Abstract
In the last decade, researchers and clinicians have reported that transplantation of bone marrow stromal cells (BMSCs) promotes functional recovery after brain or spinal cord injury (SCI). However, an appropriate scaffold designed for the injured spinal cord is needed to enhance the survival of transplanted BMSCs and to promote nerve regeneration. We previously tested a honeycomb collagen sponge (HC), which when applied to the transected spinal cord allowed bridging of the gap with nerve fibers. In this study, we examined whether the HC implant combined with rat BMSCs increases nerve regeneration in vitro and enhances functional recovery in vivo. We first evaluated the neurite outgrowth of rat dorsal root ganglion (DRG) explants cultured on HC with or without BMSCs in vitro. Regeneration of neurites from the DRGs was increased by BMSCs combined with HC scaffolds. In the in vivo study, 3-mm-long HC scaffolds with or without BMSCs were implanted into the hemisected rat thoracic spinal cord. Four weeks after the procedure, rats implanted with HC scaffolds containing BMSCs displayed better motor and sensory recovery than those implanted with HC scaffolds only. Histologically, more CGRP-positive sensory fibers at the implanted site and 5-HT-positive serotonergic fibers contralateral to the implanted site were observed in spinal cords receiving BMSCs. Furthermore, more rubrospinal neurons projected distally to the HC implant containing BMSCs. Our study indicates that the application of BMSCs in a HC scaffold in the injured spinal cord directly promoted sensory nerve and rubrospinal tract regeneration, thus resulting in functional recovery.
Keywords
Introduction
Spinal cord injury (SCI) continues to be a major health care burden. Even though its prevalence is not as high as that of other diseases(7, 29), the continued lack of effective treatment for the damage, especially treatment aiming toward a functional recovery, presents a considerable clinical challenge. Current treatment involves rigorous physiotherapy to preserve function, but not to restore areas where disability has occurred. Improvement of surgical management, inhibition of molecules that inhibit axonal regeneration, and transplantation of human stem cells for tissue renewal or neuroprotection have demonstrated promise in both laboratory and clinical settings (11, 13, 52), but widespread utilization of these techniques has yet to be achieved. The original theories concerning spinal cord repair were limited by the consensus that injured spinal cord tissue is not capable of meaningful clinical recovery. However, evidence from animal studies and human trials now suggests the potential for functional recovery (55), and strategies for cell therapy have focused on the restoration of the injured cord since the initial studies of stem cell biology two decades ago (33, 43). Bone marrow stromal cells (BMSCs) represent a potential cell source for therapeutic application in SCI patients (60). In Japan, a clinical study reported that intrathecal administration of cultured autologous BMSCs is safe and feasible for treatment of SCI (45, 46). A clinical advantage of such cells is that they may be obtained autologously, which may obviate the need for immunosuppression usually associated with transplantation. However, an obstacle that needs to be overcome is the relatively low viability of BMSCs implanted into the damaged spinal cord (34). A supportive extracellular matrix (ECM) has been demonstrated to be vital for effective, functional improvement after cell transplantation, and various methods using synthetic and natural structures have been explored (24, 30, 39). We previously demonstrated that neurites extended from dorsal root ganglia (DRG) in artificial ECM comprising a honeycomb collagen sponge (HC) in vitro, and that HC bridging between the stumps of the transected spinal cord enhanced nerve regeneration and functional recovery in rats (15).
We here propose that engraftment of BMSCs into HC scaffolds may further promote neural recovery. We hypothesize that the BMSCs will support regenerating axons with scaffolding to provide a directional matrix through which the axons can grow. Therefore, it should be possible to control axonal growth in a desired direction, which could eventually provide a method for designed regeneration. To test our hypothesis, we comparatively evaluated BMSC-engrafted HC scaffolds against HC scaffolds alone as a control in both in vitro DRG and in vivo spinal cord settings.
Materials and Methods
Preparation of Bone Marrow Stromal Cells
All animal experiments were performed following the approval of the Animal Study Committee of Tokyo Medical and Dental University and conformed to relevant guidelines and laws. The method for obtaining the BMSCs was described previously (3). BMSCs were extracted from female 11-week-old Fischer rats (Sankyo Laboratory Service, Tokyo, Japan) weighing 200–240 g. After one passage, the BMSCs were transduced with lentiviral vectors (pRRLsin.PPT.Th.CMV.MCS.Wpre) (12, 14) expressing green fluorescent protein (LV-GFP) with a multiplicity of infection of approximately 300 and cryopreserved in a storage solution (Cell Banker; Juji Field, Inc., Tokyo, Japan). BMSCs expressing GFP at passage 3 were used for the experiments. We evaluated potential differences in viability between nonlabeled and LV-GFP-labeled BMSCs using the Cell Counting Kit-8 (Dojindo Laboratory, Kumamoto, Japan) according to the manufacturer's protocol. There was no difference in cell number between nonlabeled and LV-GFP-labeled BMSCs at 3 days in vitro (data not shown).
Preparation of Honeycomb Collagen Sponge (HC)
Pieces of HC (3 × 3 × 2 mm; KOKEN Inc., Tokyo, Japan) (21) with a unidirectional pore size of 200–400 μm were presoaked in phosphate-buffered saline (PBS; Wako Pure Chemical Ind., Osaka, Japan) and centrifuged at 2,380 × g until no visible air bubbles existed in the pores. The HC pieces were then soaked overnight in PBS containing 1% fibronectin (Invitrogen, Carlsbad, CA, USA). After a PBS wash, we prepared DRG medium, which was neurobasal medium (Invitrogen) containing B27 supplement (Invitrogen), 0.5% l-glutamine, and 1% penicillin/streptomycin (Invitrogen) with 20 ng/ml nerve growth factor (NGF; Peprotech, Rocky Hill, CT, USA), and the HC pieces were soaked in the medium. For later stabilization of the HC pieces in culture plates, the wells in 12-well plates (BD Falcon, Franklin Lakes, NJ, USA) were filled with 2% agar (BM Equipment Co. Ltd., Tokyo, Japan) in PBS. We prepared a 5-mm-diameter hole in the center of the agar in each well, and a piece of HC was placed into the hole filled with DRG medium (15).
Cultivation of Adult Rat Dorsal Root Ganglia on HC with BMSCs
Lumbar DRGs were obtained from adult female Fischer rats (Sankyo Laboratory Service, Tokyo, Japan) and stored immediately in Liebovitz's L15 medium (Invitrogen) to prevent further damage arising from medium acidification during the preparation. The DRGs were cut in half between the proximal and distal roots under a microscope and washed twice in fresh L15 solution. To obtain efficient seeding of BMSCs into the HC pores, a low-pressure loading method was performed to distribute the BMSCs in the HC pores (53). Briefly, HC blocks were placed into a pressurizable glass chamber, and the air in the chamber was removed by a syringe to create a vacuum. The BMSCs were collected by centrifugation and resuspended in DRG medium. A BMSC suspension with a concentration of 2 × 105 cells per scaffold was slowly infused into the chamber, maintaining the low-pressure condition, and the HC blocks were allowed to soak in the cell suspension. The valve of the chamber was then released to normalize the pressure in the chamber. The BMSC-seeded HC blocks were left in the chamber for 10 min and then placed into the holes in the agar in the culture plates. The DRGs were carefully placed on the HC blocks in the wells of the culture plate, and the wells were filled with DRG medium. The medium was changed every 2 days and maintained for 10 days. At that time, the height of the HC blocks had increased from 2 mm to approximately 3 mm because of swelling in the medium.
Histological Evaluation of DRGs
The HC blocks were fixed in 2% paraformaldehyde (PFA; Wako Pure Chemical Ind.) in PBS after 10 days of in vitro culture, and 200-μm sections were carefully cut on a microslicer (Dosaka EM, Kyoto, Japan). After treatment with 0.2% Triton X-100 (Sigma-Aldrich, St. Louis, MO, USA) for 5 min, the sections were blocked with 5% normal goat serum (NGS; Vector Laboratories, Burlingame, CA, USA) and incubated with anti-neuronal class III β-tubulin (clone Tuj-1; 1:200; Covance, Princeton, NJ, USA) and anti-GFP polyclonal antibody (1:500; Molecular Probes, Eugene, OR, USA). The primary antibody was visualized using goat anti-mouse IgG conjugated to cyanine 3 (Cy3) (1:200; Jackson ImmunoResearch, West Grove, PA, USA) and goat anti-rabbit IgG Alexa 488 (1:400; Molecular Probes). Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen). Images were obtained on an AX 70 Olympus microscope with a digital camera (DP70; Olympus, Tokyo, Japan) connected to a computer with Olympus DP manager software. We measured a scale bar containing obtained images and the length of neuronal class III β-tubulin- positive neurites along the holes of the collagen sponges. The maximum length of the neurites from each DRG explant was evaluated from all sections containing neurites.
Scanning electron microscopy (SEM) was performed. Some samples were fixed with 2.5% glutaraldehyde (TAAB Laboratories Equipment Ltd., Berks, UK) for 24 h. These samples were postfixed with 2.0% (v/v) osmium tetroxide (TAAB Laboratories Equipment Ltd.) for 2 h. After dehydration in a graded series of ethanol and drying in a critical point drying apparatus (HCP-2; Hitachi, Tokyo, Japan) with liquid CO2, samples were sputter coated with platinum (Hitachi) and examined by SEM (S-4500; Hitachi).
Spinal Cord Injury and Cell Transplantation
For implantation, pieces of HC (1 × 1 × 3 mm) seeded with GFP-labeled BMSCs at a concentration of 3 × 104 cells per scaffold or pieces of control HC (i.e., HC scaffolds without cells) were prepared and cultured with Dulbecco's modified Eagle's medium (Invitrogen) and 10% fetal bovine serum (Invitrogen) for 3 days. Thirty-six adult female Fischer 344 rats (Sankyo Laboratory Service) weighing 130–160 g were used in the study. The rats were anesthetized by intraperitoneal injection of chloral hydrate (280 mg/kg; Junsei Chemical, Tokyo, Japan), and Eltacin (50 mg/kg; Fuji Pharma, Shizuoka, Japan) was injected intramuscularly prior to surgery. A laminectomy was performed at the T8 and T9 level, and the dura was incised longitudinally. The exposed spinal cord was hemisected at T9, followed by removal of 3 mm of hemicord on the right side. After hemostasis was achieved, an HC scaffold (HC group: n = 18) or HC scaffold combined with BMSCs (HC/BMSC group: n = 18) was implanted between the rostral and caudal spinal cord stumps. The muscles and skin were sutured in layers. The rats were given a subcutaneous saline injection and were allowed to recover in a warm cage. They received manual bladder expression twice a day until spontaneous urination was reestablished, and they were given subcutaneous saline injections and intramuscular injections of Eltacin (50 mg/kg) every day for 1 week.
Histology
Four weeks after surgery, the rats were deeply anesthetized with chloral hydrate (400 mg/kg) and transcardially perfused with a solution of 4% PFA in 0.1 M phosphate buffer (PB). The spinal cords were postfixed in 4% PFA in 0.1 M PB for 24 h and dehydrated in 30% sucrose (Wako Pure Chemical Ind.) in 0.1 M PB for 48 h. The 9-mm spinal cord segment centered on the implanted/injury site was dissected and embedded in 1.5% low-melting-temperature agarose (BM Equipment Co. Ltd., Tokyo, Japan) in PBS. The spinal cord segments were cut into 30-μm-thick horizontal sections using a cryostat (Leica Microsystems, Nussloch, Germany), and sections collected every 210 μm were used to assess the volume. A series of the sections was stained with Luxol fast blue and cresyl violet (LFB/CV; Wako Pure Chemical Ind.).
Determination of Tissue Volume
For histological evaluation of the tissue damage around the injury site, the total cross-sectional cord area and the area of spared myelin in the white matter were calculated using Scion Image software (Scion Corporation, Frederick, MD, USA). LFB was used to detect myelin; a foamy appearance, pale LFB color, and the invasion of inflammatory cells were all considered signs of demyelination. The tissue volume in each 9-mm cord segment was calculated using the following numerical formula:
(A, cross-sectional area, D, distance between sections = 210 μm, and N, total number of sections).
Immunohistochemistry
To characterize the distribution of transplanted BMSCs in the host injured spinal cord, sections from each experimental group were stained for the marker gene GFP and/or neuronal markers. After treatment with 0.2% Triton X-100 for 5 min, the sections were blocked with 5% NGS and incubated with rabbit polyclonal anti-GFP (1:1,000; Molecular Probes), mouse monoclonal anti-SMI31 (a marker for neurofilament M+H, 1:500; Covance), anti-glial fibrillary acidic protein (GFAP; a marker for astrocytes, 1:400; Sigma), or anti-myelin basic protein (a marker for myelin, 1:1,000; Covance) antibodies for 24 h. Primary antibodies were visualized using goat anti-mouse IgG conjugated to Cy3 (1:200; Jackson ImmunoResearch) for neuronal markers and goat anti-rabbit Alexa 488 (1:400; Molecular Probes) for GFP. Images were obtained on an AX70 Olympus microscope.
Quantification of Transplant Survival in the Injured Spinal Cord
To quantify transplant survival in the injured spinal cord, sections selected every 210 μm in the transplantation groups were stained with anti-GFP antibody, and the GFP-positive cells were counted in all selected sections.
The number of surviving cells was calculated as the sum of the GFP-positive cell counts in all selected sections in each animal.
Estimation of the Number of Neurofilament-, CGRP-, and 5-HT-Positive Axons in the Injured Spinal Cord
One series of mounted sections was incubated with SMI31 and rabbit polyclonal anti-GFAP (1:500, DAKO, Carpinteria, CA, USA) antibodies. Primary antibodies were visualized using goat anti-mouse IgG conjugated to Cy3 and goat anti-rabbit Alexa 647 (1:400, Molecular Probes). The implanted areas including the HC were observed in areas of the spinal cord where GFAP-positive cells were not located. The number of SMI31-positive fibers was determined by counting all labeled fibers crossing an imaginary line placed perpendicular to the centerline of the implanted area. The number of regenerated fibers was calculated as the sum of the SMI31-positive fiber counts in all selected sections in each animal. Another series of mounted sections was incubated with mouse monoclonal anti-GFAP and antibodies against either calcitonin gene-related peptide (CGRP) (1:1,000; Peninsula Laboratories Inc., San Carlos, CA, USA) or 5-hydroxytryptamine (5-HT; 1:1,000; ImmunoStar, Hudson, WI, USA) overnight at 4°C. Primary antibodies were visualized using goat anti-mouse IgG conjugated to Cy3 and anti-rabbit Alexa 647 (1:400; Molecular Probes) as the secondary antibody. The number of CGRP- or 5-HT-positive fibers was also determined as described above.
Retrograde Tracing
To assess the survival of spinal cord tracts, retrograde tracing with Fluoro-Gold (FG; Fluorochrome, Denver, CO, USA) was performed 4 weeks after transplantation (HC group: n = 6, HC/BMSC group: n = 6). A laminectomy was performed at T12, and 3 μl of 4% FG was injected bilaterally into the ventral, lateral, and dorsal columns of the white matter (total six points) using a glass pipette (Iwaki/Asahi Techno Glass, Tokyo, Japan). The brains and spinal cords were dissected 1 week after FG injection and dehydrated as described above. The cerebral cortex, brainstem, and T6-level spinal cord were cut into 30-μm-thick transverse sections, and one section every 300 μm was observed using an AX70 Olympus microscope. FG-positive neurons were counted in 2.4 mm of the cerebral cortex (eight slices), 1.5 mm of the brainstem (five slices), and 3.0 mm of the T6 spinal cord (10 slices).
Hindlimb Behavioral Tests
The Basso–Beattie–Bresnahan (BBB) locomotor rating score (4) and BBB subscores (28) were used to evaluate the recovery of hindlimb motor function. Two observers who were blind to the experimental procedures performed the BBB scoring once a week for 4 weeks. Footprint analysis was performed 4 weeks after injury. The hindpaws of the rats were inked, footprints were recorded on a paper runway with a length of 1 m and a width of 7 cm, and the limb rotation, stride length, and base of support were measured as described previously (32).
Sensory tests were also performed for mechanical and cold sensation 2 and 4 weeks after injury. The animals were placed in clear acrylic boxes (Ugo Basile Biological Research Apparatus, Comerio, Italy) and allowed to acclimate to the testing environment. The boxes were then placed on an elevated perforated plastic surface for a minimum of 30 min prior to all behavioral tests (17). A blind observer conducted the behavioral testing.
Mechanical sensitivity was measured by applying a series of calibrated von Frey filaments (0.02–15 g) to the plantar aspect of the hindpaw using the up–down method (6).
For determination of cold sensitivity, using a plastic tube connected to a 1-ml syringe (Terumo Corp., Tokyo, Japan) and without touching the skin, 100 μl of acetone (Wako Pure Chemical Ind.) was applied to the plantar surface of the foot. Acetone was applied five times to each paw with an interval of at least 30 s, and brisk foot withdrawals in response to the acetone evaporation within 10 s after the application were considered to be positive. The number of positive responses was then recorded.
Statistical Analysis
All results are expressed as the mean ± SEM, even when nonparametric for clarity. The data were analyzed using GraphPad Prism5 (GraphPad Software Inc., San Diego, CA, USA) statistical software. The two groups (HC scaffold alone vs. HC scaffold + BMSCs) were compared using the unpaired two-tailed Student's t-test or the Mann–Whitney U-test. In the behavioral testing, the score at every time point was analyzed using a two-way ANOVA followed by Bonferroni post hoc test, and p < 0.05 was considered statistically significant.
Results
Neurites Extended From DRGs Into HC Pores with or Without BMSCs
Efficient expression of GFP in BMSCs was observed at passage 3 (Fig. 1A). The cells were distributed in the HC pores 3 days after seeding by the low-pressure loading method (Fig. 1B). Bioactivity in vitro was analyzed using lumbar DRGs seeded over HC pieces with or without BMSCs. The DRGs extended their neurites toward and into the porous structure of the honeycomb scaffolding (Fig. 2A). Neuronal class III β-tubulin (Tuj-1 clone)-positive neurites were observed in the tubular HC sections after 10 days in vitro (Fig. 2B). The neurites were measured from the base of the DRG residing on the surface of the HC scaffold to their furthest discernible extension. The average maximum extension was 733.3 ± 112.4 μm in the HC pieces and 1,992.7 ± 92.3 μm in the HC pieces with BMSCs. Thus, neurite growth was increased by over twofold in the HC pieces combined with BMSCs compared to the control HC pieces (p < 0.0001) (Fig. 2C). GFP-labeled BMSCs were located alongside the neuronal class III β-tubulin (Tuj-1 clone)-positive neurites and on the inner walls of the HC pores (Fig. 2D, E).
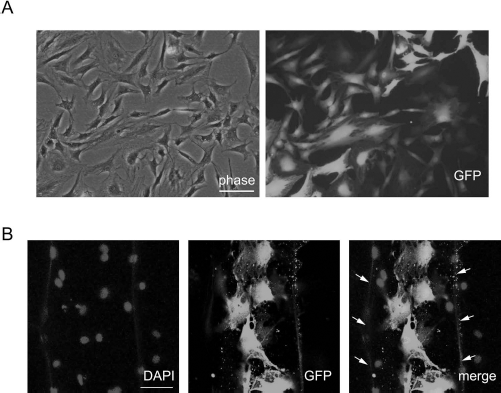
In vitro expanded BMSCs after lentiviral infection and engraftment of BMSCs in the honeycomb collagen sponge (HC). BMSCs were expanded and infected at P2 by GFP-gene-encoding lentiviruses. The cells were cultured and fixed at P3. GFP (A) was efficiently expressed in the BMSCs. The BMSCs were introduced into the HC by a low-pressure loading method and cultured for 5 days. The GFP-labeled cells (B) were attached to the wall of HC pores (arrows). Nuclei were counterstained with DAPI. Scale bar: 50 μm (A and B).

Scanning electron microscopy (SEM) micrographs of dorsal root ganglia (DRGs) on HC pieces and neurites extending from a DRG cultured with BMSCs. The DRG explant was attached to the HC scaffold and extended neurites toward and into the porous structure (arrows in A). Neuronal class III β-tubulin (Tuj-1 clone)-positive neurites were observed to enter the HC piece (B). The neurites were longer in the HC piece combined with BMSCs than in the HC piece without BMSCs (C). GFP-labeled BMSCs were distributed into the HC pores (green in D and E), and some of the cells were localized alongside the neuronal class III β-tubulin (Tuj-1 clone)-positive neurites (arrows in D). DAPI was used as a nuclear counterstain. Higher magnification view of the BMSCs showed extending processes along the neurites (E). Scale bar: 300 μm (A), 1 mm (B), 50 μm (D), and 20 μm (E). ***p < 0.0001, Mann–Whitney U-test (C).
Locomotor and Sensory Function Recovered After HC Scaffold Implantation with or Without BMSCs
The HC scaffold (HC group) or HC scaffold combined with BMSCs (HC/BMSC group) was implanted between the rostral and caudal spinal cord stumps. The BBB score on the injured side reached a plateau with an average score of 11.6 ± 0.2 at 3 weeks after injury in the HC group, whereas the BBB score was 12.6 ± 0.1 in the HC/BMSC group and 11.7 ± 0.1 in the HC group at 4 weeks after injury (p < 0.001) (Fig. 3A, B). Hindlimb coordination was significantly improved in the HC/BMSC group at 4 weeks after injury. In the BBB subscore, the HC/BMSC group displayed a better outcome than the HC group at 4 weeks (p < 0.05) (Fig. 3C). In addition, the stride length was significantly improved in the HC/BMSC group compared to the HC group (p < 0.05) (Fig. 3D). Both the base of support and external rotation angle appeared to improve in the HC/BMSC group, but the difference did not reach statistical significance (data not shown).

Locomotor and sensory recovery after HC implantation with and without BMSCs. The Basso–Beattie–Bresnahan (BBB) score at every time point was not significantly different between the HC and HC/BMSC groups by two-way ANOVA followed by Bonferroni post hoc test (A). However, in the comparison study of the BBB score at the final time point (4 weeks after injury), there was a significant difference (B). The HC/BMSC group presented a better outcome in the BBB subscore than the HC group at 4 weeks following SCI (C). The relative value for the stride length (injured side/contralateral side) in the footprint analysis was significantly improved in the HC/BMSC group (D). Cold sensation in response to acetone was observed in both groups at 4 weeks. There was a significant difference between the responses at 2 and 4 weeks in the HC/BMSC group (E). *p < 0.05, **p < 0.01, ***p < 0.001, unpaired two-tailed Student's t-test (B, D, and E), two-way ANOVA followed by Bonferroni post hoc test (C and E).
After the hemisection injury, hypoesthesia or anesthesia to mechanical and cold stimuli developed in the hindpaw on the injured side. The number of animals showing a response to the 4- to 8-g von Frey filaments in the HC/BMSC group (n = 12) increased from two (at week 2) to seven (at week 4), whereas the respective number of responsive animals increased from four to six in the HC group (n = 12). The response to acetone was also statistically increased in the HC/BMSC group 4 weeks after injury (p < 0.01) (Fig. 3E). Thus, the HC scaffold combined with BMSCs contributed to a partial recovery of hindlimb function.
HC Scaffold Implantation with BMSCs Did Not Affect the Estimated Tissue Volume but Increased Nerve Regeneration
At 4 weeks, the defect was filled with the HC scaffold, which contacted both cord stumps (Fig. 4A). We estimated the total lesion volume and amount of spared myelin in 9-mm-long horizontal sections stained with LFB/CV under light microscopy. The total tissue volume and cavity volume were not significantly different between the groups (data not shown). GFP-positive cells were observed in the implantation site of the HC/BMSC group (Fig. 4B). The average number of GFP-positive cells was 208 (minimum number of cells = 14, maximum number of cells = 711) in each animal. Regenerated fibers were also observed as SMI31-positive fibers in the GFAP-negative implanted area (Fig. 4B). A quantitative analysis showed that more SMI31-positive fibers were present in the HC/BMSC group than in the HC group (p < 0.001) (Fig. 4C, D).

HC scaffold implantation into the hemisected spinal cord and nerve regeneration in the HC scaffold with or without BMSCs. The implanted HC scaffold partially remained between the cord stumps 4 weeks after injury, but the staining intensity of luxol fast blue (LFB) was maintained in the cord adjacent to the HC implant (A). The immunohistological study showed that GFP-positive cells (green in B) had survived in the implantation site of the HC/BMSC group. The regenerated nerve fibers are shown as SMI31-positive fibers (arrows in B) in the GFAP-negative area. The number of SMI31-positive fibers was determined by counting all labeled fibers crossing imaginary lines placed perpendicular to the centerline of the implanted site (arrows in C). A higher number of SMI31-positive fibers was observed in the HC/BMSC group than in the HC group (D). Scale bar: 1 mm (A), 100 μm (B), 200 μm (C). ***p < 0.001, Mann–Whitney U-test (D).
BMSCs Increased the Number of CGRP-Positive Fibers in the Implanted Site and 5-HT-Positive Fibers Contralateral to the Implanted Site
To evaluate the growth of sensory fibers, CGRP-positive fibers were analyzed in the implanted spinal cord 4 weeks after implantation (Fig. 5A). The number of CGRP-positive fibers was higher in the HC/BMSC group than in the HC group at the implantation site (p < 0.01), but not contralateral to the implantation site (Fig. 5B). Furthermore, to examine the growth of serotonergic fibers related to functional recovery, we quantified the 5-HT-positive fibers. Few 5-HT-positive fibers were present in the implanted area, even in the HC/BMSC group (Fig. 5C). However, more 5-HT-positive fibers were observed contralateral to the implantation site (Fig. 5D); in particular, the number of 5-HT-positive fibers was higher in the HC/BMSC group than in the HC group (p < 0.01) (Fig. 5E). The nerve fiber distribution suggested that the transplanted BMSCs enhanced and attracted the growth of sensory afferents at the implantation site and serotonergic descending fibers contralateral to the implantation site.

Quantification of regenerated fibers expressing calcitonin gene-related peptide (CGRP) or 5-hydroxytryptamine (5-HT) in the HC with or without BMSCs. CGRP-positive fibers (arrows in A) were observed in HC scaffolds with GFP-positive cells (A). A quantitative analysis showed that the number of CGRP-positive fibers in the HC/BMSC group was significantly higher than that in the HC group at the implanted site (B). However, there was no significant difference contralateral to the implantation site (B). Few 5-HT-positive fibers (arrows in C) were observed in HC scaffolds with GFP-positive cells (C) at the implanted site. However, a quantitative analysis contralateral to the implanted site showed that more 5-HT-positive fibers (arrows in D) were present in the HC/BMSC group than in the HC group (E). Scale bar: 100 μm (A and C), 200 μm (D). **p < 0.01, Mann–Whitney U-test (B and E).
HC Implantation with BMSCs Increased Rubrospinal Axonal Density After Injury
The FG-labeled neurons represented the corticospinal tract, rubrospinal tract, and the short tract starting from the propriospinal neurons at the T6 segment. The red nucleus on the injured side contained a higher number of FG-positive cells in the HC/BMSC group than in the HC group (p < 0.05) (Fig. 6A, B). Thus, HC scaffold implantation with BMSCs led to axonal regeneration in at least the rubrospinal tract.

Quantification of retrograde neuronal tracing 4 weeks after implantation. Flurogold (FG)-labeled neurons were quantified in sections of the cortex, red nucleus, and T6 cord segment in both the implanted and contralateral sides (A). A higher number of FG-labeled neurons in the HC/BMSC group relative to the HC group was observed only in the red nucleus. In a representative section of a red nucleus in the HC/BMSC group, the FG-labeled neurons were present at both the implanted and contralateral side (B). Scale bar: 300 μm. *p < 0.05, Mann–Whitney U-test (A).
Discussion
We demonstrated that in vitro expanded BMSCs engrafted in a HC matrix enhanced neurite elongation in vitro and directly promoted sensory and rubrospinal tract regeneration in addition to functional recovery after SCI.
Recent studies have identified axonal recovery after SCI and have thus opened a relatively new field compared to the well-established regeneration of peripheral nerves. It remains unclear whether sufficient recovery of central nervous system tissue can occur to support meaningful recovery. For functional recovery, the damaged axons must regenerate to an adequate length and also have the potential to reconnect or relay to their original distal axons. Various artificial scaffolds have been developed and studied with or without cell therapy in SCI models (23, 38, 48, 51). We have already demonstrated that the HC matrix has the potential to aid the regrowth of axons after SCI (15). In addition to supporting regenerating axons along its entire length, the HC matrix also guides axonal regeneration through its serial tunnel structure. In the present study, the regenerated nerve fibers were clearly visible and easy to evaluate in the HC scaffold implant site. With regard to biodegrad-ability, part of the HC scaffold remained in the host cord 4 weeks after implantation (Fig. 4A). In previous studies, HC scaffolds implanted subcutaneously were visible after 8 weeks (24), and collagen filament implants applied in the transected spinal cord partly remained after 12 weeks (64). Further observation is therefore necessary to confirm that the implanted HC scaffold degrades completely and the role, if any, that the degradation or maintenance of the scaffold in the injured cord plays in functional recovery.
BMSCs supply trophic support and structural support for regenerating axons in the injured spinal cord (18, 19, 34, 35). One limitation to the use of BMSCs is that they do not survive long in the injury site after they have been implanted (1, 20, 34). For example, BMSCs transplanted in the sub-acute phase disappeared after 1 to 2 weeks in a rat contusion SCI model (20). In our study, the transplanted BMSCs survived for at least 4 weeks at the implanted site, and some cells were located along regenerated nerve fibers (Fig. 4B, C). In addition, the BMSCs clearly formed bundles along the nerve fibers into HC pores in vitro (Fig. 2E). These results suggest that BMSCs have nerve guidance ability and promote nerve regeneration both in vitro and in vivo.
Our in vivo study also demonstrated that implantation of an HC scaffold combined with BMSCs enhanced motor and sensory recovery compared to implantation of the HC scaffold alone. It should be noted that in this study, we used a two-group design (HC alone vs. HC + BMSCs) because we previously established that the HC scaffold alone provided functional benefit in this SC model (15), and the goal of the present study was thus to specifically probe the potential for BMSCs to enhance this effect, that is, to examine the effect of adding BMSCs to the HC scaffold. We did not include a BMSC-alone group, as the lesion zone where the scaffolds are placed is a cavity with no structure for suspended cells to engraft in, and thus suspended (nonscaffolded) cells placed in the cavity would likely be immediately washed away.
We initially hypothesized that the presence of surviving BMSCs may affect functional recovery after SCI. However, the number of surviving GFP-positive cells varied among animals in the present study, and there was no obvious correlation between the number of GFP-positive cells and the functional outcome 4 weeks after implantation (data not shown). Previous reports showed that transplanted BMSCs disappeared in the early phage of SCI, but provided an ECM including collagen fibrils or stimulated the production of growth factors, which resulted in the sparing of damaged tissues (18, 20, 34, 41, 42). In the present study, unexpectedly, the estimated tissue volume was not different between animals implanted with the HC scaffold engrafted with BMSCs and those implanted with the HC scaffold alone at 4 weeks after implantation. This difference between our findings and those of previous studies may be attributed to our use of an HC scaffold, which we previously showed to independently induce the regeneration of axons and functional connections, or our use of a hemisection injury model in contrast to the previous studies. Regardless of the lack of difference in tissue sparing, as mentioned above, inclusion of BMSCs in the scaffolds still enhanced functional recovery.
Our study thus suggests that the presence of sufficient surviving BMSCs in the early phase of SCI promotes eventual functional regeneration; we also consider that a longer time course of observation in our model (i.e., longer than 4 weeks) may still reveal a correlation between the number of surviving BMSCs and functional outcome by allowing more regenerating nerve fibers to reach their target neurons or axons, as the BBB scores in the HC/BMSC group still appeared to be increasing at 4 weeks. In particular, the number of SMI31-positive fibers at the implanted site of the HC/BMSC group was more than twice as high as that in the HC-alone group, which indicates that the transplanted BMSCs facilitated the regeneration of rubrospinal axons and sensory fibers to produce functional recovery. Regarding motor function, it has been reported that functional recovery after transplantation of human adult olfactory neural progenitors is correlated with rubrospinal axon regeneration and reinnervation (61). In this study, an increase in the number or extension of regenerated rubrospinal axons toward target neurons mediated by the surviving BMSCs in the HC scaffold may have thus resulted in improvement of motor functional recovery. Our observation that BMSCs enhanced the guided regeneration of axons in vivo and neurites in vitro agrees with previous findings that transplanted BMSCs provide a guidance effect to regenerating central and peripheral nervous system axons (2, 19, 36, 37, 57, 58). However, in spinal cord repair, mesenchymal stem cells (MSCs) are also thought to migrate to the injury site, modulate inflammation by secretion of various cytokines, and produce trophic factors to induce nerve regeneration (56). In particular, BMSCs produce various trophic factors such as NGF, brain-derived neurotrophic factor (BDNF), glial-derived neurotrophic factor, and vascular endothelial growth factor (9, 16, 31, 36, 59, 62), and the trophic factors induce different axonal responses depending on the phenotype of the receptor (22). Therefore, another possible explanation for the functional recovery observed in the present study is that trophic effects of the transplanted BMSCs mediated through secreted molecules could have affected the host spinal cord. Specifically, production of trophic factors by surviving BMSCs at the implanted site may have resulted in a higher number of CGRP-positive fibers at the implanted site and 5-HT-positive fibers at the contralateral site, while contributing to the observed enhancement of axonal regeneration. Regardless of whether the effects were exerted through direct contact or through secreted molecules, in our hemitransected SCI model, we consider that HC-engrafted BMSCs may have functioned more for a nerve guidance effect than for tissue preservation, particularly considering that the HC/BMSC group showed increased axonal regeneration without any difference in spared tissue volume relative to the HC-alone group.
BMSCs have been shown to differentiate into various phenotypes in vitro (5, 47, 63). BMSCs have also been suggested to differentiate into neural phenotypes upon implantation in host neural tissues, although such neural differentiation remains controversial (5, 10, 19, 27, 37). In the present study, we did not observe neuronal or glial differentiation of the surviving BMSCs in the host spinal cord at 4 weeks after implantation (data not shown). Furthermore, few surviving BMSCs expressed CD90 (data not shown), therefore suggesting that most of the surviving BMSCs did not maintain their undifferentiated, stem-like state. However, a subset of the surviving BMSCs expressed fibronectin (data not shown), which is a mesenchymal marker that has been shown to promote nerve growth (25) and to be expressed by undifferentiated donor BMSCs associating with axons in a contusion SCI model (19). It therefore appears that in the host tissue, the transplanted BMSCs do not undergo neural differentiation; rather, their therapeutic effects appear to be mediated either by donor cells that lose their undifferentiated, stem-like status to take on a phenotype with therapeutic effects on endogenous neural tissue components or by a subpopulation of donor cells that remains undifferentiated, providing a niche to enhance the proliferation, differentiation, and/or therapeutic effects of endogenous cells in the injured spinal cord (40).
MSCs can be derived from not only bone marrow but also adipose tissue (ASCs) or umbilical cord blood (UCB-MSCs). Both BMSCs and ASCs can be obtained from autologous tissues of SCI patients, thus avoiding immunological complications and ethical concerns (49). In recent comparative studies, transplanted ASCs were shown to modulate the SCI environment with increase in the BDNF expression to a higher extent than BMSCs (8, 65). In contrast to ASCs and BMSCs, UCB-MSCs are derived from perinatal tissues and are currently under initial evaluation for CNS repair (50, 56). UCB-MSCs were shown to have higher nerve regeneration and anti-inflammation activity than other types of MSCs in a comparative study in a canine SCI model (44). Although ASCs and UCB-MSCs thus offer some advantages, BMSCs may be the closest to clinical application for SCI because MSCs were originally obtained from the bone marrow, and numerous basic and clinical reports of applications of BMSCs for CNS repair have thus already been reviewed (26, 54, 60). In particular, culture techniques for BMSCs have already been optimized, and autologous BMSC transplantation has been confirmed as safe in SCI patients in previous clinical trials, thus establishing BMSCs as potentially neuroregen-erative somatic stem cells without tumorigenicity. Further in vivo studies under the same experimental conditions are needed to determine which type of MSC promotes the greatest extent of functional recovery after SCI.
In summary, we have shown that administration of an HC scaffold engrafted with BMSCs significantly enhanced functional recovery and axonal regeneration compared to treatment with the HC scaffold alone when evaluated at 4 weeks after SCI. Our in vitro and in vivo data also support that the BMSCs surviving in the lesion zone may exert their therapeutic effect by stimulating and guiding nerve fiber regeneration. A therapy that combines HC scaffolds with BMSCs for transplantation may therefore be a clinically effective approach to fill gaps or cavities in the injured spinal cord.
Footnotes
Acknowledgments
We are grateful to Prof. Y. Kuboki (Hokkaido University) for his advice and generous support. We thank Dr. P. Tsoulfas (University of Miami Miller School of Medicine) and the Miami Project Viral Vector Core for providing lentiviral vectors. We also thank D r. A. Hurtado (Kennedy Krieger/Johns Hopkins University) for advice regarding animal surgery. This work was supported in part by the Ministry of Health Labour and Welfare Sciences Research Grant, a Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science, and a Grant-in-Aid from the General Insurance Association of Japan. The authors declare no conflicts of interest.

