Abstract
Cerebral palsy (CP) is a chronic nervous system disease that severely damages the physical and developmental health of children. Traditional treatment brings about only improvement of mild to moderate CP, but severe CP still lacks effective interventions. To explore safety and efficacy of using neural progenitor cells (NPCs) to treat CP in children, we performed NPC transplantation in 45 patients with severe CP by injecting NPCs derived from aborted fetal tissue into the lateral ventricle. Gross motor function measures (GMFM), the Peabody Developmental Motor Scale-Fine Motor (PDMS-FM) test, and a unified survey questionnaire designed specifically for children with CP were used to evaluate neurological function of the patients. Motor development was significantly accelerated within the first month after cell transplantation, but the rate of improvement gradually slowed to preoperative levels. However, after 1 year, the developmental level in each functional sphere (gross motor, fine motor, and cognition) of the treatment group was significantly higher compared to the control group. No delayed complications of this therapy were noted. These results suggest that NPC transplantation is a safe and effective therapeutic method for treating children with severe CP.
Introduction
For the past 20 years, progress in medical technology in obstetrics and neonatology has increased the survival rate of critically ill newborns who were ‘eliminated through natural selection” in the past. However, despite this achievement, the treatment of infants with severe brain injuries has not changed the incidence of cerebral palsy (CP). The current incidence of CP in China is about 1.28–2.7%, which is similar to that in Western countries (20). The long-term special care and treatment required for CP patients, especially those severe cases, strain both health care and financial resources and provide little opportunity for improving quality of life. Rehabilitation training is a basic, traditional treatment for CP, but its effects on the patients with levels above IV according to the Gross Motor Function Classification System (GMFCS) appeared and developed very slowly, and these patients make up 22–34% of all cases (4,30,35). Therefore, more effective treatment measures need to be found to improve the motor function and life quality of children with severe CP.
From the 1980s to date, research institutes in various countries have investigated cell transplantation therapy for central nervous system (CNS) diseases and have suggested the safety and efficiency of this therapy on many diseases, including stroke, traumatic CNS injury, Parkinson's disease, Huntington's disease, amyotrophic lateral sclerosis, and more (2,8,9,14,21,38) in both animal and clinical research. Previous research carried out in our laboratory indicated the effectiveness of using neural progenitor cells (NPCs) to treat cortical blindness, severe hypoxic ischemic encephalopathy in neonates, and encephalopathy from severe carbon monoxide poisoning (23,24,34). These findings suggest transplantation as a potential therapy for the treatment of children with severe CP. Based on many years of research in this field, we performed NPC transplantation on 45 children with severe CP from mid-May 2005 to mid-May 2006.
Patients and Methods
Patients
A total of 94 consecutive patients visiting our hospital who met the following criteria were recruited.
Inclusion criteria included: 1) outpatients visiting the hospital from mid-May 2005 to mid-May 2006; 2) diagnosed with definite CP, including quadriplegic, diplegic, dyskinetic, and mixed CP; 3) classified to level I–V according to GMFCS; 4) making no improvement after 3–6 months of rehabilitation training before transplantation.
Exclusion criteria included: 1) brain injuries caused by other diseases such as progressive inherited metabolic disease; 2) severe congenital heart disease, cardiac functional insufficiency; 3) severe pulmonary disease, respiratory failure; 4) complicated with acute or chronic infection; 5) frequent epileptic seizures in the last 6 months; 6) diseases of the spinal cord; 7) brain tumor or other malignant tumors; 8) coagulation disorders complicated with hemorrhagic tendency; 9) diseases of immunologic deficiency or autoimmune diseases; 10) other severe pediatric diseases.
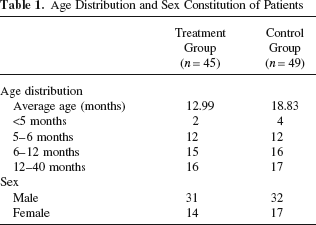
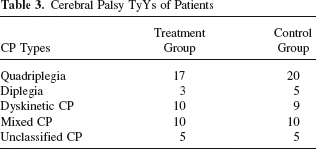
The included patients were randomly assigned to two groups (treatment group, n = 45; control group, n = 49). The treatment group received cell transplantation and rehabilitation training, and the controls only received rehabilitation training. Statistical analysis indicated that there was no significant difference in sex, age, CP types, causes and types of cerebral abnormalities shown by MRI (Tables 1–4), or developmental level according to a unified questionnaire survey of the treatment and control groups (Tables 5–8).
Age Distribution and Sex Constitution of Patients
Causes of Cerebral Palsy in Patients

Cerebral Palsy TyYs of Patients
MRI Examination of Patients

Chi-square statistics indicates that there is no significant difference in the composition of sex, age, CP categories, and brain injuries between the two groups: p > 0.05.
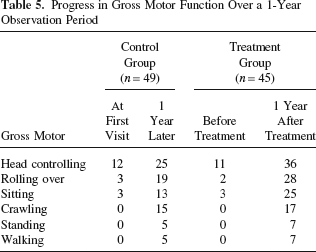
Progress in Gross Motor Function Over a 1-Year Observation Period
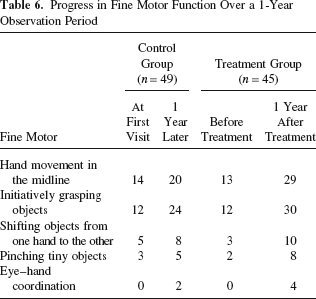
Progress in Fine Motor Function Over a 1-Year Observation Period
Progress in Cognition Over a 1-Year Observation Period

Functional Scores of Patients Over a 1-Year Observation Period
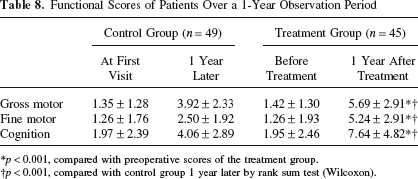
p < 0.001, compared with preoperative scores of the treatment group.
p < 0.001, compared with control group 1 year later by rank sum test (Wilcoxon).
The protocol of clinical studies was approved by the Scientific Council and Ethics Committee of Navy General Hospital and in accordance with guidelines issued by the Chinese Ministry of Health (91–006) (31). The guardians of the patients in the treatment group were fully informed about the sources, separation and cultivation of the cells, the methods of cell therapy, risks of the surgery, possible side effects, and safety measures, and an informed consent was signed. All treatment procedures were under the supervision of the hospital Ethics Committee.
Donor Selection
Fetus donors were selected from pregnant women who had requested artificial abortion during early pregnancy and consented to donate the fetus after being fully informed. Informed consent and agreement of voluntary donation was signed by all donors. Detailed family histories were obtained and inherited metabolic, chromosomal, and infectious diseases were screened for before donating. Protocols were approved by the hospital Ethics Committee.
Culture of Neural Progenitor Cells
NPCs were isolated and cultured as described previously (24). NPCs were isolated from aborted human fetal forebrain tissue with properly consent of the donors, and was cultured and propagated for 2–3 weeks in vitro. Before transplantation, the cells were strictly examined according to the technical guidelines of treatment research and quality control of human cells published by the State Food and Drug Administration (23,24,34). By using corresponding testing methods, the transplanted cells were nestin positive, and had normal karyotypes and a viability of over 95%. They were microbe free, and endotoxin level was at less than 2 EU/ml.
Cell Transplantation
Patients were placed in a supine position and given general anesthesia prior to surgery. Under the guidance of color Doppler ultrasonography, 8–10 × 106 of neural progenitor cells in 200 μl normal saline was slowly injected into one of the lateral ventricles through the unclosed fontanelle or through a hole drilled into the skull of patients with closed fontanelles, which was slowly pulled out after the injection was finished.
Rehabilitation Training
Rehabilitation training included physical and occupational therapy related to the trainings of gross motor, upper extremity function and activities of daily living, medical massage, etc. Personalized treatment programs were formulated according to the CP types and functional levels of each patient.
Evaluation of the Effects of Transplantation on Motor and Psychomotor Development
Evaluation of Motor Development of Treatment Group by Self-Control
Motor development of patients in the treatment group was evaluated by two neurologists in a double-blind fashion. The evaluation was performed 1 month before, the day before, and 1 month and 6 months after transplantation. Gross motor function was assessed with the Gross Motor Function Measure (GMFM) scale, and fine motor function was evaluated using the Peabody Developmental Motor Scale-Fine Motor (PDMS-FM) test.
Evaluation of Psychomotor Development of Treatment Group Compared with Control Group
Psychomotor evaluation of patients in the treatment group was carried out before and 1 year after cell transplantation. Patients in the control group were similarly evaluated at the beginning and end of a 1-year observation period. The evaluation used an easy-to-understand unified survey questionnaire designed to characterize the main milestones of psychomotor function at different developmental stages in children with CP. Gross motor development was represented by six functions (head controlling, rolling over, sitting, crawling, standing, and walking), fine motor development was characterized by five functions (hand movement in the midline, initiatively grasping objects, shifting objects from one hand to the other, pinching tiny objects, and eye–hand coordination), and cognitive development was based on eight functions (recognizing relatives, understanding facial expressions, using the words “hello” and “bye-bye,” using the words “mama” and “papa,” understanding simple nouns, understanding simple instructions, using words related to toilet training, and answering questions). After the questionnaires were completed, a third party was responsible for single blind analysis. The absence, partial, or complete performance of each function was scored as 0, 1, or 2, respectively. The scores of the two groups were compared.
Statistical Analysis
Data were analyzed with SPSS11.5 software. Chi-square was used to analyze enumeration data, and analysis of variance or rank sum test (Wilcoxon) was used for measurement data. The data were analyzed by a third party in the teaching and research section of statistics at Capital Medical University.
Results
Improvement of Clinical Manifestations
The earliest effect was observed in one patient 3 days after the surgery. Before transplantation, the patient was irritable, had difficulty falling asleep, and only slept for short periods of time. After the surgery, the patient could fall asleep at 10 pm and stay asleep until daylight, the irritable state improved, and abnormal posture caused by torsion spasm and opisthotonos was partially corrected. Clinical improvements in the majority of patients were observed 1 month after surgery. Patients that showed minimal improvement from rehabilitation training before surgery had improvements in head controlling and partial corrections of hyperrexplexia and abnormal postures after treatment, and had more limb function as a result. Cognitive ability of patients with mental retardation increased, showing improvement in language comprehension and communication and comprehension of simple instructions and facial expressions. The therapy did not achieve any effects on several patients initially, but they began to show clinical improvements 2–3 months after surgery. Although the improvements showed a decreasing trend 3–6 months after surgery, no regression was observed.
Evaluation of Motor Development of Treatment Group by Self-Control
After surgery total scores of GMFM and PDMS-FM increased significantly and continuously (Table 9). To discriminate the effect of cell transplantation based on age-related development and effect of rehabilitation training, the scores that improved per month were obtained to compare the development rate before and after surgery (Fig. 1). The scores improved per month were 2.22 ± 3.54 (PDMS-FM) and 0.31 ± 0.52 (GMFM) before surgery, and 2.17 ± 3.27 (PDMS-FM) and 5.35 ± 2.67 (GMFM) within 1 month after surgery, representing a substantial increase, suggesting that cell transplantation accelerated motor functional development. However, 3–6 months after surgery, the development rate decreased to the levels before surgery.

Motor developmental rate before and after surgery. The scores improved per month were 2.22 ± 3.54 [Peabody Developmental Motor Scale-Fine Motor (PDMS-FM)] and 0.31 ± 0.52 [Gross Motor Function Measures (GMFM)] before surgery, and 2.17 ± 3.27 (PDMS-FM) and 5.35 ± 2.67 (GMFM) within 1 month after surgery, representing a substantial increase; 3–6 months after surgery, the development rate decreased to the levels before surgery.
Results of GMFM and PDMS-FM Evaluation
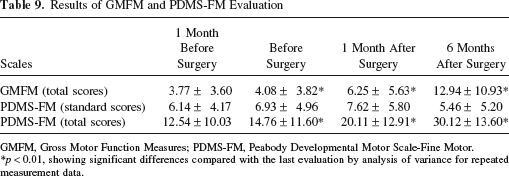
GMFM, Gross Motor Function Measures; PDMS-FM, Peabody Developmental Motor Scale-Fine Motor.
p < 0.01, showing significant differences compared with the last evaluation by analysis of variance for repeated measurement data.
Evaluation of Psychomotor Development of the Treatment Group Compared with the Control Group
Psychomotor functions evaluated before and 1 year after treatment are shown in Tables 5–7. Functional progress in the treatment group took over those of the control group after 1 year. The data were then quantified (Table 8). Statistical analysis showed that 1 year later developmental levels in the treatment group were significantly higher than those in the control group.
Complications and Side Effects
Tiny foci hemorrhage in the lobi frontalis cortex on the side of puncture occurred in one patient, manifesting in a low-grade fever and mild central type of facioplegia on the right side after surgery. The symptoms disappeared 2 weeks later after coagulant and symptomatic treatment with no sequelae. Six patients developed non-bacterial fever lasting from 1 to 7 days. According to EEG data, epileptiform activities found in 11 patients did not change after the surgery.
Discussion
In recent years, clinical studies suggest that the efficacy of NPCs in the therapy of various neural system diseases. The present study showed that NPC transplantation alleviated clinical symptoms and promoted the development of the major psychomotor functions in children with severe CP. It suggests that the procedure could be considered indicative for the severe type of the disease. In the present study, MRI revealed extensive bilateral brain damage in most patients. This made direct transplantation into the pathology too difficult to handle and hence intraventricular transplantation was selected instead. The ventricle of choice was approached under the guidance of color Doppler ultrasonography along a route through silent areas away from the blood vessels. It proved to be a relatively safe and simple procedure. Animal experiments have shown that NPCs grafted into the ventricle interact freely with neuroepithelial cells and are able to migrate into the whole brain including the injured sites through the ventricular wall (6,33,37).
Possible immune response compromising long-term survival of the implanted NPCs was a major concern in this cell therapy. However, it did not seem to have affected the outcome, probably because MHC I and MHC II molecules are sparse in NPCs and the brain is a relatively immune-privileged organ. Also, NPCs and their derived astrocytes regulate the immune response by various more sophisticated mechanisms. Implanted NPCs and their derived neurons and astrocytes do not elicit an allogenic immunological rejection (1,17,28,32) and survive for long periods of time in recipient animal brains (19,44). Postmortem histological analysis confirmed the long-term survival of grafted embryonic midbrain or striatal cells in the brain of patients with Parkinson's and Huntington's disease who received cell transplantation during their lifetime and no immunological rejection was found at the transplantation site (16,29). NPCs grafted into the brain were able to differentiate into all types of neural cells, including neurons, astrocytes, oligocytes, and more (15) and displayed site-specific differentiation (39). Derived functional neurons integrated into the neural circuitry of the host generated action potentials and received both excitatory and inhibitory inputs from neighboring cells (7). In the present study, patients who received unrelated allogeneic NPC transplantation were not treated with immunosuppressants, and none of the effective cases showed reversion of the effect, which indirectly supports the above theories.
Except for the effects of substitution, grafted cells affected the recipient brain by some complex mechanisms, such as paracrine action (10,25,26,42,43). According to our clinical observations, most patients were excited and had difficulty in falling asleep within the first 1–3 days following transplantation, which we attributed to the stimulating effects of neurotrophic factors released by NPCs. Some patients showed improvements within a few days after the surgery, which was consistent with the observations of Chen et al. (5) and Seledtsov et al. (36) after treating children with CP using olfactory ensheathing cells and fetal brain cells. They reported that most patients had abatement in manifestations within a few days after therapy. In contrast, the effects of cell transplantation on patients with stroke, Parkinson's disease, and Huntington's disease were apparent much later than in this study (18,22), probably due to the different environments in the brain of the various subjects investigated. The subjects investigated in the present study and in the study by Seledtsov et al. (36) were children of nonprogressive brain injury with permanent damage. Because developing brains have stronger plasticity and capability than adult ones, these quick effects were probably due to endogenous neural mechanism that was quickly activated by neurotrophic factors, such as glial cell-derived neurotrophic factor (GDNF), neurotrophin-3 (NT-3), brain-derived neurotrophic factor (BDNF), and neural growth factor (NGF) released by grafted NPCs (10,26,43). All these may have contributed to activating the silent synapses with early functional recovery (3,13,40). In early stages of neural development, there are a considerable number of synapses with normal anatomic structures that are kept in a silent state due to incapability of releasing neurotransmitters from presynaptic structures or absence of relevant receptors on postsynaptic membranes (12,27,41), but synapses in a silent state gradually reduce in number with development.
To date, there are no scales dedicated to the evaluation of fine motor function of CP children. In the present study, PMDS-FM was used. However, PMDS-FM was designed based on normal children, and the functions of children with severe CP were at the extreme end of the normal distribution. Even though raw scores of the patients increased after surgery, standard scores were still below 1% in the distribution and showed no obvious changes. Therefore, we obtained total scores of PMDS-FM according to the calculation of GMFM to observe the development of fine motor function of these patients before and after surgery (Table 9). Nevertheless, a more sensitive scale of evaluating fine hand function that is more suitable for CP children needs to be explored.
Comprehensive rehabilitation training had obvious effects on children with mild and moderate CP, but its effects on children with severe CP appeared and developed very slowly. The present study showed that transplantation of NPCs significantly improved the functional development of children with severe CP, and no delayed complications were noted, suggesting that NPCs transplantation was a safe and effective treatment for severe CP. But the rate of improvement decreased gradually with time after 3 months of NPC transplantation, suggesting that the effect of a single transplantation was limited and that multiple transplantations might increase the efficacy of the treatment (11). However, the appropriate window of time and course of later multiple transplantations needs further investigation.
Footnotes
Acknowledgment
The authors declare no conflicts of interest.
