Abstract
After severe spinal cord injury, spontaneous functional recovery is limited. Numerous studies have demonstrated cell transplantation as a reliable therapeutic approach. However, it remains unknown whether grafted neuronal cells could replace lost neurons and reconstruct neuronal networks in the injured spinal cord. To address this issue, we transplanted bone marrow stromal cell-derived neural progenitor cells (BM-NPCs) in a rat model of complete spinal cord transection 9 days after the injury. BM-NPCs were induced from bone marrow stromal cells (BMSCs) by gene transfer of the Notch-1 intracellular domain followed by culturing in the neurosphere method. As reported previously, BM-NPCs differentiated into neuronal cells in a highly selective manner in vitro. We assessed hind limb movements of the animals weekly for 7 weeks to monitor functional recovery after local injection of BM-NPCs to the transected site. To test the sensory recovery, we performed functional magnetic resonance imaging (fMRI) using electrical stimulation of the hind limbs. In the injured spinal cord, transplanted BM-NPCs were confirmed to express neuronal markers 7 weeks following the transplantation. Grafted cells successfully extended neurites beyond the transected portion of the spinal cord. Adjacent localization of synaptophysin and PSD-95 in the transplanted cells suggested synaptic formations. These results indicated survival and successful differentiation of BM-NPCs in the severely injured spinal cord. Importantly, rats that received BM-NPCs demonstrated significant motor recovery when compared to the vehicle injection group. Volumes of the fMRI signals in somatosensory cortex were larger in the BM-NPC-grafted animals. However, neuronal activity was diverse and not confined to the original hind limb territory in the somatosensory cortex. Therefore, reconstruction of neuronal networks was not clearly confirmed. Our results indicated BM-NPCs as an effective method to deliver neuronal lineage cells in a severely injured spinal cord. However, reestablishment of neuronal networks in completed transected spinal cord was still a challenging task.
Keywords
Introduction
Spinal cord injury (SCI) induces local neural cell death and disruption of axonal pathways. Recovery is limited, since the central nervous system (CNS) environment deters axonal growth and regeneration through the actions of myelin inhibitors and astrocytes (14,45). Among the many experimental approaches to treat SCI, cell transplantation has the potential to repair or compensate for local spinal cord damage (5,15,27,32,44).
Bone marrow stromal cells (BMSCs) constitute a possible source of cells for autologous transplantation. They can be obtained from patient bone marrow aspirates and are readily expanded in vitro, which has made them a suitable candidate for clinical applications (9,11,34). We have established a method in which neural progenitor cells can be induced from BMSCs by introduction of the Notch-1 intracellular domain (NICD) followed by culturing using the neurosphere method (10,17). These progenitor cells, that is, bone marrow-derived neural progenitor cells (BM-NPCs), successfully formed spheres that highly expressed markers related to neural progenitors. When BM-NPCs were transplanted into a rat stroke model, they were shown to differentiate into neuronal cells, reconstruct synapses with host neurons, and lead to functional recovery of the animals (17).
In the present study, we newly applied BM-NPC transplantation to a rat model of complete spinal cord transection. The aim of this study is to confirm selective neuronal differentiation of the grafted BM-NPCs in the injured spinal cord and to achieve functional recovery. A possible means of achieving recovery from the injury would be the reconstruction of disrupted neuronal circuits between grafted cells and endogenous surrounding neurons, as suggested elsewhere (1). We employed immunohistochemistry to confirm neuronal differentiation of the grafted cells in the injured spinal cord. Synaptic formation of the BM-NPCs was evaluated with synaptophysin and postsynaptic density (PSD)-95. Retrograde tracing with fluorogold (FG) was utilized to see whether the BM-NPCs extended neurites across the transected portion of the spinal cord. We also used functional magnetic resonance imaging (fMRI) of the brain using blood oxygenation level-dependent (BOLD) contrast (12,13,30,40) to test sensory recovery and detect reestablishment of ascending neurotransmission across the injury. Behavioral analysis was included to evaluate locomotor recovery.
Materials and Methods
Preparation of Marrow Stromal Cells and Neural Induction
All animal experiments were approved by the Animal Studies Ethics Committee of Tohoku University Graduate School of Medicine. Experimental procedures are presented in Figure 1. Numbers of animals used for each experiment were listed in Table 1. Rat BMSCs were isolated from adult female 10-week-old Wistar rats (CLEA Japan, Inc., Tokyo, Japan) according to methods described previously (4). Cells were maintained in α-minimum essential medium (α-MEM; Sigma, St. Louis, MO, USA) containing 10% fetal calf serum (FCS; Hyclone, Inc., Logan, UT, USA) and kanamycin (Wako Pure Chemical industries, Ltd., Osaka, Japan) at 37°C with 5% carbon dioxide. Next, the cells were transfected with a vector (pCI-neo-NICD) containing the mouse NICD (10). The NICD cDNA coded for a transmembrane region that included a small fragment of extracellular domain followed by a sequence encoding the entire intracellular domain of mouse Notch (initiating at amino acid 1,703 and terminating at the 3′-untranslated sequence).

Experimental procedure. See Materials and Methods for detailed information. fMRI, functional magnetic resonance imaging.
The Number of Animals per Experiments

BBB, Basso, Beattie, Bresnahan; BM-NPCs, bone marrow-derived neural progenitor cells; fMRI, functional magnetic resonance imaging.
This fragment was subcloned into a pCI-neo vector (Promega, Madison, WI, USA) and was transfected with BMSCs using Lipofectamine LTX (Invitrogen, Carlsbad, CA, USA) and selected using G418 (Invitrogen) for 5 days according to the manufacturer's instructions (10).
Induction of BM-NPCs
After G418 selection, rat NICD-transfected cells were washed and cultured in α-MEM containing 10% FCS for 2 days for recovery. The efficacy of NICD transfection was 98.8 ± 0.8%, which is consistent with the previous report (10). During the expansion and recovery of transfected cells after G418 selection, green fluorescent protein (GFP) lentivirus (provided by Dr. D. Trono, Lausanne, Switzerland) was added to the culture medium for labelling purposes (29,39). Efficacy of GFP transfection was calculated three times. After recovery, the cells were washed and cultured in neurobasal medium supplied with B27 supplement (Invitrogen), 20 ng/ml of basic fibroblast growth factor and epidermal growth factor (both R&D Systems, Minneapolis, MN, USA) (17) at a cell density of 100,000 cells/ml on low cell-binding dishes (Nalgene Nunc, Rochester, NY, USA). After 8 days, generated spheres, namely BM-NPCs, were resuspended in Neurobasal medium to a concentration of approximately 30,000 cells/ml and transplanted to the injured rat spinal cord.
Immunocytochemistry
Spheres were fixed with 4% paraformaldehyde in 0.1 mol/L phosphate-buffered saline (PBS; both from Wako Pure Chemical Industries), collected by centrifugation, embedded in optimal cutting temperature (OCT) compound (Sakura Finetek Japan, Tokyo, Japan), and cut into 10-μm-thick sections using a cryostat (CM1850; Leica, Wetzlar, Germany). Samples were incubated with blocking solution containing 5% normal goat serum (Vector Laboratories, Burlingame, CA, USA), 0.1% Triton X-100 (Sigma), and 0.3% bovine serum albumin (BSA; Sigma) in 0.1 mol/L PBS at room temperature for 30 min. Samples were then incubated with primary antibodies in blocking solution overnight at 4°C. After three washes with 0.1 mol/L PBS, samples were incubated with secondary antibodies for 2 h at room temperature, followed by counterstaining with 4′,6-diamidino-2-phenylindole (DAPI) (for nuclear staining, 1:500; Sigma). The following primary antibodies were used for immunocytochemistry: anti-sex-determining region Y box 2 (Sox2; rabbit IgG, dilution 1:5,000; Chemicon, Temecula, CA, USA), anti-neurogenic differentiation (NeuroD; rabbit IgG, 1:200; Chemicon), anti-nestin (mouse IgG, 1:400; BD Pharmingen, San Jose, CA, USA), and anti-musashi (rabbit IgG, 1:200; Millipore, Billerica, MA, USA), anti-neuron-specific nuclear antigen (NeuN) (mouse IgG, 1:200; Chemicon), anti-glial fibrillary acidic protein (GFAP) (mouse IgG, 1:300; Sigma-Aldrich), and anti-oligodendrocyte marker 4 (O4; mouse IgM, 1:20; Millipore). These primary antibodies were detected with Alexa 488-conjugated anti-rabbit IgG or anti-mouse IgG antibodies (Molecular Probes, Invitrogen, Eugene, OR, USA) or Alexa 546-conjugated anti-rabbit IgG (1:500; Molecular Probes) or biotin-conjugated anti-mouse IgM (1:500; Jackson ImmunoResearch, West Grove, PA, USA) and streptavidin Alexa Fluor 488 (1:500; Molecular Probes) and streptavidin Alexa Fluor 680 (1:200; Molecular Probes). Percentages of immunopositive cells were calculated by comparing the cell numbers with the number of DAPI-positive cells. Cells in five fields, each including 100–500 cells, were counted in three independent cultures. Results were averaged and expressed as mean ± SEM.
Complete Transection of the Midthoracic Spinal Cord (T9)
Adult, 10-week-old female Wistar rats weighing 200 ± 20 g underwent complete transection of the spinal cord at the midthoracic level. Under isoflurane anesthesia (Wako Pure Chemical Industries), a laminectomy was performed at the T8–9 level. The spinal cord was exposed and transected completely using microscissors. Possible remaining adhesions were cut with a scalpel, and the rostral and caudal stumps were carefully lifted to verify complete transection. The dural incision was left open. Muscle and skin were sutured separately. The urinary bladder was emptied manually twice daily during the first week and once daily thereafter for 8 weeks.
Transplantation of BM-NPCs
Nine days after SCI rats were randomly assigned to groups receiving vehicle (vehicle group; n = 14) or induced neural progenitor cells (BM-NPC group; n = 10). Based on a previous report, the timing of transplantation was chosen to avoid delivering cells in an acute inflammatory stage following the injury or in chronic stage in which glial scar tissues would hinder regeneration of axons (31). The numbers of animals used in each experiment are shown in Table 1. Rats were reanesthetized and the thoracic spinal cord was carefully reexposed. Four injections were made with a depth of 1 mm, at 2 mm rostral and caudal of the lesion, and 0.5 mm left and right from the midline. At each site, 2.5 μl of cell suspension or vehicle (α-MEM; Sigma) was infused stereotactically using a Hamilton microsyringe attached to a glass micropipette at the rate of 0.5 μl/min (Hamilton, Reno, NV, USA). A total of 300,000 BM-NPCs were delivered to the spinal cord. The needles were left in the place for 1 min following each injection to prevent cells leaking from the site (2).
Immunohistochemical Analysis
Eight weeks after SCI, animals were anesthetized with an overdose of pentobarbiturate (Wako Pure Chemical Industries) and perfused transcardially with 4% paraformaldehyde in 0.1 mol/L PBS. Spinal cords were removed and embedded in OCT compound, and axial or sagittal slices were cut. Each spinal cord slice was cut into 10-μm sections using a cryostat. In three animals in each group, sagittal sections were used for neurofilament staining. The other immunohistochemical analyses were performed with axial slices. For immunostaining, the sections were washed with PBS and incubated with 5% normal goat serum, 0.3% Triton X-100, and 0.3% BSA in PBS (blocking solution) at room temperature for 30 min to block nonspecific binding. The slides were incubated with primary antibodies diluted in the blocking solution and incubated overnight at 4°C. After three washes with PBS containing 0.05% Triton X-100, the slides were incubated with secondary antibodies for 2 h at room temperature, followed by counterstaining with DAPI, which was diluted in PBS containing 0.1% Triton X-100. Sections were immunolabeled with the following primary antibodies: anti-neuron-specific class III β-tubulin (antibody name: Tuj-1; mouse IgG, 1:200; Sigma-Aldrich), anti-NeuN (mouse IgG, 1:200; Chemicon), anti-GFAP (mouse IgG, 1:300; Sigma-Aldrich), anti-O4 (mouse IgM, 1:20; Millipore), anti-synaptophysin (mouse IgG 1:1000; Chemicon), anti-PSD-95 (mouse IgG2a 1:100; Chemicon), anti-neurofilament (rabbit IgG, 1:200; Chemicon), and anti-GFP (chicken IgG, 1:1,000; Abcam, Philadelphia, PA, USA). Secondary antibodies were anti-mouse Alexa Fluor 568 (1:500; Molecular Probes), biotin-conjugated anti-chicken IgG (1:200; Jackson ImmunoResearch), anti-rabbit Alexa Fluor 568 (1:500; Molecular Probes), and streptavidin Alexa Fluor 488 (1:500; Molecular Probes). After immunolabeling, the samples were inspected under a confocal microscope system (C1si; Nikon, Tokyo, Japan). Using three animals in the BM-NPC group, transplanted cells in 20 fields in each cryosection were counted to confirm the survival of GFP-positive transplanted cells and evaluated the differentiation of BM-NPCs in the injured spinal cord. Results were averaged and expressed as mean ± SEM.
Tracing Study
To label regenerated axons, FG (Fluorochrome, Denver, CO, USA) was injected into the spinal cord 4 days before the rats were killed (41). Using four rats in each group, 4% FG was injected into the spinal cord 10 mm caudal to the transected site over a period of 3 min using a microsyringe (24). After transcardiac perfusion, sections in the axial plane in the thoracic spinal cord including rostral and caudal ends of the transected site, cervical spinal cord, and the brain were stained with anti-FG (rabbit IgG, 1:200; Millipore) as a primary antibody and Alexa 568-conjugated anti-rabbit IgG (1:500, Molecular Probes) as a secondary antibody. Numbers of FG-positive grafted cells were counted at the rostral boundary of the transected spinal cord up to 5 mm from the transected stump. Results were averaged and expressed as mean ± SEM.
Locomotor Scale
Motor function in the hind limbs of all animals (n = 24) was assessed using the Basso, Beattie, Bresnahan (BBB) locomotor rating scale on the day after injury and each week for 8 weeks after injury (6). Hind limb function was scored from 0 (flaccid paralysis) to 21 (normal gait) as a blind basis. The BM-NPC and vehicle groups were compared using multiple measurement analysis of variance (ANOVA) followed by Tukey's test. All values are given as mean ± SEM. For comparison, we also applied Mann– Whitney U test to evaluate BBB locomotor scale. A value of p < 0.05 was considered statistically significant.
Functional MRI
Animal Preparation
Rats subjected to spinal cord transection and BM-NPC transplantation (n = 6) or vehicle injection (n = 6) underwent brain fMRI 8 weeks after injury, as described (25,40,42). Rats were first anesthetized with isoflurane (2.5% during induction and intubation) mixed with oxygen (30%) and air (70%). Rats were intubated and mechanically ventilated using a rodent ventilator (SAR-830AP Ventilator; CWE, Ardmore, PA, USA). A pair of small needle electrodes (NE-224S; Nihon-Koden, Tokyo Japan) was implanted subcutaneously in the left hind limb of each animal to deliver electrical stimulation. To confirm correct placement of the electrodes, a short sequence of current pulses (0.5 mA) was applied outside the magnet to evoke light muscle twitches. Next, a bolus of α-chloralose (20 mg/kg) (Sigma-Aldrich) was injected through the tail vein catheter, and 10 min later, the isoflurane was discontinued. Anesthesia was continued with α-chloralose infusion (20 mg/kg/h), and pancuronium bromide (2 mg/kg/h; Sigma-Aldrich Japan, Inc., Tokyo, Japan) was added for muscle relaxation. Rectal temperature was monitored and maintained at 37 ± 0.5°C using a water-circulating pad (CLEA Japan, Inc.) during the experiment.
fMRI Recordings
All MRI data were acquired using a 7T Bruker PharmaScan system (Bruker Biospin, Karlsruhe, Germany) with a 38-mm diameter birdcage coil. Prior to all MRI experiments, we first performed global magnetic field shimming inside the core and later completed shimming at the region of interest (ROI) by using a point-resolved spectroscopic protocol (37). Line width (full width at half maximum) at the end of the shimming procedure ranged from 15 to 20 Hz in the ROI (~300 μl). For the rat experiment, BOLD signals were obtained using gradient echo planar imaging (EPI) with the following parameters: repetition time (TR) = 1500 ms, echo time (TE) = 15 ms, spectral band width (SBW) = 250 kHz, field of view (FOV) = 25 × 14 mm2, matrix size = 125 × 70, number of slices = 7, slice thickness = 1 mm, slice gap = 0 mm, number of volumes = 427, and number of dummy scans = 4.
Electrical Stimulation
A block design paradigm consisting of 10 blocks was employed, in which each block comprised 13 image packages of stimulation followed by 27 image packages of the resting condition. Electrical pulses were produced using a generator (SEN-3401; Nihon Koden) and an isolator (SS-203J, Nihon Koden). Pulsed currents of 10.0 mA with 0.3 ms duration and a constant frequency of 3 Hz were first delivered to the left hind limb of animals.
fMRI Data Analysis
Using statistical parametric mapping software (SPM2, Wellcome Department of Cognitive Neurology, London, UK), we normalized an individual rat's brain to the rat brain atlas template using the T2-weighted images (38). Spatial smoothing was performed using a Gaussian kernel of 0.6 mm full width at half maximum. Single-subject analysis was performed with a critical T value for each voxel calculated for the significance level of p < 0.001. In response to the hind limb stimulation, volumes of significant clusters in cortex were compared between the groups using the Student's t test, after normal distribution of the data sets were confirmed using Kolmogorov–Smirnov test. We counted fMRI signals in bilateral somatosensory cortex. Activations were counted separately in the hind limb territory of the primary somatosensory cortex as well as the cortical areas medial and lateral to the hind limb area as defined in the rat atlas (36). Time course of BOLD signals (%) was depicted for the BM-NPC transplantation groups using a voxel [2.6, −2.0, −1.0] located in the original hind limb territory of primary somatosensory cortex (36). A single time course was created by averaging across all stimulation periods as described elsewhere (35). All values are given as mean ± SEM.
Results
Grafted BM-NPCs Survived and Differentiated Into Neuronal Lineage Cells in the Injured Spinal Cord
In neurosphere culture, the NICD transfected BMSCs formed spheres. We examined the expression of nestin, NeuroD, Sox2, and musashi to determine whether these spheres contained cells with neural progenitor cell markers. Spheres contained high percentages of cells positive for nestin (62.5 ± 2.1%), NeuroD (82.3 ± 2.1%), Sox2 (87.8 ± 1.4%), and musashi (81.9 ± 1.4%) (Fig. 2), while none of the cells were positive for NeuN (not shown). In addition, spheres did not contain cells positive for GFAP and O4 (not shown) suggesting that, consistent with the previous report, cells committed to the glial fate were not present in the sphere (17,28).

Induction of bone marrow-derived neural progenitor cells (BM-NPCs). (A) A phase contrast microscopy demonstrating BM-NPCs forming spheres on a low cell-binding dish. (B–E) Immunofluorescence images. Expression of nestin (B), neurogenic differentiation (NeuroD) (C), sex-determining region Y box 2 (Sox2) (D), and musashi (E) was confirmed. DAPI, 4′,6-diamidino-2-phenylindole. Scale bars: 50 μm.
Next, BM-NPCs were transplanted into the injured spinal cord. Over 7 weeks after transplantation, no tumor formation was observed in any of the 10 spinal cords by inspection or histological analysis. Continuity of the spinal cord parenchyma was confirmed macroscopically and by neurofilament immunohistochemistry (Fig. 3A, B). Grafted cells were recognized in vivo by positive labeling of GFP. After transplantation of BM-NPCs 2 mm rostral and caudal to the transected portion of the spinal cord, grafted cells were confirmed to be located as far as 6 mm from the center of the injury (Fig. 3C–F). The total numbers of GFP-labeled cells were 8.02 ± 0.71 × 103 in the injured spinal cord. Efficacy of GFP transfection was 45.0 ± 4.4% in vitro. Accurate assessment of donor cell survival can only be pursued by counting all of the GFP signals in every spinal cord section. In this sense, there is a limitation of assessing the true measure of donor cell survival in our count, but the ratio of transplanted cells that survived in the injured spinal cord was estimated to be roughly 5.9%.
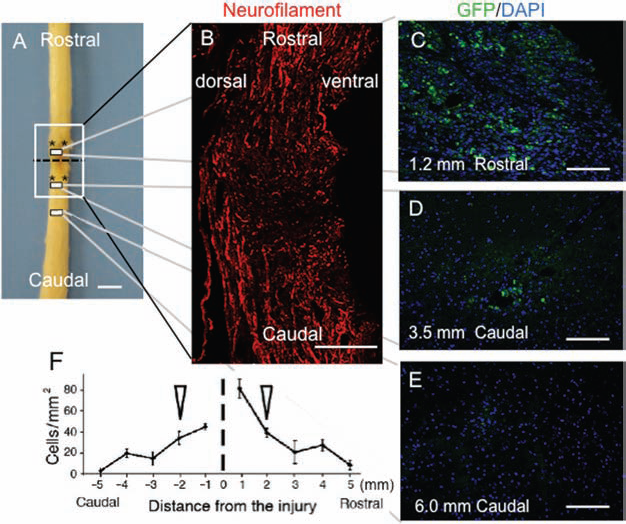
Transplanted bone marrow-derived neural progenitor cells (BM-NPCs) in the injured spinal cord 8 weeks after transection. (A) Schematic drawing of a representative spinal cord from the BM-NPC group showing sites of transection (dotted line) and transplantation (*). (B) Representative longitudinal section stained for neurofilaments corresponding to the white box in (A), showing anatomical continuity of spinal cord parenchyma at the injury site. (C–E) Distribution of green fluorescent protein (GFP)-positive transplanted cells at sites 1.2 mm rostral (C), 3.5 mm caudal (D), and 6.0 mm caudal (E) to the injury center in the dorsal spinal cord. Positions of the panels are indicated by small boxes in (A). (F) Numbers of GFP-positive cells between 5 mm caudal and 5 mm rostral to the injury center. The dotted line and arrowheads indicate the transected portion and the site of cell transplantation, respectively. Note that the GFP-positive cells were incorporated into the host spinal cord. DAPI, 4′,6-diamidino-2-phenylindole. Scale bars: 2 mm (A), 250 μm (B), 100 μm (C–E).
Importantly, BM-NPCs survived and showed low capacity for differentiating into astrocytes in the injured spinal cord. The frequencies of βIII tubulin (antibody Tuj-1)- and NeuN-positive cells among GFP-positive cells were 77.2 ± 2.6% (Fig. 4A–E) and 36.0 ± 3.2% (Fig. 4F–I), respectively. Nestin-positive cells were observed among the GFP-positive cells at the ratio of 9.7 ± 2.0% (Fig. 4J–M), while the ratio of GFAP-positive cells was 2.9 ± 0.9% (Fig. 4N–Q). O4 positive cells could not be detected (not shown). These observations suggested that the majority of the transplanted BM-NPCs became neuronal markerpositive cells. Moreover, the frequency of nestin-positive cells, which was 62.5 ± 2.1% in vitro and 9.7 ± 2.0% in vivo, indicated advancement of differentiation of the transplanted cells within the injured spinal cord, as nestin is potential indicator of neural differentiation (43). Spheres contained no cells positive for NeuN, while the frequency of NeuN-positive cells among GFP-positive cells increased in vivo, which also supported differentiation of the grafted cells. Transplanted cells were also found to express the synaptic marker synaptophysin (Fig. 5). Among GFP-positive cells, 24.2 ± 2.2% were positive for synaptophysin. It was noted that synaptophysin was localized predominantly next to GFP-positive transplanted cells in a punctate pattern (Fig. 5). To further assess the synaptic formation between the transplanted cells and the host cells, immunohistochemistry for PSD-95 was performed (8,22). PSD-95 labeling was adjacent to the synaptophysin-positive signal in the transplanted cells (Fig. 6).

Differentiation of green fluorescent protein (GFP)-positive bone marrow-derived neural progenitor cells (BM-NPCs) in the spinal cord 8 weeks after injury. (A–Q) Cells labeled with neuron-specific class III β-tubulin (antibody: Tuj-1) (A–E) and neuronal nuclei (NeuN) (F–I) were encountered more frequently than those labeled with nestin (J–M) or glial fibrillary acidic protein (GFAP) (N–Q). Arrowheads, positively labeled cells. Arrows, GFP-positive and Tuj-1-negative cells. DAPI, 4′,6-diamidino-2-phenylindole. Scale bars: 20 μm.

Synaptophysin expressed around GFP-positive bone marrow-derived neural progenitor cells (BM-NPCs). Photomicrographs depicting synaptophysin (A), DAPI (B), GFP (C), and a merged image (D) staining of transplanted cells in the spinal cord. This finding suggests the synapse formation between the transplanted cells and host cells. Arrowheads indicate positively labeled cells. Abbreviations are as in Figure 4. Scale bars: 20 μm.

Postsynaptic density (PSD-95) localized adjacent to synaptophysin on GFP-positive bone marrow-derived neural progenitor cells (BM-NPCs). Photomicrographs depicting GFP (A), PSD-95 (B), DAPI (C), and synatophysin (D) staining of cells in the spinal cord. This finding suggests synapse formation between the transplanted cells and host cells. Arrowheads indicate positively labeled cells. Abbreviations are as in Figure 4. Scale bars: 50 μm.
Fluorogold Tracing Detected Extension of Neurites From BM-NPCs Across the Transected Site in the Spinal Cord
Four days before the animals were sacrificed, we injected the retrograde tracer FG 10 mm caudal to the transected site, so that FG would be taken up through the terminals of the regenerated neurites (Fig. 7A). Strikingly, in the BM-NPC group, we detected FG-labeled grafted cells in the rostral as well as caudal spinal cord in all four rats (Fig. 7B–G). The presence of cells double positive for FG and GFP rostral to the transected portion indicated that the grafted cells extended their neurites across the transected portion to reach the caudal spinal cord. The site of injection was carefully examined to ensure that no FG could have reached the transected portion by diffusion (24). The number of cells labeled with FG and GFP at the rostral boundary of the transected spinal cord was 5.17 ± 0.75/mm2 in the BM-NPC group. FG-labeled BM-NPCs were located rostrally 1.8 ± 0.18 mm from the transection stump to a maximum of 4.6 mm. In the vehicle group, no FG-labeled cells were found in the spinal cord rostral to the transected site. In any of the injured animals with or without treatments (n = 4, each group), no FG-labeled cells were found in the cervical spinal cord or in the brain sections.

Expression of the retrograde tracer Fluorogold (FG) injected 10 mm caudal to the transected site of the spinal cord. (A) Schematic drawing of a representative spinal cord taken from the bone marrow-derived neural progenitor cell (BM-NPC) group, showing sites of FG injection (red arrow). Red dotted lines indicate the supposed routes by which FG was transported from the terminals of extended neurites and crossed the transected portion. (B–G) Green fluorescent protein (GFP)-positive BM-NPCs, transplanted rostral (B–D) and caudal (E–G) to the injured site, were also positive for FG in representative sections obtained 1.6 mm rostral and 0.4 mm caudal to the transected site. Abbreviations, schematic drawings, and arrowheads are as described in Figures 2 and 3. Scale bars: 2.0 mm (A) and 20 μm (B–G).
Behavioral Analysis Indicated Improved Hind Limb Locomotor Function After BM-NPC Transplantation
BBB locomotor scores for the BM-NPC and vehicle groups were determined each week for 8 weeks after injury (Fig. 8). Improvements in hind limb motor function were significantly greater in the BM-NPC group than in the vehicle group after 2 weeks (p < 0.01) and over 3–8 weeks (p < 0.001, repeated measures ANOVA followed by post hoc Tukey's test). Mann–Whitney U test also indicated significant functional recovery between 2 and 8 weeks (Fig. 8). Eight weeks after the injury, averaged BBB scale of injured animals was 3.31 in the control group while 5.80 in BM-NPCs transplantation group. Following the treatments, rats showed symptoms of recovery; they showed extensive movements of the three joints in the hind limbs. The difference between these groups was statistically significant (p < 0.001, post hoc Tukey's test; p < 0.01, Mann–Whitney U test).

Basso, Beattie, Bresnahan (BBB) locomotor scale. Open-field locomotor hind limb function of all rats was tested on the day after injury and then weekly for 8 weeks postoperatively. Results are shown separately for groups transplanted on day 9 with bone marrow-derived neural progenitor cells (BM-NPCs) (n = 10) or injected with vehicle (n = 14). Repeated measures ANOVA followed by the post hoc Tukey's test as well as Mann–Whitney U test showed significant differences between the groups. Two weeks after injury, BBB scores of the BM-NPC group were significantly higher than those of the vehicle group. *p < 0.01, **p < 0.001 post hoc Tukey's test. #p < 0.05, ##p < 0.01 Mann–Whitney U test.
Functional MRI Was Not Able to Provide Evidence of Reconstruction of Local Neuronal Networks After BM-NPC Transplantation
In response to electrical stimulation to the hind limbs, we counted fMRI signals in the bilateral somatosensory cortex in both BM-NPC and vehicle groups. BOLD signal changes were as high as 2.4% in the original hind limb territory in the BM-NPC group (Fig. 9). In the contralateral hind limb territory (HL) as defined in Figure 9B (36), volumes of activation in the BM-NPC group were significantly larger than those in the vehicle group (p = 0.014, Student's t test) (Fig. 9C). Particularly, we noticed that cortical activation in spinally injured rats was not confined to the original hind limb territory in the primary somatosensory cortex (Fig. 9D). Signals extended more medially and even to the ipsilateral side of stimulation. Total volumes of cortical activation were 1.53 ± 0.83 and 0.41 ± 0.17 mm3 in the BM-NPC and vehicle groups, respectively, while the difference was not statistically significant (p = 0.11, Student's t test).

Cortical fMRI signals in response to hind limb stimulation 8 weeks after spinal cord transection. (A) Time course of blood oxygenation level-dependent (BOLD) signals (%) in a voxel in the original hind limb territory in the bone marrow-derived neural progenitor cell (BM-NPC) transplantation group (n = 6). Gray bars indicate the period of stimulation. (B) Schematic drawing of a rat brain to illustrate medial cortical areas (dark gray), the hind limb territory of the primary somatosensory cortex area (HL) and lateral cortical areas (light gray), in which cortical activations were counted. (C) Volumes of functional magnetic resonance imaging (fMRI) signals in vehicle and BM-NPC groups. Significant differences between the BM-NPC and vehicle injection groups were noted in the signals in the contralateral cortex and the hind limb territory of the contralateral side to the stimulation (*p < 0.05). Note that broad cortical areas were activated in response to hind limb stimulation 8 weeks after spinal cord injury. (D) A representative cortical sensory map from the BM-NPCs group, demonstrating neuronal activation in various cortical areas outside of the original hind limb territory in the primary somatosensory cortex. Signals were observed on the left side (L), which was ipsilateral to the stimulation as demonstrated in a coronal section (left) at bregma −1.4 mm and a horizontal section (right). The color scale indicates T statistics calculated by statistical parametric mapping software (SPM2). The coronal sections are superimposed on a schematic brain section from the same level [reproduced with permission of Elsevier from the Paxinos and Watson atlas (36)].
Discussion
In this study, we report that transplantation of BM-NPCs promoted functional recovery of the spinally injured animals evidenced by improved locomotor scores. The grafted cells survived and committed predominant neuronal differentiation in the injured spinal cord. Immunohistochemistry using synaptophysin and PSD-95 as well as the FG tracing indicated synaptic formation of the BM-NPCs with the surrounding neurons and extension of the neurites across the transected portion in the spinal cord. As a consequence, neuronal networks were partially restored 7 weeks posttransplantation, which was represented as increased cortical signals in fMRI of the BM-NPC transplanted animals. However, the representation of the cortical signals was diverse and altered when compared to the original sensory map. Therefore, reestablishment of neuronal networks by transplantation of BM-NPCs was not fully accomplished.
Here, we demonstrated that transplantation of BM-NPCs was an efficient method in supplying neuronal lineage cells in a severely injured spinal cord. Majority of sphere-derived cells cultured with several trophic factors were reported to express neuronal markers such as βIII tubulin (Tuj-1) and microtubule associated protein 2 in a high ratio (98.5% and 95.7%, respectively) and GFAP in a low ratio (0.7%) (17). These findings clearly indicated that cells in the BM-NPC-derived spheres tended to differentiate into neurons and were not initially committed to the glial fate. In the same study in which BM-NPC-derived spheres were transplanted into the rat stroke model, BM-NPCs predominantly differentiated into NeuN-expressing postmitotic neurons (79.5%), and only a small population of the transplanted cells expressed GFAP (1.9%) (17). In the present study, we found that the rate of the differentiation of the transplanted GFP-positive cells into the neuronal fate detected by Tuj-1-positive cells was 77.2%; however, that of postmitotic neurons positive for NeuN was only 36.0%. The rate of differentiation into GFAP-positive astrocytes was 2.9%, similar to that observed in the transplantation into the rat stroke model. These observations indicate that, although the ratio of the differentiation into postmitotic neurons was dependent on the disease models, the BM-NPC-derived sphere cells predominantly differentiate into the neuronal fate after transplantation.
In general, the environment of the spinal cord is not permissive for survival and neuronal differentiation of the transplanted cells (19,27). Exogenous neural stem cells undergo proliferation and differentiate mainly into astrocytes in the spinal cord (33). When naive neural stem cells were transplanted into the injured spinal cord, 84% of the cells were GFAP-positive 9 weeks after transplantation, while only 4% of the cells had neuronal characteristics (18). Even when cells are shown to differentiate into neuronal lineage cells in vitro, the degree of neural induction in vivo may turn out to be less than expected after transplantation into the injured spinal cord (7). Our findings indicate that BM-NPCs are a reliable source of neuronal cells that can be used in cell transplantation therapy for SCI. Previously, we transplanted BM-NPCs and naive BMSCs in a rat stroke model and compared the results (17), in which BM-NPCs demonstrated much higher efficacy in survival, differentiation, and integration into the host brain. Having established the difference between BM-NPCs and naive BMSCs, we solely used BM-NPCs in the current protocol.
As Abematsu et al. demonstrated recently, grafted neuronal cells may establish neuronal connectivity by making synaptic connections with endogenous surrounding neurons (1). In this study, immunohistochemistry for synaptophysin and PSD-95 revealed the adjacent localization of synaptophysin and PSD-95 in the same GFP-positive transplanted cells (Fig. 6). Synaptophysin and PSD-95 are markers for presynaptic vesicles and post synaptic density, respectively, suggesting that the transplanted cells constructed synaptic connections and received synaptic inputs from host and/or surrounding neurons.
In our model, retrograde axonal tracing showed the presence of cells double positive for FG and GFP in the spinal cord rostral to the transected portion. However, no FG-positive cells were detected in cervical spinal cord or brain sections, when we performed histological analyses 4 days following the injection of FG. In a previous report using a matrix seeded with BMSCs to fill the gap of the spinal cord transection injury, FG-labeled neurons were detected in the cortex and in the brainstem a week after the FG injection (22). With regard to the time between the FG injections and histological analyses, 4 days might not be long enough for the retrograde tracers to reach the brain stem through the severely injured spinal cord. Therefore, the purpose of the FG tracing was focused on evaluating grafted BM-NPCs located rostral to the transected stump. To avoid confusion in interpreting results, we only injected FG caudally and 10 mm from the stump. In this way, we could minimize the possibility that the tracer would diffuse and go across the transected portion (22). Results of the FG thus indicated that the grafted cells survived, differentiated, and might have extended neurites across the transected portion of the spinal cord.
Furthermore, the FG-labeled BM-NPCs localized 4.6 mm rostral to the spinal cord stump. Recently, we demonstrated the capacity of migration of BM-NPCs in a rat model of stroke (17). In spinal cord, various studies using in vivo MRI tracking systems have observed that the transplanted cells could actually migrate long distances. In one of these study, injected live cells traveled as far as 9 mm in 8 weeks following the transplantation (23). Although it is possible that dispersion might have transferred the grafted cells a few millimeters, the aforementioned information suggested that the BM-NPCs had the capacity to migrate in the spinal cord.
In this study, transplantation of BM-NPCs to the completely transected adult rat spinal cord led to a significant, albeit modest improvement of hind limb motor function as determined by BBB scoring. Eight weeks after the injury, the BBB score of injured animals was 3.3 without BM-NPC transplantation, while with the transplantation, it improved to 5.8. In other studies, BMSC-derived Schwann cells or olfactory mucosa were transplanted into similar complete spinal cord transection models in Wistar rats. In those studies, BBB scores were 3.6 and 2.6 in the controls, while 7.0 and 4.6 in the treatment groups, respectively (3,20). Degrees of improvement in BBB scores by BM-NPC transplantations were therefore comparable to those of the other studies. Importantly, to judge degrees of locomotor recovery by the BBB scores needs caution, since functional improvement may also depend on the strains used or the types of treatments applied. For instance, in Sprague–Dawley strains, BBB scores were lower and between 0 and 2 following complete transection of the spinal cord (13,26,41). In one of these studies, 10 weeks posttransplantation of olfactory ensheathing cells, BBB scores improved to 4.3 from 1.0 (26).
FMRI also demonstrated an increase of BOLD signals in cortex cerebri in response to electrical stimulation of a hind limb. In particular, cortical activations were not only observed in the original somatosensory cortex, rather the BOLD signals had an abnormal and diverse distribution, including ipsilateral activity. Together, the addition of the BM-NPCs to the injury area might lead to the reestablishment of a degree of ascending and descending neurotransmission across injury, while we could not confirm reconstruction of the disrupted neuronal networks by the distribution of recovered cortical activations in fMRI.
One of the reasons for such ambiguity could partly rely on the end point of this study, which was 7 weeks posttransplantation. In the current methods, we set the end point mainly based on the time course of locomotor recovery. As shown in Figure 8, BBB scores improved and reached a plateau 5–6 weeks after the transplantation. In another experiment using fMRI to evaluate sensory recovery in a rat model of SCI, fMRI was performed 9 weeks posttransplantation, which demonstrated recovery of cortical responses (18). Moreover, in our previous experiment applying BMSC-derived Schwann cells to a rat model of complete spinal cord transection, it was 6 weeks posttransplantation when the axonal regeneration was histologically confirmed (20). However, as we demonstrated previously, reorganization in cerebral cortex may take place over 6 months long after SCI (13). After severe thoracic SCI, cortical connections from the spared forelimbs expanded and took over the deafferented hind limb areas (13,16). If such deafferentation plasticity occurs, the corresponding somatosensory cortex may no longer be available to receive or process recovered ascending sensory information. In this sense, the time allowed for recovery following the complete transection could be too short and limit the value of fMRI in this study.
Another possible explanation for the diverse cortical signals in fMRI is misdirection of sensory inputs through the reconstructed neuronal networks. After transplantation of BM-NPCs, new synaptic pathways relaying peripheral inputs to cortical areas were formed in the injured spinal cord. However, they could be different from those in the original ascending tracts. In such a situation, recovered sensory inputs would not connect with the original hind limb territory, but with various cortical areas.
Besides specific neuronal cell induction, another possible strategy for treating SCI includes transduction of neural stem cells into the oligodendrocytic lineage to enhance myelination (18,21). Using this approach, myelination of spared axons led to recovery of conduction velocity and promoted functional recovery in SCI. Hypothetically, however, if axons are completely severed in the damaged spinal cord, as occurs in our injury model and a model used by others (1), enhancement of myelination with oligodendrocytes may have limited effectiveness. In this situation, a strategy in which added neuronal cells may serve as a form of interneuron providing a link across the injury may be a more reliable approach. Depending on the degree of injury, one may need to select or combine different types of cell transplantation to achieve the best functional recovery.
We conclude that delayed grafting of BM-NPCs into the injured spinal cord was effective in providing neuronal lineage cells. If an efficient neuronal induction in the injured spinal cord was feasible, reconstruction of disrupted neuronal circuits between grafted cells and endogenous surrounding neurons can be a possible means of achieving recovery from SCI. Depending on the degrees of SCI, we can select or combine different types of cell transplantation to achieve the best functional recovery in SCI and other neural degenerative diseases occurring in the spinal cord.
Footnotes
Acknowledgments
We thank Ms. Ryoko Mamiya for her technical assistance and Dr. Mitsunobu Matsubara and Dr. Kuniyasu Niizuma (Tohoku University) for their useful inputs to our project. This work was supported by Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (NIBIO). This work was supported by Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (NIBIO). The authors declare no conflict of interest.
