Abstract
Interaction between chemokine stromal cell-derived factor 1 and the CXC chemokine receptor 4 (CXCR4) governs the sequestration and mobilization of bone marrow stem cells. We investigated the therapeutic potential of TG-0054, a novel CXCR4 antagonist, in attenuating cardiac dysfunction after myocardial infarction (MI). In miniature pigs (minipigs), TG-0054 mobilized CD34+CXCR4+, CD133+CXCR4+, and CD271+CXCR4+ cells into peripheral circulation. After isolation and expansion, TG-0054-mobilized CD271+ cells were proved to be mesenchymal stem cells (designated CD271-MSCs) since they had trilineage differentiation potential, surface markers of MSCs, and immunosuppressive effects on allogeneic lymphocyte proliferation. MI was induced in 22 minipigs using balloon occlusion of the left anterior descending coronary artery, followed by intravenous injections of 2.85 mg/kg of TG-0054 or saline at 3 days and 7 days post-MI. Serial MRI analyses revealed that TG-0054 treatment prevented left ventricular (LV) dysfunction at 12 weeks after MI (change of LV ejection fraction from baseline, −1.0 ± 6.2% in the TG-0054 group versus −7.9 ± 5.8% in the controls). The preserved cardiac function was accompanied by a significant decrease in the myocardial expression of TNF-α, IL-1β, and IL-6 at 7 days post-MI. Moreover, the plasma levels of TNF-α, IL-1β, and IL-6 were persistently suppressed by the TG-0054 treatment. Infusion of TG-0054-mobilized CD271-MSCs reduced both myocardial and plasma cytokine levels in a pattern, which is temporally correlated with TG-0054 treatment. This study demonstrated that TG-0054 improves the impaired LV contractility following MI, at least in part, by mobilizing MSCs to attenuate the postinfarction inflammation. This insight may facilitate exploring novel stem cell-based therapy for treating post-MI heart failure.
Keywords
Introduction
Despite recent advances in reperfusion strategies and pharmacotherapy, myocardial infarction (MI) remains a leading cause of morbidity and mortality. In patients that survive MI, the subsequent left ventricular (LV) remodeling and heart failure (HF) remain as major health problems (48). Inflammation and immune responses are vital in adverse remodeling and HF (2, 3, 32). Therefore, immunomodulation exhibits potential for treating postinfarction HF.
Cell therapy for postinfarction myocardial dysfunction is an emerging field of investigation (19). Various cell types have been considered as candidates, including hematopoietic stem cells (HSCs), endothelial progenitor cells (EPCs), and mesenchymal stem cells (MSCs) (45). Numerous studies have attributed the beneficial effects of stem cells on post-MI cardiac function to anti-inflammation (15) or immunomodulation (8) rather than tissue regeneration (5, 39). Among various stem cells, MSCs are well known for immunomodulation and are expected to alleviate detrimental inflammatory reactions following cardiac injuries (51). The cluster of differentiation 271 (CD271) antigen has been reported as one of the most selective markers (24) to define a subset of MSCs with differentiation potential (23, 27, 31) and immunosuppressive capability (27, 28). However, preparing and locally delivering stem cells is time consuming and demanding in terms of technique (9). Direct mobilizing stem cells from bone marrow (BM) to injured tissues is considered to be a viable alternative approach to treat post-MI HF. In this study, the therapeutic effect of endogenous stem cells rather than transplantation of cultured stem cells was investigated.
CXC motif chemokine receptor 4 (CXCR4), a seventransmembrane G-protein-coupled receptor, is broadly expressed on stem cells in BM (42). Through the interaction between CXCR4 and stromal cell-derived factor 1 (SDF-1), stem cells are sequestered in the BM in the resting state and mobilized when this interaction is disrupted (40). Several small molecule compounds have been developed to disrupt SDF-1/CXCR4 interaction and thereby mobilized BM stem cells. A majority of the compounds target CXCR4 (13). One of the CXCR4 antagonists, AMD3100, was reported to mobilize HSCs and EPCs, whereby it promotes cardiac functional recovery after MI (25, 26). The therapeutic effect of AMD3100 was thought to be primarily augmenting endothelial protective function and neovascularization (25, 26). TG-0054, another CXCR4 antagonist, was reported to mobilize BM HSCs (22) and is under clinical trials for cancer therapy (35, 36). Little is known about MSC mobilization and recruitment to cardiac damage sites by CXCR4 antagonists. Here we evaluated the effect of TG-0054 on MSC mobilization and correlated it with post-MI myocardial function and changes in proinflammatory cytokines. The results show that TG-0054 mobilized MSCs as well as HSCs accompanied by reduction of tumor necrosis factor (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), and improved left ventricular ejection fraction (LVEF).
Materials and Methods
Animals
Adult Taiwanese Lanyu miniature pigs (minipigs, 4–6 months, 25–40 kg), procured from the Animal Propagation Station, Livestock Research Institute (Taitung, Taiwan), were maintained in the Laboratory Animal Center of National Taiwan University. Twenty-two minipigs (female = 17, male = 5) were used in accordance with the university guidelines for animal care. The experimental protocol was approved by the Institutional Animal Care and Use Committee (approval numbers 20090194 and 20110196).
CXCR4 Antagonist
TG-0054, also known as burixafor, was a selective CXCR4 small-molecule antagonist (49) and provided by TaiGen Biotechnology Co., Ltd. (Taipei, Taiwan). A Phase I study to evaluate the pharmacokinetics and phar-macodynamic effects of TG-0054 in healthy participants was completed (34). A Phase II study is currently being conducted to assess the therapeutic effect of HSC mobilization of TG-0054 alone or in combination with granulocyte colony-stimulating factor (G-CSF) in patients with multiple myeloma, non-Hodgkin's lymphoma, or Hodgkin's disease (36).
Animal Study Protocol
A TG-0054 dose of 2.85 mg/kg per pig was converted from the Food and Drug Administration's guidelines (50) based on the dose of 3.14 mg/kg used in Phase II clinical studies (35, 36). Twenty-two healthy, age-matched minipigs were blindly randomized to receive intravenous dual doses of TG-0054 (n = 12) or saline (equivalent volume to TG-0054, n = 10), receiving the first dose at 3 days and the second dose at 7 days post-MI (Fig. 1). We designed the dual-dose TG-0054 regimen based on several considerations. A major consideration is timing of cell mobilization. Clinical studies have shown that the beneficial effects of BM-derived progenitor cell therapy on the recovery of myocardial contractility are confined to patients who were treated more than 4 days but less than 7 days after MI (44). On the other hand, when comparing benefits of BM cell therapy at 3 days, 7 days, and 14 days post-MI, cell treatment at 3 days post-MI resulted in improved LV function, but delayed therapy at 7 days and 14 days did not (55). Therefore, we administered the first dose of TG-0054 at 3 days post-MI to serve as an early intervention and a second dose at 7 days post-MI as deferred therapy.
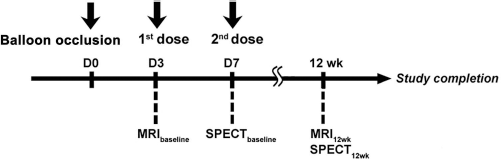
Study protocol of animal experiment. Twenty-two miniature pigs (minipigs) were randomized to receive intravenous dual doses of TG-0054 (n = 12) or saline (equivalent volume to TG-0054, n = 10). Magnetic resonance imaging (MRI) analyses were performed at 3 days (prior to the first dose treatment, baseline) and 12 weeks post-MI. 201Tl SPECT analyses were conducted at 7 days (prior to the second dose treatment) and 12 weeks post-MI.
Porcine Model of Coronary Artery Occlusion and Reperfusion
The minipigs underwent overnight fasting, were anesthetized using Zoletil (5 mg/kg; Virbac, Taiwan) and atropine (0.6 mg/kg; Taiwan Biotech Co., Ltd., Taiwan), and maintained using isoflurane (3%/1.5 L/min; Halocarbon Laboratories Inc., USA) by ventilator after intubation. The minipigs were placed in the supine position, and their body temperature was maintained in the normal range (36–37°C) by using a heating blanket. In sterile conditions, a carotid arterial cutdown was performed for vascular access by using 6F vascular sheaths (St. Jude Medical, USA). After systemic heparinization [150 IU/kg, Bodene (Pty) Limited, South Africa], a coronary angiography was performed using a mobile C-arm (GE stenoscop 6000; GE Medical Systems, USA) to determine the optimal location of the occlusion and assess the coronary artery size. Depending on the visual estimate of vessel size, an appropriately sized angioplasty balloon catheter (diameter, 3.0 ± 0.5 mm; length, 10 ± 2 mm; Boston Scientific, USA) was fluoroscopically guided into the left anterior descending artery (LAD) at the level distal to the first major diagonal branch. The balloon was inflated to occlude the middle LAD at 6–8 atm for 157 ± 17 min. Thereafter, the angioplasty balloon was deflated, and the restoration of normal coronary flow was documented using angiography. The localization of the coronary occlusion and patency of the first diagonal branch were confirmed using a contrast injection and ECG ST-segment elevation. During the procedure, blood pressure, heart rate, and ECG were continually recorded using a cardiac electrophysiologic monitor (Prucka CardioLab; GE Medical Systems, USA). In addition, 2% lidocaine (4–8 ml total; Recipharm Monts, France) was administered during the coronary artery occlusion to manage arrhythmias. Episodes of ventricular fibrillation were immediately terminated using electric cardioversion delivered at 200 Joules (TEC-7200K; Nihon Kohden, Japan). After the surgical procedure, the arterial sheath was removed, and the carotid incisions were closed in layers, followed by animal recovery.
Time Course Analysis of HSC and MSC Mobilization
Peripheral blood (PB) was collected from the minipigs at baseline and 0.5 h, 1 h, 2 h, 3 h, 6 h, and 24 h after the saline or TG-0054 injection. Red blood cells were removed from the sample using ACK lysing buffer [NH4Cl 150 mM (Wako Pure Chemical Industries, Ltd.); KHCO3 10 mM (J. T. Baker); EDTA 0.01 mM (Sigma-Aldrich)]. The remaining leukocytes were collected, labeled using primary antibodies to porcine CD34 (YST01; R&D, USA), human CD133 (AC133; Miltenyi Biotec, Germany), and human CD271 (ME20.4; Miltenyi Biotec), and analyzed using a FACS Calibur instrument and Cell Quest software (Becton Dickinson). The frequency of CD34+, CD133+, or CD271+ events in the PB was expressed as the percentage of positive cells among all leukocytes after electronic gating on the viable cells. The number of positive cells per microliter of cells was calculated by multiplying the frequency of CD34+, CD133+, or CD271+ events by the total leukocyte count. To evaluate the mobilization effects, the fold increase from the baseline was calculated as (events at postinjection times)/(events at baseline). Similarly, anti-porcine CXCR4 (ab2074; Abcam, UK) was used to confirm the coexpression of CXCR4 on mobilized CD34+, CD133+, and CD271+ cells.
Expansion and Characterization of MSCs
Peripheral blood mononuclear cells (PBMCs) were isolated using density gradient centrifugation (Ficoll-Paque; GE Healthcare Bio-Sciences AB, UK). PBMCs were collected from the minipigs at baseline and then 0.5 h, 1 h, 2 h, 3 h, 6 h, and 24 h after saline or TG-0054 injection. One proportion of PBMCs underwent immuno magnetic selection of CD271+ cells (CD271+PBMCs) by using IMag™ Anti-Phycoerythrin Magnetic Particles-DM (BD Bioscience, USA). To generate MSCs by plastic adherence (PA-MSCs), the overall PBMCs were incubated with MSC medium comprising αMEM (Gibco, USA) and 10% FBS (Gibco) and deposited in 75-mm2 flasks (Corning, USA), yielding a cell density of 2 × 105 cells/cm2. After 24 h, the adherent cells were cultured in the MSC medium supplemented using basic fibroblast growth factor and epithelial cell growth factor (10 ng/ml each; R&D Systems), which were changed every 3 days. To acquire CD271-MSCs, the CD271+PBMCs were expanded in the same medium as that for the generation of PA-MSCs. The MSC colonies were defined as adherent, clonogenic, nonphagocytic, and fibroblastic (designated as colony-forming units-fibroblastic: CFU-Fs) (6). The MSC CFU-Fs were enumerated after 8 days of incubation by using a Nikon ECLIPSE TS100 microscope (Lin Trading Co., Ltd., Taiwan). Colony-forming efficiency (CFE) was defined as the number of colonies for every 106 cells seeded. Furthermore, both PA-MSCs and CD271-MSCs from passage 3 were labeled with primary antibodies to cell surface antigens: CD11R3 (2F4/11; AbD Serotec, UK), CD14 (M-M9; VMRD, USA), CD44 (IM7; Abcam), CD45 (K252-1E4; AbD Serotec), CD90 (5E10; Becton Dickinson), and CD105 (MEM-229; Abcam), or corresponding isotype controls.
Analysis of the Differentiation Potentials of MSCs
Both PA-MSCs and CD271-MSCs from passage 4 were induced toward the adipogenesis, osteogenesis, and chondrogenesis pathways as previously described (38). Stained adipocytes (Oil red O; Sigma-Aldrich) and osteoblasts (Alizarin red S; Sigma-Aldrich) were examined on Nikon ECLIPSE TS100 microscope. Chondrocytes subjected to immunohistochemical staining (collagen type II; Millipore, USA) were examined on Zeiss Axiophot 2 microscope.
One-Way Mixed Lymphocyte Reaction (MLR)
We used MLR as a cell model to investigate immunological activity following MI. The allosuppressive potential of PA-MSCs, CD271-MSCs, and CD271+PBMCs was tested in parallel against BM-MSCs in each MLR assay, as we described previously (20). Briefly, PBMCs from two unrelated minipigs were cultured alone or mixed with autologous, mitomycin-C (25 μg/ml; Kyowa Hakko Kogyo Co., Ltd.)-treated MSC at an MSC/PBMC ratio of 1:2 (5 × 104 MSCs/1 × 105 PBMCs) in a 96-well round-bottom plate (BD Bioscience). To measure cell proliferation, cells were pulsed on the fourth day of coculture with 1 mCi/well [3H]thymidine (PerkinElmer, USA) for 18 h. Radioactivity was counted using a MicroBeta Filter Mate-96 Harvester and a 1450 MicroBeta TriLux (both from PerkinElmer). Responder cell proliferation was measured by [3H]thymidine uptake and expressed as cpm. The supernatants of MLR were harvested after 5 days of culture for quantification of TNF-α, IL-1β, and IL-6.
Delivery of TG-0054-Mobilized CD271-MSCs
A single dose of TG-0054 increased the number of circulating CD271+ cells from 14.3 ± 6.9 cells/μl to 40.6 ± 15.6 cells/μl in the time course analysis. On average, blood accounts for 7% of animal body weight. Therefore, we determined the dosage of intravenous CD271-MSCs based on the following equation: (41–14 cells/μl) × 1,000 μl/ml × 1,000 ml/kg × 7% = 1.89 × 106 MSCs/kg. The infused MSC formulation comprised 2.5 × 106 CD271-MSCs/ml in MSC medium. The minipigs received a dual-dose intravenous infusion of MSC suspension delivered at a rate of 2 ml/min; the first and second doses were administered at 3 days and 7 days post-MI, respectively.
Magnetic Resonance Imaging (MRI) Analysis
To investigate the therapeutic effects of TG-0054 on global LV function, MRI analyses were performed at 3 days (prior to the first dose treatment, baseline) and 12 weeks post-MI. Cardiac MRI images were acquired using a 1.5-T MRI system (Sonata; Siemens, Erlangen, Germany) comprising a four-channel phased array torso coil. Detailed procedures regarding LV function and mass analysis were performed as previously described (47). Briefly, cine MRI was first performed using a segmented balanced steady-state gradient echo-pulse sequence with a retrospective ECG R-wave trigger. The scanning parameters were TR/TE, 3.0/1.6 ms; flip angle, 50°–70°; bandwidth, 975 Hz/pixel; matrix size, 216 × 256; and pixel size, 1.25 mm × 1.25 mm. Multiple short-axis slices were prescribed from the mitral orifice to the LV apex with a slice thickness of 7 mm and a 3-mm gap. Approximately 10–12 short-axis slices were obtained, depending on the cardiac size. The images were ECG-gated and acquired during free breathing. A user-interface graphics program was used to determine the endocardial and epicardial contours of the LV in the short-axis view at each level, using a gray-level autocontouring algorithm provided by MatLab 7.9 (MathWorks, Natick, MA) and computing the area enclosed by each contour. The LV volumes for each time were subsequently determined using Simpson's rule to generate the volume–time curve of the LV. This rule is equivalent to the sum of the areas of the corresponding levels multiplied by the slice thickness. The end-diastolic volume (EDV) and end-systolic volume (ESV) were assessed based on the maximal and minimal values of the volume–time curve. These values were normalized according to body surface area and used to calculate the LVEF. The LV mass was calculated as the difference between the LV epicardial volume at the end diastole and LVEDV, multiplied by the density of the myocardium (1.05 g/ml).
201Thallium Single-Photon Emission Computed Tomography (201Tl SPECT) Analysis
To evaluate the myocardial viability under the influence of TG-0054 treatment, 201Thallium single-photon emission computed tomography (201Tl SPECT) analyses were conducted at 7 days (prior to the second dose treatment) and 12 weeks post-MI (Fig. 1). The myocardial viability was assessed using 201Tl SPECT rest- redistribution scintigraphy. SPECT images were obtained with a dual-head gamma camera (Millenium; GE Medical Systems, Milwaukee, WI, USA) as previously described (52). 201Tl was purchased from Institute of Nuclear Energy Research, Atomic Energy Council, Taiwan. The regional SPECT was assessed using the 17-segment model and the semiquantitative scoring system of defect of severity and extent, as recommended by the American Heart Society of Nuclear Cardiology. Each segment was scored based on the severity of tracer uptake by using a 5-point scoring system (0 = normal, 1 = equivocal, 2 = moderate, 3 = severe, and 4 = apparent absence of tracer uptake). The summed rest scores (SRS) of the 17 segmental rest scores were subsequently calculated.
Analysis of Vascular Density
The vessels in the cardiac histology specimens were stained using immunohistochemistry to assess the von Willebrand factor (vWF; DakoCytomation, USA). The number of vessels per 1 mm2 in the peri-infarct zone were counted in five randomly chosen fields per slide in a blinded manner and averaged for statistical analysis.
Cytokine Analyses
The plasma levels of TNF-α, IL-1β, and IL-6 at specified times were measured using commercially available ELISA kits (R&D Systems). The detection limits of the ELISA kits were 23.4 pg/ml for TNF-a, 39 pg/ml for IL-1β, and 18.8 pg/ml for IL-6. Myocardial expression of TNF-α, IL-1β, and IL-6 was determined by real-time quantitative PCR.
Real-Time Quantitative PCR
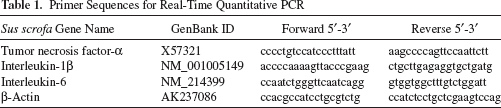
Total RNA was isolated from the infarcted border zone of the myocardial tissues by homogenization followed by TRIzol extraction (Invitrogen, USA); the RNA was subsequently purified using an RNeasy Mini Kit (Qiagen, Germany) and reversely transcribed to cDNA by using MMLV reverse transcriptase (Promega, USA). The real-time PCR reactions were performed in triplicates on the Roche LightCycler Instrument 1.5 by using the LightCycler® FastStart DNA MasterPLUS SYBR Green I kit (Roche) and oligonucleotide primers (Table 1) specific to target genes. The mixture was incubated at 95°C for 10 min followed by 40 cycles of 95°C for 10 s, 60°C for 15 s, and 72°C for 10 s. The data were analyzed by calculating the crossing point (CP) value, or the turning point that corresponded to the first maximum of the second derivative curve, using the LightCycler3 data analysis software. Porcine β-actin served as a reference gene. The expression fold of a target gene in the experimental groups (the saline group, the TG-0054 group, or the TG-0054-mobilized MSC group) relative to that in the control was calculated using he formula: 2-ΔΔCP where ΔCP=CPtarget gene -CPβ-actin and ΔΔCP = ΔCPaline, -ΔCPcontrol, -ΔCPTG-0054 -ΔCPcontrol or ΔCPTG-0054-mobilized MSC - ΔCPcontrol.
Primer Sequences for Real-Time Quantitative PCR
Statistical Analysis
All data were expressed as mean ± SD. Statistical analysis was carried out using the Prism 5 software (GraphPad Software, San Diego, CA, USA). Group differences were assessed by t-test, and three data sets were compared using one-way ANOVAs with Bonferroni's multiple comparison for measuring significance. Values of p < 0.05 (*), p < 0.01(**), or p < 0.001 (***) were considered statistically significant.
Results
Mobilization of CXCR4+ Stem Cells by TG-0054
To ensure that TG-0054, which was shown to mobilize HSCs in mice and humans (11, 22), was effective in mobilizing CXCR4+ stem cells in minipigs, we analyzed PB cell components after TG-0054 treatment (two intravenous doses of 2.85 mg/kg TG-0054 of 72 h apart). TG-0054 increased PB white blood cell (WBC) counts, which peaked at 0.5 and 1 h after the first dose and the second dose, respectively (Fig. 2A). In a similar pattern, TG-0054 increased CXCR4+ cells in WBCs (Fig. 2B, C). We then determined the stem cell markers, including CD34, CD133, and CD271, in TG-0054-mobilized-CXCR4+ cells at 1 h posttreatment. A large percentage of CXCR4+ cells expressed not only the HSC marker CD34+ (Fig. 3A) but also MSC markers CD133+ and CD271+ (Fig. 3B, C). Time course analysis of stem cell mobilization revealed that, after the first dose, CD34+, CD133+, and CD271+ cells in PB peaked at 0.5-1 h. CD34+ cells returned to baseline at 2 h, while CD133+ and CD271+ cells lingered and returned to baseline at 24 h and 6 h, respectively (Fig. 3A-C). These results suggest that TG-0054 mobilizes not only HSCs but also MSCs.

TG-0054 mobilizes CXCR4+ cells. (A) Mobilization of white blood cells (WBCs) by TG-0054. (B) Representative flow cytometry plots of TG-0054-mobilized WBCs and increases of CXCR4+ cells in TG-0054-mobilized WBCs. In (B), shaded areas indicate staining with chemokine CXC motif receptor 4 (CXCR4) antibodies, and open areas represent isotype controls. (C) Mobilization of CXCR4+ cells by TG-0054. Data were collected at 0, 0.5, 1, 2, 3, 6, and 24 h after treatment in healthy minipigs treated with two doses of TG-0054 (2.85 mg/kg, n = 3) or saline (equivalent volume to TG-0054, n = 3). Two injections per cycle were separated by 72 h. *p < 0.05, **p < 0.01 versus saline.

TG-0054 mobilizes HSCs and MSCs. Representative flow cytometry plots and time course analysis of TG-0054-mobilized (A) CD34+, (B) CD133+, or (C) CD271+ cells in WBCs. Data were collected at 0, 0.5, 1, 2, 3, 6, and 24 h after treatment in healthy minipigs treated with two doses of TG-0054 (2.85 mg/kg, n = 6) or saline (equivalent volume to TG-0054, n = 3). Two injections per cycle were separated by 72 h. *p < 0.05, **p < 0.01, ***p < 0.001 versus saline.
Mobilization of MSCs by TG-0054
In view of increased CD133+ and CD271+CXCR4+ cells by TG-0054, we ascertained MSC mobilization by assessing the properties of PA-MSCs and CD271-MSCs from control and TG-0054-treated PB. PA-MSCs were morphologically similar to BM-derived MSCs (BMMSCs) (Fig. 4A). CD271-MSCs, collected by immunomagnetic isolation and expansion, displayed similar differentiation property as PA-MSCs (Fig. 4B). Both PA-MSCs and CD271-MSCs tested negative for hematopoietic markers, such as CD11b, CD45, CD34, and CD14, and positive for CD271, CXCR4, CD90, CD44, and CD105 (Fig. 4C). Colony formation of PA-MSCs was increased from CFE 0.05 ± 0.02 before treatment to 0.92 ± 0.25 after the first dose of TG-0054 and to 3.18 ± 0.39 following the second dose of TG-0054 (Fig. 4D). These findings confirm that TG-0054 is effective in mobilizing MSCs from BM.
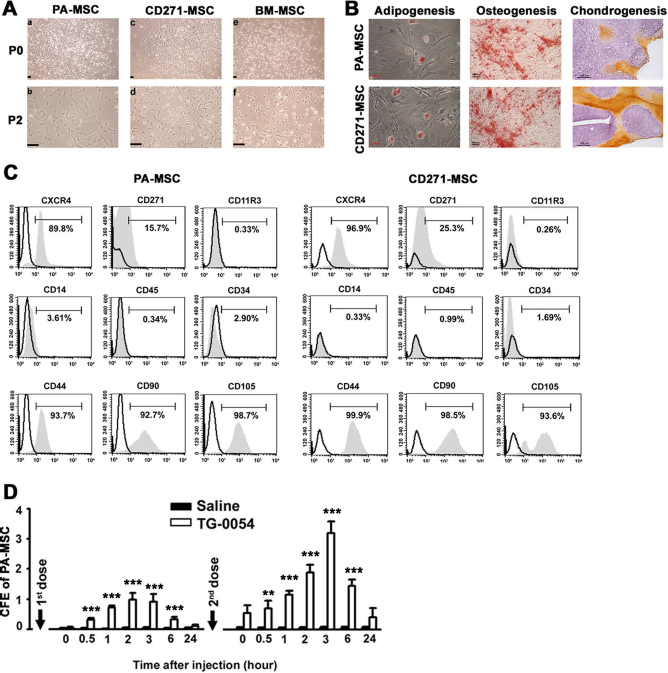
TG-0054-mobilized MSCs have colony-forming and differentiation potential. (A) Morphologic features of MSCs generated by plastic adherence (PA-MSCs) (a, b), MSCs derived from CD271+PBMCs (CD271-MSCs) (c, d), and BM-MSCs (e, f) at the primary culture (P0) and later passage (P2). (B) Differentiation of PA-MSCs and CD271-MSCs into adipogenic (Oil red O staining), osteogenic (Alizarin red S staining), and chondrogenic (immunohistochemical staining of collagen II) cell lineages. (C) Phenotypic characteristics of TG-0054-mobilized PA-MSCs and CD271-MSCs. In (C), shaded areas indicate staining with primary antibodies to cell surface antigens, and open areas represent isotype controls. (D) CFE (number of colonies/106 PBMCs) of PA-MSCs. Scale bar: 100 μm. **p < 0.01, ***p < 0.001 versus saline.
Immunomodulatory Effects of TG-0054-Mobilized MSCs
We evaluated the suppressive effects of PA-MSCs on the allogeneic proliferation of responder PBMCs, which were isolated from the minipigs before MI and 3 days post-MI. The proliferative capacity of the responder PBMCs increased post-MI (Fig. 5A), implying an immune activation in response to myocardial injury. Furthermore, the PA-MSCs significantly reduced the [3H]thymidine incorporation of allostimulated responder PBMCs at a magnitude equivalent to that of BM-MSCs (Fig. 5B). CD271+PBMCs and CD271-MSCs suppressed allogeneic PBMC proliferation (Fig. 5B). In the presence of PA-MSCs, CD271-MSCs, or CD271+PBMCs, the levels of TNF-α, IL-1β, and IL-6 were decreased in allogeneic MLR (Fig. 5C). These findings suggest that CD271-MSCs as well as PA-MSCs are competent in immunomodulation.
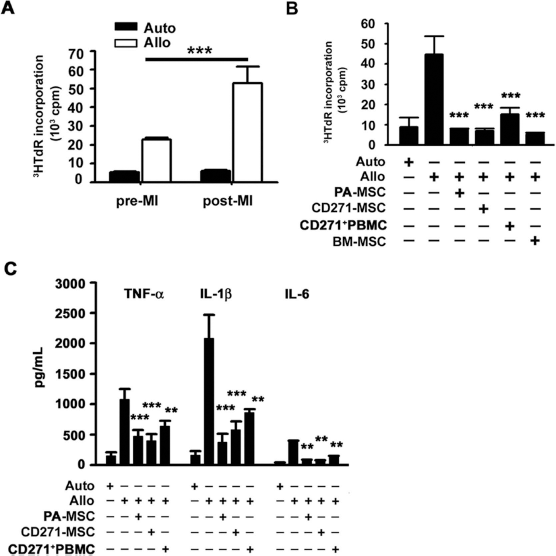
TG-0054-mobilized MSCs exert immunomodulatory effects. (A) Allogeneic-stimulated proliferation of PBMCs isolated from minipigs before (pre-MI) and 3 days after MI. (B) Proliferation of PBMCs and (C) levels of inflammatory cytokines in the allogeneic-simulated mixed lymphocyte cultures in the presence of various types of stem cells. **p < 0.01, ***p < 0.001 versus Allo.
Effects of TG-0054 on Cardiac Function and Remodeling
At 12 weeks post-MI, the EDV (Fig. 6B) and ESV (Fig. 6C) increased significantly in both groups. In the saline-treated group (n = 10), the LVEF significantly declined from 54 ± 8% at the baseline to 46 ± 10% at 12 weeks post-MI (p = 0.002, Fig. 6D). By contrast, the TG-0054-treated group (n = 12) demonstrated a preserved LVEF (47 ± 10% at the baseline versus 46 ± 12% at 12 weeks, p = 0.587) (Fig. 6D). The difference in the LVEF changes from the baseline (ΔLVEF) between the groups were statistically significant (p = 0.014) (Fig. 6D). Although the LV systolic function was preserved, treating MI pigs with TG-0054 did not significantly attenuate the LV hypertrophy, which is considered to contribute the increase in LV mass from the baseline (Fig. 6E).

TG-0054 preserves post-MI LV systolic function. (A) Representative cine MRI images obtained from different cardiac phases at the middle LV from different groups at different time points. (B) EDV, (C) ESV, (D) LVEF, and (E) LV mass over time in saline-treated (Saline) and TG-0054-treated (TG-0054) groups. P indicates the p value for difference in the LVEF changes from the baseline (ΔLVEF) between the groups.
Influences of TG-0054 on Myocardial Viability and Angiogenesis
To evaluate the effects of TG-0054 on myocardial viability, 201Tl SPECT was performed at 7 days and 12 weeks post-MI. The SRS changes from the baseline to the 12-week follow up (ΔSRS) did not significantly differ between the groups (Fig. 7B). Moreover, the vessel density in the peri-infarct myocardium was not significantly increased in response to TG-0054 treatment (173 ± 69/mm2 vs. 157 ± 61/mm2, p = 0.287) (Fig. 7C).

The influences of TG-0054 on myocardial viability and vascular density. (A) Representative short-axis (short), vertical long-axis (vertical), and horizontal long-axis (horizontal) SPECT images obtained from different groups at different time points. (B) Changes from the baseline of SRS (ΔSRS) in saline- or TG-0054-treated minipigs. (C) Photographs of the vol Willebrand factor (vWF)-stained sections in the peri-infarct myocardium from the saline- and TG-0054-treated groups. The numbers of vWF-positive vessels in each area were manually counted in each section for immunohistochemistry. Scale bar: 100 μm.
Inhibition of Proinflammatory Cytokines by TG-0054 Treatment
We suspected that TG-0054 may preserve myocardial function through MSC-mediated modulation of proinflammatory cytokine production. We determined the myocardial mRNA expression of TNF-α, IL-1β, and IL-6 at 7 days post-MI. Compared to the saline group, TNF-α, IL-1β, and IL-6 mRNA levels were markedly reduced in the TG-0054 group (Fig. 8). We also analyzed plasma levels of TNF-α, IL-1β, and IL-6 in TG-0054 versus saline groups. TNF-α level was not significantly different between the two groups at 3 days post-MI but was significantly lower in the TG-0054 group than that in the controls from 7 days post-MI till the end of the study (Fig. 9A). TG-0054 reduced IL-1β and IL-6 in a time-dependent manner parallel to the modulation of TNF-α (Fig. 9A).
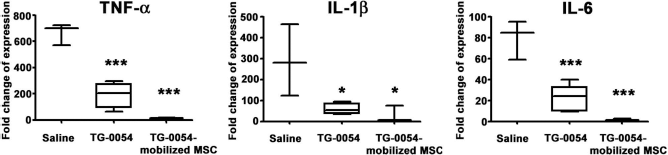
Both TG-0054 and TG-0054-mobilized CD271-MSCs inhibit post-MI myocardial proinflammatory cytokines. Myocardial mRNA levels of TNF-α, IL-1β, and IL-6 at 7 days post-MI in minipigs after receiving two doses of saline (n = 3), TG-0054 (n = 5), or TG-0054-mobilized CD271-MSCs (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001 versus saline.
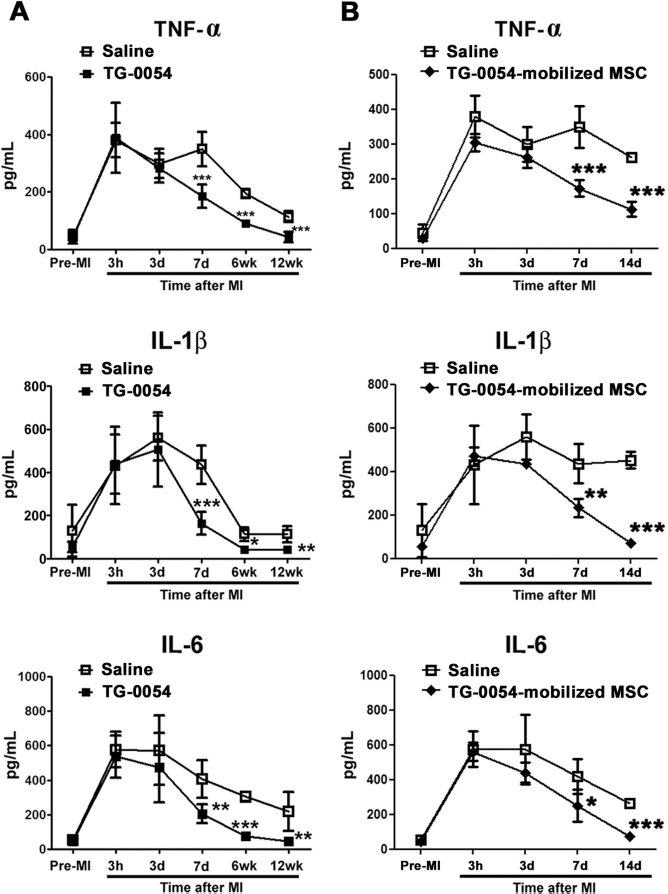
Both TG-0054 and TG-0054-mobilized CD271-MSCs suppress plasma proinflammatory cytokines. (A) Plasma levels of TNF-α, IL-1β, and IL-6 at pre-MI (n = 6), 3 h (n = 6), 3 days (n = 6), 7 days (n = 6), 6 weeks (n = 3), and 12 weeks (n = 6) post-MI in minipigs after receiving two doses of saline or TG-0054. (B) Plasma levels of TNF-α, IL-1β, and IL-6 at pre-MI (n = 3), 3 h (n = 3), 3 days (n = 3), 7 days (n = 5), and 14 days (n = 3) post-MI in minipigs after receiving two doses of saline or TG-0054-mobilized CD271-MSCs. *p < 0.05, **p < 0.01, ***p < 0.001 versus saline.
Suppression of Proinflammatory Cytokines by TG-0054-Mobilized CD271-MSCs
We hypothesized that the MSC egress yielded by TG-0054 treatment accounts for the inhibition of proinflammatory cytokines. Therefore, a short-term study was designed to investigate the alteration of inflammatory cytokines by TG-0054-mobilized CD271-MSCs. Compared to the saline group, TNF-α, IL-1β, and IL-6 mRNA levels were significantly reduced in the TG-0054-mobilized MSC-treated group (Fig. 8). The intravenous infusion of the first dose of CD271-MSCs reduced the plasma levels of TNF-α (349 ± 60 pg/ml vs. 172 ± 21 pg/ml, p < 0.001), IL-1β (436 ± 89 pg/ml vs. 234 ± 38 pg/ml, p = 0.001), and IL-6 (419 ± 100 pg/ml vs. 251 ± 84 pg/ml, p = 0.019) compared with those in the saline group at 7 days post-MI (Fig. 9B). After the second dose, levels of all three cytokines were further decreased in the TG-0054-mobilized MSC-treated group and significantly lowered than those in the saline group at 14 days post-MI (Fig. 9B). The modulatory effect of CD271-MSC infusion on cytokine levels correlated temporally with that of TG-0054 treatment (Fig. 9A). These results support the notion that the anti-inflammatory effect of TG-0054, at least in part, can be attributed to the egress of CD271-MSCs from BM.
Discussion
TG-0054 is a CXCR4 antagonist and was reported to mobilize BM HSCs (22). It is being evaluated for treating human BM malignancies (35, 36). In this study, we addressed its effect on mobilizing MSCs and its therapeutic potential for postinfarction HF in a porcine model. Our findings confirm that TG-0054 is active in mobilizing HSCs in minipigs and demonstrate that it also mobilizes CXCR4+ MSCs including the CD271-MSCs. The CD271-MSC is a subset of BM-derived MSCs possessing immunomodulating and allosuppressive properties (28). Our results show that porcine CD271-MSCs are morphologically similar to BM-MSCs or PA-MSCs. CD271-MSCs express common MSC markers and can be driven to differentiate into adipocytes, osteoblasts, and chondrocytes. Thus, through blocking CXCR4 interaction with its ligands, TG-0054 mobilizes CXCR4+ HSCs as well as MSCs. It mobilizes a considerable number of CD271-MSCs, which can be isolated and expanded in vitro for potential therapeutic application.
Administration of TG-0054 at the two-dose protocol starting at 3 days post-MI results in significant improvement of cardiac contractile function without an apparent effect on cardiac structural change. We also found that TG-0054 reduces the production of key proinflammatory cytokines. The results suggest that the TG-0054-induced improvement in systolic function may be attributed to the suppression of post-MI inflammation and subsequent myocardial remodeling.
Inflammatory responses and cytokine elaboration are activated after MI and may directly and indirectly compromise LV performance levels (37). It has been reported that the circulatory levels of inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, correlate with HF progression, severity, and mortality (7). The circulating inflammatory mediators may be released from the failing heart or from circulating leukocytes, platelets, endothelial cells, and the lung or liver (53). In this study, the expressions of inflammatory cytokines in the myocardium and circulation were substantially reduced in the acute postinfarction period following TG-0054 treatment. Moreover, the downregulation of systemic inflammation persisted throughout the follow-up period. However, our data are insufficient to support a cause–effect relationship between reduction of inflammatory cytokines and improvement of postinfarction cardiac function. Genetic manipulation in a murine model may be useful in elucidating the causal relationship and its underlying mechanisms.
We found a correlation between the reduction of inflammatory cytokine production, increase in circulating MSCs, and improvement in cardiac function. BM-derived MSCs suppress allogeneic immune reaction and the consequent release of inflammatory responses (51). We provide in vitro evidence that TG-0054-mobilized CD271-MSCs reduced alloimmune-mediated lymphocyte proliferation as well as TNF-α, IL-1β, and IL-6 production. Furthermore, infusion of CD271-MSCs in vivo reduced both myocardial and plasma levels of TNF-α, IL-1β, and IL-6. These findings suggest that TG-0054 controls post-MI inflammation through mobilizing MSCs including the CD271+ subset of MSCs.
The anti-inflammatory effects of TG-0054 may be mediated by pathways other than MSC mobilization. Certain studies have reported that CD34+ stem cells purged by G-CSF exhibit immune regulatory activity (16, 41). CXCR4 antagonist-mobilized autologous HSCs suppressed allogeneic MLR and prolonged islet allograft survival (14). In addition, CXCR4 antagonists, such as AMD3100, at doses insufficient to mobilize stem cells, were shown to alleviate neuroinflammatory responses in a mouse model of focal cerebral ischemia (21, 43). Neutralizing antibody targeting CXCR4 was shown to interrupt the costimulation effects of SDF-1 on CD4+ T-cells (33). Thus, the beneficial effects of TG-0054 observed in the present study are multifaceted. A CXCR4 blockade and mobilized cell-mediated immunomodulation are not mutually exclusive.
Neovascularization is a hallmark response of the myocardium to MI (37). Another CXCR4 antagonist, AMD3100, has been reported to preserve post-MI cardiac function by enhancing EPC-mediated neovascularization (25); however, in the present study, the efficacy of TG-0054 regarding LVEF preservation yielded no significant enhancement of vascular density in the peri-infarct myocardium (Fig. 7). More recent studies suggest that proinflammatory cytokines elaborated during MI can set the stage for subsequent angiogenesis. In a postinfarction model, expression of vascular endothelial growth factor (VEGF) along with expression of TNF-α and inducible nitric oxide synthase was noted (18). This finding suggests that angiogenic factors are released at the same time as proinflammatory cytokines. Therefore, attenuation of post-MI inflammation by TG-0054 may blunt the potential angiogenic effects of the mobilized stem cells. The upregulation of angiogenic factors is usually immediate in response to ischemia (17, 29). Further studies to quantify VEGF expression in the peri-infarct myocardium in the early stage after MI will be helpful to confirm the net effects of TG-0054 on post-MI angiogenesis.
It is interesting that antagonizing (25) and promoting (46, 54) SDF-1/CXCR4 both have been reported to preserve myocardial function after MI. In the BM, stem cells are anchored by SDF-1/CXCR4 binding. Intravenous delivery of TG-0054, as shown in this report, transiently disrupted SDF-1/CXCR4 interaction and purged the BM-derived CXCR4+ stem cells into circulation. In the infarcted heart, a SDF-1 gradient toward the injured tissue will facilitate local SDF-1/CXCR4 interaction and attract the circulating stem cells. A growing body of evidence suggested that the myocardial SDF-1 expression was rapidly upregulated and maintained for 1 week after MI (4, 30). Therapeutic strategies, including myocardial injection of exogenous protease-resistant SDF-1 (46), adenovirus-mediated SDF-1 transduction of the infarcted myocardium (1), and overexpression of CXCR4 in transplanted MSCs (54), have been demonstrated to drive endogenous or transplanted stem cells toward injured myocardium and improve cardiac function (46, 54).
A reversible CXCR4 antagonist that offers a transient release of stem cells from BM without compromising the recruitment of stem cells within the injured myocardium may be important for our therapeutic strategy. In this study, we released stem cells from BM by intravenous injection of dual-dose TG-0054, on 3 days and 7 days post-MI. Based on the time course analysis of stem cell mobilization (Fig. 3), the blockade of SDF-1/CXCR4 interaction in the BM are of short duration. The preservation of LV function by TG-0054 is in agreement with the findings using AMD3100 (25), another reversible CXCR4 blockade, suggesting that a short term of stem cell mobilization is beneficial. By contrast, continuous or chronic CXCR4 blockade was reported to exacerbate postinfarction LV remodeling (12, 25). This is probably related to the long-term disruption of the myocardial driving chemotaxis of mobilized stem cells through systemic delivery. In addition, G-CSF activates extracellular proteases that irreversibly cleave cell-surface CXCR4 to release BM-derived stem cells (10). However, the efficacy results from REVIVAL-2 clinical trial have been disappointing (56), perhaps because the docking of G-CSF-mobilized cells in the ischemic region is impaired. Therefore, the transient efflux of stem cells into the circulation may have to be complemented by homing signals at the injured tissue. Both the release and the recruitment of stem cells should be balanced by manipulating the SDF-1/CXCR4 interaction for cell therapy in ischemic HF.
Conclusions
In summary, CXCR4 antagonist TG-0054 protects the post-MI porcine heart from the progressive deterioration of systolic function and is associated with attenuation of myocardial and systemic inflammation. This immunomodulatory effect is at least partially attributed to the mobilization of MSCs. This insight may facilitate exploring novel stem cell-based therapy for treating post-MI HF.
Footnotes
Acknowledgments
This study was supported by research grants from the National Research Program for Biopharmaceuticals (NRPB, 101TM1009), National Health Research Institutes (NHRI-CS-099-PP-13 and NHRI-CS-100-PP-16), and Ministry of Science and Technology (NSC-101-2321-B-400-012, MOST 103-2321-B-002-101, and MOST 103-2321-B-002-083), and assisted in part the 3rd core facility at National Taiwan University Hospital (NTUH). We thank C. C. Yu (Laboratory Animal Center, National Taiwan University College of Medicine) and C. C. Liu (Department of Nuclear Medicine, NTUH) for their technical assistance. M.C.H. is the cofounder and a shareholder of TaiGen Biotechnology Co., Ltd., Taipei, Taiwan. Y.H.H. is an employee of TaiGen Biotechnology Co., Ltd., Taipei, Taiwan.
