Abstract
Neonatal asphyxia is an important contributor to cerebral palsy (CP), for which there is no effective treatment to date. The administration of human cord blood cells (hUCBCs) is emerging as a therapeutic strategy for the treatment of neurological disorders. However, there are few studies on the application of hUCBCs to the treatment of neonatal ischemia as a model of CP. Experiments and behavioral tests (mainly motor tests) performed on neonatal hypoxia/ischemia have been limited to short-term effects of hUCBCs, but mechanisms of action have not been investigated. We performed a study on the use of hUCBCs in a rat model of neonatal hypoxia/ischemia and investigated the underlying mechanism for therapeutic benefits of hUCBC treatment. hUCBCs were intravenously transplanted into a rat model of neonatal hypoxia ischemia. hUCBCs increased microglia temporarily in the periventricular striatum in the early phase of disease, protected mature neurons in the neocortex from injury, paved the way for the near-normalization of brain damage in the subventricular zone (SVZ), and, in consequence, significantly improved performance in a battery of behavioral tests compared to the vehicle-treated group. Although the transplanted cells were rarely observed in the brain 3 weeks after transplantation, the effects of the improved behavioral functions persisted. Our preclinical findings suggest that the long-lasting positive influence of hUCBCs is derived from paracrine effects of hUCBCs that stimulate recovery in the injured brain and protect against further brain damage.
Keywords
Introduction
Cerebral palsy (CP) is estimated to occur in 2.0–2.5 per 1,000 live births worldwide (43). The term “cerebral palsy” refers to a group of permanent disorders in the development of movement and posture, symptomatic complex, and nonprogressive conditions (24). Children with CP develop spastic paresis, ataxia, disorders of sensory motor coordination, and cognitive impairments (35). The etiology of CP is diverse, and diagnosis is limited. Perinatal infections or inflammation and hypoxia/ischemic insults are frequently associated with preterm and term CP (19,36,59). Despite advances in child care, there is an increase in the incidence of CP, which may be due to an increased survival rate of infants. To date, there are no effective therapies for CP (38).
Human umbilical cord blood cells (hUCBCs) have many advantages over embryonic stem cells in that they are an abundant source of stem and progenitor cells and they are easily obtainable and reliable; furthermore, they do not raise ethical questions and there is no concern about tumor formation. Therefore, the transplantation of hUCBCs is highly attractive for regenerative medicine (3,14,29,37,48). There are several reports that hUCBCs treatment lessens neurological dysfunction in a rat model of neonatal cerebral ischemia and stroke. hUCBCs were initially investigated as a therapy in models of adult stroke (12). Subsequently, hUCBCs were developed for testing their therapeutic effect on CP (31,42). Although there are many studies showing the effect of hUCBCs on the adult stroke model, there is relatively little research on the neonatal ischemia model. Furthermore, the mechanisms by which transplanted hUCBCs are effective on both stroke and CP are largely unknown.
The purpose of the present study is to (1) accurately measure functional efficacy of hUCBC transplantation and (2) investigate mechanisms of therapeutic effect by hUCBC transplantation. We performed an extensive battery of behavioral tests, including motor performance, learning and memory, and anxiety tests, to measure the integrative functional recovery in a rat model of neonatal hypoxia/ischemia, a possible rodent model of CP. To elucidate the underlying mechanisms by which the therapeutic effects of hUCBCs are produced, systematic in vitro analyses of secreted factors from hUCBCs were measured. Furthermore, in vivo assays were conducted to systematically investigate the effects of hUCBCs on the stimulation of endogenous stem cells and the induction of chemokines.
Here, our findings demonstrate that the hUCBC-treated group exhibits (1) functional recovery in a battery of behavioral tests; (2) an increase in microglia in the periventricular striatum at 1 week after transplantation, probably due to the enhanced chemokine effects by hUCBCs; (3) protection of mature neurons in neocortex; and (4) reduction of brain damage in the subventricular zone (SVZ). We also show that chemokines secreted from hUCBCs may direct SVZ cells toward the injured area in the early stages of disease.
Materials and Methods
Surgical Procedure in a Rat Model of Neonatal Ischemia and hUCBCs Transplantation
All animal procedures were approved by the CHA University Institutional Animal Care and Use Committee (IACUC) in accordance with the Guide for the Care and Use of Laboratory Animals. On postnatal day 7, Sprague–Dawley (SD) rat pups (P7, n = 60) were anesthetized with inhaled isoflurane (Hana Pharm. Co., Ltd.; induction 5%, maintenance 1.5%) and subjected to the unilateral ligation of the left common carotid artery followed by exposure to the hypoxia environment (8% O2, 92% N2 at 37°C) for 90 min with the intention of damaging the left hemisphere. Anesthetized rat pups received hUCBCs (n = 24, 1 × 107/200 μl) or phosphate-buffered saline (n = 24, PBS 200 μl) through the external jugular vein 24 h after hypoxia/ischemia insult (52). hUCBCs and vehicle groups were treated daily with cyclosporine A (10 mg/kg IP, Chong kun dang Pharm., Seoul, Korea) as an immunosuppressant for a month, and afterwards half of the recommended dose (5 mg/kg IP) was used until they were sacrificed. Sham group underwent surgical procedure without the unilateral ligation of the common carotid artery and exposure to the hypoxic environment.
Preparation and Intravenous Transplantation of hUCBCs
hUCBCs were obtained ex utero using sterile syringes containing anticoagulant following term births of healthy donors. Ten samples from 10 donors were collected after obtaining written informed consent documents approved by the CHA University, Bundang CHA Hospital IRB (Institutional Review Board). hUCBCs obtained from each patient were delivered as a pack. Each pack of hUCBCs contained approximately 50–150 ml. Anticoagulant-treated hUCBCs were processed within 24 h after collection. For preparation of mononuclear cells (MNCs), hUCBCs were separated on Ficoll-Paque density gradient solution (Amersham Biosciences) and centrifuged at 400 x g for 40 min. About 1 × 108 of the MNCs were collected per pack and washed twice with PBS/ EDTA. Rats subjected to hypoxia/ischemia insult were randomly divided into either a cell treatment group or a PBS treatment group. Using an insulin syringe, hUCBCs (1 × 107 MNCs/200 μl) or PBS (vehicle, 200 μl) were transplanted into the external jugular vein 1 day after the induction of neonatal ischemia.
Conditioned Medium (CM) Preparation in the Human UCBC-Isolated MNCs
In order to quantify cytokines and chemokines secreted by MNCs from hUCBCs, MNCs were isolated from hUCBCs using Ficoll-Paque density gradient solution (Amersham Biosciences). Conditioned medium (CM-MNC) was obtained from MNCs cultured for 3 days under serum-free culture conditions using insulin–transferrin–selenium (ITS) supplemented Dulbecco's modified Eagle's medium (DMEM). The CM was filtered at 0.42 μm and kept at −70°C until use.
MRI Analysis
The 4.7 T preclinical magnetic resonance imaging (MRI) instrument (BioSpec 47/40, Bruker BioSpin, Ettlingen, Germany) has a 40-cm horizontal bore magnet interfaced to an AVANCE console and equipped with a 12-cm gradient set (BGA-12), capable of providing 720 mT/m with a slew rate of 6,000 T/m/s. A birdcage coil that had a 70-mm inner diameter was used to transmit and receive the radio frequency (RF) signal. Scout images were acquired in three planes with a gradient-echo sequence to determine the appropriate positioning for the study. A fast spin-echo, rapid acquisition with relaxation enhancement (RARE) sequence was used for the MR images. A sequence with a 256 × 256 matrix was obtained with the following parameters: effective echo time = 90 ms, repetition time = 5,000 ms, echo train length = 8, field of view = 2 × 2 cm2, number of averages = 2. Whole-brain coverage was obtained with 1-mm-thick axial slices with no gap. Each animal was anesthetized with isoflurane (5% induction, 1.5% maintenance) in mixed N2O and O2 (7:3). The rectal temperature was monitored, and temperature was maintained with a warm air blower.
Evaluation of Disruption of Blood–Brain Barrier (BBB)
A 2% solution of Evans blue (Sigma Aldrich; 4 ml/kg) was injected into the external jugular vein of animals (n = 3 per group) 1 day after insult to confirm the disruption of BBB. Animals were perfused with PBS, and the brains were extracted from the skull 1 h after systemic injection of Evans Blue to identify the leakage of the dye.
Human Fetal Neural Progenitor Cells Culture and CM Preparation
Human neural precursor cells (NPCs) were generated from human ventral midbrain tissue from naturally aborted 14-week-old fetus with the mother's consent for the purpose of comparing cytokines/chemokines released by hUCBCs. Samples were harvested and supplied by Bundang CHA Hospital according to NIH and local IRB guidelines. All experiments were approved by the ethics committee of CHA University/Hospital. Human fetal brain NPCs were cultured as described previously (49). The conditioned media (CM-NPC) were collected from NPCs cultured during 3 days of differentiation without medium changes using ITS medium. The CM was filtered at 0.42 μm and kept at −70°C until use.
Histopathology, Immunohistochemistry, and Microscopy
Animals were sacrificed under pentobarbital (50 mg/ kg IP) anesthesia and perfused with phosphate-buffered saline and 4% formaldehyde through the left ventricle at 1 and 10 weeks after transplantation (1 week, n = 4 per group; 10 weeks, n = 4 per group). However, human nuclei (HN) immunoreactivity was measured at 1, 3, and 10 weeks. To process for immunohistochemistry, nonspecific binding was blocked with 10% normal goat serum in 0.3% Triton X-100 (USB Corporation) and PBS for 1 h. The 30-μm cryosections from whole-brain blocks were incubated overnight at 4°C with the primary antibody. The following primary antibodies were used: ionized calcium-binding adaptor molecule 1 (Iba-1; 1:1,000, Wako Chemicals), doublecortin (DCX; 1:200, Cell Signaling), Nestin (1:200, Abcam), human nuclei (1:200, Chemicon), neuronal nuclei (NeuN; 1:1,000, Chemicon), growth-regulated oncogene-α (GRO-α; 1:200, Abcam), and macrophage inflammatory protein-1α (MIP-1α; 1:200, Nobus Biologicals). Sections were then incubated with the appropriate secondary antibody conjugated with Alexa Fluor 488 (1:400, Invitrogen) and Alexa Fluor 594 (1:400, Invitrogen) for 1 h at room temperature. Fluorescence was detected, analyzed, and photographed with Zeiss LSM laser scanning confocal photomicroscope (Carl Zeiss). Every 20th section of each brain was analyzed to count immunoreactive cells, and total number of immunoreactive cells were estimated by means of Abercrombie's correction (1).
The tissue loss volume induced by insult was determined in a series of 30-μm-thick brain sections stained with hematoxylin and eosin (10 weeks, n = 10 per group). Using indirect method, the tissue loss volume was expressed as a volume percentage of lysed ipsilateral hemisphere compared to the intact contralateral hemisphere (12).
Assay of Human Growth Factors, Cytokines, and Chemokines
To investigate profiles of cytokines and chemokines in CM-NPC and CM-MNC, CM were analyzed with a Milliplex® assay (human cytokine/chemokine panel I kit; Millipore Corp.) using Luminex 200 (Luminex, TX, USA), which allows the simultaneous quantification of 42 human cytokines/chemokines (detailed information in Table 1). ITS medium was used as a control.
Levels of Cytokines in Secretome Analysis of hUCBCs and NPCs

In the in vitro secretome analysis, large amounts of IL-6, IL-8, IP-10, MCP-1, RANTES, MCP-3, GRO-α, and G-CSF were predominantly detected in human umbilical cord blood cells (hUCBCs) compared to human fetal neural progenitor cells (NPCs) and control (ITS medium). ∗Higher expression in hUCBCs than other groups. IL, interleukin; CCL, chemokine (C-C motif) ligand; CXCL, chemokine (C-X-C motif) ligand; IP-10, interferon gamma-induced protein 10; MCP, monocyte chemotactic protein; MIP, macrophage inflammatory protein; RANTES, regulated upon activation, normal T-cell expressed and secreted; GRO, growth-regulated oncogene; MDC, macrophage-derived chemokine; sCD40L, soluble CD40 ligand; sIL-2Ra, soluble IL-2 receptor a; EGF, epidermal growth factor; FGF, fibroblast growth factor; Flt-3, fms-like tyrosine kinase-3; G-CSF, granulocyte-colony stimulating factor; GM-CSF, granulocyte-macrophage stimulating factor; PDGF, platelet-derived growth factor; TGF, transforming growth factor; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.
RNA Isolation
At 1 and 7 days after transplantation, rats were sacrificed under pentobarbital (50 mg/kg IP) anesthesia, and brains were extracted (n = 3 in each group at 1 day; n = 3 in each group at 1 week). cDNA obtained from only the ipsilateral hemisphere was collected for analyzing the expression of chemokine-related genes by RT-PCR using the primers shown in Table 2. Total RNA was isolated from homogenates of each ipsilateral hemisphere brain (damaged) using TRI reagent (Invitrogen). cDNA was synthesized from 1 ug of total RNA using Superscript II reverse transcriptase kit (Invitrogen), oligo-d(T)12-18 primer (Invitrogen), DTT, and dNTPs according to the manufacture's recommended protocol. Denaturation of RNA was performed at 65°C for 5 min and the reverse transcriptase reaction at 42°C for 60 min, then at 70°C for 10 min. For the amplification of cDNA generated by the reverse transcriptase reaction, PCR reaction (Bioneer) was carried out with chemokine and GAPDH primer under the following conditions: 95°C for 30 s, 60°C for 30 s, and 72°C for 40 s with 28–30 cycle number. RT-PCR band intensity was measured via Image J program (National Institutes of Health) to detect the extent of mRNA expression. Relative ratio between the expression levels of chemokines and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was calculated, and a group comparison analysis was carried out.
Primers Used for Semiquantitative RT-PCR

Behavioral Tests
Forty Sprague–Dawley rats (4–8 weeks, n = 15 for the hUCBC group, n = 15 for the vehicle group, and n = 10 for sham group) were used for behavioral tests. Since neonatal hypoxic/ischemic injury results in the dysfunction of numerous brain regions, a battery of behavioral tasks were administered to each rat. Each behavioral task was administered at various time points over 10 weeks. Briefly, the cylinder test for motor function (11) was carried out between 3 and 6 weeks after transplantation. To measure associative learning and memory, passive avoidance was performed 10 weeks posttransplantation. Anxiety tests such as light/dark exploration and forced swim test (16) were performed from 8 to 10 weeks after transplantation. Rats were acclimated in the experiment room at least 1 h prior to testing. Rats were euthanized for immunohistochemistry at 1 and 10 weeks. All the behavioral assays were performed and coded with the double blinded design.
Motor Tasks
Basic Motor Test
First of all, basic motor function was examined prior to a battery of behavioral tests at 5 weeks after transplantation, using tendency to avoid a brightly lit area. The apparatus was equipped to reduce confounding anxiety behaviors or other unnecessary behaviors prior to entering the dark room. Rats were placed in a small acrylic start box (3 × 5 × 12 cm) and allowed to explore for 30 s. After 30 s, the guillotine door, which separate two compartments, was lifted. The latency to move into dark compartment through a narrow opening was recorded.
Cylinder Test
Each rat was placed in a transparent cylinder, and the rat was observed for 5 min to evaluate somatosensory motor function. The number of times the forelimb touched the Plexiglas wall during full rearing was recorded for 5 min. The following parameters were checked: (1) use of forelimb simultaneously, (2) use of forelimb independently, and (3) an asymmetry use of forelimb to contact with wall. The results are presented as a proportion of the use of impaired forelimb relative to total forelimb use.
Anxiety or Depression Tasks
Forced Swim Test
Forced swim test is an effective tool for assessing the neurobiology of anxiety in an animal disease model. A cylinder was filled with water to a depth that did not allow animals to touch the bottom with their tails. During 2 min of total 6-min test, rats were allowed to swim freely in a transparent cylinder (diameter 20 cm, height 40 cm) containing ~23°C water in the 30-cm-deep cylinder. For the remaining 4 min, the time spent making the minimum movements necessary to keep the rat's head above water was measured. After the test, the rats were dried with the paper towel and placed on a warm pad. All tasks were recorded by a camera above the cylinder.
Light/Dark Exploration
This test was conducted to test the level of anxiety using the ethological conflict between the tendencies of rodent to explore a novel environment and to avoid a brightly lit, open area. Each rat was placed in a lit room (60 W white bulb, 35 cm above the floor, 21 × 21 × 21 cm), facing away, and allowed to explore the compartment for 30 s. After 30 s, a guillotine door was lifted to make a small opening in the dividing compartment. The time to stay in the light room and the number of transitions between the lit and dark compartments were measured for 5 min.
Passive Avoidance
The passive avoidance test is conducted using the Avoidance system (JEUNG DO Bio & Plant CO., Ltd.), which consists of a two-compartment chamber (each side 21 × 21 × 21 cm) separated by a compartment. The dark chamber of the two chambers was equipped with a scrambled electric shock generator. Each rat was placed individually in the light compartment of the two compartments and the lid was closed. The trial began with a 30-s adaptation period in which the rat was allowed to explore the compartment. After 30 s, the guillotine door was opened. The gate remained open until the animal entered the dark chamber completely. In the first trial, the rat was subjected to an electric shock (1 mA) upon entering the dark chamber. On day 2, to test retention, the rat was tested using the same parameters except that the training shock was not used. The retention tests show how well the rat “remembers” the shock in the dark compartment. The latency to enter the shock room was measured.
Statistics
Statistical analyses were conducted on a CHA University mainframe computer using the Statistical Analysis System (SAS; SAS Korea, Inc., Seoul, South Korea), version Enterprise 4.0.
Basic motor function analysis, brain volume assay, and data of image intensity were analyzed using either t test or two-way variance analysis followed by LSD (Fisher's least significant difference) post hoc tests. All other measures were analyzed using a mixed model analysis of variance procedure (SAS, PROC MIXED) to account for the random effect of rats. The correlation between the cylinder test and NeuN staining was analyzed using the Pearson correlation coefficient.
Results
Typical Features of Neonatal Ischemia Rat Model
Survival rate of this animal model and transplantation was 80%. After transplantation, there was no difference in survival rate between hUCBC group and vehicle group until 10 weeks.
Confirmation of Infarct Area by MRI
MRI coronal images of rat brain at 1 day after hypoxia ischemia revealed infarcted areas (a relatively bright region compared to surrounding) in both ipsilateral and contralateral regions. The affected areas included neocortex, striatum, thalamus, and hippocampus. Only neocortex regions were infarcted on the side contralateral to the injury. The severity of infarction was decreased from anterior to posterior brain (the most anterior part of MRI brain images is presented in the lowest, rightmost panel, Fig. 1A).

Typical features of a rat model of neonatal ischemia. (A) Transverse representative magnetic resonance (MR) images of a rat brain acquired 1 day following hypoxia/ischemia (HI) show high signal intensity, indicative of infarction, extended into the contralateral neocortex from the affected hemisphere. (B) Evans Blue dye was injected intravenously at 1 day after insult. Extravasation of Evans Blue into brain parenchyma showing hypoxia/ischemia-induced disruption of blood–brain barrier (BBB) was observed only in HI. (C) Hematoxylin and eosin stain (H&E) images of brain at 10 weeks after HI show the most affected areas of the brain were the striatum, neocortex, and hippocampus. Abnormal enlargement of ventricle was seen (asterisks). (D) There is no significant difference in the volume of the tissue loss between the two groups.
Disruption of BBB
In contrast to the adult brain, the P7 rat brain has an immature BBB in terms of permeability, and insults can easily disrupt the BBB (34).
Extravasation of Evans Blue dye into the brain parenchymal was observed in three of the three insulted brains. The most Evans Blue-visible area was superficial cortex, indicating severe vascular damage (Fig. 1B).
Tissue Loss Induced by Hypoxia Ischemia Insults
As seen in representative images of H&E analysis (Fig. 1C), tissue loss was observed in striatum, neocortex, and hippocampus. Ventromegaly (asterisks) was seen in both groups. The results showed selective vulnerability of brain tissue in response to hypoxia/ischemia damage. No significant difference was seen in the proportion of tissue loss between hUCBC-treated (n = 10) and vehicle-treated (n = 10) groups (Fig. 1C, D).
Presence of hUCBCs in Ipsilateral Area around Ventricle
The typical features of neonatal ischemia rat model were examined. Infarct area was confirmed by MRI (Fig. 1A), and disruption of the BBB was observed using Evans Blue staining (Fig. 1B). The assay of tissue loss induced by hypoxic/ischemic insult showed the selective vulnerability of brain tissue in response to hypoxia/ischemia damage (Fig. 1C, D). For the identification of transplanted hUCBCs in brain, human nuclei (HN) antibody immunoreactivity was examined in the periventricular region. Many HN-positive cells costaining with Nestin or DCX were found 1 week after transplantation in the periventricular region on the side of the brain ipsilateral to the insult, but only in the hUCBC group (Fig. 2A, B). The presence of HN-positive cells decreased dramatically over time. At 3 and 10 weeks after treatment, very few HN-positive cells were seen in the same region (Fig. 2C, D).

Detection of transplanted cells in brain. (A) costain of Hu-nuclei and nestin; (B) costain of Hu-nuclei and DCX. Quite a few cells immunoreactive for Hu-nuclei (HU, red) were seen in the periventricular striatum at 1 week after transplantation. Some of the HU-positive cells were costained with nestin or doublecortin (DCX) (A, B). HU-positive cells were rarely detected in the periventricular striatum at 3 weeks (C) and 10 weeks (D) after transplantation. The white arrows point to the HU-positive cells costained with nestin or DCX. Scale bar: 20 μm.
Protective Effect by hUCBCs on Mature Neocortical Neurons
The loss of NeuN reactivity, a marker of mature neurons, following CNS damage has been reported (27,30,56). To determine the effects of hUCBCs on the damaged brain, NeuN immunoreactivity in cortex was analyzed by staining every 20th section of each whole brain. Immnohistochemistry results at 1 week showed that there was no apparent difference in NeuN-positive cells between hUCBC and vehicle groups (Fig. 3A–D) after transplantation. However, at 10 weeks after transplantation, significantly fewer NeuN-expressing cells were detected even in the contralateral region of the vehicle-treated group than in that of the hUCBC group, indicating that global hypoxia ischemia at postnatal day 7 affected the contralateral region [t(0.05, 6)) = 2.16, p < 0.05]. In the total number of NeuN on both sides, there was a significant difference between the two groups [t(0.05, 6) = 2.16, p < 0.05] (Fig. 3E–I). In comparison to the data at 1 week, NeuN-positive cells were significantly reduced in both groups at 10 weeks after transplantation compared to those at 1 week posttransplantation. Mature neurons in the cell-treated group marginally outnumbered those in the vehicle group in the neocortex ipsilateral to injury, to be exact, around somatosensory cortex of one section from a pool of 20 sections [t(0.05, 6) = 1.29, p > 0.1] (Fig. 3I). The maintenance of the higher NeuN expression for a longer period following hypoxia/ischemia in the cell treatment group than in the vehicle group suggests that hUCBCs protect mature neurons in the neocortex from progressive injury.

Protective effects of human umbilical cord blood cells (hUCBCs) on mature neurons damaged by HI insult. There is no significant difference in the number of neuronal nuclei (NeuN)-positive cells of neocortex between hUCBCs and vehicle groups at 1 week after transplantation (A–D). In comparison to the 1 week data, NeuN-positive cells were reduced in both groups at 10 weeks after transplantation. Despite the total reduction of mature cells, significantly more NeuN-expressing cells were found in the hUCBC group than in the vehicle group on both sides of neocortex [contralateral side: 1,008.75 ± 108.82 for cell group, 417 ± 115.74 for vehicle group, t(0.05, 6) = 2.16, p < 0.05; total number: 1,810.25 ± 290.01 for cell group, 790.5 ± 196.7 for vehicle group, t(0.05, 6) = 2.16, p < 0.05] (E–I). The difference in ipsilateral regions was not significant [801.5 ± 193.14 for cell group, 373.5 ± 96.24 for vehicle group, t(0.05, 6) = 1.29, p > 0.1]. Scale bar: 50 μm. ∗p < 0.05.
hUCBCs Triggered the Activity of Microglia in the Early Phase of Disease
To examine the effects of transplanted hUCBCs on the activity of microglia, immunoreactivity for Iba-1, a panmicroglial marker, was investigated in the periventricular striatum, which is vulnerable to insults during perinatal period (Fig. 4). There was a significantly higher number of Iba-1-positive cells in the ipsilateral region of the hUCBC group compared to that of the vehicle group at 1 week after transplantation (115.22 ± 17.84 for vehicle group vs. 152.50 ± 27.31 for hUCBC group, p < 0.05), but not significant group difference in contralateral region (80.22 ± 7.03 for vehicle group vs. 71.00 ± 3.79 for cell group, p > 0.2) (Fig. 4B–E, J, K). At 10 weeks after transplantation, the two groups showed no significant difference in the number of Iba-1-positive cells in both regions (Fig. 4F–K). The Iba-1 staining indicates that the transplantation of hUCBCs following hypoxic/ischemic insults on neonatal rat brain triggered increased activity of microglial cells in the striatum during the early phase of disease.

The effect of hUCBCs on the activity of microglia in the periventricular striatum. Representative section showing periventricular striatum (white dotted squares) where Iba-1 immunoreactivity was investigated (A) (Con: contralateral, Ipsil: ipsilateral). There was a significantly higher number of cells immunoreactive for ionized calcium-binding adaptor molecule-1 (Iba-1) in the ipsilateral region of the hUCBC group than in that of vehicle group at 1 week after transplantation (p < 0.05), but not in contralateral region (B–E, J, K). At 10 weeks after transplantation, there was no statistically significant group difference in the number of Iba-1-positive cells (F–K). Scale bars: 50 μm. ∗p < 0.05. ST, striatum; SE, septal nucleus.
Sparing of Endogenously Induced Progenitor Cells in Periventricular Region
The cell-treated group showed more DCX-detectable cells in the SVZ at 1 week after treatment. In the hUCBC-treated group, DCX-positive cells were spread and extended to the periventricular region. In contrast, the DCX cells in the vehicle-treated group were restricted to the SVZ. As in the 1-week data, there were more DCX-reactive cells in the SVZ at 10 weeks in the cell-treated group than in the vehicle group. Interestingly, in the cell-treated group, DCX-positive cells were detected even in the striatum and septal nucleus (Fig. 5B–I).

Representative section showing regions (white dotted squares) where DCX and nestin immunoreactivity was investigated (A) (Con.: contralateral, Ipsil: ipsilateral). A higher number of DCX-positive cells were found in subventricular zone (SVZ) of hUCBC group than of vehicle group both at 1 and 10 weeks after transplantation. In the cell treatment group, DCX-positive cells were spread to the periventricular region at 1 week (asterisks in C and G). At 10 weeks after transplantation, aggregates of DCX-positive cells were detected only in striatum of the hUCBC group (arrows in E and I). At 1 week, many nestin-expressing cells were spread over the affected striatum adjacent to ventricle of hUCBC group (asterisk in O) with rare detection of nestin in the same region of vehicle group. Nestin-positive cells were rarely seen in both groups at 10 weeks after transplantation (L, M, P, Q). LV, lateral ventricle; ST, striatum; SE, septal nucleus. Scale bars: 50 μm.
Also, more Nestin-positive cells were detected in the cell-treated group than in the vehicle group, especially in the ipsilateral region at 1 week after transplantation. In particular, similar to DCX results, Nestin-positive cells in the hUCBC group spread to the striatum, whereas the cells were restricted to the ventricular region in the vehicle group. Nestin-positive cells were not expressed at 10 weeks posttransplantation in either group (Fig. 5J–Q).
In Vitro Secretome Analyses
The secreted factors from human fetal NPCs, a well-known NPC source for the brain therapeutic approach, were further compared with those from hUCBCs to investigate whether hUCBCs had more beneficial functions than fetal NPCs to modulate immune reactivity in the damaged brain. Interestingly, there were marked differences between hUCBCs and human fetal NPC, as evident in the levels of interleukin (IL)-6, IL-8, GRO-α, granulocyte colony stimulating factor (G-CSF), and monocyte chemotactic protein (MCP-1) (Table 1). Taking into account the secretome results in which hUCBCs secreted significantly higher levels of cytokines and chemokines than fetal NPCs, hUCBCs could play an important role as an immune modulator in restoring the damaged brain, in particular from neonatal hypoxia/ischemia and stroke.
Homing of Transplanted Cells Toward Infarct Border and Upregulation of Chemokines
To investigate the possible effects of the two secreted proteins GRO-α and MIP-1α on CNS, human nuclei-positive cells were costained with GRO-α or MIP-1α in the hUCBC-transplanted CP rats. Consistent with the results from in vitro secretome analysis, GRO-α and MIP-1α were highly distributed in the lesion area of the vehicle-treated group, with little detection of the two secreted proteins in the contralateral corresponding region at 1 day after transplantation. The expression of these chemokines decreased dramatically at 1 week, although transplanted cells remained in the area. Also, human nuclei-staining cells were observed only in the injured area at 1 and 7 days after transplantation. Some of the transplanted cells were costained with GRO- α and MIP-1α (Fig. 6A).

Increased levels of chemokines, secreted by hUCBCs, are responsible for migration of progenitor cells. (A) Growth-regulated oncogene-α (GRO-α) and macrophage inflammatory protein-1α (MIP-1α) were distributed in perilesion of cell-treated group, with little detection of the two secreted proteins in the corresponding contralateral region at 1 day after transplantation. Few cells expressing these two chemokines were detected in the same region at 7 weeks after transplantation. Some of the chemokine-immunoreactive cells were colabeled with HU nuclei (white arrows). (B) Semiquantitative RT-PCR measurement of gene expression of endogenously induced chemokines in rat brain. (C–H) Relative ratio between the expression levels of chemokines and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The transplantation of hUCBCs resulted in a significant increase in MIP-1α expression compared to vehicle group at 1 day after cell transplantation [t(0.05, 4) = 3.03, p < 0.05]. Also, the levels of other chemokines measured in the present study tended to be higher in the hUCBC group than in the vehicle group except for stromal-derived factor (SDF). At 1 week, there was no obvious difference between the two groups. MCP, monocyte chemotactic protein; RANTES, regulated upon activation normal T-cell expressed and secreted. Scale bars: 20 μm. ∗p < 0.05.
In an attempt to evaluate the effects of transplanted hUCBCs on endogenously induced chemokines in the brain, gene expressions of GRO-α, MIP-1α, MCP-1, MCP-3, CCL5 (regulated and normal T-cell expressed and secreted; RANTES) and stromal-derived factor-1 (SDF-1) were measured semiquantitatively in the ipsilateral region at the level of mRNA, using ratios between densities of chemokines and the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH). hUCBC group expressed a significantly higher level of MIP-1α than vehicle group at 1 day after transplantation [t(0.05, 4) = 3.03, p < 0.05] (Fig. 6B–H).
Obviously, at 7 days after transplantation, the five chemokines' gene expressions were decreased in both groups compared to the results of 1 day. In the changed levels between the two time points (7 days–1 day), there was a significant difference only in MIP-1α [–0.86 ± 0.03 for the vehicle group vs. –1.04 ± 0.05 for the hUCBC group, t(0.05, 4) = 2.86, p < 0.05, data not shown]. It is the most likely explanation that the increased levels of chemokines at 1 day following transplantation correlate with direct (the potential paracrine effects of the hUCBCs themselves) and indirect impacts (endogenously induced chemokine expression by hUCBCs).
Functional Recovery by hUCBCs Transplantation
Neonatal ischemia produces a great diversity of motor, cognitive, and possibly emotional changes due to global brain damage. Several behavioral abilities were assessed in a battery of tests conducted from 1 to 10 weeks after transplantation. Motor, learning/memory, and anxiety tasks were used, and each task was administered at different time points (see Fig. 7A for details). To examine animals' basic motor function and exclude factors influencing specific brain function, ambulatory ability was measured by examining the latency to simply walk from a lit compartment to a dark compartment. There was no significant difference between the three groups in the basic motor function test, indicating no visual impairments and olfactory disorders that would affect motor, learning and memory, and anxiety tests (Fig. 7B). The two groups did not differ significantly in body weight across age throughout the 10-week period [F(2, 17) = 0.4, p > 0.9] (Fig. 7C).

(A) Experimental plan. (B) Measurement of latency to enter into dark compartment to evaluate basic motor function. There was no significant difference between the three groups in the basic motor function test. (C) Change in weight over time. The two cerebral palsy (CP) groups did not differ significantly in body weight across age throughout the 10-week period [F(2, 17) = 0.4, p > 0.9]. (D) Cylinder test to evaluate sensory motor function. In sham group, the proportion of paw usage is close to that of opposite forelimb. Cell-treated group showed an improvement in the usage of the impaired paw compared to the vehicle-treated group (p < 0.03), although the proportion of usage is always less than that of sham group during the cylinder test (p < 0.001). The vehicle-treated group showed preferential use of the less affected forelimb. (E) Passive avoidance test to measure the latency to enter the dark room with (first day) and without electric shock (second day). There were no between-group differences in the latency to enter during the first day, whereas the vehicle-treated group showed reduced latency on the second day (p < 0.02). (F) Forced swim test to assess depression. There is a marginally significant between-group difference in the immobility time between hUCBC-treated group and the vehicle-treated group [F(2, 23) = 3.56, p < 0.06]. (G) Light/dark exploration revealed that there is a marginally significant difference in time spent in lit room between the hUCBC-treated group and the vehicle-treated group [F(2, 23) = 3.76, p < 0.06]. Rats given hUCBCs spent more time in the light room. ∗p < 0.05, ∗∗∗p < 0.001, #p < 0.06.
The injury affected contralateral movements, as confirmed by the asymmetric use of the unimpaired paws. To investigate somatosensory function, the cylinder test was performed. There is a main effect of Group [F(2, 25) = 10.47, p < 0.0005)] (Fig. 7D). In the sham group, the proportion of paw usage was close to that of the opposite forelimb. The hUCBC-treated group showed a greater improvement in the usage of the impaired paw than the vehicle-treated group (p < 0.03), although for both groups, the proportion of usage was significantly less than that of the sham group during the cylinder test (p < 0.001). The vehicle-treated group preferred use of the unaffected forelimb (Fig. 7D).
To further investigate associative learning and memory, the passive avoidance test was administered at 10 weeks after transplantation. The parameter measured in this test was the difference between the time required to enter into a dark room with electric shock on the first day test and the latency to enter a dark room without electric shock in the second test as a measure of learning and memory retention. The interaction between Group and Session was significant [F(2, 30) = 5.36, p < 0.03] (Fig. 7E). There was no group difference in the latency on the first day, where as the vehicle-treated CP group showed shorter latency on the second day (p < 0.02). Post hoc analysis showed that the difference of the latencies was significantly longer for the hUCBC-treated group than the vehicle-treated group (p < 0.04) and that the cell-treated animals did not differ from the sham group (Fig. 7E), indicating that the cell-treated group showed improved performance in the retention phase of the passive avoidance test.
In the forced swim test, immobility time was checked as a standard measurement of depression. As shown by Figure 7F, there was a marginally significant difference in the immobility time necessary for floating and keeping their heads above water between cell-treated CP group and sham CP group [F(2, 23) = 3.56, p < 0.06] (Fig. 7F), indicating less depressed behavior for the hUCBC-treated group. There was no difference between sham and hUCBC groups.
The time spent in either the light compartment or dark compartment was measured during the transition between the two rooms in light/dark exploration task. There was a marginally significant main treatment effect [F(2, 23) = 3.76, p < 0.06] (Fig. 7G) in the total time to stay in the lit compartment. Contrast analysis revealed that there was a marginally significant difference in the parameters between the hUCBC-treated group and the vehicle-treated group (p < 0.06).
Correlation Between the Number of NeuN in Neocortex and Preferential Use of Paws
To evaluate how directly the result of immunohistochemistry is related to performance on cylinder test, we measured the correlation between the total number of NeuN in both sides of neocortex and the proportion of usage of the affected paw in the cylinder test (n = 4 per group). In the hUCBC group, a strong positive correlation existed between the two variables (r = 0.97731, p = 0.02) (Fig. 8A), but not in the vehicle group (r = 0.25, p > 0.74) (Fig. 8B). As shown in scatterplot, hUCBC-treated rats tended to exhibit greater usage of the impaired paw in the cylinder test and an increase in the number of NeuN, whereas the vehicle-treated rats had a tendency to display lower usage of impaired paw in the cylinder test and a concomitant decrease in the number of NeuN (Fig. 8C).
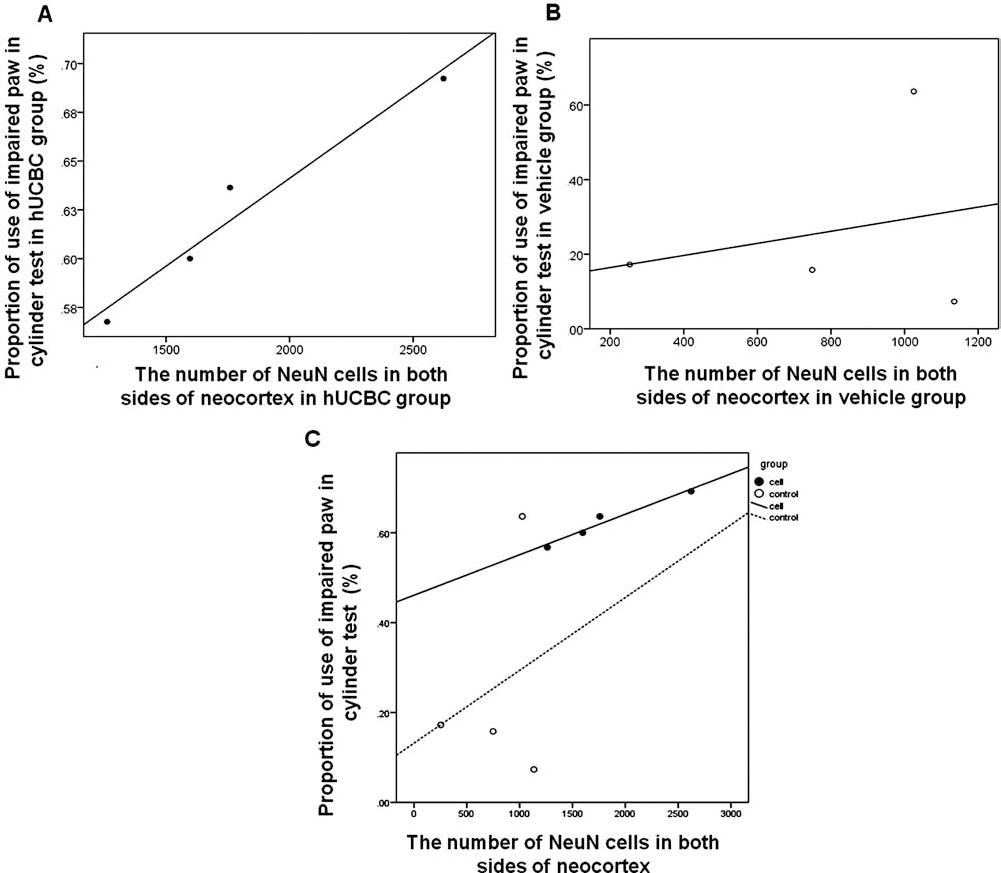
Correlation between mature neurons in neocortex and usage of the impaired paw. There is a strong positive correlation between the number of neuronal nuclei (NeuN) in both sides of neocortex and the use of the impaired forelimb in the hUCBC group (A) r = 0.9731, p < 0.02), but not in the vehicle group (B) r = 0.25018, p > 0.74). As seen in the distribution of scattered dots (solid dots, hUCBC-treated rats; empty dots, vehicle-treated rats), cell-treated groups showed greater usage of the impaired paw in the cylinder test and an increase in the number of NeuN+ cells on both sides of the neocortex, and vice versa in the vehicle group (C).
This suggests that the density of NeuN in neocortex is closely associated with the recovery of the impaired paw and that hUCBCs protect mature neurons, consequently improving the functional recovery of the impaired forelimb.
Discussion
The aims of this study were as follows: (1) analysis of behavioral features that are manifested in a rat model of neonatal ischemia with and without cell treatment, using motor, anxiety, and learning/memory tests up to 10 weeks after transplantation; (2) evaluation of the therapeutic effects on the disease model; and (3) a better understanding of the mechanism by which the therapeutic effects are produced.
Integration, Migration, and Differentiation of Transplanted Cells in Damaged Host Brain Area (Ipsilateral) and Stimulation of Endogenous Neurogenesis
Transplanted cells were detected in the injured area at 1 week after cell treatment, while there were few injected cells in the same area after 3 weeks. Despite the poor survival of hUCBCs in the injured area, the beneficial effects such as enhanced endogenous neurogenesis, protection of neurons, and an improvement in behavioral functions lasted until 10 weeks. Accordingly, it can be concluded that the continued existence of hUCBCs is not vital for exerting the effects advantageous to the recovery. It is arguable whether the presence or long-term survival of transplanted cells after passing out of BBB is essential to repair the damaged brain. Some studies have demonstrated that transplanted cells are rarely observed in brain parenchyma, irrespective of functional recovery in the cell treatment group (12,55). The entry of intravenously injected hUCBCs into brain parenchyma was shown not to be a requisite qualification for the functional recovery in a rat model of stroke (8).
The extravasation of the Evans Blue dye into the brain parenchyma was observed only in the neonatal ischemia model, whereas sham-operated rats did not show any indication of the extravasation of the dye. Human nuclei-positive cells were predominantly observed in septal nucleus and striatum adjacent to ventricle of the hUCBC group at 1 week, indicating that hUCBCs are able to penetrate BBB and migrate toward the lesion area.
A higher number of DCX-expressing cells, characteristic of neuronal precursor and immature neurons, were observed in periventricular striatum of hUCBC group than in the vehicle group at 1 and 10 weeks after transplantation, indicating that hUCBCs affect the brain environment in a long-term fashion. The cells immunoreactive for nestin, a neural progenitor marker, were detected more in periventricular striatum of hUCBC group than in that of vehicle group at 1 week after transplantation with no nestin-positive cells in both groups at 10 weeks.
Taken together, the administration of hUCBCs preserved and proliferated the endogenously induced neural precursor cells, which may provide neurogenesis-prone environment in order to repair the damaged brain region. Neural progenitor cells may have generated from the SVZ, which is a source of new neurons in the postnatal and adult brain (4).
However, more comprehensive studies on neurogenesis need to offer a convincing interpretation, because there was not enough empirical evidence in our study that neural precursor cells matured and integrated into brain.
Even though there was no difference between the two groups in the levels of NeuN expression in the cortex at 1 week, a higher expression of NeuN was observed at 10 weeks posttransplantation in hUCBC group than in vehicle group. Abundant mature neurons were present throughout the cortex, ipsilateral to the injury, in the hUCBC group compared to the vehicle group. In the contralateral region of the vehicle-treated group, fewer NeuN-expressing cells were detected than in the hUCBC group. Mature neurons, but not immature neurons, are immunopositive for NeuN, which is expressed in the nucleus and cell body (56). NeuN staining was used to examine the phases of infarction in a rat model of stroke. Loss of NeuN immunoreactivity occurred in a time-dependent manner in striatum and cortex following middle cerebral artery occlusion, and NeuN-positive cells in the cortex increased slightly at the later time point (27). Previous studies have reported that a decrease in NeuN immunostaining was commonly observed with neurological insults (13,30,57). Thus, the reduction of NeuN-expressing cells may be an index of neuronal injury, considering the difference in the staining pattern between injured and noninjured regions. The clear distinction in the expression of NeuN between the two groups was seen, not only in ipsilateral region but also in the contralateral region. MRI images in our experiment, showing that the contralateral neocortex was severely affected, supported the observation that hypoxia ischemia insults can affect both ipsilateral and contralateral regions. These results parallel one study that shows that hypoxic ischemia-induced injury is progressive with no recovery and that the damage extends to the contralateral hemisphere over time, as evidenced by MRI (33). We showed that NeuN staining following CNS degeneration may be a useful marker to evaluate the severity of the damage. The higher NeuN expression for a longer period following hypoxia/ischemia in the cell treatment group than that in the vehicle group suggests that hUCBCs may protect mature neurons from progressive injury or may induce neurogenesis.
hUCBCs Exert Paracrine Factors and Enhance the Activity of Microglia in the Striatum in the Early Phase of Disease
Our data demonstrated that hUCBC transplantation produced functional recovery and histological improvement. Other groups have also demonstrated that transplanted hUCBCs were able to differentiate into neuronal cells in the host brain (18,25,61). Interestingly, as seen in Figure 2, injected cells were rarely detected from 3 weeks posttransplantation. However, functional recovery persists for at least 10 weeks posttransplantation. Therefore, the mechanism by which injected hUCBCs sustain beneficial effects in the injured host brain needs to be elucidated. Several studies have suggested an indirect influence of hUCBCs on the host tissue that leads to the functional improvement (31,39,40,51). We propose here that injected hUCBCs secrete neuroprotective, immune modulatory, or angiogenic factors, thereby providing beneficial effects in the host tissue for up to at least 10 weeks posttransplantation.
The Transient Upregulation of Microglia Following the Transplantation of hUCBCs
Microglial cells in the brain are at the heart of chronic neurodegeneration and can be activated for a prolonged period of time. If not suspended, the chronic activation of microglia results in progressive neuronal injury, which is present in CNS diseases, by releasing toxic factors (cytokines and reactive oxygen species) (6,7,32). This negative effect of chronic activation of microglia is thought to play a role in the pathological features seen in our animal model. In contrast to the deleterious influence, microglia can play a positive role by producing neurotrophic factor (21,26,50).
Some studies have shown that, in a neonatal cerebral ischemia model, the administration of anti-inflammatory agents such as minocycline could exert protective effects without influencing the activity of microglia, whereas attenuating the activity of microglia is indispensable for protecting the injured brain in adult stroke, implying that the response of microglia is age-dependent. The roles of microglia during the progress of disease are highly controversial (15,17,53). We examined the expression pattern of cells positive for Iba-1, a pan-microglia marker, after transplantation of hUCBCs. Interestingly, hUCBCs increased the number of microglia cells in the affected brain during the early phase, which, in terms of therapeutic potential, is thought to be inconsistent with the previous studies showing that an increase in microglia leads to severe brain damage (54).
At 10 weeks after transplantation, neither group showed a significant difference in the number of Iba-1-positive cells. Considering the early rises in chemokines following transplantation, an increase in microglia may be an inevitable consequence due to ability of chemokines to attract immune-related cells. Whether hUCBC-driven microglias have helpful impacts on the damaged brain or not remains unclear by the current evidence. However, the results of long-term histological improvements and better performance in behavioral tests in hUCBC group imply that transient upregulation of microglia may promote brain recovery to a certain extent.
Chemokines
hUCBCs secreted substantial amounts of chemokines, interleukins, and growth factors compared with negative control (ITS medium) and fetal stem cells. Many of these secreted factors were inflammatory factors and chemokines that mediate cellular migration and intercellular communication. Specifically, three chemokines, MCP-1, MIP-1α, and GRO-α, were more highly secreted from hUCBCs than from control and fetal stem cells. MCP-1, MIP-1α, and GRO-α have been well characterized in regulating and directing adult SVZ-derived progenitor cell migration following brain injuries, where they act chemotactically and contribute to the homing of newly generated progenitor cells (20,28,58). Furthermore, MCP-1 is associated with the induction of angiogenesis (46,47). As shown in Figure 4A, GRO-α and MIP-1α were expressed in the injured brain of the CP rat model, and most of them were colabeled with injected hUCBCs, indicating the secretion from hUCBCs. As shown in the results of semiqualitative RT-PCR, the transplantation of hUCBCs brought about a significant increase in MIP-1α expression, relative to the vehicle group at 1 day after cell transplantation. Also, the levels of other chemokines measured in the present study tended to be more in the hUCBC group than in the vehicle group, except for SDF.
One recent study reported that MCP-1, MIP-1α, and GRO-α were upregulated in the affected brain region during only the first 2–3 days following insults (20), suggesting that any compensating action from brain injury is transient as the self-healing defense is not sufficient to protect the brain from further injury or regenerate the injured tissue. Temporary rise in chemokines is probably a contributing factor in enhancing brain plasticity and functional recovery. Thus, extending this healing process may be an approach to treat brain injuries. Our data demonstrated that the transplantation of hUCBCs exerted therapeutic effects through indirect factors such as secreted chemokines.
Improvement of Behavioral Function
Patients with CP exhibit a broad range of cognitive impairment and affective disorders from dementia and executive dysfunction to more subtle memory loss (2,9,10,60). The most common phenotype of CP is spasticity in the upper and lower extremities. Other manifestations of CP include movement disorder, muscle weakness, impairment of peripheral sensibility disturbances of cognition, communication, perception, behavior problems, and a seizure disorder (5,22,23). A potential therapy using hUCBCs in a possible rodent model of CP has been controversial because functional recovery reported in most of the published papers, to our knowledge, has been limited to one or a few motor tasks (31,42).
Therefore, in the present study, extensive behavioral paradigms were used to measure integrative functional recovery from the brain disorder until 10 weeks after transplantation. Functional recovery of impaired behavioral phenotypes by hUCBCs in the cylinder test correlated well with an enhanced brain environment demonstrated by immunohistochemistry analysis. The regions where neuronal injuries are seen following hypoxia/ ischemia insults are dependent on gestation age (44). In preterm period, white matter injury is a clear manifestation of perinatal brain injury referred to as periventricular leukomalacia (45). However, in term infants, hippocampus, cerebral cortex, and cerebellar Purkinje cells are the affected sites. As a secondary effect of white matter injury, axonal malfunction, and gray matter injury has been observed (41). In spite of several damaged areas, as in a rat model of neonatal ischemia, behavioral validation is confined to motor properties such as spastic paralysis. Given that the known causes of CP are limited (43), several examinations such as tests of cognitive function, anxiety, and depression are required for early diagnosis of perinatal brain injury.
Summary
Even though the scattered transplanted cells with neural phenotypes were detected around periventricular region at 1 week after transplantation and only few cells were present at 10 weeks posttransplantation, there was a long-lasting positive influence of hUCBCs in vivo. These long-lasting effects appeared to be derived from (1) paracrine effects of hUCBCs that stimulated recovery in the injured brain and reduced progressive brain damage, (2) an increase in microglia during the early phase, (3) an increased expression of antigen related to endogenous stem/progenitor cells compared to vehicle group, and (4) secretion of chemokines responsible for directing SVZ cells toward the injured area by transplanted cells. Consequently, an improved behavioral performance was observed in the hUCBC-treated group compared with the vehicle-treated group in several behavioral tests up to 10 weeks posttransplantation.
Footnotes
Acknowledgments
This work was funded by the KOSEF (grants 2011-0029342, 2011-0013280, and 2011-0019485) and CHA Bio & Diostech. We are grateful to Dr. TeaJong Yoon for helping the MRI study and Yun-hwa Jeong and Clinical Statistics Center of CHA University for analyzing statistics. The authors declare no conflicts of interest.
