Abstract
Interleukin-10 (IL-10) gene transduction into allogeneic smooth muscle cells (SMCs) was evaluated to improve the long-term benefits of allogeneic cell transplantation into infarcted myocardium. Allogeneic cells, including SMCs, have been demonstrated to restore cardiac function and repair the infarcted myocardium, but late rejection of the transplanted cells by the host immune system may reverse the benefits of cell therapy. In a rat myocardial infarction model, three groups of rats were injected with either unmodified autologous, unmodified allogeneic, or allogeneic + IL-10 SMCs into the infarct region. Three weeks later, most of the allogeneic cells were rejected, whereas autologous cells were engrafted in the myocardium. IL-10 gene transduction of the allogeneic SMCs significantly improved the cell survival. To understand the mechanism of this improved survival, we evaluated the host immune responses against the SMCs. Allogeneic SMCs expressing IL-10 decreased leukocyte-mediated cytotoxicity in coculture, decreased the number of cytotoxic CD8+ T-cells, and increased the number of CD4+CD25+ regulatory T-cells in vitro and in vivo. Furthermore, IL-10 prevented the production of antidonor antibodies by the recipients against the allogeneic SMCs. Transplantation of unmodified autologous SMCs, but not unmodified allogeneic SMCs, significantly improved fractional shortening and left ventricular dimensions compared to the media-injected control group. However, IL-10 gene-enhanced allogeneic SMCs improved ventricular function, increased wall thickness, and decreased scar length in association with their enhanced survival. We conclude that IL-10 gene-enhanced cell therapy with allogeneic SMCs prevents detrimental alloimmune responses in the recipient, thereby increasing the survival of transplanted allogeneic SMCs and more effectively restoring cardiac function.
Introduction
Allogeneic (unrelated donor) cells isolated from healthy, young volunteers may be the best choice to insert into the injured myocardium to enhance cardiac repair and regeneration because these cells have excellent proliferative and regenerative potential. However, rejection remains a formidable problem for allogeneic cell transplantation (12). Ex vivo adjuncts to prevent allogeneic cell rejection would represent a major advancement in the field. In this study, we evaluated ex vivo interleukin-10 (IL-10) transduction to prevent detrimental immune responses after cell implantation.
Clinical trials have been initiated, and preliminary data with a limited number of patients suggested that allogeneic cells might be an alternative candidate for cardiac transplantation (10). Allogeneic mesenchymal stromal cells (MSCs) are believed to be both immunoprivileged and immunosuppressive (6). After implantation into the injured myocardium, these cells differentiate into endothelial, smooth muscle, and myogenic cells (29), but following differentiation, they may be rejected (12). Cardiac stem cells may provide more potent cardiac repair than MSCs (25), but allogeneic cardiac stem cells are rapidly rejected from the infarcted heart, limiting their therapeutic capacity (37). Cardiomyocytes derived from either embryonic stem cells or induced pluripotent stem cells have been demonstrated to engraft in the infarcted heart and induce cardiac repair (15,32), but these allogeneic cells are also eventually rejected. The benefits of cell therapy to restore ventricular function after an extensive myocardial infarction (MI) would be greatly enhanced if the grafted cells could be rendered immunoprivileged.
Cytokines have been shown to play a critical role in modulating the host immune response to allograft transplantation. IL-10 decreases the proliferation of cytotoxic T-cells and natural killer (NK) cells and promotes immune tolerance (35). Adenovirus-mediated delivery of the IL-10 gene to donor lungs ameliorated ischemia-reperfusion injury and improved posttransplant allograft function in a mouse model (11). Therefore, IL-10 transduction offers a unique opportunity to render cardiac regenerative cells immunoprivileged to prolong and enhance their reparative capacity.
Gene-enhanced cell therapy has distinct advantages over gene therapy (7). Ex vivo cell transduction is safe, and the amount and timing of the gene product can be regulated in the transduced cells. Clinical trials of cell-based gene therapy have been accomplished without major safety concerns (34,41). Therefore, IL-10 gene-enhanced cell therapy may be a promising approach for the long-term engraftment of allogeneic cells.
The most appropriate cell type for cardiac repair after an MI remains controversial. MSCs that have been differentiated toward myogenic cells restore ventricular function better than unmodified MSCs (2,3). Myogenic cells may be more potent than undifferentiated stem cells (25), and pluripotent stem cells induced to become myogenic cells have been proposed for cardiac repair (32). Somatic cells of myogenic origin, including smooth muscle cells (SMCs), which retain the distinct capability to remodel the surrounding tissue, may be the ideal candidates (16,22). Myogenic cell transplantation has been proposed as a way to modify the stiff, fibrotic infarct region to a more elastic, contractile tissue (9,40). Restoring the elastic properties of the infarct region can prevent progressive cardiac dilation and ventricular dysfunction following an MI (18,23). Previous studies have established that SMC transplantation into the heart after an MI improves ventricular function (16). SMCs are easy to obtain and culture, and they secrete angiogenic factors such as fibroblast growth factors, vascular endothelial cell growth factor, and nitric oxide (1,14).
In this proof-of-principle evaluation, we have used SMCs to avoid the potential immunomodulating effect of stem or progenitor cells. We hypothesized that IL-10 gene transduction of allogeneic SMCs would suppress the host immune response and prevent rejection of transplanted SMCs to improve cardiac function after an MI. If we are able to prevent rejection of SMCs, we hope that this approach might be beneficial to the variety of new allogeneic myogenic cells being considered for cardiac repair.
Materials and Methods
Animals
Male Wistar rats (allogeneic SMC donors) and female inbred Lewis rats (cell recipients and autologous cell donors) were procured from Charles River Laboratories (St-Constant, QC, Canada). The male green fluorescent protein (GFP)-positive Wistar rats used as donors in some experiments were originally obtained from the YS Institute, Inc. (Utsunomiya, Japan), and subsequently bred at our institution. All animal procedures were approved by the Animal Care Committee of the University Health Network, and all animals received humane care in compliance with the Guide for the Care and Use of Laboratory Animals (NIH, revised 1996).
Smooth Muscle Cell Isolation and Culture
SMCs were isolated from rat aortas employing 0.2% trypsin (Life Technologies, Carlsbad, CA, USA) and 0.1% collagenase (Worthington Biochemical, Lakewood, NJ, USA) digestion as described previously (16,20). SMCs were counted and culture expanded in Iscove's modified Dulbecco's medium (Life Technologies), with 10% fetal bovine serum (FBS; Life Technologies), 100 U/ml penicillin G (Life Technologies), and 100 μg/ml streptomycin (Life Technologies).
IL-10 Constructs and Transduction
A human IL-10 adenoviral construct was used to overexpress the IL-10 gene in allogeneic SMCs, and a LacZ recombinant adenovirus was used to assess the transduction efficiency. The HD-Ad vector expressing the reporter gene LacZ (HD-Ad-CMVlacZ) was previously described (26,36). The empty HD-Ad vector (C4HSU-HD-Ad) used as a control in the in vitro experiments testing IL-10 expression from SMCs was described by Koehler et al. (13). The HD-Ad vector for human IL-10 expression was constructed similarly to HD-Ad-CMVlacZ, but the CMV promoter and poly A signal were taken from the pMP6a plasmid (27). All vectors were produced and purified as described (13,36).
Optimization of IL-10 gene expression was accomplished using different multiplicities of infection (MOIs 12.5, 25, 50, 100, and 200) for 1—5 days. To study the time course of IL-10 release by SMCs, the cells were transduced with IL-10 at MOI 25 for 48 h, and IL-10 levels were measured in the cell culture medium at 7, 14, 21, 28, and 35 days. The IL-10 released from the cells was detected by ELISA (R&D Systems, Minneapolis, MN, USA), and cell viability was assessed by the MTT assay (Sigma-Aldrich Canada, Oakville, ON, Canada). (Two independent experiments were performed; n = 4 in each experiment.)
In Vivo Myocardial Infarction Model and Transplanted Allogeneic SMCs Survival
MI was induced in female Lewis rats by ligating the left anterior descending coronary artery. One week later, a suspension (50 μl/rat) of unmodified autologous or allogeneic (with or without IL-10) GFP+ or GFP- SMCs (3 × 106 cells/rat) prepared in FBS-free medium were transplanted around the infarct area at three different sites over the entire infarct and border zone region as described previously (12). This procedure was consistent throughout the study in all animals. GFP+ SMCs were used for transplanted cell survival studies. For all other studies (host immune response, morphometric assessment) GFP- cells were used. The control group received cell culture medium alone. The survival of transplanted SMCs was analyzed at 1 and 3 weeks after cell implantation. The number of Y chromosomes (representing the number of transplanted male SMCs in the female recipient hearts) was quantified by real-time PCR as described previously (39). Briefly, genomic DNA was isolated from the frozen heart samples, and TaqMan real-time PCR (Life Technologies) was performed to detect the Sry3 gene to quantify the number of cells. Cell survival was also analyzed at 3 weeks by immunohistochemical staining of the myocardium with an anti-GFP antibody (Life Technologies). DAPI (Sigma-Aldrich) was used to stain the nuclei. (Two independent experiments were performed; n = 4 in each experiment.)
Cardiac Function and Morphometry
Cardiac function was evaluated by echocardiography and pressure—volume analysis. Echocardiography was performed with the ACUSON SEQUOIA C256 system (Siemens Medical Solutions, Malvern, PA, USA) before and 1 week after MI (before cell transplantation) and 3 weeks after cell transplantation. Left ventricular (LV) internal dimensions at end systole and end diastole and fractional shortening were measured to assess heart function. For pressure—volume analysis, the carotid artery was exposed, and a calibrated Millar and conductance pressure—volume catheter was inserted into the LV cavity through the carotid artery (Harvard Apparatus, Holliston, MA, USA). Data were collected using Millar PV software (Harvard Apparatus). End-systolic and end-diastolic ventricular volumes and ejection fraction as well as the minimum and maximum dP/dt were calculated as load-dependent indices of ventricular function. Then pressure—volume measurements were made during inferior vena caval occlusion to calculate the load-independent measurements of preload recruitable stroke work and end-systolic elastance (8).
At 3 weeks after cell transplantation, hearts were fixed in 10% formaldehyde (Sigma-Aldrich), embedded in paraffin, cut into 5-mm-thick sections, and stained with Masson's trichrome (performed at the University Health Network's Pathology Research Program Laboratory, Toronto, ON, Canada) for morphometric assessment. Scar length was measured along the middle of the scar between the edges of the border of the infarct. Scar thickness was expressed as the average wall thickness measured at the middle of the scar and along each edge. (Two independent experiments were performed; n = 4 in each experiment.)
Host Immune Response
Immunohistochemistry was employed to evaluate leukocyte infiltration in the myocardium at 3 weeks after cell implantation. Briefly, hearts were arrested with potassium chloride (Sigma-Aldrich), and frozen tissue was embedded in Tissue-Tek OCT (Sakura Finetek, Inc., Torrance, CA, USA). Each sample was cut into 5-mm-thick sections that were fixed with 2% paraformaldehyde (Sigma-Aldrich), permeabilized with 0.2% Triton X-100 (Sigma-Aldrich), and immunostained for CD8 (559976; BD Biosciences, Mississauga, ON, Canada) or double stained for CD4 (1:200, 554843; BD Biosciences) and CD25 (1:200, 554846; BD Biosciences). Nuclei were stained with DAPI (Sigma-Aldrich). Host immune responses were also determined by measuring antidonor alloantibody production in the recipient blood 3 weeks after cell transplantation. The serum derived from recipient blood in the different groups was diluted (1:10) and incubated with autologous and allogeneic SMCs in vitro for 2 h. After incubation, the SMCs were stained with PE-conjugated anti-rat IgG1 (1:200; BD Biosciences) to detect the antidonor alloantibodies present in the serum against SMCs. The alloantibody titer was also measured in recipient serum by our own custom-made ELISA. The assay was standardized by titrating the different concentrations of antigen and antibodies. Finally, each well was coated with diluted serum (1:10) samples (antigen) from different groups in a total volume of 100 ml of 0.05 M carbonate buffer (pH 9.6; Sigma-Aldrich) and incubated for 24 h at 4°C. Unbound antigen was removed by decanting the plate solution, and wells were blocked for 1 h with 1% bovine serum albumin (Bioshop) in 0.1 M phosphate-buffered saline (PBS, pH 7.2; Sigma-Aldrich) at room temperature. The wells were washed three times with 200 ml of PBS containing 0.05% (v/v) Tween-20 (Sigma-Aldrich). Coated wells were then incubated with primary antibody (1:5,000), anti-rat IgG (Jackson ImmunoResearch, West Grove, PA, USA) for 2 h at room temperature. The plate was again washed three times with PBS and incubated with peroxidase-labeled secondary antibody (1:10,000; Santa Cruz Biotechnology, Dallas, TX, USA), for 2 h at room temperature. Wells were washed three times as described above, and color was developed by addition of 100 ml of substrate solution in each well for 30 min, and finally 100 ml of stop solution was added to each well (R&D Systems). Optical density of each well was measured by plate reader (Biotek Instruments, Winooski, VT, USA) at 450 nm, and the correction wavelength used was 570 nm. (Two independent experiments were performed; n = 4—8 in each experiment.)
Mixed Leukocyte-Mediated Cytotoxicity
Mixed peripheral blood leukocytes (cytotoxic leukocytes) were isolated from Lewis rats using HISTOPAQUE 1083 (Sigma-Aldrich) and cocultured with autologous cells, unmodified allogeneic cells, or allogeneic IL-10 SMCs in a 10:1 ratio. After 72 h, leukocyte-mediated cytotoxicity in the SMCs was assessed by measuring the lactate dehydrogenase (LDH) release from the damaged cells (Cytotoxicity Detection Kit; Roche Applied Science, Mannheim, Germany). The culture medium was collected (without cells) and incubated with the reaction mixture from the kit. LDH activity is directly proportional to optical density measured at 490 nm with a reference filter of 620 nm. (Two independent experiments were performed; n = 5 in each experiment.)
Leukocyte Proliferation and Suppression Assays
Mixed peripheral blood leukocytes were cocultured with autologous or allogeneic SMCs with or without IL-10 (10:1) in the presence of 10 μg/ml phytohemagglutinin (PHA, Sigma-Aldrich). After 72 h, BrdU (10 μM; Cell Signaling Technology, Danvers, MA, USA) was added and the coculture incubated for another 24 h. Leukocyte proliferation was evaluated with the BrdU Cell Proliferation Assay Kit (Cell Signaling Technology), which detects BrdU incorporation into DNA during cell proliferation. The magnitude of the absorbance at 450 nm for the developed color is proportional to the quantity of incorporated BrdU and cell proliferation. Leukocyte proliferation was also assessed by immunofluorescent assessment of the BrdU uptake by the cells using a BrdU detection antibody (Ab6326; Abcam, Cambridge, UK). DAPI was used as a nuclear stain. (Two independent experiments were performed; n = 5 in each experiment.)
CD4+ and CD8+ T-Cell Quantification
Mixed peripheral blood leukocytes were cocultured with autologous or allogeneic SMCs with and without the IL-10 construct (10:1) in the presence of 10 μg/ml PHA. After 72 h of coculture, leukocytes were stained with anti-CD4 (1:200) and anti-CD8 (1:200) antibodies to analyze the expression of CD4 and CD8 by immunofluorescence and flow cytometry. (Two independent experiments were performed; n = 4 in each experiment.)
CD4+ T-Cell Isolation, Flow Cytometry, and CD25 Expression
CD4+ T-lymphocytes were isolated from a single cell suspension of the total leukocyte population using the MagCellect Rat CD4+ T-Cell Isolation Kit (R&D Systems). The purity of CD4+ T-cells was determined by flow cytometry. CD25 mRNA and protein expression in CD4+ T-cells were measured by RT-PCR (primers were F: 5′-aacggcaccatcctaaactg-3′ and R: 5′-caggaagcctcactctctgg-3′) and Western blot (anti-CD25 was SC-666 from Santa Cruz, and anti-β-actin was A1978 from Sigma-Aldrich). CD4+CD25+ T-cells in the mixed leukocyte population were quantified by flow cytometry. Cell surface molecules were stained with fluorescence-conjugated anti-CD4 and anti-CD25. (Two independent experiments were performed; n = 3 in each experiment.)
Real-Time PCR Array Analysis
Real-time PCR analysis was performed on whole RNA isolated from leukocytes cocultured with unmodified or IL-10-expressing allogeneic SMCs. A commercial kit purchased from SABiosciences (Frederick, MD, USA) was used to analyze rat genes implicated in immune tolerance and T-cell anergy (RT2 Profiler PCR array: PARN-074A). Those restricted genes that were reproducibly differentially expressed in experimental versus control groups were subsequently analyzed by RT-PCR using fresh independent RNA samples. The RT-PCR primers used were as follows: CTLA4 F: 5′-agtgacccaaccttcagtgg-3′; CTLA4 R: 5′-gtctg aatctgggcatggtt-3′; CD401g F: 5′-cttccagcgagca-3′; CD401g R: 5′-attcgctgccctg-3′; PDCD1 F: 5′-cacagtgtcagagggagcaa-3′; PDCD1 R: 5′-ggggtctccaggattctctc-3′; EGR2 F: 5′-catctctgcg cctagaaacc-3′; EGR2 R: 5′-ccagagaggaggtggaagtg-3′; EGR3 F: 5′-gacaatctgtaccccgagga-3′; EGR3 R: 5′-atacatggcctccac gtctc-3′; GADPH F: 5′-atgactctacccacggcaag-3′ and GADPH R: 5′-ggatgcagggatgatgttct-3′. (Two independent experiments were performed; n = 4 in each experiment.)
Statistical Analyses
Data were analyzed with GraphPad Prism (GraphPad, San Diego, CA, USA) and expressed as mean ± SD. Comparisons between two groups were made with a two-tailed Student's t-test. Comparisons among multiple groups were performed with either one-way or two-way ANOVA. When F values were significant, group differences were specified with Tukey or Bonferroni posttests. Differences were considered statistically significant when p < 0.05.
Results
IL-10 Levels in SMCs
We first analyzed basal IL-10 levels in SMCs by ELISA. IL-10 was not found in either the medium collected from SMC cultures or in SMC lysates (Fig. 1A). An adenoviral construct was used to express IL-10 in SMCs, and optimization of IL-10 gene expression and cell viability was accomplished using varying MOIs (Fig. 1B, C). During the time course of IL-10 release, an MOI of 25 for 48 h achieved a significant amount of IL-10 (99.43 ± 17.65 pg/ml) release by the SMCs at day 35, and cell viability was >90%, so this titer was chosen for subsequent experiments (Fig. 1C, D). Control MSCs were transduced with empty adenovirus.
SMCs do not produce endogenous IL-10, but can be optimized for IL-10 expression using an IL-10 adenovirus construct. (A) Basal IL-10 levels were measured by ELISA in SMC culture medium and lysate after 24 h of cell culture. Leukocytes (positive control) released IL-10 into the culture medium, but IL-10 was not detected in the SMC culture medium or lysate (n = 6 per group). Data were derived from two independent experiments. (B, C) Optimization of IL-10 expression from an adenovirus construct was done using varying MOIs for 1 to 5 days. The IL-10 release from the cells was detected by ELISA (B), and cell viability was assessed by the MTT assay (C). (D) To study the time course of IL-10 release by SMCs, the cells were transfected with the adenoviral IL-10 vector at an MOI of 25 for 48 h, and IL-10 levels were measured in the cell culture medium by ELISA at 7, 14, 21, 28, and 35 days. Data shown are representative of two independent experiments (n = 4 in each experiment).
IL-10 Prevented the Rejection of Transplanted Allogeneic SMCs
We transplanted unmodified autologous, unmodified allogeneic, and allogeneic + IL-10 SMCs into infarcted rat hearts and assessed cell survival in the myocardium at 1 and 3 weeks. One week after implantation, cells were detected in both the autologous and allogeneic groups, and IL-10 gene-enhanced therapy significantly increased the allogeneic cell survival (Fig. 2A). After 3 weeks, nontransduced allogeneic cells were not detected in the myocardium, but IL-10 gene enhancement significantly increased cell survival (Fig. 2A, B).
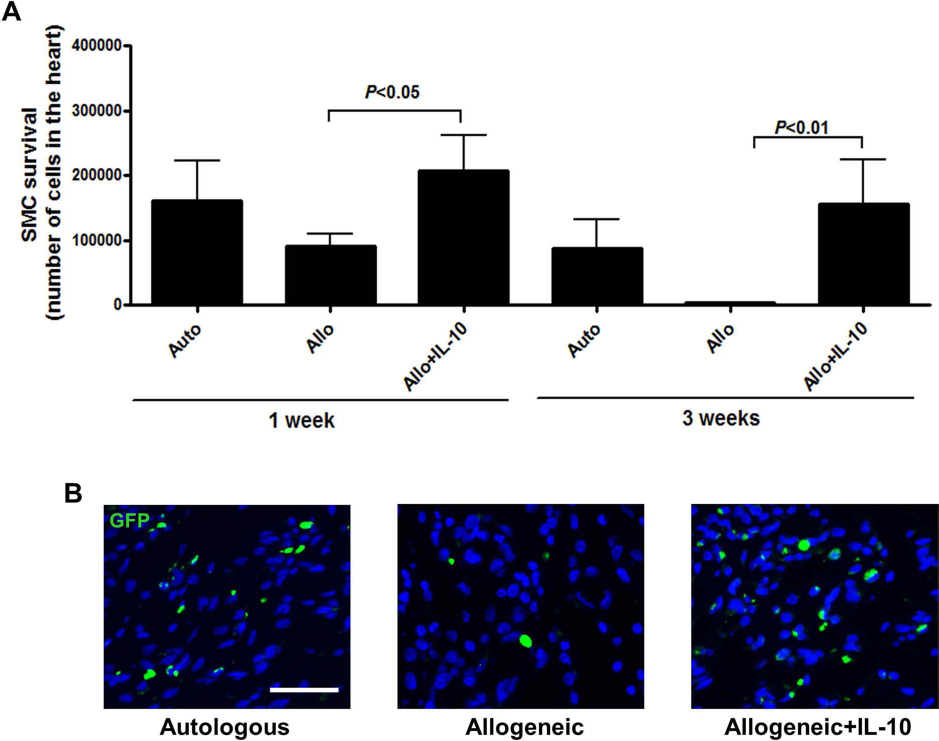
IL-10 prevented rejection of allogeneic SMCs in the heart. Unmodifed autologous (from male Lewis rats) and allogeneic (from male Wistar rats) either unmodified or transfected with IL-10 SMCs were transplanted into the infarct area of female Lewis rat hearts 1 week after MI. (A) Cell survival at 1 and 3 weeks after transplantation was determined by quantifying the number of Y chromosomes in the female myocardium by real-time PCR. (B) At 3 weeks after transplantation, GFP+ cells were detected by staining myocardial sections with an anti-GFP antibody (scale bar: 50 μm). Data shown are representative of two independent experiments (n = 4 in each experiment).
SMCs Expressing IL-10 Restored Post-MI Cardiac Function and Improved Scar Morphology
To determine the effects of SMC transplantation on cardiac function, we performed echocardiography before MI, 1 week after MI (before cell transplantation), and 3 weeks after cell transplantation. Coronary artery ligation produced a precipitous fall in fractional shortening in all animals at 1 week, with no significant differences among groups (Fig. 3A). However, 3 weeks after cell transplantation, unmodified autologous SMCs and IL-10-expressing allogeneic SMCs prevented further deterioration of heart function. The autologous SMC and the IL-10-expressing allogeneic SMC groups had significantly improved fractional shortening compared to the media-injected control group. The unmodified allogeneic SMCs did not improve fractional shortening. Furthermore, we also observed a significant difference in fractional shortening between unmodified allogeneic SMCs and IL-10-expressing allogeneic SMCs (p < 0.05) (Fig. 3A).

IL-10-expressing allogeneic SMCs preserved heart function as effectively as unmodified autologous SMCs 3 weeks after implantation. Unmodified autologous and allogeneic SMCs and allogeneic SMCs expressing IL-10 were transplanted into female Lewis rats following MI. (A) Fractional shortening, determined by echocardiography, was preserved by unmodified autologous and IL-10-transduced SMCs compared to nontransduced allogeneic cells or media control. (B—H) LV volumes improved in the naive autologous SMC and IL-10-expressing allogeneic SMC groups compared to the media control group, but there was no difference observed in LV volumes between the unmodified allogeneic SMC group and IL-10-expressing allogeneic SMC group. (B) LV end-systolic volume. (C) LV end-diastolic volume. (D) Ejection fraction. (E) dP/dt max. (F) dP/dt min. (G) Preload recruitable stroke work. (H) End-systolic pressure—volume relationship (ESP, end-systolic pressure, ESV, end-systolic volume). Data shown are representative of two independent experiments (n = 4 in each experiment).
Cardiac function was further assessed by pressure—volume analyses 3 weeks after cell implantation. LV volumes improved in the unmodified autologous SMC and IL-10-expressing allogeneic SMC groups compared to the media control group, but there was no difference observed in LV volumes between the unmodified allogeneic SMC group and IL-10-expressing allogeneic SMC group (Fig. 3B, C). Load-dependent indices of function (ejection fraction, maximum and minimum dP/dt) demonstrated a similar response (Fig. 3D—F). Load-independent indices, including preload recruitable stroke work and end- systolic pressure—volume relationship (systolic elastance), were also improved with autologous SMC transplantation, and IL-10-transduced allogeneic SMCs compared with the media control and unmodified allogeneic SMC groups (Fig. 3G, H).
Unmodified autologous SMC and IL-10-expressing allogeneic SMC groups showed decreased scar length and increased scar thickness compared to the media control group, but there was no difference observed in scar length and scar thickness between the naive allogeneic SMC group and the IL-10-expressing allogeneic SMC group (Fig. 4A—C).

IL-10 gene-enhanced allogeneic SMCs prevented scar thinning and dilatation. Unmodified autologous and allogeneic SMCs and allogeneic SMCs expressing IL-10 were transplanted into female Lewis rats following MI. Scar length and thickness were assessed 3 weeks later. (A) Representative heart sections stained with Masson's trichrome illustrate the scar (scale bar: 2 mm). (B) Scar length. (C) Scar thickness. The naive autologous SMC and IL-10-expressing allogeneic SMC groups had decreased scar length and increased scar thickness compared to the media control, but there was no difference observed in scar length and scar thickness between the naive allogeneic SMC group and the IL-10-expressing allogeneic SMC group. Data shown are representative of two independent experiments (n = 4 in each experiment).
IL-10 Prevented Allogeneic Leukocyte-Mediated Cytotoxicity in SMCs
To discover the mechanisms responsible for the IL-10-mediated increase in allogeneic SMC survival, we cocultured SMCs with unmodified or IL-10-expressing allogeneic leukocytes and assessed the alloimmune response by measuring the LDH release (cytotoxicity) from the damaged SMCs. Cytotoxicity was significantly greater for unmodified allogeneic SMCs compared to autologous cells and IL-10-transduced allogeneic SMCs (p < 0.001) (Fig. 5A). However, there was no difference observed in leukocyte proliferation after coculture with any of the three SMC cell types (Fig. 5B, C).

IL-10 prevented allogeneic leukocyte-mediated cytotoxicity in SMCs. SMCs were cocultured with naive autologous or transduced or nontransduced allogeneic leukocytes. IL-10-transduced SMCs maintained an immunoprivileged environment in the coculture. (A) Leukocyte-mediated cytotoxicity (LDH release) was significantly greater with allogeneic SMCs compared to autologous SMCs (p < 0.001), and the cytotoxicity was reduced with IL-10 gene-enhanced cell therapy (n = 5). (B, C) BrdU uptake was employed to determine leukocyte proliferation and was detected by immunofluorescence using a FITC-labeled antibody (B, scale bar: 50 μm) and by ELISA (C, n = 5). Control is leukocyte activated with PHA, without SMCs. Data shown are representative of two independent experiments.
Next we analyzed the phenotype of the leukocytes following 72 h of SMC coculture. The number of CD4+ and CD8+ T-cells was determined by immunostaining (Fig. 6A) and flow cytometry (Fig. 6B). Cocultures with autologous, nontransduced allogeneic and allogeneic SMCs + IL-10 had no effect on the number of CD4+ T-cells (Fig. 6C). Unmodified autologous and allogeneic SMCs did not alter the number of CD8+ T-cells, but IL-10-expressing allogeneic SMCs significantly decreased the number of cytotoxic CD8+ T-cells after coculture (p < 0.001) (Fig. 6C).

IL-10 expression in SMCs was associated with a decrease in the number of CD8+ T-cells. SMCs were cocultured with naive autologous or transduced or nontransduced allogeneic leukocytes, and the effect of SMCs on CD4+ and CD8+ T-cells was assessed by immunofluorescence and flow cytometry. (A) Representative immunofluorescent micrographs of CD4+ and CD8+ T-cells in the mixed leukocyte population (scale bar: 50 μm). (B) Representative flow cytometry plots of CD4+ and CD8+ T-cells in the mixed leukocyte population. (C) Quantification of flow data; the percentage of CD4+ T-cells was not different among groups, but the percentage of CD8+ T-cells was significantly reduced with the IL-10-transduced SMCs compared to other groups (p < 0.001). Control is leukocyte activated with PHA, without SMCs. Data shown are representative of two independent experiments (n = 4 in each experiment).
IL-10 Increased the Regulatory T-Cell Population in Coculture
Another mechanism to promote immune tolerance is the induction of host regulatory T (Treg) cells, which suppress alloreactive cytotoxic T-cells. Flow cytometry was employed to count the number of CD4+CD25+ Treg cells in the leukocyte population after coculture with SMCs. The number of Treg cells was similar after coculture with unmodified autologous and allogeneic SMCs, but IL-10-expressing allogeneic SMCs increased the Treg cell population (Fig. 7A, B). The CD25 mRNA and protein expression in the purified CD4+ T-cell population increased after CD4+ T-cell coculture with IL-10-expressing SMCs (Fig. 7C, D).
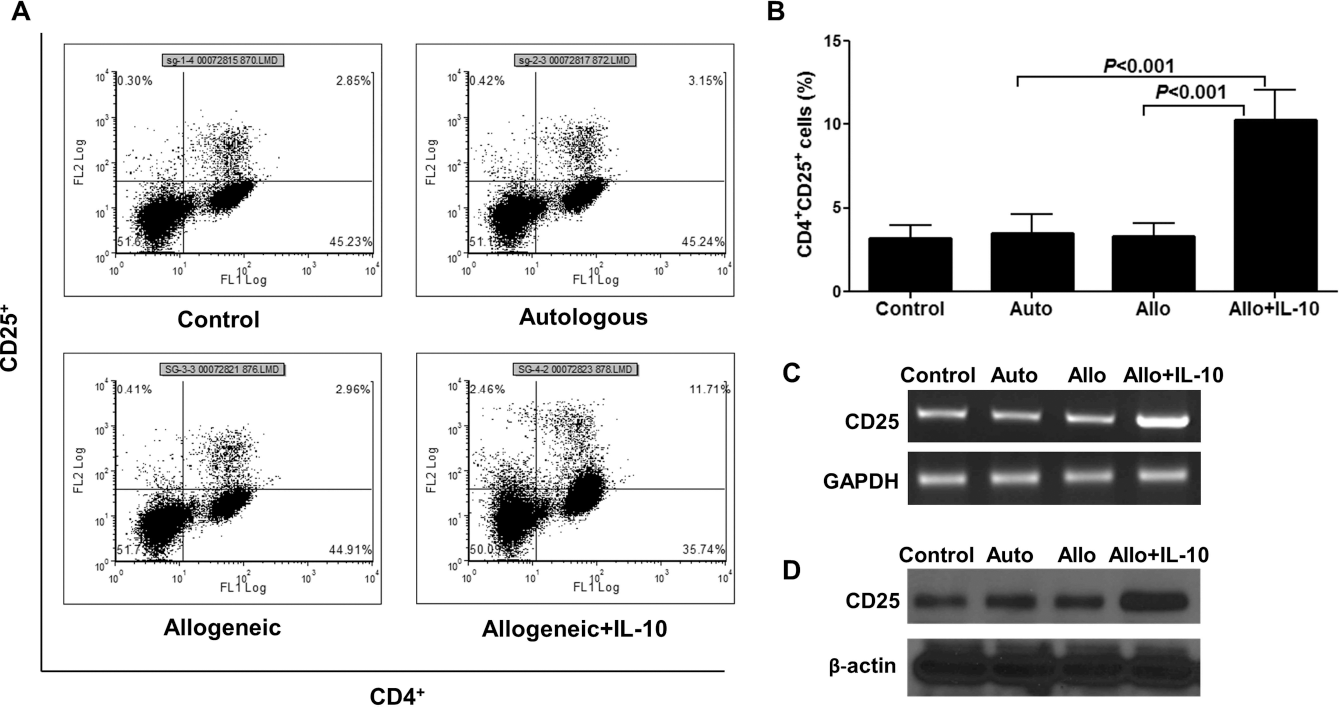
IL-10 expression in SMCs was associated with induction of Treg cells. SMCs were cocultured with naive autologous and transduced or nontransduced allogeneic leukocytes, and the effect of SMCs on Treg cell induction was assessed. (A) Representative flow cytometry plots of CD4+CD25+ T-cells in the mixed leukocyte population. (B) Quantification of flow data; the percentage of CD4+CD25+ T-cells was significantly greater in the IL-10-transduced SMC group than the other groups (p < 0.001). Control is leukocyte activated with PHA, without SMCs. (C, D) CD4+ T-cells isolated from the total leukocyte population were cocultured with SMCs. CD25 expression by RT-PCR (C) and Western blot (D) was greater in the IL-10-transduced SMC group compared to the other groups. Control is T-cell activated with PHA. Data shown are representative of two independent experiments (n = 3 in each experiment).
To determine the mechanism of IL-10-mediated induction of Treg cells in the mixed leukocyte population, we screened transcripts of 84 genes related to immune tolerance in leukocytes after coculture with SMCs. This PCR array indicated that the mRNA expression of PDCD1, CD40lg, CTLA4, EGR2, and EGR3 was significantly increased in leukocytes after coculture with IL-10-expressing allogeneic SMCs in comparison to SMCs without IL-10 (Table 1). To more precisely determine the mRNA expression of these five genes, RT-PCR using specific primers was performed (Fig. 8A). Interestingly, CTLA4 expression increased significantly in leukocytes cocultured with the IL-10-expressing allogeneic SMCs, whereas PDCD1, CD40lg, EGR2, and EGR3 expression did not change, with or without IL-10 treatment (Fig. 8B).

IL-10 gene-enhanced SMCs increased leukocyte CTLA4 expression after coculture. (A) CTLA4, CD40lg, PDCD1, EGR2, and EGR3 mRNA expression by RT-PCR. (B) Quantification of mRNA expression. Data shown are representative of two independent experiments (n = 4 in each experiment). Control is leukocyte activated with PHA, without SMCs. IL-10 gene-enhanced cell therapy increased CTLA4 compared to the other groups.
Immune Tolerance-Related Gene Expression in Control Leukocytes (Cocultured With Unmodified Allogeneic SMCs) and Leukocytes Cocultured With IL-10-Expressing Allogeneic SMCs by RT2 Profiler Real-Time PCR Array

IL-10 Limited the In Vivo Alloimmune Response
We analyzed the in vivo host immune response against transplanted SMCs 3 weeks after cell implantation. No significant differences were found in the number of cytotoxic CD8+ T-cells infiltrating the myocardium after naive autologous or allogeneic SMC transplantation (Fig. 9A). However, IL-10 transduction of the allogeneic SMCs decreased the number of CD8+ T-cells in the heart. We also analyzed the number of CD4+CD25+ Treg cells in the infarct area. Similar to our in vitro data, both nontransduced autologous and allogeneic SMC transplantation had no effect on Treg cell numbers in the heart. However, IL-10 gene transduction of allogeneic SMCs significantly increased the number of Treg cells in the myocardium (p < 0.001) (Fig. 9B).

IL-10 expression in transplanted allogeneic SMCs modulated the host immune response. Naive autologous SMCs, nontransduced allogeneic SMCs, and IL-10-expressing allogeneic SMCs from male rats were transplanted into the infarct area in female rats 1 week after MI. Control is medium without SMCs. DAPI was used as a nuclear stain (blue). (A) CD8 expression by immunohistochemistry in myocardial sections 3 weeks after cell transplantation (scale bar: 20 μm, n = 4). (B) Colocalization of CD4 (green) and CD25 (red) was evaluated by immunohistochemistry in myocardial sections 3 weeks after cell transplantation (scale bar: 50 μm, n = 4). (C) Antidonor alloantibodies were detected in recipient blood 3 weeks after cell transplantation. Recipient serum was incubated with autologous and allogeneic SMCs for 2 h, and the SMCs were stained with PE-conjugated anti-rat IgG. Representative micrographs illustrate IgG expression (scale bar: 100 μm). Alloantibody titers (IgG levels) were measured in recipient serum by ELISA and were significantly reduced in IL-10 transduced allogeneic SMCs compared to nontransduced allogeneic SMCs and similar to the unmodified autologous SMCs (p < 0.001, n = 8). Data shown are representative of two independent experiments.
We investigated whether the transplanted IL-10-expressing allogeneic SMCs elicited any long-term adaptive immune responses by altering the antidonor alloantibody production by the host against the transplanted cells. At 3 weeks after cell transplantation, blood collected from the autologous cell recipients contained no alloantibodies that reacted with autologous SMCs, but alloantibodies were detected against unmodified allogeneic SMCs. The alloantibody titers were significantly lower in recipients of IL-10-expressing allogeneic SMCs (p < 0.001) (Fig. 9C). Thus, IL-10-transduced SMCs maintained the immunoprivilege of the transplanted allogeneic cells and mitigated both cellular and humoral host immune responses following cell implantation.
Discussion
Here we demonstrated that IL-10 gene-enhanced cell therapy increased survival of transplanted allogeneic SMCs in the injured myocardium and improved heart function. The present study also revealed that IL-10 overexpression in allogeneic SMCs decreased the number of cytotoxic CD8+ T-cells, decreased the level of cytotoxicity caused by alloreactive leukocytes, and increased the number of Treg cells in vitro and in vivo.
Cell therapy improves ventricular function primarily through paracrine effects, and some studies suggest that the benefits are proportional to the number of engrafted cells (28). Bone marrow-derived MSCs have been employed for cardiac repair in both preclinical and clinical studies. However, myogenic differentiation of these cells may enhance their beneficial effects (2,3). Autologous cells may have limited regenerative capability in aged patients with multiple comorbidities (5). Allogeneic cells from young, healthy donors offer the benefits of improved cardiac function and immediate availability (10). Allogeneic cell products can be consistently expanded, banked, and readily available for transplantation. However, the limited survival and engraftment of allogeneic cells may reduce their long-term benefits (12). To attain sustained therapeutic benefit, the immunological barriers must be overcome to increase transplanted cell survival. IL-10 gene transduction of allogeneic SMCs has been proposed to suppress the host immune system, enhance survival of the allogeneic cells, and improve ventricular function after an MI.
SMCs are a myogenic cell type that contributes to cardiac repair by multiple mechanisms. We have previously demonstrated that transplanted SMCs limited LV dilation, improved heart function following coronary occlusion (16), and maintained the integrity of the extracellular matrix (9). SMCs can be easily harvested from a peripheral vein of a healthy donor and readily expanded in vitro to provide an off-the-shelf product for cell therapy. However, the major benefit of SMCs in these studies was the proof of concept that IL-10 transduction could reduce rejection of myogenic cells that were not inherently immunomodulatory.
IL-10 is a pleiotropic, anti-inflammatory cytokine. It inhibits the activation and proliferation of cytotoxic T-cells, B-cells, and NK cells (35). In the present study, IL-10 gene transduction of SMCs decreased the number of cytotoxic CD8+ T-cells and reduced the cytotoxicity caused by leukocytes toward the allogeneic SMCs in coculture. In the in vivo study, IL-10 gene enhancement decreased the number of infiltrating CD8+ T-cells and the antidonor alloantibody titers in allogeneic cell recipients. Thus, IL-10 maintained the immunoprivileged environment of allogeneic SMCs, mitigated the alloimmune response in the myocardium, and enhanced the survival of transplanted allogeneic SMCs in the heart. Several studies have reported that exogenous IL-10 improves graft tolerance and survival (11,33). IL-10 gene therapy of donor lungs improves the posttransplant graft function in a mouse model (11). IL-10 therapy promotes allograft acceptance and survival by inhibiting the activation of antigen-presenting dendritic cells and by inducing apoptosis of cytotoxic T-cells (11,24).
An important mechanism responsible for immune tolerance of allografts is the induction and stimulation of Treg cells (4). Tregs secrete inhibitory soluble factors such as IL-35, IL-10, and TGF-β and regulate cytotoxic T-cell-mediated immune responses (4,31). Tregs also promote immunomodulation by inducing apoptosis of cytotoxic T-cells through the production of cytolytic effector molecules such as granzyme A/B, perforin, and galectin (31). In the present study, we found a significantly greater number of CD4+CD25+ Tregs with IL-10 gene-enhanced cell therapy both in vitro and in vivo. The increased Treg population may have suppressed the cytotoxic CD8+ T-cells and the alloimmune responses and contributed to the observed increase in survival of transplanted IL-10-expressing allogeneic SMCs. Li et al. demonstrated in a liver transplant model that an increase in the number of Tregs was associated with increased expression of the T-cell inhibitory molecule cytotoxic T-lymphocyte antigen 4 (CTLA4) (19).
In this study, increased CTLA4 expression was seen in leukocytes after coculture with IL-10-expressing SMCs. CTLA4 (expressed on the surface of T-cells) is a negative regulator of cytotoxic T-cell function, and promotes Treg cell production (30,38). Fusion proteins of CTLA4 have been reported to promote the conversion of naive CD4+ CD25- T-cells into CD4+CD25+ Treg cells (30). CTLA4 ligation antagonizes early T-cell activation by IL-2 production, cell cycle progression, and T-cell antigen receptor signaling (21). Treatment with antibodies against CTLA4 prevented the induction of liver transplant tolerance and was associated with increased activity of donor-specific T-cells and NK cells (19). We observed that IL-10 overexpression in SMCs increases the CTLA4 levels in leukocytes after coculture. Thus, in our study, CTLA4 probably mediates the conversion of cytotoxic T-cells toward Tregs and downregulates the activation of CD8+ T-cells. Further studies using CTLA4-blocking antibodies are required to confirm the role of CTLA4 in IL-10-mediated tolerance of allogeneic SMCs in the heart.
This work has some limitations. In the present study, we have not specifically compared SMCs with and without the GFP label. In previous work, we were not able to detect any differences in functional outcomes between animals transplanted with GFP+ and GFP- MSCs, nor have we found differences after transplanting labeled and unlabeled SMCs (16,17). Also, an empty adenoviral vector control was not employed for the in vivo studies. The vector may have increased the immunogenicity of the cells resulting in an underestimation of the effects of IL-10. In addition, IL-10 transfection of autologous cells was not evaluated in this study. Cytokine therapy alone has been shown to improve outcomes in this model. Future studies will be required to determine the long-term effects of IL-10-transduced SMCs and to evaluate cell survival and cardiac function at 6 months and 1 year. In addition, future studies will be required to conclusively determine the mechanisms responsible for the improved survival and functional benefit achieved with the transduced cells.
In summary, this study demonstrated that IL-10 gene-enhanced cell therapy mitigated alloimmune responses, increased survival of allogeneic SMCs in the heart, and improved cardiac function after MI compared to controls. IL-10 gene-enhanced cell therapy provides a new opportunity to deliver highly regenerative allogeneic cells to fulfill the promise of cell transplantation to restore ventricular function after an MI in aged, debilitated patients.
Footnotes
Acknowledgments
We thank Rongqi Duan for her technical assistance in producing and purifying the viral vectors. This work was supported by grants from the Canadian Institutes of Health Research (MOP102535 and RMF111623 to R.K.L.). Dr. Ren-Ke Li holds a Canada Research Chair in Cardiac Regeneration. Dr. Sanjiv Dhingra was supported by a postdoctoral fellowship from CIHR. The authors declare no conflicts of interest.
