Abstract
Bone marrow stromal cell (BMSC) transplantation has shown promise for repair of the spinal cord. We showed earlier that a BMSC transplant limits the loss of spinal nervous tissue after a contusive injury. Here, we addressed the premise that BMSC-mediated tissue sparing underlies functional recovery in adult rats after a contusion of the thoracic spinal cord. Our results reveal that after 2 months BMSCs had elicited a significant increase in spared tissue volumes and in blood vessel density in the contusion epicenter. A strong functional relationship existed between spared tissue volumes and blood vessel density. BMSC-transplanted rats exhibited significant improvements in motor, sensorimotor, and sensory functions, which were strongly correlated with spared tissue volumes. Retrograde tracing revealed that rats with BMSCs had twice as many descending brainstem neurons with an axon projecting beyond the contused spinal cord segment and these correlated strongly with the improved motor/sensorimotor functions but not sensory functions. Together, our data indicate that tissue sparing greatly contributes to BMSC-mediated functional repair after spinal cord contusion. The preservation/formation of blood vessels and sparing/regeneration of descending brainstem axons may be important mediators of the BMSC-mediated anatomical and functional improvements.
Introduction
Contusion of the adult rat thoracic spinal cord causes immediate locomotor and sensory impairments of the hindlimbs (37,62). Spontaneous recovery in locomotor ability reaches plateau levels a few weeks after a contusion (6,11,36,53). Sensory function remains impaired (32,68) with only small improvements months after trauma (8).
Transplantation of repair-promoting cells into the contused spinal cord has been explored as an intervention to restore function over what is spontaneously observed (22,28,51,52,56,60,62). Bone marrow stromal cells (BMSCs) are among the candidate cell types for spinal cord repair (56,67). BMSCs are relatively easy to obtain, which warrants their promise for clinical application (25,45,65).
BMSC transplantation into the contused rat spinal cord improves overground walking (15–17,29,30,33,40, 50,71,72). However, this particular gain in motor function was not observed in several other studies (2,58, 61,69). Little is known about the effects of BMSC transplants on contusion-induced sensory impairments. Himes and colleagues (29) showed that BMSC transplantation into the contused adult rat spinal cord does not affect thermal hyperalgesia.
The mechanisms underlying BMSC-mediated repair of the contused spinal cord remain elusive. Cell replacement is doubtful as grafted BMSCs survive poorly (30,46,67) and their differentiation into neural cells is moot (12,41,42,47). Another possible repair mechanism is neuroprotection resulting in tissue sparing in the contused segment. Larger spared tissue volumes are often (23,48,49,62) but not always (21,63) accompanied by improved functional outcome.
In the present study, we tested the premise that BMSC-mediated tissue sparing underlies functional recovery in adult rats with a contused spinal cord. Previously, we showed that BMSC transplantation into the 3-day-old contused adult rat spinal cord elicits tissue sparing (46). We employed this paradigm and investigated the contribution of tissue sparing in functional outcomes after two months. Moreover, blood vessel formation and descending brain stem axon sparing/regeneration were explored for their role in tissue sparing and functional restoration.
Materials and Methods
Ethics and Surgical Approval
Rats used in this study were housed pre- and post-surgery according to the National Institutes of Health and the United States Department of Agriculture guidelines. Air in the cages was continuously refreshed and water and food were available ad libitum. At all times during the experiment, rats were kept within a double-barrier facility. All animal procedures were approved by the Institutional Animal Care and Use Committee at the Johns Hopkins University and the University of Pittsburgh.
BMSC Culture and Lentiviral Transduction
BMSCs were harvested from the marrow of femurs of adult female Sprague-Dawley rats (n = 6, 200–230 g; Harlan, Indianapolis, IN, USA) according to previously described methods (3,57). Lentiviral vectors (LVs) encoding for green fluorescent protein (GFP) were used at an multiplicity of infection (MOI) of 150 to transfect BMSCs at passage 0 to enable identification after transplantation (46). BMSCs from the third passage were used for transplantation and of these 63% expressed GFP as determined by FACScan/FACSorter (Becton Dickinson Immunocytometry Systems Biosciences, San Jose, CA, USA). The cells used for transplantation expressed the typical BMSC markers, cluster of differentiation 90 (CD90) and CD105, but not blood cell markers, CD34 and CD45, or the immune cell marker, human leukocyte antigen (HLA)-DR (57).
Spinal Cord Contusion
Adult female Sprague-Dawley rats (n = 24, 200–230 g; Harlan) were sedated with intraperitoneal injections of 60 mg/kg ketamine HCl (Phoenix Pharmaceuticals, St. Joseph, MD, USA) and 0.4 mg/kg medetomidine (Domitor®, an α-2-adrenergic agonist; Orion Corporation, Espoo, Finland). The back was shaved and cleaned with Betadine and 70% alcohol, and Lacrilube ointment was applied to the eyes. The lower thoracic (T) spinal column was exposed, the T9 lamina was removed, and the underlying T10 spinal cord was contused using the Infinite Horizon Impactor (Precision Systems and Instrumentation, LLC, Lexington, Kentucky, USA) at 200 kdyn (57,59). Two rats with recorded force and displacement that deviated over 5% from the intended values were excluded. Two rats died before receiving the contusive impact. The wound site was rinsed with phosphate-buffered saline (PBS) with 0.1% gentamicin (Abbot Laboratories, North Chicago, IL, USA), the muscles were closed in layers using 5.0 sutures, and the skin was closed with Michel wound clips. Rats received a subcutaneous injection of 1.5 mg/kg atipamezole hydrochloride (antisedan®; an α-2-adrenergic antagonist; Pfizer Inc., New York, NY, USA) to reverse the sedative and analgesic effects of medetomidine. Ten milliliters lactated Ringer's solution was injected subcutaneously and 6 mg/kg gentamicin (Abbott Laboratories) intramuscularly. The rats were kept at 37°C until fully awake and were then returned to their cages. Between contusion and transplantation 3 days later, rats received daily 5 ml Ringer's solution (subcutaneously), 6 mg/kg gentamicin (Abbot Laboratories; intramuscularly), and 0.03 mg/kg buprenorphin (Buprenex®; Reckitt Benckiser Pharmaceuticals Inc., Richmond, VA; subcutaneously) and the bladder was emptied manually twice per day.
BMSC Transplantation
Three days postcontusion, rats were tested using the Basso-Beattie-Bresnahan rating scale (BBB test) (5,6); three rats were removed from the study as their BBB scores were higher than 5 (11,46). The remaining rats were sedated with intraperitoneal injections of 60 mg/kg ketamine HCl (Phoenix Pharmaceuticals) and 0.4 mg/kg medetomidine (Orion Corporation). The T10 spinal cord segment was reexposed and 1 × 106 BMSCs in DMEM (n = 9) or DMEM alone (control group; n = 8) (total volume was 5 μl in both cases) was injected into the contusion epicenter using a Hamilton syringe with a pulled glass needle attached (tip diameter: 150 μm) fixed within a micromanipulator (62). The cells were injected over a 5-min period using an electronic micropump and the needle was kept in place for an additional 3 min. The wound site was closed in layers. The rats were kept at 37°C until fully awake and then returned to their cages. The following 3 days, rats received daily 5 ml Ringer's solution (subcutaneously), 6 mg/kg gentamicin (Abbot Laboratories; intramuscularly), and 0.03 mg/kg buprenorphin (Buprenex®; Reckitt Benckiser Pharmaceuticals Inc.; subcutaneously). The bladder was emptied manually twice per day until reflex voiding occurred (~2 weeks). Throughout the remainder of the experiment rats were monitored daily; all rats survived without a need for treatment for pain or distress.
Testing of Locomotor Function
Overground locomotor recovery of automated hind limb movements was assessed using the BBB test (5,6) once a week for 8 weeks postinjection over a 4-min period by two testers unaware of the treatments. Rats were familiarized with the open field and baseline values were confirmed.
Higher motor functions were assessed using the BBB subscore (39). In the predominant paw we determined position [internal/external at initial contact (IC) and liftoff (0 points), parallel at IC and internal/external at liftoff or vice versa (1 point), parallel at IC and liftoff (2 points)], toe clearance [none (0 points), occasional (1 point), frequent (2 points), consistent (3 points)], tail position [down (0 points), up (1 point)], and trunk instability [yes (0 points), no (1 point)]. Scores were summed for a possible maximum score of 7 (39).
Locomotion pattern was assessed using footprint analysis [modified from de Medinaceli et al. (19)]. Before (baseline) and at 4 and 8 weeks postinjection, the forepaws were dipped in red ink and the hind paws in blue ink and footprints were recorded during walking across a paper track (100 cm long, 7 cm wide) and used to measure angle of hind paw rotation, hind limb base of support, and hind limb stride length. Average values per paw were calculated from at least five sequential steps. Values for both hind paws were averaged. Hind paw rotation was defined by the angle formed by the intersection of the line through the prints of the third digit and the metatarsophalangeal joint and the line through the central pad parallel to the walking direction. Hind limb base of support was determined by measuring the core to core distance of the hind paws central pads. Hind limb stride length was defined by the distance between the central pads of two consecutive prints. At the time of footprint testing all rats exhibited weight support.
Sensorimotor function of the hind limbs was assessed before (baseline) and at 4 and 8 weeks postinjection using horizontal ladder walking (38). Rats walked across a 100-cm-long horizontal ladder three times each test. The passages were videotaped and played back in slow motion for accurate evaluation. Only the middle 60 cm of the ladder was used for measurements. Small (foot or part of foot), medium (foot and part of lower leg), and large (full leg) slips were counted and individually and collectively expressed as a percentage of the total number of steps.
Testing of Sensory Function
Thermal hyperalgesia (i.e., an increased response to a nocuous stimulus) was measured at 4 and 8 weeks postinjection by assessing the withdrawal response time to a heat source applied using a Hargreaves' heat source (27). Rats were acclimated for 5 min before measurements in a Plexiglas test box with an elevated mesh floor. The radiant heat source with constant intensity was aimed at the midplantar area of each hind paw and the time (in seconds) until limb withdrawal was recorded. The heat source automatically shuts down at 15 s to prevent injury. The resting interval between measurements was 20 min. All rats withdrew their limb before the cut-off time was reached. Baseline measurements were taken before contusion. Three middle measurements out of five of each paw were averaged and expressed as a percentage of the baseline.
Mechanical allodynia (i.e., a pain-related response to a normally innocuous stimulus) was measured at 4 and 8 weeks postinjection by assessing the force to the hind paw resulting in limb withdrawal. Rats were acclimated for 5 min before measurements in a Plexiglas test box with an elevated mesh floor. An electronic von Frey anesthesiometer (13) was applied perpendicularly to the midplantar area of each hind paw and the pressure increased until limb withdrawal. The force (in grams) at withdrawal was recorded. Before contusion baseline measurements were taken. Three middle measurements out of five of each paw were averaged and expressed as a percentage of the baseline.
Retrograde Neuronal Tracing
After the last functional tests were completed, rats were injected with 1.2 μl fast blue (FB) at 7 mm caudal to the contusion epicenter (between vertebrae T10 and T11) to retrogradely label neurons with an axon projecting caudal to the contusion/transplant. The tracer was injected in six deposits of 0.2 μl using a glass needle (approx. diameter: 150 μm) attached to a 5-μl Hamilton syringe held within a micromanipulator (31,62).
Histological Procedures
One week after retrograde tracer injections, rats were anesthetized with an intraperitoneal injection of 60 mg/ kg ketamine HCl (Phoenix Pharmaceuticals) and 0.4 mg/kg medetomidine (Domitor®, Orion Corporation). After deep sedation was confirmed, the heart was exposed and 0.1 ml heparin (500 IU; Henry Schein, Melville, NY, USA) was injected into the left ventricle. Next, 300 ml saline followed by 500 ml 4% paraformaldehyde in phosphate buffer (PB; 0.1 M, pH 7.4) was pumped through the cardiovascular system. The spinal cord and brain were removed and postfixed overnight in the same fixative at 4°C. The brain stem, motor cortex, and a 10-mm-long spinal cord segment centered at the contusion site were transferred to phosphate-buffered 30% sucrose. These tissue blocks were embedded in Shandon M-1 Embedding Matrix (Thermo Electron Corporation, Pittsburgh, PA) and cut on a Leica CM 1950 cryostat. The spinal cord segment was cut into 10 series of 20-μm-thick horizontal sections and the brain stem and motor cortex were cut into 10 series of 40-μm-thick transversal sections. The sections were mounted onto glass slides and stored at −20°C until further processing. One series of sections was stained with cresyl violet to demonstrate the cytoarchitecture of the contused segment. Another series was stained with luxol fast blue to demonstrate presence of myelin.
Immunohistochemistry
Four series of cryostat sections were permeabilized with 0.3% Triton X-100 in PB (0.1 μM, pH 7.4), immune blocked with 5% normal goat serum in PB at room temperature for 30 min, and then immunostained (31). The following primary antibodies were used: polyclonal rabbit antibodies against glial fibrillary acidic protein (GFAP, 1:200; Incstar Corp., Stillwater, MN), and monoclonal mouse antibodies directed against rat endothelial cells [RECA-1 (20), 1:100; AbD Serotec, Raleigh, NC]. Then, the sections were washed with PBS and incubated for 2 h at room temperature with goat-anti-mouse IgG or goat-anti-rabbit IgG coupled to Alexa Fluor® 594 (1:200; Molecular Probes). Staining specificity was confirmed by omitting the primary antibody. Stained sections were covered with glass slips with Vectashield with DAPI (Vector Laboratories, Inc., Burlingame, CA) and kept refrigerated until analysis.
Measurement of Spinal Cord Tissue Sparing
Cresyl violet-stained sections were used to determine in a blinded fashion the volume of spared tissue in each spinal cord using the Cavalieri estimator function of Stereo Investigator® (MicroBrightField Inc.). The Gundersen Coefficient of Error was ≤0.05 for all measurements. In each 10th section the volume of spared tissue in a 3.4-mm-long segment centered at the contusion epicenter was determined. Within the analyzed segment, tissue was considered spared if it lacked cavities, areas with densely packed small cells resembling infiltrated neutrophils and lymphocytes, and neurons with darkly stained cytoplasmic Nissl bodies (46,62,63). The volume of spared tissue was expressed as a percentage of the average volume of a comparable uninjured spinal cord segment.
Measurement of Blood Vessel Density
One series of sections was stained with RECA-1 antibodies (20) and used to determine the blood vessel density (RECA-1-positive blood vessels per tissue surface area) using Stereo Investigator® (MicroBrightField Inc.) in a blinded fashion. In each 10th section, at 20x magnification, three areas were analyzed: the epicenter, rostral to the epicenter, caudal to the epicenter. Within each area, three samples were taken (320 × 420 μ m each) in which all RECA-1-positive structures were outlined. In most cases, the measuring area of the three contiguous samples was larger than the width of the spinal cord in the epicenter but smaller than the width just rostral and caudal to the epicenter. Therefore, we normalized the blood vessel surface area for the measured tissue surface area to get the blood vessel density (blood vessel area per 0.1 mm2). The values per level in each section were averaged to give the blood vessel density per level per rat.
Quantification of GFP-Positive BMSCs and FB-Positive Neurons
One series of spinal cord cryostat sections was covered with a glass slip with Vectashield with DAPI (Vector Laboratories, Inc.) and used to determine the number of GFP-positive BMSCs in the contusion using Stereo Investigator® (MicroBrightField Inc.). In each 10th section with GFP-positive cells, the transplanted area was outlined manually at 4x magnification and covered with a 250 × 250-μm grid. At 60x magnification with oil immersion, GFP-positive cells were marked using the optical fractionator with a 60 × 60-μm counting frame. BMSC numbers were corrected for the 63% transduction rate (46).
One series of brain stem and motor cortex sections was covered with a glass slip with Vectashield (Vector Laboratories, Inc.). FB-positive cells in the red nuclei, reticular formation, vestibular nuclei, raphe nuclei, and motor cortex (hind limb area) were counted by an investigator unaware of the treatments. FB-labeled neurons counted in the gigantocellular reticular formation and raphe nuclei were summed because these nuclei are difficult to distinguish from each other. All numbers were multiplied by 10 to obtain total numbers of FB-positive cells per nucleus (62).
Statistical Analysis
Student t-test and, in case of multiple time points, repeated measures ANOVA followed by Tukey's post hoc test were used to determine statistical differences between groups. Differences were accepted as statistically significant at p < 0.05. Correlation analysis was used to determine the relationship between variables and expressed by the coefficient of determination (r2) and the significance level (p). Relationships were considered strong when r2 > 0.25 and accepted when p < 0.05.
Results
BMSC Transplants Elicited Tissue Sparing
Spared tissue volume in a 3.4-mm-long segment centered at the contusion (Fig. 1A) was determined in BMSC-transplanted rats (Fig. 1B) and control rats (Fig. 1C) at 8 weeks postinjection. With a BMSC transplant the volume of spared tissue relative to that of a similar spinal cord segment in an uninjured/untreated rat was 24.8 ± 1.4% (SEM), which represented a significant 66% increase (p < 0.01) in tissue sparing compared to controls (14.9 ± 1.6%) (Fig. 1D). The tissue that was spared in BMSC-transplanted rats consisted mostly of white matter (97.03 ± 2.6%) and little of gray matter (2.97 ± 2.6%) (Fig. 1E). In three out of nine BMSC-transplanted rats, GFP-positive BMSCs were present in the contusion at 8 weeks postinjection (Fig. 1F). Some of these BMSCs were round and other elongated (Fig. 1G). The total numbers of BMSCs in these three rats were 3,937 (0.4% of injected number of cells), 11,460 (1.1%), and 13,429 (1.3%). In all rats, the contusion epicenter was surrounded by GFAP immunostaining, which was used to identify the contusion epicenters (Fig. 1H, I).

Bone marrow stromal cells (BMSCs) increased the amount of spared tissue in the contusion epicenter. Spared tissue volumes were determined at 8 weeks posttransplantation within a 3.4-mm-long spinal cord segment centered at the contusion (A) in BMSC-transplanted rats (B) and controls (C). BMSCs elicited a neuroprotective effect as the spared tissue volume in transplanted rats was significantly (p < 0.01) 66% larger than in controls (D). In BMSC-transplanted rats, 97 ± 2.6% of the spared tissue consisted of white matter (E). Three out of nine transplanted rats had green fluorescent protein (GFP)-positive BMSCs in the contusion (F), which exhibited largely variable morphologies (G). Glial fibrillary acidic protein (GFAP)-positive astrocytes were present in the contusion epicenter in all rats (H). In most rats, GFAP-positive astrocytes were found within tissue strands in the epicenter (I). Note (I) is outlined in (H). Scale bars: 500 μm (B, C, H), 250 μm (F, I), 50 μm (G).
Rats with BMSCs Demonstrated Improved Motor and Sensorimotor Functions
Overground walking ability (Fig. 2A) was not different (repeated measures ANOVA) between treatment groups. Both groups had a final score of about 11 (Fig. 2B), indicating frequent to consistent weight-supported plantar stepping without fore limb–hind limb coordination.
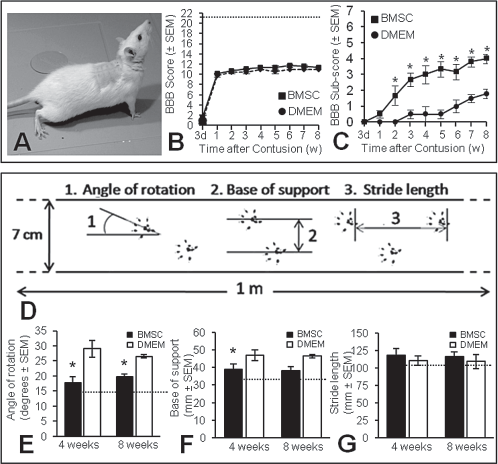
BMSCs improved locomotion-related features/pattern but not overground walking. With the Basso-Beattie-Bresnahan (BBB) test (A) we found that BMSC-transplanted and control rats exhibited similar overground walking abilities throughout survival (B). The baseline (21) is indicated by the horizontal broken line. The BBB subscore revealed that higher motor functions in BMSC-transplanted rats were significantly (p < 0.0001) better than in controls (C). Footprint analysis was used to assess locomotion pattern by measuring the angle of hind paw rotation, base of support, and stride length (D). Hind paw rotation in BMSC-transplanted rats was significantly (p < 0.01) improved by 39% at 4 weeks and 26% at 8 weeks postinjection compared to controls (E). At 8 weeks, external rotation of the hind paws in BMSC-transplanted rats was increased compared to baseline (15 ± 2.1; horizontal broken line). Base of support was 17% improved (p < 0.01) in rats with BMSCs compared to controls at 4 weeks but similar at 8 weeks postinjection (F). At the 8-week time point, the base of support in rats with BMSCs was 12% larger than baseline (34.2 ± 2.2; horizontal broken line). Stride length was similar between groups at both measuring time points (G) and not different from baseline (105 ± 8.3; horizontal broken line in G).
Higher locomotor functions were significantly different (repeated measures ANOVA) between groups [F(1, 126) = 240.6, p < 0.0001]. The final score was 4.0 ± 0.3 (SEM) in rats with BMSC and 1.8 ± 0.3 in controls (Fig. 2C).
Footprint analysis (Fig. 2D) revealed a significant difference between groups in angle of hind paw rotation [repeated measures ANOVA: F(1, 20) = 28.5, p < 0.0001] (Fig. 2E) and hind limb base of support [F(1, 20) = 15.6, p < 0.001] (Fig. 2F) but not hind limb stride length (p = 0.29) (Fig. 2G). The angle of rotation of rats with BMSCs was 38.6% smaller [mean = 17.8; 95% Cl (15.8, 19.8)] at 4 weeks and 25.7% smaller [mean = 19.8; 95% Cl (18.8, 20.8)] at 8 weeks postinjection compared to controls [mean = 29.0; 95% Cl (26.3, 31.7) and mean = 26.5; 95% Cl (26.0, 27.0), respectively] (Fig. 2E). At 8 weeks postinjection, external rotation of the hind paws was increased in BMSC-transplanted rats compared to baseline (Fig. 2E). The base of support of BMSC-transplanted rats was 17.0% smaller [mean = 39.0; 95% Cl (36.0, 42.0)] at 4 weeks postinjection compared to controls [mean = 47.0; 95% Cl (45.0, 49.0)] and similar at 8 weeks postinjection (Fig. 2F). At the later time point, the base of support in transplanted rats was 12.3% larger than baseline (Fig. 2F).
Horizontal ladder walking (Fig. 3A) was significantly different between groups [repeated measures ANOVA: F(1, 24) = 24.8, p < 0.0001]. The number of slips was expressed as a percentage of the total number of steps needed to cross the measuring area [average of both groups: 14.4 ± 0.2 (SEM) at 4 weeks and 15.0 ± 0.3 at 8 weeks]. For total slips, the improvement in rats with BMSCs was 66.0% at 4 weeks [mean = 32.1; 95% Cl (28.9, 35.3)] (Fig. 3B) and 69.6% at 8 weeks [mean = 21.4; 95% Cl (18.8, 24.0)] (Fig. 3C) compared to controls.

BMSCs improved horizontal ladder walking performance. The numbers of small, medium, and large slips (A) at 4 and 8 weeks postinjection were quantified and expressed as a percentage of the total number of steps used to cross the ladder. Rats with BMSCs performed significantly (p < 0.0001) better than controls at 4 weeks (B) and 8 weeks (C) postinjection. BMSC-transplanted rats made 66% less total mistakes at 4 weeks and 70% less at 8 weeks posttransplantation.
BMSC-Transplanted Rats Had Improved Sensory Functions
Thermal hyperalgesia (Fig. 4A) was significantly decreased in rats with BMSCs compared to controls [repeated measures ANOVA: F(1, 0.41) = 53.5, p < 0.0001] (Fig. 4B). The improvement was 43.7% [mean = 10.2; 95% Cl (9.9, 10.5)] at 4 weeks and 24.6% [mean = 7.6; 95% Cl (7.3, 7.9)] at 8 weeks postinjection [mean = 7.1; 95% Cl (6.7, 7.5) and mean = 6.1; 95% Cl (5.8, 6.4), respectively] (Fig. 4B).

BMSCs decreased thermal hyperalgesia and mechanical allodynia. Thermal hyperalgesia was assessed using a Hargreaves' heat source (A), which revealed that rats with a BMSC transplant had significantly (p < 0.0001) longer hind paw withdrawal response times (i.e., improved tolerance) than controls (B). The improvement was 44% at 4 weeks and 25% at 8 weeks after BMSC transplantation. At 8 weeks postinjection, thermal hyperalgesia in BMSC-transplanted rats was 49% above baseline (15 ± 1.8; horizontal broken line). With an electronic von Frey anesthesiometer (C) we found that mechanical allodynia in BMSC-transplanted rats was significantly (p < 0.0001) improved compared to controls (D). The improvement in tolerance to pressure to the hind paws was 35% at 4 weeks and 52% at 8t weeks after BMSC transplantation. At the latest time point, mechanical allodynia in rats with BMSC was 49% above baseline (150 ± 13.5; horizontal broken line).
Mechanical allodynia (Fig. 4C) was significantly decreased in rats with BMSCs compared to controls [repeated measures ANOVA: F(1, 24) = 26.9, p < 0.0001] (Fig. 4D). The improvement was 34.7% at 4 weeks postinjection [mean = 96.9; 95% Cl (94.2, 99.6)] and 51.7% [mean = 88.0; 95% Cl (85.8, 90.2)] at 8 weeks postinjection [mean = 72.3; 95% Cl (70.0, 74.6) and mean = 58.2; 95% Cl (56.5, 59.9), respectively] (Fig. 4D).
Table 1 provides an overview of the functional test results.
Functional Improvements at 4 and 8 Weeks After BMSC Transplantation Into the Contused Adult Rat Spinal Cord

An upward arrow represents a statistically significant improvement in test performance of BMSC-transplanted rats compared to controls. A dash represents no difference between BMSC-treated rats and controls. BBB, Basso-Beattie-Bresnahan; BMSC, bone marrow stromal cell.
Rats with BMSCs Had Higher Blood Vessel Density in the Contusion Epicenter
The density of blood vessels was assessed in the contusion epicenter, the white matter in the epicenter, and outside of the epicenter (Fig. 5A). In the epicenter, blood vessels were mostly larger and with fewer branches (Fig. 5B) than outside of the contused area (Fig. 5C). The blood vessel density in the epicenter was significantly (p < 0.001) 6.5-fold higher in BMSC-transplanted rats than in controls (Fig. 5D). Outside of the epicenter there were no significant differences between groups (Fig. 5D), which was in agreement with previous reports (66). The latter value was similar as that found in the epicenter in BMSC-transplanted rats and in the spinal cord of uninjured rat (Fig. 5D). In uninjured rats the average blood vessel density was 4,922 ± 397 (SEM) in the spinal cord, 8,299 ± 645 in gray matter only, and 1,545 ± 220 in white matter only. The blood vessel density in epicenter white matter in BMSC-transplanted rats was significantly (p < 0.01) 4.5-fold higher than in controls and 2.3-fold higher than in uninjured rats (Fig. 5E).
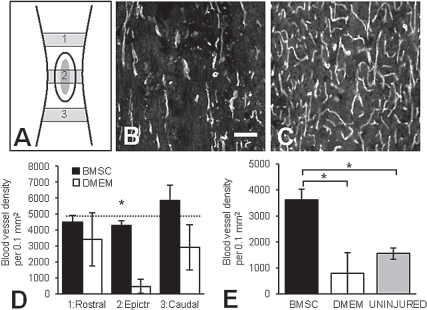
BMSCs increased blood vessel density in the contusion epicenter. Endothelial cell (RECA-1) antibodies were used to identify blood vessels rostral, within, and caudal to the contusion epicenter (A). The blood vessels in the epicenter were generally longer, more straight, and with fewer branches (B) than outside the epicenter (C). In the epicenter blood vessel density was significantly (p < 0.001) higher in rats with BMSCs than in controls (D). Outside the epicenter the blood vessel density in both groups was similar and comparable to that in uninjured rats (average indicated by horizontal broken line). In white matter only in the epicenter, the blood vessel density was significantly (p < 0.01) higher in BMSC-transplanted rats than in controls or uninjured rats (E). Scale bar: 35 μm (in B, C). Numbers in (A) represent rostral to epicenter (1), epicenter (2), and caudal to epicenter (3).
Rats with BMSCs Had More Brain Stem Neurons Projecting Beyond the Contusion
Retrograde FB tracing was used to identify brain stem and motor cortex neurons with an axon beyond the contusion site. Figure 6 provides examples of FB-labeled neurons present in the nucleus raphe obscurus (Fig. 6A, B) and the reticular formation (Fig. 6C, D). In transverse brain stem sections FB-labeled neurons were plotted and quantified in the red nucleus (Fig. 6E), reticular formation (Fig. 6F), vestibular nuclei (Fig. 6G), gigantocellular reticular nucleus (Fig. 6G), and raphe nuclei (Fig. 6H). No labeled neurons were found in the hind limb area of the motor cortex. The total number of FB-labeled brain stem neurons in rats with BMSCs was 7,650 ± 765, a significant twofold increase (p < 0.05) over controls (Fig. 6I). There were 2.2-fold more labeled neurons in the reticular formation (p < 0.05), threefold more in vestibular nuclei (p < 0.05), and twofold more in raphe nuclei (including the gigantocellular reticular formation; p < 0.05) in rats with BMSCs compared to controls (Fig. 6I). The number of FB-labeled neurons in the red nuclei was similar in both groups (Fig. 6I). With correlation analysis a strong significant association was identified between tissue sparing and total brain stem neurons (r2 = 0.57, p = 0.01).
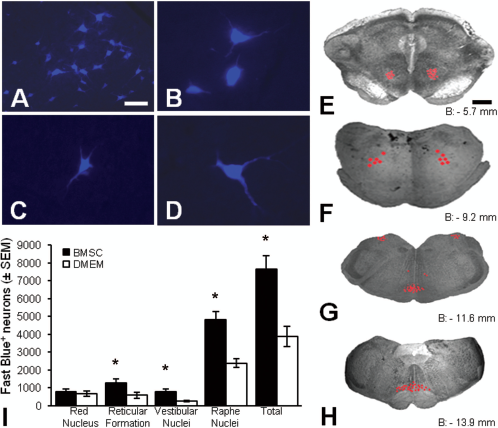
BMSCs increased the number of brain stem axons caudal to the contusion. We used retrograde fast blue (FB) tracing to identify brain stem and motor cortex neurons projecting beyond the contusion. Labeled neurons were found in several brain stem nuclei including the nucleus raphe obscures, shown here at 10X (A) and 20x (B), and the reticular formation, shown here at 20x (C, D). FB-labeled neurons (represented by red dots) were found in the red nucleus (E), reticular formation (F), vestibular and gigantocellular reticular nuclei (G), and the nucleus raphe obscurus and nucleus raphe pallidus (H). The level of brain stem sections shown in (E–H) is provided relative to bregma (B). Rats with BMSCs had significantly (p < 0.05) more FB-labeled neurons in their brain stem than controls (I). Significantly higher numbers (p < 0.05) were found in the reticular formation, vestibular nuclei, and raphe nuclei, but not the red nucleus (I). On average the increase in FB-labeled neurons in BMSC-transplanted rats was 2.3-fold. Scale bars: 50 μm (A), 25 μm (B–D), 1 mm (E–H).
Relationships Between Tissue Sparing, Motor/Sensory Functions, Blood Vessel Density, and Brain Stem Neurons Projecting Beyond the Contusion
Tissue Sparing and Motor/Sensory Functions (Table 2)
A strong significant functional relationship was present between tissue sparing and BBB subscore (r2 = 0.87, p < 0.001), angle of rotation (r2 = 0.81, p = 0.001), horizontal ladder walking (r2 = 0.74, p = 0.001), and thermal hyperalgesia (r2 = 0.56, p = 0.013). Relationships between tissue sparing and BBB, hind limb stride length and base of support, and mechanical allodynia were not significant.
Relationship Between Tissue Sparing and Motor/Sensory Functions at 8 Weeks After BMSC Transplantation Into the Contused Adult Rat Spinal Cord

Correlation analysis was employed to examine the functional relationships between the outcome measures at 8 weeks postinjection. The table provides the coefficient of determination (r2, top) and the significance level (p-value, bottom). Strong statistically significant relationships between spared tissue volumes and neuron numbers (r2 > 0.25, p < 0.05) are shown in bold. BBB, Basso-Beattie-Bresnahan.
Motor/Sensory Function and Brain Stem Neurons (Table 3)
Strong significant functional relationships were found between raphe neurons and the BBB subscore (r2 = 0.78, p = 0.002), angle of rotation (r2 = 0.68, p = 0.007), and horizontal ladder (r2 = 0.54, p = 0.02). Strong relationships were also found between the total brain stem neurons and the BBB subscore (r2 = 0.76, p = 0.002), angle of rotation (r2 = 0.51, p = 0.03), and horizontal ladder (r2 = 0.54, p = 0.02). Reticular neurons had a strong significant relationship with BBB (r2 = 0.60, p = 0.008) and BBB subscore (r2 = 0.50, p = 0.03).
Relationship Between Brain Stem Neurons and Motor/Sensory Functions
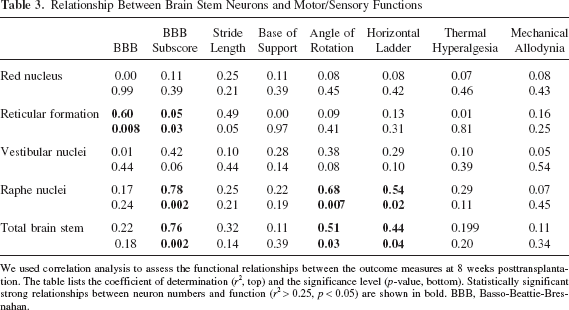
We used correlation analysis to assess the functional relationships between the outcome measures at 8 weeks posttransplantation. The table lists the coefficient of determination (r2, top) and the significance level (p-value, bottom). Statistically significant strong relationships between neuron numbers and function (r2 > 0.25, p < 0.05) are shown in bold. BBB, Basso-Beattie-Bresnahan.
Tissue Sparing and Blood Vessel Density
We found a strong significant functional relationship between spared tissue volumes and blood vessel density in the epicenter in general (r2 = 0.67, p = 0.004) and in white matter only (r2 = 0.47, p = 0.04).
Discussion
We found that a BMSC transplant in the contused spinal cord of adult rats elicits tissue sparing that correlated strongly with improvements in higher motor functions, hind paw rotation, horizontal ladder walking, and thermal hyperalgesia. The data also revealed that rats with BMSCs elicited an angiogenic response in the contusion epicenter that correlated with tissue sparing. Retrograde tracing revealed that rats with BMSCs had significantly more brain stem neurons with an axon projecting beyond the contusion that correlated with improved motor functions but not sensory functions. Together, the data indicate that BMSC transplants in the adult rat contused spinal cord result in tissue sparing, which is differentially involved in functional restoration. Blood vessel preservation/formation and descending brain stem axon sparing/regeneration appear important mediators of the BMSC-mediated improved outcomes.
Transplantation of BMSCs into the contused adult rat spinal cord resulted in a significant 66% increase in spared tissue volume in the injury epicenter. This finding is consistent with previous studies (2,33,46). Tissue sparing was determined at 8 weeks posttransplantation. At this time point almost all BMSCs had died; few were present in only three of the transplanted rats. Therefore, it is unlikely that the transplanted cells contributed significantly to the volume of spared tissue. We showed previously that BMSCs transplanted into the contusion at 3 days postinjury survive poorly with 50% lost after 1 week and almost all lost after 4 weeks (46). Interestingly, the relative short duration of BMSC presence in the contusion is sufficient to elicit a significant tissue sparing effect. The neuroprotective effect of BMSC transplants is not present with delayed (7 and 21 days) injection paradigms (46), which indicates the existence of a specific (sub)acute therapeutic window for BMSC-mediated neuroprotection.
The mechanisms underlying BMSC-mediated tissue sparing are largely unknown. In search for mediators of this effect we identified blood vessels to be a possible candidate. After injury the loss of blood vessels decreases the spinal cord blood flow, which correlates with nervous tissue loss (24). We show here that rats with BMSCs transplants have significant higher blood vessel densities in the contusion epicenter and that this was strongly associated with spared tissue volumes. Transplanted BMSCs secrete the proangiogenic vascular endothelial growth factor (VEGF) (14,44), which is known to be involved in protection (70) and formation (43) of blood vessels in injured nervous tissue. Through both mechanisms, BMSCs could have contributed to tissue sparing in the contusion epicenter.
A positive relationship between blood vessel density and spared tissue volumes in the injured spinal cord has been proposed before for adult rats (66) but not for adult guinea pigs (9). The latter study revealed a correlation between blood vessel density and secondary loss of function, indicating a role of hypervascularity in secondary pathology (9). Our present results are in variance with this idea as they show greater improvements in neurological function in rats with higher blood vessel density in the contusion. Further studies are required to elucidate the role of hypervascularity in spinal cord repair.
BMSC transplantation resulted in improved motor, sensorimotor, and sensory functions, which, with the exception of mechanical allodynia, strongly correlated with spared tissue volumes. In fact, based on our findings, over 70% of the variability in higher motor and sensorimotor function and 56% of that in thermal hyperalgesia can be attributed to a linear relationship with spared tissue volume. These results highlight a significant involvement of tissue sparing in functional restoration after a spinal cord contusion and emphasize the relevance of treatment that focus on tissue sparing as an early intervention in spinal cord repair strategies.
The behavioral improvements in our study seemed more pronounced during the first month after transplantation compared to the second month. Currently, it is unknown whether functional improvements due to BMSC transplantation remain present permanently. It is possible that the deteriorating general health due to spinal cord injury affects function negatively. Also, benefits from the BMSC transplant may be temporary due to the poor transplant survival. Future studies will be required to address these important clinically relevant issues.
The precise mechanisms underlying the relationship between spared tissue and motor and sensory functions are poorly understood. Tissue sparing likely includes a variety of cellular consequences which each exert their specific effect on behavioral recovery. We examined more closely to what extent descending axons from brain stem neurons were involved in the observed functional improvements. Different types of descending brain stem axons are involved in motor function (7,34,35). We demonstrated in this study that the large majority of spared tissue in BMSC-transplanted rats is white matter. Thus, it is possible that increased amounts of spared tissue reflect more spared axons descending to lower spinal cord segments involved in motor functions. We found here that rats transplanted with BMSCs had a twofold increase in number of (retrogradely FB-labeled) brain stem neurons projecting their axon beyond the contusion compared to controls. The correlative analysis further indicated that these brain stem neurons were involved in over 40% (coefficient of determination, r2 > 0.4) of the observed recovery in higher motor and sensorimotor function, but did not contribute to recovery in sensory functions (r2 < 0.2).
Among the brain stem neurons, raphe neurons were strongly correlated with higher motor functions and sensorimotor function and reticular formation neurons with higher motor functions and overground walking. Descending raphe spinal axons have been implicated in initiating and modulating motor behaviors through the central pattern generator (4,26). Reticulospinal axons have been associated with flexion during the swing phase and extension during the stance phase (54), which may explain their association with walking and higher motor functions.
BMSC-transplanted rats exhibited significantly improved higher motor functions as revealed with the BBB subscoring scale (4,39) and footprint analysis. Transplantation of BMSCs also significantly enhanced horizontal ladder walking with fewer small, medium, and total slips, indicating enhanced integration of sensory and motor inputs (38). Interestingly, the combined improvements did not result in improved overground walking as examined using the BBB test. A lack of improved walking in rats with a BMSC transplant was reported previously by some (2,58,61,69) but not by others (29,30,50). The absence of improved walking could reflect the complexity of properly rating increasing degrees of fore limb–hind limb coordination. This convolution has been recognized and resulted in the design of the BBB subscoring scale to increase the sensitivity of the BBB rating scale for rats that lack coordination (4,39). An alternative explanation for the lack of improved walking in the present study is that mechanisms underlying recovery in overground walking are in part different from those underlying recovery of higher motor and sensorimotor functions. This possibility needs to be considered because elucidating the different mechanisms would be imperative for the design of more effective and comprehensive spinal cord repair approaches.
BMSC-transplanted rats exhibited less sensitivity to mechanical and thermal stimuli to the hind paws, which is an improvement over the typically observed contusion-induced hypersensitivity. To our knowledge only one study has investigated the effects of a BMSC transplant on sensory function in rats with a contused spinal cord. Himes and colleagues (29) reported that transplantation of BMSCs did not affect thermal hyperalgesia. A possible explanation for this discordance with our study is the difference in time of BMSC transplantation: 3 days postinjury in our study and 7 days postinjury in Himes et al. (29). We have previously shown that transplantation of BMSCs into a contused adult rat spinal cord at 7 days postinjury leads to significantly less tissue sparing than transplantation at 3 days after injury (46). We show in the present study that tissue sparing is correlated with thermal hyperalgesia reduction. Thus, less tissue sparing after a 7-day delayed BMSC transplantation may have caused the observed lack of improvements in thermal hyperalgesia in Himes et al. (29).
The injury-induced inflammatory response may contribute to the development of thermal hyperalgesia (18). BMSC transplantation has been associated with reduced inflammation (1,10). Thus, it is possible that in our study the decrease in thermal hyperalgesia in transplanted rats reflects a BMSC-mediated decrease in inflammation, which may also contribute to tissue sparing (55). On the other hand, we showed that BMSC transplantation into a 3-day-old spinal cord contusion does not lead to a decrease in the number of macrophages (57). Further studies are needed to elucidate the interactions between inflammation, thermal hypersensitivity, and tissue sparing following spinal cord injury and repair.
The present and previous studies suggest that optimal BMSC neuroprotective effects are obtained within a 3-day (or acute) transplantation paradigm, suggesting clinical translation would favor allograft transplantation, because culturing an autograft would take more time. On the other hand, a human bone marrow biopsy, combined with cell sorting strategies, might provide an adequate amount of BSMCs to enable timely autograft transplantation. Additional research will be needed to investigate the applicability and efficacy of such autografts.
Footnotes
Acknowledgments
The authors thank Dr. Gerald Gebhart for his valuable discussion, the viral vector core facility of The Miami Project to Cure Paralysis for provided lentiviral vectors, and the animal core facility of the International Center for Spinal Cord Injury for animal surgeries, maintenance, and behavioral testing. Financial support was provided by LUMC (grant # 30229/5000), the Netherlands Organization for Scientific Research (grant # 017.001.265), Hugo W. Moser Research Institute at Kennedy Krieger, and the Department of Physical Medicine and Rehabilitation at the University of Pittsburgh. The authors declare no conflicts of interest.
