Abstract
Transplant arteriosclerosis (TA) remains the major limitation of long-term graft survival in heart transplantation despite the advances in immunosuppressants. Mesenchymal stem cells (MSCs) have been demonstrated to suppress allogeneic immune responses by numerous in vitro studies. However, the immunomodulatory effects of MSCs in vivo are controversial and the underlying molecular mechanisms are not conclusive. In this study, we investigated the therapeutic potential of autologous bone marrow-derived MSCs on TA in a porcine model of femoral artery transplantation. MSCs or saline were injected into the soft tissue surrounding the arterial grafts immediately postanastomosis. Four weeks after transplantation, neointimal formation increased significantly in untreated allografts compared with the MSC-treated grafts as assessed by intravascular ultrasound (maximum luminal area stenosis: 40 ± 12% vs. 18 ± 6%, p < 0.001). Grafts harvested at 4 weeks showed dense perivascular lymphocyte infiltration accompanied by significant intimal hyperplasia in the untreated but not in the MSC-treated allografts. Serial angiographic examination showed that all of the untreated allografts became occluded at the 8th week whereas the majority of the MSC-treated grafts remained patent at the 12th week posttransplantation (n = 12 each group, p < 0.001). Quantitative PCR analysis revealed that Foxp3 expression was comparable between the untreated and the MSC-treated groups. However, expression of interleukin-10 (IL-10), interferon-γ (IFN-γ), and indoleamine 2,3-dioxygenase (IDO) was increased significantly in the MSC-treated allografts compared with that in the allograft controls (p = 0.021 for IL-10, p = 0.003 for IFN-γ, and p = 0.008 for IDO). In conclusion, local delivery of autologous MSCs alleviates TA by inducing allograft tolerance via enhanced expression of IL-10, IFN-γ, and IDO but not Foxp3-positive cells in the vessel wall. These results suggest that MSCs induce immune tolerance by activating the type 1 regulatory T-like cells.
Introduction
Heart transplantation is one of the established treatment modalities for end-stage heart failure. However, the long-term survival of recipients is limited mainly due to transplant arteriosclerosis (TA), which is characterized by accelerated concentric intimal hyperplasia diffusely involving the entire coronary vascular bed (15). TA is a chronic inflammatory disease that develops in the setting of conventional immunosuppression regimens that are effective in the treatment of acute rejection to the myocardium of allografts. So far, there is no effective therapy for TA. Once TA complication occurs, a repeat heart transplantation will be necessary, which is costly and limited by the graft availability. Thus, it is important to investigate new strategies for TA therapy in order to improve the survival of heart transplantation recipients.
Mesenchymal stem cells (MSCs) isolated from bone marrow (BM) are nonhematopoietic stromal cells that are able to differentiate into multiple lineages, such as adipocytes, osteocytes, and chondrocytes (19). In addition to their regenerative potential, BM-derived MSCs have been demonstrated to suppress immune responses in vitro by inhibiting the proliferation of activated T cells, preventing the development of cytotoxic T cells, promoting the generation of regulatory T cells (Tregs) (1,9,21), and modulating the maturation or function of dendritic cells (DCs) (3).
Multiple potential molecules underlying the immunosuppressive effect of MSCs have been proposed based on studies in vitro (26). MSCs inhibit allogeneic lymphocyte proliferation by secreting immunosuppressive factors including hepatocyte growth factor (HGF), interleukin-10 (IL-10), prostaglandin E2 (PGE2), and transforming growth factor-β1 (TGF-β1), or through expression of indoleamine 2,3-dioxygenase (IDO) upon stimulation with interferon-γ (IFN-γ) (1,4,8,9,23). IDO suppresses T-cell responses and promotes immunological tolerance via tryptophan depletion and the production of proapoptotic downstream metabolites (17).
It is not entirely clear whether MSCs function in vivo as they do in vitro. The immunosuppressive effect of MSCs has been examined in several rodent models of heart transplantation and the results are conflicting. Intravenous administration of MSCs was reported to have a modest effect on prolonging graft survival in some studies (11,27), and accelerating graft rejection in others (14). When combined with low-dose mycophenolate (20) or rapamycin (11), MSCs seemed to be capable of inducing long-term myocardial allograft tolerance, but not effective in the prevention of TA (20). Moreover, MSCs delivered via systemic infusion were found to be only transiently expressed in animal models of cardiac transplantation (5,20) and therefore might have little time to secret immunomodulatory factors. It has been suggested that induction of tolerance in the target tissue of immune damage does not have to be systemic and localized therapy may circumvent the requirement for large number of regulatory cells (24). Thus, local delivery of immunomodulatory cells may be an alternative to alleviate allograft rejection.
In the cardiac transplantation model, the therapeutic potential of MSCs on TA and the underlying mechanisms could not be analyzed without the confounding of immunosuppressants that are required to prevent acute myocardial rejection. Thus, in the present study, we investigated the therapeutic effect of MSCs on TA in a porcine model of femoral artery transplantation without concomitant immunosuppressants. Our results indicate that local administration of autologous MSCs effectively prevents TA through enhanced expression of IL-10, IFN-γ, and IDO but not forkhead box P3 (Foxp3).
Materials and Methods
Porcine MSC Preparation and Characterization
Mononuclear cells were isolated from porcine BM aspirate by using density gradient centrifugation (Ficoll-Paque, GE Healthcare Bio-Sciences AB). After overnight incubation, nonadherent cells were washed out with changes of the medium and the attached cells were expanded in MSC medium consisting of α-minimum essential medium (αMEM, Invitrogen) and 10% fetal bovine serum (Invitrogen) supplemented with 10 ng/ml basic fibroblast growth factor and 10 ng/ml epithelial cell growth factor (R&D). The cells were confirmed as stem cells by differentiation into adipogenic, osteogenic, and chondrogenic lineages as described previously (19). To trace the injected cells in vivo, one third of MSCs were labeled with 100 μg/ml of superparamagnetic iron oxide (SPIO) particles (Resovist®, Schering AG) at 37°C overnight. On the day of transplantation, cells were trypsinized from culture wares, washed with αMEM, and then resuspended in Iscove's modified Dulbecco's medium (Invitrogen) at a density of 3 × 106/ml.
Flow Cytometry
MSCs isolated from six different animals (passage 3) were labeled with primary antibodies to cell surface antigens: CD11R3, CD44, CD45, src-like adaptor I (SLA I), SLA II (AbD Serotec), CD34 (Santa Cruz Biotechnology), CD90 (Becton Dickinson), CD105 (Abcam), or corresponding isotype controls. Single-color staining was performed using a fluorescein isothiocyanate- or phycoerythrin-conjugated secondary antibody and analyzed using a FACS Calibur instrument and with Cell Quest software (Becton Dickinson).
One-Way Mixed Lymphocyte Reaction (MLR)
Peripheral blood mononuclear cells were isolated by gradient centrifugation and contaminating red blood cells were eliminated using ACK lysing buffer (NH4Cl 150 mM, KHCO3 10 mM, and EDTA 0.01 mM). For conditioned medium preparation, MSCs autologous to responder were plated at a density of 2 × 103/cm2 and grew in MSC medium described above to 50% confluence; the medium was then changed to RPMI-1640 medium (Invitrogen) containing 15% heat-inactivated adult porcine serum (Invitrogen), and culture supernatant was harvested after 72 h. Prior to the MLR, MSCs and stimulators were rendered mitotically inactive by treatment with mitomycin-C (25 μg/ml, Kyowa Hakko Kogyo Co, Ltd.). A coculture was made by mixing 1 × 105 stimulator and responder cells per well (1:1) in 96-well, round-bottomed plates (Becton Dickinson) in 0.2 ml of RPMI-1640 medium supplemented with 15% heat-inactivated adult porcine serum. For inhibition experiments, 5 × 104 MSCs or 0.1 ml of conditioned medium were added to the coculture at the beginning of the reaction. Each combination of responders and stimulators was run in triplicate, and each responder was set up against autologous simulator as a negative control. On the fourth day of coculture, plates were pulsed with 1 μCi of [3H]thymidine (PerkinElmer) for a further 18 h. Cells were then harvested and [3H]thymidine incorporation was assessed using a MicroBeta FilterMate-96 Harvester (PerkinElmer) and a 1450 MicroBeta TriLux (PerkinElmer). Proliferation was expressed as the incorporated radioactivity in counts per minutes.
Pig Model of Femoral Artery Transplantation
Forty minipigs (20—25 kg) bred in Laboratory Animal Center of National Taiwan University were used in the study according to the Guidelines for Animal Care. The experimental protocol was approved by the Institutional Animal Care and Use Committee (approval number 20060178). Animals were initially sedated by intramuscular injection of Zoletil (5 mg/kg, Virbac Laboratories, France), followed by endotracheal intubation. Anesthesia was maintained throughout the procedure by mechanical ventilation with isoflurane (1—3%).
For allograft transplantation (n = 30), femoral arteries of both recipient and donor pigs were dissected and heparin (5,000 IU, Leo Pharmaceutical Products, Denmark) was administered intravenously prior to transplantation. A 1-cm vascular segment was excised from a femoral artery of the recipient pig, and then replaced with a 3-cm femoral artery segment harvested from the donor pig. Grafts were implanted in an end-to-end fashion anastomosis with continuous 8-0 prolene sutures. The limb ischemia time was less than 30 min (time from clamp to reperfusion) for each extremity. Autologous MSCs (3 × 106 in 1 ml divided into 10 injections for each graft for Allo + MSC group, n = 15) or saline (1 ml divided into 10 injections for Allo group, n = 15) were injected into the skeletal muscle surrounding the vascular grafts. The incision wound was closed in layers. Autograft transplantation (Auto group, n = 10) was performed by inserting the femoral artery with a segment of graft harvested from contralateral femoral artery of the same animal, followed with saline injection. Following operation, all animals received oral prophylactic cephalexin 250 mg/day for 5 days.
Angiography and Intravascular Ultrasound (IVUS)
Graft patency was assessed at the 4th, 8th, and 12th weeks following transplantation by angiography. The pigs were anesthetized as described above. A 6-F vascular sheath was introduced into the carotid artery and selective angiography was performed through a 6-F catheter engaged at the external iliac artery upstream to the femoral arterial graft. For IVUS assessment, under the guidance of a 0.014-inch soft wire, the IVUS catheter (Atlantis™ SRPro 2.5F, 40-MHz, Boston Scientific Corp, Fremont, CA) was advanced so that the IVUS transducer was positioned distal to the arterial graft. The IVUS transducer was pulled back at a constant speed of 0.5 mm/s along the length of the vessel to the ostium of common femoral artery. The IVUS records were digitized and the maximum area stenosis in the middle of the vascular grafts was analyzed with the resident software (Galax system, Boston Scientific Co.) (16).
Magnetic Resonance Imaging (MRI) Analysis
At 3 days and 4 and 12 weeks after cell delivery, MRI scanning was performed on a 1.5-T MR system (Siemens Sonata, Erlangen, Germany). The animals were examined in the supine position with bilateral thighs covered by using a four-element phased-array surface coil for signal reception. For tracking SPIO particles in the lower limbs, images were obtained consecutively using an axial T2-weighted fast spin echo [repetition time (TR)/echo time (TE)/flip angle: 6730 ms/104 ms/150°; field of view (FOV): 300 mm; scanning matrix: 192 × 256, slice thickness: 5 mm, 30 slices], an axial T1-weighted fast spin echo (TR/TE/flip angle: 502 ms/9.1 ms/150°; FOV: 300 mm), and an axial T2*-weighted gradient-echo (TR/TE/flip angle: 1170 ms/22 ms/30°; FOV: 300 mm) sequence.
Collection of Arterial Graft Specimens
Eight (Auto: n = 2, Allo: n = 3, and Allo + MSC: n = 3) grafts were harvested from the study animals at 4 weeks after transplantation for early stage histological examination. The remaining grafts (n = 32) were harvested when they were found to be occluded on angiograms or at the end of the study (12 weeks posttransplantation). The grafts were cut into 3-mm segments and then snap frozen in liquid nitrogen for RNA isolation, or fixed with 10% buffered formalin for histological analysis.
Real-Time Quantitative PCR
Total RNA was isolated from graft tissues by homogenization followed by Trizol extraction (Invitrogen), purified by RNeasy Mini Kit (Qiagen), and reversely transcribed to cDNA using MMLV reverse transcriptase (Promega). Real-time PCR reactions were performed in triplicates on the Roche LightCycler Instrument 1.5 using LightCycler® FastStart DNA MasterPLUS SYBR Green I kit (Roche) and oligonucleotide primers (Table 1) specific to target genes. The mixture was incubated at 95°C for 10 min followed by 40 cycles of 95°C for 10 s, 60°C for 15 s, and 72°C for 10 s. Data were analyzed by calculating the crossing point (CP) value, the turning point corresponding to the first maximum of the second derivative curve, using the LightCycler3 data analysis software. Pig β-actin served as a reference gene. The expression fold of a target gene in the allograft relative to that in the autograft was calculated by the formula: 2−ΔΔCP where ΔCP = CPtarget gene − CPβ-actin and ΔΔCP = ΔCPallograft − ΔCPautograft.
Primer Sequences for Real-Time Quantitative PCR

DFCI pig gene index.
GenBank ID.
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum concentrations of IL-10 and IFN-γ were determined in duplicate with commercially available ELISA kits for detection of porcine cytokines (Quantikine Porcine Immunoassays, R&D). The minimum limits of detection were: IL-10, 31 pg/ml and IFN-γ, 39 pg/ml.
Histological Analysis
Formalin-fixed vascular grafts were embedded in paraffin, cross-sectioned, deparaffinized, rehydrated, and subjected to hematoxylin and eosin (H&E) or orcein staining. Intimal hyperplasia of vascular grafts was determined from the computer images of orcein-stained cross sections. The area surrounded by internal elastic lamina (IELA) and the luminal area (LA) were calculated by an image analysis program (NIH Image). The severity of intimal hyperplasia was calculated by the formula: [(IELA − LA)/IELA] x 100%. For Prussian blue staining of SPIO particles, tissue sections were incubated with 5% potassium ferrocyanide in 10% hydrochloric acid for 30 min and counterstained with nuclear fast red (Sigma). For immunohistochemical analysis, tissue sections were subjected to antigen retrieval by heating in 10 mM sodium citrate buffer (pH 7.2) for 10 min in a microwave, then treated for 5 min in 3% hydrogen peroxide to block endogenous peroxidase activity. Tissue sections were incubated with antibodies to CD3 (Vertana), CD14 (Novocastra), CD20, smooth muscle cell αalpha-actin (α-SMA, Dako), collagen type II (Millipore), IDO (Santa Cruz Biotechnology), IL-10, and IFN-γ (R&D) overnight at 4°C. The application of isotype antibodies to tissue sections was used in negative controls. Signals were developed in a 30-min incubation step with the polymer-HRP detection kit QD420-YIK (BioGenex) followed by 3,3′-diaminobenzidine detection. Slides were counterstained with hematoxylin.
Statistical Analysis
All data are expressed as mean ± SD. Statistical analysis was performed using the Mann-Whitney test or Student t-test for continuous variables and chi-square analysis for categorical variables, respectively. Values of p < 0.05 were considered statistically significant.
Results
Characterization of Porcine MSCs
Cells were isolated from porcine BM and selected based on their adherence to plastic. After 7 days of culture, the adherent cells had a spindle-like appearance (Fig. 1A,a). FACS analysis revealed that these BM-derived cells were negative for hematopoietic cell markers CD11R3, CD34, and CD45, positive for the cell adhesion molecule CD44, progenitor cell markers CD90, and CD105, and MHC class I but not MHC class II (Fig. 1B). They were able to differentiate into osteocytes, chondrocytes, and adipocytes (Fig. 1A,b—d). Both MSCs and their conditioned medium were able to suppress proliferation of allogeneic peripheral mononuclear cells in MLRs (Fig. 1C). Taken together, the characteristics of our cells fulfill the recommended definition of MSCs for their application to the immunomodulation therapy of solid organ transplantation (7).

Characteristics of porcine mesenchymal stem cells (MSCs). (A) The morphology of porcine MSCs examined by phase contrast microscopy (a) and their differentiation into chondrogenic (b, immunohistochemical staining of collagen II), adipogenic (c, oil red staining), and osteogenic (d, von Kossa staining) cell lineages. Black bars denote 100 mm for all panels. (B) Phenotypic analysis of porcine MSCs by flow cytometry. Open areas indicate staining with the specific antibodies, whereas the gray regions represent staining with isotype control antibodies. SLA: swine leukocyte antigen (C). The immunosuppressive effects of porcine MSCs (a) and conditioned media (CM) from 72-h MSC culture (b) on peripheral blood mononuclear cell proliferation in mixed lymphocyte reactions. Results are expressed as counts per minutes (cpm) from four independent experiments each performed in triplicate. Auto: autologous response, Allo: allogeneic response. #p = 0.007 between Allo and Allo + MSC, *p < 0.001 between Allo and Allo + CM.
MSCs Attenuate Neointima Formation in Transplanted Vessels
We investigated the immunosuppressive effect of autologous MSCs on TA in a porcine model of femoral artery transplantation. To assess the intimal hyperplasia in the early stage of TA (16), we performed IVUS examination 4 weeks after transplantation. As shown in Figure 2A, intimal hyperplasia was observed diffusely along the untreated allograft but not in the autograft, indicating that the neointimal formation was resulted from allogeneic transplantation but not surgical injury. Intimal hyperplasia was not evident in the MSC-treated allografts. The extent of maximum area stenosis in the MSC-treated allografts was significantly lower than that in the untreated allografts at 4 weeks postarterial transplantation (18 ± 6% vs. 40 ± 12%, p < 0.001) (Fig. 2B).

Neointima formation in vascular grafts at early stage. (A) Intravascular ultrasound (IVUS) analysis of intimal thickening of autografts (a, b), untreated (c, d) and MSC-treated (e, f) allografts at the 4th week after transplantation. Representative images of cross (a, c, e) and longitudinal (b, d, f) sections of the grafts are illustrated. White triangles mark the sites of anastomoses and white arrows indicate the sites of intimal hyperplasia. Auto, autografts; Allo, untreated allografts; Allo + MSC, MSC-treated allografts. (B) Percentage of maximum area stenosis in autografts (n = 8), untreated (n = 7), and MSC-treated (n = 10) allografts assessed by IVUS at the 4th week after transplantation. #p < 0.001 between Auto and Allo, ‡p < 0.001 between Auto and Allo + MSC, *p < 0.001 between Allo and Allo + MSC. (C) Histological examination of autografts (a, d), untreated (b, e) and MSC-treated (c, f) allografts at the 4th week posttransplantation with hematoxylin and eosin (H&E) staining (a—c) and CD3 (d—f) immunohistochemical staining. White arrows mark the boundaries of the external elastic lamina, gray arrows mark the internal elastic lamina, and black arrows mark CD3+ cells. Scale bars: 100 μm for all panels. (D) The number of infiltrating cells (a) and the percentage of CD3+ cells (b) in the autografts (n = 2), untreated (n = 3) and MSC-treated (n = 3) allografts at the 4th week after transplantation. Each bar denotes counts from 10 cross-sectional images per graft. #p < 0.001 between Auto and Allo, *p < 0.001 between Allo and Allo + MSC.
Histological examinations revealed increased cell infiltration in the intimal and perivascular regions of the untreated allograft versus autograft (Fig. 2C,b vs. 2C,a, and 2D,a). MSC treatment resulted in a significant reduction of cell infiltration (Fig. 2C,c and 2D,a). Immunohistochemical analysis showed that a vast majority of the infiltrating cells in the untreated allografts were CD3+ lymphocytes that were significantly reduced by MSC treatment (Fig. 2C,e vs. 2C,f, and 2D,b). These results indicate that autologous MSCs prevent TA by attenuating alloantigen-induced inflammatory reactions and intimal hyperplasia.
MSCs Protect the Long-Term Patency of Transplanted Vessels
The long-term effects of MSCs on arterial grafts were evaluated by serial angiographic examinations up to 12 weeks. The results revealed patent anastomoses in all autografts (n = 8) throughout the study period of 12 weeks (Fig. 3A, B). On the contrary, only 58% (7/12) of the allografts in the control group were patent at the 4th week and all of them became occluded by the end of the 8th week posttransplantation (Fig. 3A, B). Notably, MSC treatment significantly improved vascular patency. Up to 83% (10/12) of the MSC-treated allografts remained patent 12 weeks after transplantation (p < 0.001 between untreated and MSC-treated allografts) (Fig. 3A, B).
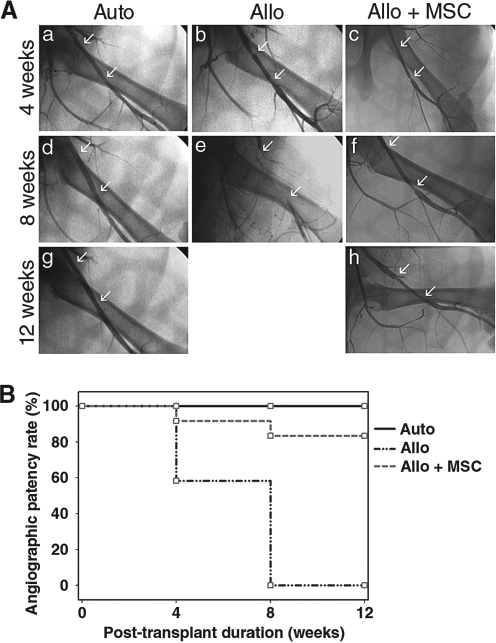
Time course analysis of graft patency. (A) Patency of vascular grafts was assessed periodically by angiography at 4 (a—c), 8 (d—f), and 12 (g, h) weeks posttransplantation or until total occlusion of the grafts was noted. Representative serial images of autografts (a, d, g), untreated (b, e), and MSC-treated allografts (c, f, h) are illustrated. White arrows mark the anastomotic sites of arterial grafts. Angiography was not applicable to untreated allografts at 12 weeks as total occlusion of the grafts was noted at 8 weeks posttransplantation. (B) Angiographic patency rate of autografts (n = 8), untreated (n = 12), and MSC-treated (n = 12) allografts. p < 0.001 between Auto and Allo, p = 0.139 between Auto and Allo + MSC, and p < 0.001 between Allo and Allo + MSC.
Histological examination of the vascular grafts at the time of occlusion or at the study end point (12 weeks after transplantation) confirmed the angiographic findings (Fig. 4A). Compared to autografts, the untreated allograft was completely occluded by intimal hyperplasia, which was enriched with α-SMA-positive smooth muscle cells and possibly myofibroblasts (Fig. 4A, B). The histological findings of the untreated allograft are consistent with the vascular changes of human TA. The intimal hyperplasia of the allograft was significantly attenuated by MSC treatment (Fig. 4B), as were the excessive α-SMA-positive cells (Fig. 4A). These findings indicate that locally delivered MSCs not only suppress TA in the early stage posttransplantation, but also improve long-term graft patency rate by controlling intimal hyperplasia.
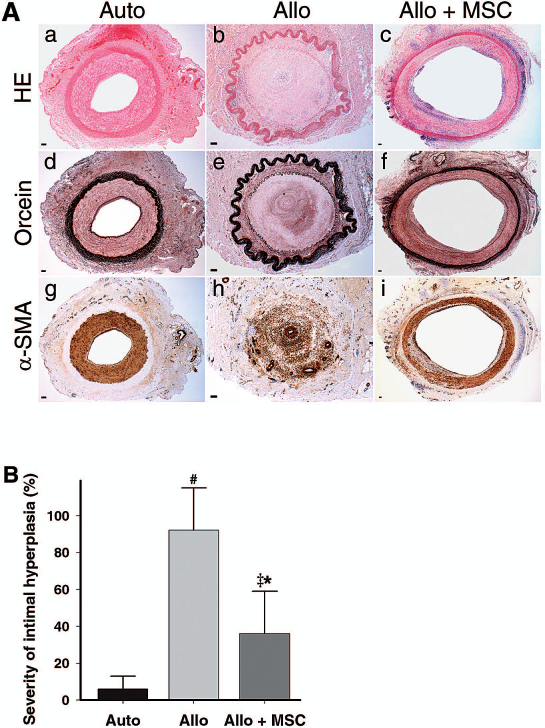
Histological examination of vascular grafts. (A) Representative cross-sectional images of autografts (a, d, g), untreated (b, e, h) and MSC-treated (c, f, i) allografts at the study end point (12 weeks posttransplantation) or at the time of occlusion with H&E (a—c), orcein (d—f), and smooth muscle cell alpha actin (α-SMA, g—i) immunohistochemical stainings. Black and white arrows mark the internal and external elastic laminae, respectively. Scale bars: 100 μm for all panels. (B) Severity of intimal hyperplasia determined from orcein-stained histological sections by an image analysis program at the study endpoint or at the time of occlusion. #p < 0.001 between Auto and Allo, ‡p = 0.003 between Auto and Allo + MSC, *p < 0.001 between Allo and Allo + MSC.
Localization of MSCs in the Tissue of Recipients
To track the injected SPIO-loaded MSCs in vivo, we did serial MRI scanning of MSC-treated allografts from 3 days to 12 weeks after transplantation. As shown in Figure 5A, most SPIO particles were localized to the soft tissue adjacent to the vascular grafts. Histological examinations at 4 or 12 weeks after transplantation revealed localization of the SPIO-loaded cells to the vascular adventitia (Fig. 5B) and in skeletal muscle surrounding the vascular grafts (Fig. 5C).

Localization of intramuscular injected MSCs in tolerant recipients. (A) In vivo localization of superparamagnetic iron oxide (SPIO)-loaded MSCs by magnetic resonance imaging analysis 3 days (a), 4 weeks (b), and 12 weeks (c) after transplantation. Yellow arrows indicate the detected SPIO particles on the T2*-weighted gradient-echo images. (B) Prussian blue staining of SPIO-loaded cells in the perivascular region of an allograft harvested at the 4th week posttransplantation. SPIO particles were identified in the boxed area (a, 40x), which is also shown in higher magnification (b, 630x). (C) H&E (a, 40x) staining of the perivascular soft tissue (Ve, vessel; Ct, connective tissue; Sk, skeletal muscle) of an allograft harvested 12 weeks after transplantation. The brown SPIO particles found in the boxed area (a) were confirmed by Prussian blue staining (b, 200x and c, 630x). Gray arrows indicate the capillaries (c). Scale bars: 100 μm for all panels.
Expression of Immune Modulatory Factors in the Vascular Grafts
To determine whether Treg expansion was involved in MSC-induced allograft tolerance, we analyzed Foxp3 mRNA expression by real-time quantitative PCR in untreated allografts harvested at the time when vascular occlusion was noted and in MSC-treated allografts and autografts harvested 12 weeks after transplantation. Compared to autografts, Foxp3 expression levels were increased in both untreated (5.0 ± 3.2 fold) and MSC-treated (6.5 ± 2.8 fold) allografts, and there was no significant difference between these two groups (p = 0.310). By contrast, IL-10, IFN-γ, and IDO were significantly higher in the MSC-treated allografts than in the untreated allografts (p = 0.021 for IL-10, p = 0.003 for IFN-γ, and p = 0.008 for IDO) (Fig. 6A), whereas HGF and TGF-β1 were not different (data not shown). To determine whether IL-10 and IFN-γ are elevated in circulation, we measured their serum protein levels by ELISA. Neither cytokine was detectable in any of the experimental groups, suggesting that they are increased locally by MSC treatment.
Immunohistochemical analysis of the MSC-treated allograft revealed that IL-10 and IFN-γ expressions were increased mostly in the perivascular cells while IDO expression was diffusely distributed extending from the intimal to the perivascular region (Fig. 6B).

Expression of immunomodulatory factors in vascular grafts. (A) Expression of interleukin-10 (IL-10), interferon-γ (IFN-γ), and indoleamine 2,3-dioxygenase (IDO) in untreated (n = 10) or MSC-treated (n = 10) allografts relative to autografts by real-time quantitative PCR analysis. Grafts were harvested once found to be occluded on angiograms or at the end of the study (12 weeks posttransplantation). Data are presented as box plots: the boundaries of a box indicate the 25th and 75th percentiles, the line within a box marks the median, and whiskers above and below a box indicate the 90th and 10th percentiles. *p = 0.021 for IL-10, p = 0.003 for IFN-γ, and p = 0.008 for IDO. (B) Immunohistochemical staining of the MSC-treated allografts harvested 12 weeks posttransplantation using antibodies to IL-10 (a, d), IFN-γ (b, e), and IDO (c, f). The inset in (c) is the matched isotype control. Lumen is oriented toward the bottom right corner on each image. Black and white arrows mark the internal and external elastic laminae, respectively. Scale bars: 100 μm for all panels.
Discussion
Our findings indicate that local injection of autologous MSCs alone at the time of femoral artery transplantation is effective in preventing TA in a porcine model. The beneficial effect is attributed to the immunosuppressive action of MSCs. The exact mechanism underlying the immunosuppressive effect of MSCs is not entirely clear. Our results suggest that MSCs act in situ to exert immunosuppressive activities through paracrine actions. We show by serial MRI tracking and histological analysis that the injected MSCs are retained at the sites adjacent to the vascular transplants. Based on the in vivo data, our findings imply that MSCs situated adjacent to the transplant may produce soluble factors that suppress immune cell proliferation. In agreement with previous reports (1,9,23), our results show that the MSC conditioned medium suppresses proliferation of allogeneic mononuclear cells. Since the locally injected MSCs remain detectable at 12 weeks after transplantation, it is reasonable to speculate that MSCs prolong vascular allograft patency via the sustained secretion of soluble factors.
It is controversial whether the immunomodulatory effects of MSCs depend on their MHC genotypes. Previous studies in rodent models showed that only MSCs derived from the donor or a third party that partially shared MHC with the donor are effective in induction of tolerance to cardiac allografts (5,20). However, other studies claimed that infusion of MSCs, derived from either the recipient or a third party, could achieve long-term graft tolerance (11). In the present study, we demonstrate the protective effects of autologous MSCs. Our finding is supported by the recent report that systemic infusion of syngeneic crude BM mononuclear cells protected aortic allografts from intimal hyperplasia in rats (10). These results have important clinical implications. The recipient autologous BM-derived MSCs are much more convenient to procure than the MSCs from the donor or a third party. Furthermore, the allogeneic MSCs may not be entirely immunoprivileged (2,5,20).
Recent studies have suggested a role of Tregs in MSC-mediated immunosuppression (5,11). In our pig model of femoral artery transplantation, IL-10 was found to be more abundant in MSC-treated allografts than in the untreated ones (Fig. 6A). However, the expression levels of Foxp3 mRNA were comparable between these two groups. These results suggest that the inducible type 1 regulatory T (Tr1)-like cells, instead of the naturally occurring Foxp3+ Tregs, are involved in MSC-induced allograft tolerance. Tr1-like cells are chronically activated by persistent antigens, produce large amounts of IL-10 and IFN-γ, and are able to suppress antigen-specific immune responses both in vitro and in vivo (12,13,28). Interestingly, in addition to IL-10, IFN-γ expression was significantly increased in MSC-treated vascular allografts compared to untreated controls in our animal model. Furthermore, both cytokines are localized to the infiltrating mononuclear cells at the perivascular site. It remains to be confirmed that the infiltrating cells are Trl-like cells.
IFN-γ is generally considered as a proatherogenic factor in TA (18,22,25). However, in the immune system inflammation frequently elicits counterregulatory (anti-inflammatory) pathways. Recent studies have shown that IFN-γ might help limit excessive T-cell activation at local sites of inflammation through the induction of IDO expression (4,8). Our immunohistochemistry analysis provides evidence for increased IFN-γ expression by MSC treatment in the perivascular mononuclear cells accompanied by elevation of IDO expression primarily in the medial layer. These findings are in keeping with the recent report that IFN-γ-induced IDO expression in human vascular smooth muscle cells inhibits allogeneic T-cell response in vitro and in vivo (6). More importantly, blocking of IDO with 1-methyl tryptophan was reported to abrogate MSC-induced long-term graft acceptance in a rat heart transplantation model (20). Taken together, IFN-γ-mediated IDO expression within the local environment of the transplant could indeed prolong graft survival by inducing immunological tolerance.
In conclusion, our results provide strong evidence for controlling allograft intimal hyperplasia and maintaining vascular patency by MSCs through an IFN-γ—IDO mechanism. We postulate that MSCs stimulate the Trl-like cells that express IL-10 and IFN-γ, and IFN-γ in turn induces IDO expression in the vascular smooth muscle cells. Through the immunosuppressing actions of these molecules, MSCs maintain graft patency. Local infusion of autologous MSCs represents a new strategy for treating TA including that secondary to heart transplantation.
Footnotes
Acknowledgments
This work was partially supported by a research grant from National Health Research Institutes (NHRI-CS-099-PP-13). We thank C-C. Yu from Laboratory of Animal Center, National Taiwan University College of Medicine, M-Y. Su and S-Y. Wei from Department of Medical Imaging and Radiology, and P-L. Lai from Department of Pathology, National Taiwan University Hospital for technical assistance. The authors declare no conflicts of interest.
