functional recovery in a mouse model of SCI by using a liposome-mediated IFN-β gene delivery method (11). IFN-β is a type I IFN that exerts pleiotropic biological effects (3) and acts as a cell cycle regulator to control the reentrance of cells undergoing aberrant cell cycle progression into a senescence-like state (16). Recently, it has been proven that IFNs block the constitutive activation of the MEK-ERK signaling pathway (32,39,40,48), which is activated at the site of SCI (11). IFN-β also blocks the infiltration of neutrophils and proinflammatory cytokines into sites of injury and stimulates the expression of anti-inflammatory cytokines (7). In order to clarify the novel function of IFN-β in the suppression of glial scar formation in this study, we attempted to identify the profile changes in gene expression that occur during astrocytic gliosis. This systematic processing revealed the statistically significant activation of many intriguing pathways (p < 0.05) (Fig. 1). Of these, the TLR4 pathway, which plays an important role in immune regulation in astrocytes (8) and autoregulatory apoptosis in cells bearing TLR4 (14,17) was of particular note. TLR4 can be detected in astrocytes in sites of SCI (17). The autoregulatory apoptosis of activated astrocytes is known to be initiated by TLR4 and proceeds by at least two pathways, one of which is IFN-β activation (14). Indeed, TLR4 ligation is essential for the autoregulatory apoptosis of cells bearing this receptor, which ultimately acts to regulate glial scar formation (17). This function requires the presence of IFN-β. Furthermore, TLR4 deletion significantly impairs the normal progression of SCI repair and functional recovery (17). Our results demonstrate that TLR4 inhibition results in impaired functional recovery despite the administration of F3.CD.IFN NSCs.
Therefore, we can conclude from our findings that the combination of TLR4 and IFN-β plays a crucial role in regulating post-SCI inflammation and gliosis.
Cytosine Deaminase Gene as a Fail-Safe Modulator of Uncontrolled Proliferation of NSCs
The CD gene examined in this study encodes bacterial enzyme that catalyzes the deamination of nontoxic 5-FC into the highly toxic 5-FU. We have previously reported improved outcomes in animal models of brain tumors after therapy with this “suicide gene” (12,13,21,41). We, therefore, applied this strategy in SCI models to investigate the additive efficacy of NSC-delivered CD and IFN-β. NSCs have the ability to migrate to sites of SCI, at which they induce the apoptosis of the surrounding reactive astrocytes via the bystander effect. Thus, we anticipated that the combination of the actions of IFN-β with the 5-FC/CD-mediated bystander effect would exert greater suppression of reactive astrocytes than either treatment alone. However, this combination therapy was not found to be superior to IFN-β alone in vivo or in vitro in this study. Rather, our findings show that the addition of 5-FC to astrocytes treated with F3.CD.IFN did not cause any greater decrease in astrocyte number than treatment with F3.IFN alone. We speculate that sustained expression of IFN-β from F3.CD.IFN is required to produce an inhibitory effect in reactive astrocytes. Further, as NSCs have neuroprotective functions, including the secretion of neurotrophic factors, the presence of NSCs, themselves, may provide a partial explanation for functional recovery and neuroregeneration after SCI. The F3 NSC line can produce an unlimited number of differentiated cells in vitro and in vivo and could therefore be used in transplantation. However, serious problems could arise when transplanted NSCs proliferate uncontrollably. In order to avoid such problems, we generated an F3 human NSC line expressing both the therapeutic IFN-β gene and the CD suicide gene. The expression of the CD gene provides a fail-safe guard to allow removal of cells in cases of uncontrolled proliferation of grafted cells.
Noninvasive Monitoring of Axonal Regeneration by Diffusion Tensor Imaging
The evaluation of axonal fibers is important to assess the severity of SCI and the efficacy of treatment; however, conventional methods such as tracer injection (e.g., biotinylated dextraamine) in brain parenchyma are technically demanding and highly invasive (28,36). Because histological examinations are required to evaluate tracer studies, it has been impossible to evaluate axonal fibers in living animals and follow the sequential growth of these axonal fibers in the same animal. In order to evaluate axonal fibers in vivo, we sought a novel method capable of noninvasive evaluation in clinical applications. With this in mind, we determined that MRI is suited for use in the assessment of the state of SCIs. Conventional T1- and T2-weighted MRI of the spinal cord only shows ambiguous images of the various spinal structures, making it difficult to identify the complicated array of directionally oriented nerve fibers. However, DTI is one of the most versatile MRI modalities for longitudinal evaluations of CNS disorders and is capable of following the orientation of nerve fibers and tracing specific neural pathways such as the CST (20,30,33). In this study, we chose a dorsal transection model because the disruption and regeneration of axons after a transection injury can be more easily evaluated than that following a contusion injury (31,46). Because of the ability to visualize longitudinal axonal tracts in variously oriented neural fibers, DTI has tremendous potential for use in the diagnosis and evaluation of spinal cord diseases. Several researchers have reported successful DTI in the human spinal cord (4,5). To our knowledge, this is the first report of DTI-based visualization of axonal regeneration in the spinal cord of small rodents. Significant longitudinal axonal regeneration was confirmed by DTI in F3.CD.IFN-β NSC-treated mice 4 weeks after injury.
Conclusions
In conclusion, mice that received IV administration of genetically engineered NSCs transduced with IFN-β gene after SCI exhibited extensive suppression of glial scar formation in the lesioned spinal cord. This effect was shown to require the expression of both TLR4 and IFN-β. Significant neurobehavioral and electrophysiological recovery was attained, as measured by the BMS for locomotion, inclined plane test, and transcranial MEPs. Axonal regeneration was also visualized noninvasively using DTI. Our results suggest that IFN-β delivery by IV injection of genetically engineered NSCs inhibits glial scar formation after SCI and promotes functional recovery through the functions of TLR4. This novel NSC-based therapy of delivering IFN-β shows promise for the treatment of patients suffering from SCI.
Footnotes
Acknowledgments
This work was supported, in part, by grants from a Grant-in-Aid (B) for Scientific Research from the Ministry of Health, Labor, and Welfare, Japan (A.N.). The authors thank Dr. S. Saito, Ms. S. Shibata, Ms. C. Kajiwara, and Ms. Miho Senda for their help. The authors declare no conflict of interest.
