Abstract
Mesenchymal stromal cells from the human amniotic membrane (i.e., human amniotic mesenchymal stromal cells [hAMSCs]) of term placenta are increasingly attracting attention for their applications in regenerative medicine. Osteochondral defects represent a major clinical problem with lifelong chronic pain and compromised quality of life. Great promise for osteochondral regeneration is held in hydrogel-based constructs that have a flexible composition and mimic the physiological structure of cartilage. Cell loading within a hydrogel represents an advantage for regenerative purposes, but the encapsulation steps can modify cell properties. As pectin gels have also been explored as cell vehicles on 3D scaffolds, the aim of this study was to explore the possibility to include hAMSCs in pectin gel. Immobilization of hAMSCs into pectin gels could expand their application in cell-based bioengineering strategies. hAMSCs were analyzed for their viability and recovery from the pectin gel and for their ability to differentiate toward the osteogenic lineage and to maintain their immunological characteristics. When treated with a purposely designed pectin/hydroxyapatite gel biocomposite, hAMSCs retained their ability to differentiate toward the osteogenic lineage, did not induce an immune response, and retained their ability to reduce T cell proliferation. Taken together, these results suggest that hAMSCs could be used in combination to pectin gels for the study of novel osteochondral regeneration strategies.
Keywords
Introduction
Osteochondral defects resulting from congenital defects, pathological disorders, and/or traumatic injury represent a major clinical problem with lifelong chronic pain and compromised quality of life 1,2 . Due to its poor regenerative capacity, stratified architecture, and disparate biochemical properties, the challenge remains to replicate robust integration of cartilage and subchondral bone and the complex stratified tissue 3,4 .
Bioengineered scaffolds are being widely investigated for osteochondral regeneration strategies. Various scaffolds, such as porous sponges 5 –7 , meshes, and hydrogels 8 , have been prepared from biodegradable synthetic and natural polymers for tissue engineering applications 9 and have been shown to influence cell functions and improve cartilage, osteochondral, and bone regeneration. Among the different properties of scaffolds, a flexible structure mimicking the physiological structure of cartilage is essential. In this perspective, great promise for osteochondral regenerative medicine is entrusted in hydrogel-based structures 8,10,11 that can be obtained by the use of natural polymers. Among them, pectins, a family of polysaccharides found in the primary cell walls of plant cells, are commonly used in drug delivery and wound-healing applications 12 –15 . Embedding cells in a hydrogel has several advantages. First, considering cell delivery for regenerative purposes, the presence of a gel has the advantage of confining cells in a specific site and fitting/adapting them to the specific defect, while at the same time insulating and protecting the cells from the surrounding microenvironment 16 . Second, it can also allow processing techniques which are commonly used to obtain graded scaffolds, such as preparing cell-loaded hydrogel sheets, impregnating porous structures with the cell-loaded gel, and production of multilayered scaffolds 17 .
Current developments in tissue engineering strategies for articular cartilage regeneration focus on the design of supportive 3-dimensional (3-D) scaffolds and their use in combination with cells from different sources 18,19 . In this scenario, scaffolds allow delivery and retention of the cells at the defect site. Mesenchymal stromal cells (MSCs) have been widely investigated in tissue engineering strategies for articular cartilage regeneration. MSCs possess extensive proliferation potential and proven capability to differentiate along the osteochondral pathway 20 . In addition to proliferation and multilineage differentiation potential, MSCs possess immune-modulatory and anti-inflammatory functions, which may enhance their regenerative power 20 .
While MSCs from bone marrow are widely used, more recently, MSCs from human term placenta have gained attention due to their unique properties. After birth, the placenta is usually discarded as nothing more than biological waste, and thus placental tissue is easily accessible in a noninvasive way. Furthermore, compared to MSCs from other sources such as bone marrow and adipose tissue, high cell recovery is possible from placental tissues. Human placenta is composed of a fetal part, including the amnion, chorion, umbilical cord, and a maternal part, termed decidua. Within these components, different cell subpopulations with mesenchymal characteristics may be isolated, and as established by the consensus of the First International Workshop on Placenta-Derived Stem Cells, they are referred to human amniotic mesenchymal stromal cells (hAMSCs) and human chorion mesenchymal stromal cells 21 . Nowadays, it has been widely demonstrated that some cells from the human placenta possess some stem/progenitor cell potential which is thought, at least in part, to be due to the early origin of placental tissues during embryogenesis and prior to gastrulation 21,22 . In addition, placental MSCs, and more specifically MSCs from the amniotic membrane, possess intrinsic immune-modulatory properties which do not require licensing with inflammatory stimuli as suggested with MSCs from bone marrow 23 .
The aim of this study is to evaluate hAMSC in vitro properties after pectin gel treatment to establish whether hAMSCs could be ideal candidates for cell-based bioengineering strategies using pectin gels as cell vehicles on 3-D scaffolds. First, we investigated hAMSC recovery and cell viability after loading into a pectin gel. Then, we investigated whether hAMSCs retained their unique functional properties, such as the inability to mount an immune response, ability to decrease the proliferation of activated peripheral blood mononuclear cells (PBMCs), and to differentiate toward the osteogenic lineage. We conclude that pectin gel treatment does not alter hAMSCs and does not influence their immunological properties nor osteogenic differentiation, suggesting that hAMSCs could be used in combination with pectin gels obtained in the reported conditions for the study of novel osteochondral regeneration strategies.
Materials and Methods
Ethics Statements
All samples used in this study were obtained after informed consent from donors. For human term placenta and peripheral blood samples, written informed consent to collect placenta and isolate human amniotic-derived cells was obtained from each single donor according to the guidelines of the Ethics Committee (Comitato Etico Provinciale) of the province of Brescia.
Human MSC Isolation, Growth, and Cryopreservation
Human term placentas (n = 12) were processed within approximately 8 h after birth. The amnion was manually separated from the chorion and washed extensively in saline sodium chloride 0.9% with 100 U/mL penicillin and 100 µg/mL streptomycin (herein referred to as P/S, both from Sigma-Aldrich, St. Louis, MO, USA) and 2.5 mg/mL amphotericin B (Sigma-Aldrich). Afterward, the amnion was cut into small fragments (∼3 × 3 cm2) which were decontaminated by a brief incubation in saline sodium chloride 0.9% + 2.5% Betadine − 0.25% Iodopovidone (Betadine-Esojod 10%, Ecolab-Esoform, Venice, Italy) and 3 min in phosphate-buffered saline (PBS; Sigma-Aldrich) containing 500 U/mL penicillin, 500 µg/mL streptomycin, 12.5 µg/mL amphotericin B, and 1.87 mg/mL Cefamezin (Teva, Italy). The amnion fragments were then incubated for 9 min at 37 °C in HBSS (Sigma-Aldrich) containing 2.5 U/mL dispase (VWR, Milan, Italy). After a 3 to 5 min resting period at room temperature in Roswell Park Memorial Institute (RPMI) 1640 medium (Sigma-Aldrich) supplemented with 10% heat-inactivated fetal bovine serum (FBS, Sigma-Aldrich), 2 mM L-glutamine (Sigma-Aldrich), and P/S, the fragments were digested with 0.94 mg/mL collagenase (Roche, Mannheim, Germany) and 0.01 mg/mL DNase (Roche) for 2.5 h at 37 °C. Amnion fragments were removed, and mobilized cells were passed through a 100-µm strainer (BD Falcon, Bedford, MA, USA) and collected by centrifugation at 300× g for 10 min. The cell suspension was filtered a second time with a 70-µm strainer (BD Falcon). Cells were frozen in 90% heat-inactivated FBS and 10% dimethyl sulfoxide (DMSO) (Sigma-Aldrich) in liquid nitrogen until use.
To obtain hAMSCs at passage 1, freshly isolated or thawed cells were plated in flasks (Corning Inc, Corning, NY, USA) at a density of 10,000 cells/cm2 in CHANG Medium C supplemented with 2 mM L-glutamine and P/S. Upon reaching subconfluency, adherent cells were washed in PBS, detached with 0.25% trypsin (Sigma-Aldrich), and then cultured for subsequent experiments.
Cell viability and recovery were analyzed using the trypan blue exclusion assay.
hAMSC Loading into the Pectin Gel
Low-methoxyl pectin (CU701, kindly provided by Herbstreith & Fox, Nuremberg, Germany) was dissolved overnight in a sterile NaCl (10.8 mg/mL; Sigma-Aldrich) − NaHCO3 (2.52 mg/mL; Sigma-Aldrich) solution at a final concentration of 22.5 mg/mL. Calcium phosphate (CaP; kindly provided by Eurocoating SpA [Italy], hydroxyapatite granules, in the range 20 to 40 μm) was suspended in water to obtain a final concentration of 23 mM.
CaP suspension (0.4 mL) was mixed with pectin solution (2 mL) and 5 × 105 hAMSCs were added by resuspension in 0.4 mL of complete medium (DMEM [Sigma] + 20% FBS), using a dual-syringe system. Afterward, 0.4 mL aliquots of the hAMSC–pectin gel suspension were plated in 24-well plates (Corning) and incubated for 30 s with 0.1 mL of 2% w/v of calcium gluconate (Sigma). Complete medium (0.5 mL) was added to each well and the plates were incubated at 37 °C and 5% CO2 for different time points (1, 3, 24, 48, 72, and 168 h). As controls, hAMSCs were mixed and plated in complete culture medium (Dulbecco’s modified Eagle’s medium (DMEM) + 20% FBS) using a dual-syringe system or, to mimic the gel consistency of pectin, in Matrigel™ Basement Membrane Matrix (BD Biosciences). At the end of incubation, 1 mL of PBS + 25% ethylenediaminetetraacetic acid (EDTA) 0.5 M was added to each well to dissolve the pectin gel. Prior to immunological and differentiation assays, CaP was depleted using density gradient centrifugation (Histopaque; Sigma-Aldrich) due to its interference in these assays.
Osteogenic Differentiation
For the treatment conditions described above, hAMSCs (1 × 104/cm2) were seeded in DMEM + 20% FBS + P/S + L-glutamine. Three days later, culture medium was substituted with Stempro Osteo/Chondrocyte Diff Basal Medium with osteogenic supplement (Life Technologies). For controls, culture medium was substituted with fresh medium. Culture medium was changed twice a week.
Osteogenic differentiation was assessed after 14 d and calcium deposits were visualized using the Alizarin Red (Sigma-Aldrich) and Von Kossa (Bio Optica) stainings. Cells were fixed in 10% formalin and 2% Alizarin Red pH 4.2 was added, left for 25 min, and afterward cells were washed with distilled water. Alternatively, cells were fixed in 10% formalin and 5% silver nitrate (Sigma-Aldrich) was added for 1 h under ultraviolet light, washed with water, and incubated with 5% sodium thiosulfate (Sigma-Aldrich) for 5 min and washed once more.
Immunological Studies
The ability of hAMSCs to either induce a T-cell response or inhibit the proliferation of PBMCs activated with anti-CD3 was performed as previously described 24 . PBMCs were obtained from heparinized whole blood samples or buffy coats from healthy subjects using density gradient centrifugation (Histopaque; Sigma-Aldrich).
PBMCs (1 × 105/well in a 96-well plate) were cultured with hAMSCs (PBMC:hAMSC ratios 1:1, 1:0.5, 1:0.1, 1:0.02, and 1:0.004) or were stimulated with 125 ng/mL (final concentration) α-CD3 (Orthoclone OKT3, Janssen-Cilag) in the presence of hAMSCs (PBMC:hAMSC ratios 1:1, 1:0.5, and 1:0.1). All conditions were performed in triplicate in DMEM (Sigma-Aldrich) supplemented with 20% heat-inactivated FBS, 2 mM L-glutamine, and P/S. After 3 d of culture, lymphocyte proliferation was assessed by adding 0.67 μCi of [3H]-thymidine (INC Biomedicals) per well. Sixteen to 18 h after the addition of thymidine, cell cultures were harvested with a FilterMate Harvester (PerkinElmer), and thymidine incorporation was measured using a microplate scintillation and luminescence counter (Top Count NXT; PerkinElmer).
Lymphocyte proliferation was examined in the presence of irradiated (3,000 cGy) hAMSCs (0.1 × 105/well, 0.5 × 105/well, and 1 × 105/well) in contact with activated PBMCs.
Statistical Analysis
All data are expressed as mean ± standard error of the mean (SEM). All statistical analyses were performed using GraphPad Prism version 6.00 for Windows (Graph-Pad Software, San Diego, CA, USA). Analysis of cell recovery and viability was performed using an analysis of variance (ANOVA) or unpaired t-test. Comparison of counts per minute values between hAMSCs and PBMC activated with α-CD3 was performed using an ANOVA.
Results
hAMSC Recovery and Viability After Loading into Pectin Gel
We investigated whether loading hAMSCs into pectin gel (pectin and CaP) altered cell recovery and viability. First, we cultured hAMSCs in pectin gel or in complete culture medium (DMEM + 20%FBS) for different time intervals, from 1 h up to 7 d. As an additional control to mimic the gel consistency of pectin, hAMSCs were also cultured in Matrigel™ basement membrane matrix. After incubation in EDTA to remove pectin gel, residual fragments of the gel were observed and the concentration of EDTA was progressively increased up to 5 M during the development of the cell recovery protocols, indicating a strong interaction of the pectin chains with Ca++ cross-linker. Sodium citrate was not effective for the dissolution of the gels.
We detected a 50% drop in cell recovery after 1 h of incubation within the pectin gel; this did not decrease further and remained constant for up to 7 d after incubation (Fig. 1A). In addition, no effects on hAMSC viability were observed for up to 24 h after incubation within pectin gel when compared to untreated hAMSCs at time 0 (T0; Fig. 1B). There was a small, but significant, decrease in hAMSC viability only 48 and 72 h after incubation in pectin gel, but viability began to increase again 7 d after incubation within pectin gel when compared to untreated hAMSCs at T0 (Fig. 1B). Cell viability decreased from 83% at T0 (before loading into pectin gel) to 58% after a 7-d treatment with pectin gel. Cell recovery was similar between all the considered time points, thus subsequent studies were performed using a 3-h treatment within the pectin gel.

Human amniotic mesenchymal stromal cell (hAMSC) recovery and viability after loading into pectin gel. Panels (A) and (B) show hAMSC recovery (percentage vs. time 0 set to 100%) and viability, respectively, at passage 0 analyzed at different time points (T) after loading into pectin gel (circles, bold line) or after culture in complete medium (DMEM + 20% fetal bovine serum; squares) or in Matrigel™ Basement Membrane Matrix (triangles). For (A) and (B), data are expressed as mean ± standard error mean of ≥3 different hAMSC donors. Since no significant alterations were observed starting from 1 h up to 168 h after incubation with pectin, the time point of 3 h was chosen for the analyses shown in panels (C) and (D); bars represent mean values. A P value of less than 0.05 was considered statistically significant: *P < 0.05, **P < 0.01, and ***P < 0.001 versus pectin gel.
The recovery and viability of hAMSCs cultured in complete culture medium decreased continuously up to 24 h after culture, likely due to the stress induced by the dual-syringe system used to mix the cell suspension, but then slowly began to increase until the end of the analysis (168 h; Fig. 1A). hAMSC recovery after treatment with Matrigel was similar to those after treatment with pectin gel (Fig. 1B).
Clinical use of MSCs often requires cell expansion to obtain a high number of cells for injection into patients. Thus, we analyzed the effects of immobilization within pectin gel on culture-expanded hAMSCs. As shown in Fig. 1, pectin gel did not alter the recovery (Fig. 1C) or viability (Fig. 1D) of hAMSCs at passage 1, thus subsequent experiments were performed using hAMSCs at passage 1.
Immunological Properties and Osteogenic Differentiation Capabilities of hAMSCs After Loading into Pectin Gel
Since the low immunogenicity and immunomodulatory properties of hAMSCs are well-documented 24 –32 and have been linked to their use in allogeneic settings and therapeutic effects in preclinical models of inflammatory-based diseases 33 , we next sought to investigate whether loading into pectin gel altered these properties. Pectin gel loading had no effect on the inability of hAMSCs to induce a T cell proliferative response (Fig. 2A). Also, as shown in Fig. 2B, hAMSCs and pectin gel-loaded hAMSCs were both able to significantly inhibit anti-CD3-activated PBMCs when cultured in contact with each other at the 2 highest hAMSC concentrations used (activated PBMC:hAMSC ratios of 1:0.5 and 1:1). No significant differences were observed in the ability to inhibit T cell proliferation between hAMSCs alone and hAMSCs loaded into pectin gel (Fig. 2B).
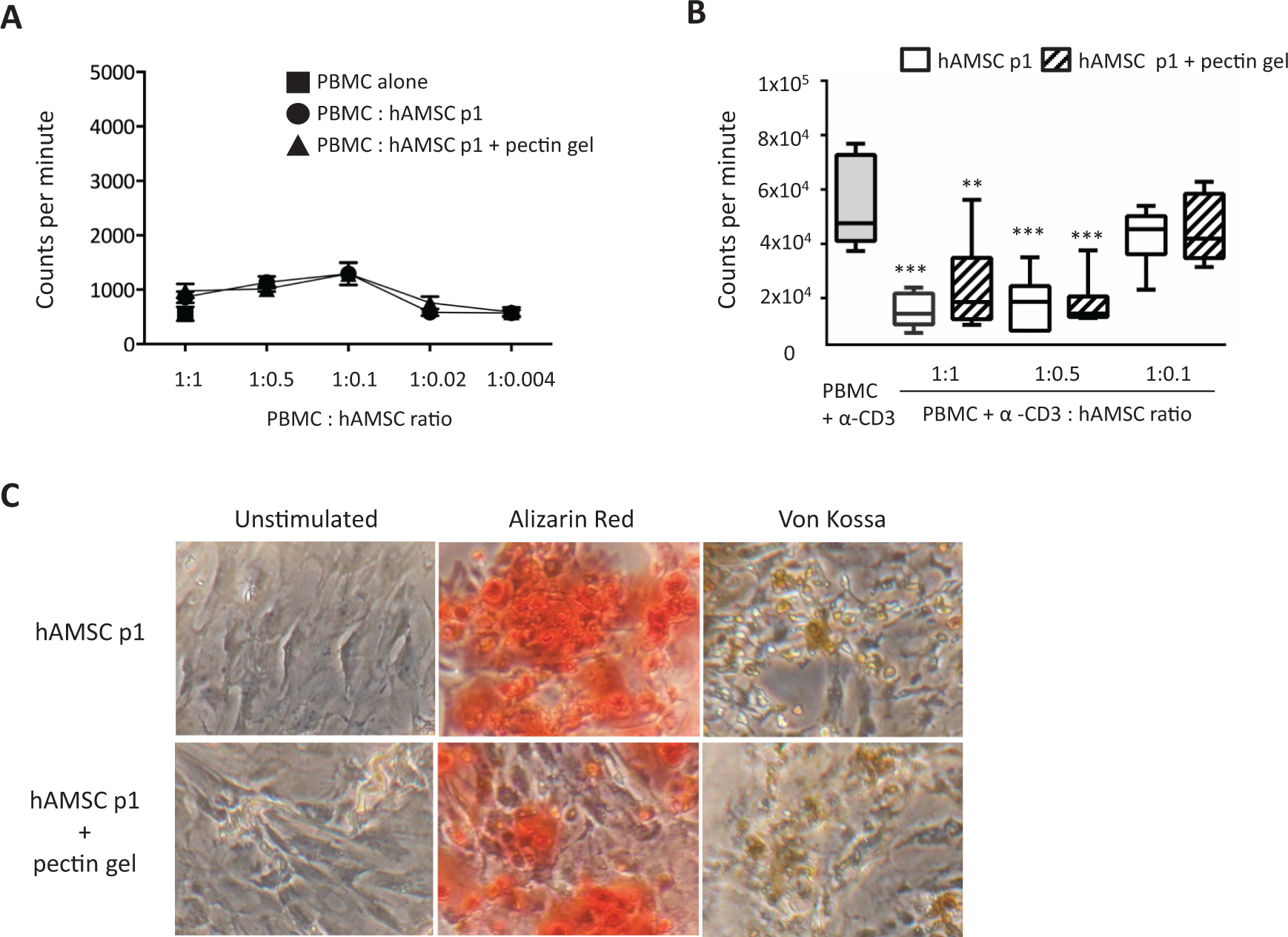
Human amniotic mesenchymal stromal cells’ (hAMSCs) functional properties after incubation with pectin gel. (A) Proliferation of peripheral blood mononuclear cells (PBMCs) alone, in presence of hAMSCs, at passage 1, or in presence of hAMSCs passage 1 (p1) recovered from pectin gel dissolution. Data are expressed as mean ± standard error mean (SEM) of ≥4 different hAMSC donors. (B) Proliferation of PBMCs activated with anti-CD3 in presence of hAMSCs at passage 1 or in presence of hAMSCs (p1) recovered from pectin gel dissolution. Data are expressed as mean ± SEM of ≥6 different hAMSC donors. Comparison of counts per minute (cpm) values between untreated and treatment hAMSCs and PBMCs activated with anti-CD3 was performed using a t-test. A P value of less than 0.05 was considered statistically significant (**P < 0.01 and ***P < 0.001 vs. PBMCs + anti-CD3). (C) hAMSCs (passage 1) alone or recovered from pectin gel dissolution were analyzed for their ability to differentiate toward the osteogenic lineage. hAMSCs were photographed 14 d after culture in culture media (unstimulated) or differentiation media (stained with Alizarin Red and von Kossa).
We also investigated the differentiation ability of hAMSCs toward the osteogenic lineage after loading within pectin gel using Alizarin Red and von Kossa stainings (Fig. 2C). There were no qualitative differences in the degree of differentiation between untreated hAMSCs and hAMSCs loaded into the gel.
Discussion and Conclusions
This is the first study investigating MSCs from the amniotic membrane of human term placenta as potential cell candidates in 3-D scaffolds using pectin gel cell carrier. MSCs from the amniotic membrane present unique characteristics; they are easily accessible in a noninvasive way, and they possess intrinsic immune-modulatory properties which do not require licensing with inflammatory stimuli as suggested with MSCs from bone marrow 34 . Thus, amniotic membrane MSCs could represent attractive candidates in tissue engineering strategies for ostechondral defect regeneration in combination to 3-D scaffolds.
Cell immobilization, and ultimately the limited exposure to cell culture media, may alter the properties of cells such as the cell viability. Recently, pectin gels have shown interesting properties toward bone regeneration 15 and promotion of mineralization 35 . Pectin microcapsules 15 and injectable gels 36 modified with arginylglycylaspartic acid (RGD)-containing peptides have demonstrated their ability to maintain viability of cells embedded within the gels, both in vitro and in vivo 36 . The properties of the pectin gels can be controlled by finely tuning the cross-linking process 37 . Among pectin gels, CaPs/pectin gels have been previously produced by exploiting the double effect of cross-linker and filler to produce biocomposite gels 38,39 or by incorporating nano-hydroxyapatite to support osteoblast growth 40 . The addition of pectin in bone cements, based on CaPs, has been shown to improve the adhesion and proliferation of human adipose tissue-derived stem cells 41 .
Herein, we sought to investigate whether hAMSCs and CaP/pectin gel that includes CaP as a biomimetic compound could be compatible partners in cell-based bioengineering strategies. Thus, the focus of this study was to define whether pectin gel did not alter viability and characteristics of amniotic cells.
When hAMSCs were encapsulated in the pectin gel, cell recovery dropped by 50% after 1 h of incubation, and the recovery remained stable (50%) at all considered time points. Since no further decrease of cell viability was observed during the incubation, the drop in cell viability after 1 h is likely due to a possible incomplete dissolution of the gel, thus causing an underestimation of the number of cells, as it was possible to observe fragments and deposits after the centrifugation step. The gel environment was able to support cell viability in agreement with the previous research on different pectin gel formulations 15,36,40,41 . Interestingly, the immunological properties of hAMSCs, that is, their inability to induce a T cell response and their ability to inhibit the proliferation of PBMCs activated with anti-CD3, were retained thus indicating that amniotic cells were able to produce the modulatory bioactive molecules even after loading into pectin gel. In addition, the presence of extracellular calcium deposits indicates a mature osteoblast cell function, supporting the hypothesis of the retention of the capability of hAMSCs to differentiate toward the osteogenic lineage. These aspects could be considered key features to exploit the potential of amniotic MSCs toward the development of novel osteochondral regeneration strategies, using a carrier that does not alter hAMSCs intrinsic properties, such as their immunological and differentiation properties. The possibility to embed hAMSCs within a gel is a fundamental achievement to expand the potential applications of these cells in tissue regeneration.
Footnotes
Acknowledgment
The authors would like thank Fondazione Poliambulanza Istituto Ospedaliero, Brescia (Italy), and the Cariplo Foundation (grant no. 2012-0842) for their support.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
The study includes human term placenta and peripheral blood samples from human subjects. All samples used in this study were obtained after informed consent from donors. Written informed consent to collect placenta and isolate human amniotic-derived cells was obtained from each single donor according to the guidelines of Ethics Committee (Comitato Etico Provinciale) of the province of Brescia.
Statement of Informed Consent
All samples used in this study were obtained after informed consent from donors. Written informed consent to collect placenta and isolate human amniotic-derived cells was obtained from each single donor according to the guidelines of Ethics Committee (Comitato Etico Provinciale) of the province of Brescia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Fondazione Poliambulanza Istituto Ospedaliero and the Cariplo Foundation (grant no. 2012-0842).
