Abstract
The aim of this study was to investigate the effect of aging and timing of left ventricular ischemic injury on the availability and functionality of stem cells. We studied young and aged male inbred Lewis rats that were used as donors of bone marrow mononuclear cells (BM-MNCs), divided in four experimental groups: controls, sham operated, 48 h post-myocardial infarction (MI), and 28 days post-MI. In vitro studies included flow cytometry analysis, hematopoietic colony-forming capacity, and invasion assays of migration capacity. BM-MNCs from these groups were transplanted in female rats after MI induction. Late engraftment was evaluated by real-time PCR of the SRY chromosome. Percentage of CD34+/CD45+low cells was similar among different experimental groups in young rats, but was significantly higher in aged animals (p < 0.001), particularly 28 days post-MI. KDR+/CD34+ cells were increased 48 h after MI and decreased 28 days post-MI in young animals, while they were profoundly reduced in the aged group (p < 0.001). Triple staining for CD44+/CD29+/CD71+ cells was similar in different groups of aged rats, but we observed an intense increase 48 h post-MI in young animals. Colony-forming units and cytokine-induced migration were significantly attenuated 28 days after the MI. Late engraftment in infarcted transplanted female hearts was present, but considerably heterogeneous. Finally, recovery of left ventricular systolic function in transplanted female recipients was significantly influenced by donors' BM-MNCs groups (p < 0.01). We have demonstrated that aging and timing of myocardial injury are factors that may act synergistically in determining stem cell availability and function. Such interaction should be considered when planning new cell therapy strategies for acute and chronic ischemic heart disease in the clinical arena.
Introduction
Cell therapy has emerged in the last decade as a potentially ground-breaking strategy to treat cardiovascular disease (24). In 2001, Orlic et al. published two land-mark studies showing that locally delivered (20) and cytokine-mediated translocation of bone marrow cells (21) could generate de novo myocardium. The newly formed tissue occupied approximately 68% of the infarcted segment and substantially decreased mortality rates. These remarkable results were partially replicated by several groups of investigators using different progenitor cell types in distinct experimental models of heart dysfunction (4,7,12,14,15,17,27,30,32,33,37). Data from small clinical studies that used primarily intracoronary bone marrow mononuclear cells (BM-MNCs) after an acute myocardial infarction, however, were not as impressive. Overall, increments in left ventricular ejection fraction (LVEF) attributed to stem cell therapy were modest or nonsignificant (1,13,16,28). Indeed, Lunde et al. found a marginal decrease in LVEF assessed by magnetic resonance imaging 6 months after cell transplantation in a small randomized trial (16). Reasons for these discrepancies and disappointing results are unclear and certainly complex, but might be related to stem cell type, abundance, quality, and function, as well as timing of the intervention.
Recently, a group of investigators published intriguing findings suggesting that BM-MNCs from patients with chronic ischemic cardiomyopathy are profoundly limited in their ability to migrate and form colonies. This impaired cellular functionality was also associated to reduced neovascularization capacity in vivo (9). Factors responsible for this functional impairment are uncertain, but aging, cardiovascular atherosclerotic risk factors and left ventricular pump dysfunction may be intrinsically involved. Although stem cell characterization implies in extensive and even indefinite self-renewal capacity, there is recent evidence suggesting that functional properties of hematopoietic stem cells may in fact profoundly altered with aging (2,8,18). Importantly, taken together these findings may limit the therapeutic potential of most cell therapy strategies in the cardiovascular setting.
In the present protocol, we sought to investigate the role of aging and timing of left ventricular damage on the abundance and functionality of stem cells in an experimental model of acute and chronic myocardial infarction, using young and aged male Lewis rats as BM-MNCs donors.
Materials and Methods
Animals
Male and female inbred Lewis rats were obtained from CEMIB-UNICAMP (Campinas, Brazil) and used as donors (males) and recipients (females) of BM-MNCs. All procedures were approved by the Ethics Committee of Hospital de Clinicas de Porto Alegre (HCPA), and performed according to the principles of the Brazilian College on Animal Experimentation (www.cobea.org.br), the Council for International Organizations of Medical Science, and the US National Institutes of Health. A subgroup of male Lewis rats was aged for 12 months at the animals' facilities of HCPA, given food and water ad libitum in day and night cycles of 12 h.
BM-MNC Donors
Young (8–10 weeks old) and aged (12 months old) male inbred Lewis rats were used as donors for BM-MNCs analysis, divided in several experimental groups (6–8 animals/group): 1) control animals, 2) sham-operated animals, 3) acute myocardial infarction (MI) animals, and 4) chronic ischemic heart failure. In the acute MI animals, BM-MNCs were collected 48 h after left anterior descending artery ligation, while in the chronic ischemic heart failure animals BM-MNCs were harvested 28 days after MI induction.
BM-MNC Recipients
Female inbred Lewis rats (8–10 weeks old) were transplanted with BM-MNCs derived from the 8 experimental groups described above (4 groups of young rats and 4 groups of aged rats), immediately after induction of an acute MI.
Myocardial Infarction Model
MI was induced according to a procedure previously described in the literature (23) and adapted in our laboratory. In brief, animals were placed in dorsal decubitus and anesthetized with xilazine (0.67 mg/kg) and ketamine (0.33 mg/kg) administered intraperitoneally. Following orotracheal intubation, animals underwent mechanical ventilation (60 movements/min and a tidal volume of 1.5 ml) with a Harvard ventilator (Model 683, Harvard Medical, MA). A surgical incision was performed in the skin along the left sternal margin, divulsion of pectoralis and transverse muscles was made, thoracotomy was performed at the third intercostal space, without exteriorization of the heart. The left anterior descending coronary artery (LAD) was identified and ligated with a 6-0 mononylon suture, between the left atrial appendage margin and the pulmonary artery. Next, the thoracic cavity was closed with a 5-0 mononylon suture, muscles were repositioned, and the skin sutured. All animals received analgesia after surgery (dipirone 0.1 ml, IM). Sham-operated rats underwent the same procedure, except for LAD ligation.
Isolation of BM-MNCs
Donors (male Lewis rats) were individually killed and both femurs and tibias were removed in sterile conditions. All connective tissue was removed and bones were placed in culture medium [Iscove's modified Dulbecco's medium (IMDM, Gibco) with 10% fetal bovine serum (Gibco), 1% penicillin (10,000 U/ml), and 1% streptomycin (10,000 μg/ml, Gibco)]. Proximal and distal margins of the tibia and femur were then clipped to expose the marrow; bone marrow cells were recovered by the “flushing procedure” using a 22-gauge needle and a 5-cc syringe filled with culture medium. Cell suspension was washed twice and transferred to a sterile tube. BM-MNCs were isolated by centrifugation through a Ficoll-plaque Plus (Amersham Biosciences) density gradient. Briefly, cells suspension was loaded on a 50% Ficoll gradient, according to the manufacturer's indications, and centrifuged at 1500 rpm for 15 min. Fat and serum layers were discarded, and the band of low-density white cells was collected, washed twice, and studied for cell viability with trypan blue. These BM-MNCs were used for the in vitro and in vivo studies.
In Vitro Studies
Flow Cytometry Analysis
Approximately 1 × 106 BM-MNCs were prepared. They were place in sterile tubes and washed two times by centrifugation at 2000 rpm for 5 min at 4°C. BM-MNCs were then resuspended in 200 μl of PBS and incubated for 20min at 4°C with anti-CD45/FITC (CALTAG lab) and anti-CD34/PE (Santa Cruz Biotechnologies); anti-KDR (Abcam) and anti-CD34/PE (Santa Cruz Biotechnologies). We also performed triple staining for anti-CD44/FITC (Abcam), anti-CD71/PE (AbD serotec), and anti-CD29/PECy5 (BioLegend). All assays were conducted using concentration of antibodies that were recommended by manufacturers. Phycoerythrin-PE and FITC mouse anti-rat IgG1, IgG2a, and IgM were used as isotype controls. After incubation of antibodies, lysis solution (1 ml) was added for 15 min to hemolize erythrocytes; then 1 ml of PBS was added for 15 min to stop the hemolytic treatment. Cells were collected and washed with PBS by centrifugation and 500 μl of PBS was added to prepare the cell suspension. Analysis was carried out with the BD FACS-Calibur flow cytometry system with the one-laser system that is capable of detecting three fluorochromes excited by the 488–nm laser in a multiparameter manner. The samples were read in the cellquest and PAINT-A-GATE softwares.
Hematopoietic Colony-Forming Unit
BM-MNCs were seeded in methylcellulose (Methocult GF R3774, Stem Cell Technologies, Vancouver) containing Iscove's medium, fetal bovine serum, bovine serum albumin, mercaptoethanol, and recombinant cytokines (stem cell factor, granulocytic-macrophage colony stimulating factor, and interleukin-3). Cells were plated in triplicate at 1 × 104 cells per plate and cultures were incubated at 37°C and 5% CO2. After 10 days of incubation, cultures were removed and the plates were studied under phase-contrast microscopy. Granulocyte-macrophage, isolated granulocyte, and total colony-forming units were counted (Fig. 1). Colony forming units were counted whenever aggregation was greater than 50 cells. Counting was performed by a technician that was blinded for the assignment of experimental groups.

Representative examples of different types of colony-forming units.
Migratory Capacity (Invasion Assay)
Invasion assay assessing the ability of cells to invade a synthetic basement membrane was performed in invasion chambers with 8 μm pore size PET membrane (cell culture insert, BD Bioscience) separating the upper and lower chambers. The top surface of the membrane was coated with Matrigel matrix [BD Matrigel™ Basement Membrane Matrix High Concentration (HC) Phenol Red Free]. The chamber was placed in a 24-well culture dish containing 500 μl of culture medium, 50 ng/ml of vascular endothelial growth factor (VEGF, Biosource International), and 100 ng/ml of stromal cell-derived factor 1 (SDF-1, Biosource International). After 24 h of incubation at 37°C and 5% CO2, the noninvading cells on the top surface of the filter membrane were removed with a cotton swab and the transmigrated cells found on the bottom surface were counted, as previously described (9). Counting was performed by a technician that was blinded for the assignment of experimental groups. The number of transmigrated cells was counted under a microscope at 400× magnification and the mean number of cells per field in 10 random fields was recorded and used for statistical analysis.
In Vivo Studies
Cell Transplantation Procedure
Recipients (female Lewis rats) were infarcted as described above. Immediately after operation, 1 × 106 BM-MNCs from each experimental group were resuspended in 70 μl of culture medium without serum and were injected at 5–6 points in the border zone of the infarcted area. For the cell transplant procedure, each male donor rat had one or two reciprocating female recipients. All female recipients were euthanized 28 day after the MI to search for the presence Y chromosome in the transplanted heart.
SRY Chromosome Analysis by Real-Time PCR
Genomic DNA was extracted using the Purelink genomic DNA kit (Invitrogen) and quantified with Quant-it DSDNA assay kit. Triplicate real-time PCR reactions were performed using 20 ng of each genomic DNA. Real-time PCR reactions were performed using the ABI PRISM 7500 PCR System (Applied Biosystems), the TaqMan Gene Expression Assay-SRY (Applied Biosystems), and the Rat GAPD (GAPDH), as an endogenous control (Endogenous Control VIC/MGB Probem primer Limite, Applied Biosystems) to normalize the amount of DNA. The cycling conditions (n = 50) were 2 min at 50°C, 10 min at 95°C for polymerase activation and denaturation, 15 s at 95°C for annealing, and 1 min at 60°C for extension. For relative quantification of SRY amplification, specific calibration (from young control animals) was used for each segment of the ventricle analyzed (on an animal per animal comparison).
Echocardiography
Donor animals underwent two-dimensional Doppler echocardiography after the surgical procedures to attest that MI was induced and sham procedures did not affect left ventricular (LV) systolic function. Echocardiographic studies were performed under light intraperitoneal anesthesia and spontaneous respiration. Fundamental and harmonic cardiac imaging was obtained by an ultrasonographer experienced in rodent imaging, using an EnVisor HD System (Philips Medical, Andover, MA) and a 12–13 MHz linear transducer. Animals were placed in the left lateral decubitus position (45°) in a warm environment to obtain cardiac images. M-mode, two-dimensional, and Doppler images from the parasternal and apical views were obtained and recorded on digital format for off-line measurements. Echocardiography-derived parameters used for the current analysis included LV fractional area change and MI size, using a previously validated protocol (19). LV systolic function was estimated by calculating the fractional area chance (FAC), calculated as: FAC = (end-diastolic transverse area – end-systolic transverse area/end-diastolic transverse area) × 100. The final FAC is reported as a percentage and represents the mean from three transverse planes (basal, medial, and apical). In addition, on each echocardiographic transverse plane the arc corresponding to the segments with infarction (AI)—representing segments showing hypokinesis, akinesis, and/or dyskinesis—and to the total perimeter of the endocardial border (EP) was measured at end-diastole. Infarction size (IS) was estimated as previously described as: IS (%) = (AI/EP) × 100. The final value of myocardial infarction size for each animal is reported as the mean IS obtained from the same transverse planes. As previously reported (19), there is an excellent agreement between echocardiography and histology for infarct size assessment in the rodent experimental model (r = 0.88 between echo and histology findings). After undergoing echocardiography, animals were killed and underwent thoracotomy for heart and lung removal.
Statistical Analysis
Continuous variables with normal distribution were expressed as average ± SD and compared using Student's t-test or variance analysis (ANOVA). Post hoc analysis using the Tukey test was applied whenever appropriate. General linear models (GLM) were used to evaluate differences between young and aged animals and to evaluate the potential interaction between age and timing of MI. Categorical variables were expressed as frequencies and percentages. For comparisons between different groups, the chi-square test or Fisher's exact test was used. A multivariate model was also created to assess independent predictors for the migration analysis and the colony-forming unit assay. A bi-caudal value of p < 0.05 was considered statistically significant. Analyses were performed using SAS for Windows statistical package (version 8.0).
Results
Donors (Male Inbred Lewis Rats)
Perioperative surgical mortality for MI induction was approximately 40%, mostly related to anesthesia hypersensitivity and infarct-related events. As expected, left ventricular systolic fractional area change was substantially reduced in infarcted animals when compared to control and sham-operated rats [0.41 ± 0.10% for the 28 days post-MI group vs. 0.49 ± 0.13% for the 48 h post-MI group vs. 0.64 ± 0.08% for the sham-operated group vs. 0.69 ± 0.03% for the control group; p < 0.0001, pos hoc analysis showing significant differences between infarcted animals versus non-infarcted animals (shams and controls)]. No segmental abnormalities were identified in sham-operated and control animals, while the average infarct size from the remaining infarcted animals was 50 ± 14.9%, representing a large amount of damaged myocardium.
Flow Cytometry Analysis
Hematopoietic Stem Cell Markers
To investigate whether the number of hematopoietic stem/progenitor cells is reduced in BM-MNCs of young and aged rats with early and late MI, we determined the percentage of stem progenitor cells characterized by CD34 and CD45 expression. Overall, CD34+/CD45+low cells were marginally increased after 28 days post-MI. The percentage of CD34+/CD45+low BM-MNCs was similar among different experimental groups in young rats, but in aged rats the number of CD34+/CD45+low was significantly higher (p < 0.001), particularly 28 days after the MI (Fig. 2). We observed a significant interaction between age and experimental groups in this analysis (p < 0.001 for GLM interaction term).
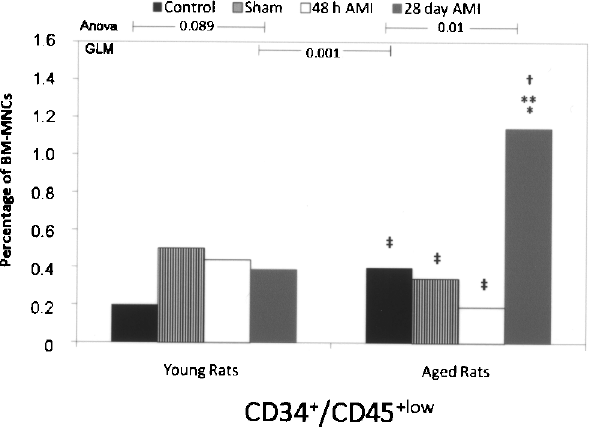
Flow citometry analysis for CD34+/CD45+low in young and aged animals according to experimental groups (control, sham, acute MI, and chronic MI). Differences between groups were evaluated by post hoc analysis using the Tukey test: *significant difference between controls and other groups; **significant difference between shams and other groups; †significant difference between acute MI and other groups; ‡significant difference between chronic MI and other groups. GLM procedure was applied to identify differences between young and aged animals, as depicted. Interaction term between age and timing of MI was statistically significant (p < 0.001).
Endothelial Progenitor Cell (EPC) Markers
The percentage of endothelial progenitors in the BM-MNCs was determined by the expression of KDR and CD34 (Fig. 3). Overall, EPCs were significantly increased after an acute MI when compared with control and sham animals; however, the percentage of KDR+/CD34+ cells was substantially reduced 28 days after the MI. In young animals, KDR+/CD34+ were increased 48 h after the MI and decreased 28 days after the MI, when compared to control and sham-operated animals. Notably, EPCs were profoundly reduced in the aged group (p < 0.001). Aged animals had a 7.8-fold reduction in KDR+/CD34+ cells 48 h after the MI and a sevenfold reduction 28 days after the MI (Fig. 3B). For this analysis, we observed a tendency of interaction between age and experimental groups (p = 0.06 for GLM interaction term).
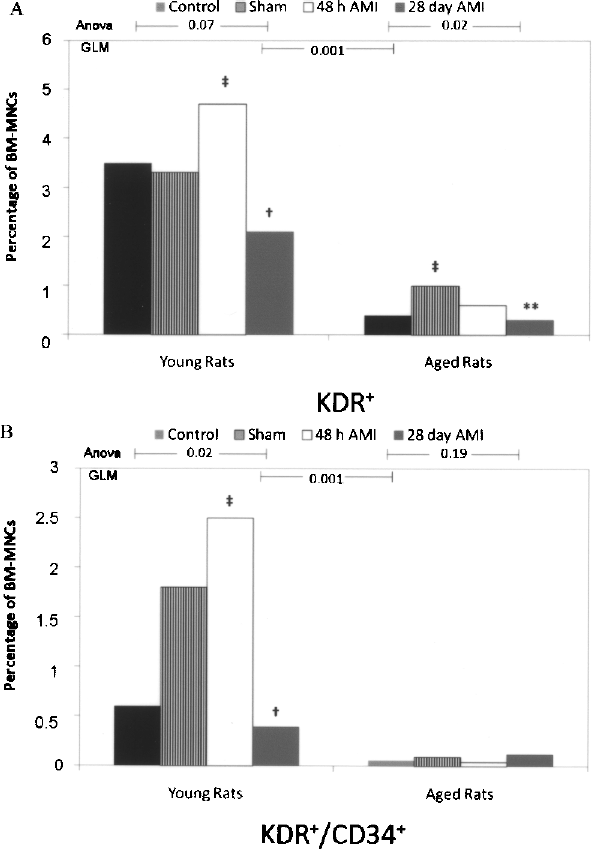
Flow cytometry analysis for total KDR+ (A) and KDR+/CD34+ (B) in young and aged animals according to experimental groups (control, sham, acute MI, and chronic MI). Differences between groups were evaluated by post-hoc analysis using the Tukey test: *significant difference between controls and other groups; **significant difference between shams and other group; †significant difference between acute MI and other groups; ‡significant difference between chronic MI and other groups. GLM procedure was applied to identify differences between young and aged animals, as depicted. Interaction term between age and timing of MI was marginally statistically significant (p = 0.06).
Mesenchymal Stem Cells Marker
Although there is not a consensual set of markers for mesenchymal stem cells detection in vivo or in the bone marrow, for the purpose of this study we used the concomitant expression of CD44, CD29, and CD71 as potential indicators of this lineage. Individually, expression of these markers was age-dependent and significantly diverse according to experimental groups (Fig. 4). For instance, CD71+ cells were more than threefold increased 48 h post-MI when compared to sham-operated rats in young animals, while in aged animals they were increased only 28 days post-MI. Triple staining (CD44+/CD29+/CD71+ cells) (Fig. 4C) was similar in aged rats irrespective of the experimental groups, but we observed an intense increase of CD44+/CD29+/CD71+ cells 48 h post-MI in young animals (p < 0.001, post hoc analysis depicting significant differences to all other groups) (Fig. 4C). For triple staining, we observed a significant interaction between age and experimental groups (p < 0.001 for GLM interaction term).

Flow cytometry analysis for total CD44+ (A), CD 41+ (B), and CD44+/CD71+/CD29+ (C) in young and aged animals according to experimental groups (control, sham, acute MI, and chronic MI). Differences between groups were evaluated by post hoc analysis using the Tukey test: *significant difference between controls and other groups; **significant difference between shams and other group; †significant difference between acute MI and other groups; ‡significant difference between chronic MI and other groups. GLM procedure was applied to identify differences between young and aged animals, as depicted. Interaction term between age and timing of MI was statistically significant (p < 0.001).
Colony-Forming Units and Migration Assay
The functional capacity of progenitor cells in the bone marrow aspirates was determined by measuring the colony-forming activity. The same profile of colony-forming capability was observed in young and aged animals. Overall, we observed a significant increase in the absolute number of colonies after the sham procedure and 48 h post-MI. This amplified functionality was completely abolished after 28 days post-MI (Fig. 5A). The same findings were observed in analysis stratified for the granulocytic-monocytic- and monocytic-forming units. A similar profile of migration response to SDF-1α and VEGF was observed in young and aged animals. However, migration was also significantly attenuated after 28 days post-MI (Fig. 5B). No interaction between age and experimental groups was identified for migration and colony-forming capacity.

Colony-forming units (A: all types; B: granulocytic-monocytic + granulocytic) and migration capacity (C) in young and aged animals according to experimental groups (control, sham, acute MI, and chronic MI). Differences between groups were evaluated by post hoc analysis using the Tukey test: *significant difference between controls and other groups; **significant difference between shams and other group; †significant difference between acute MI and other groups; ‡significant difference between chronic MI and other groups. GLM procedure was applied to identify differences between young and aged animals, as depicted. Interaction term between age and timing of MI was not statistically significant (p > 0.50).
No significant association was observed between colony-forming and migration capacity (correlation coefficient <0.10, p = 0.76). In a multivariate model adjusted for age, percentage of KDR+/CD34+ cells, percentage of CD34+/CD45+low cells and experimental groups, migration capacity was independently associated only to timing of the MI, indicating a substantial decrease in the ability to migrate in the 28 days post-MI group (β coefficient −70.8; p = 0.003; model r2 = 0.33). Conversely, in a multivariate model adjusted for the same variables, granulocytic-monocytic-forming units were independently associate to experimental groups [sham surgery (p = 0.03) and 48 h post-MI (p = 0.01)] and aging (p = 0.07, model r2 = 0.46).
SRY Chromosome Analysis (Real-Time PCR)
For SRY chromosome analysis, 28 days after MI induction and cell transplantation, animals were killed and the left and right ventricles from female recipient hearts were separated. Heart tissue from the left ventricle was further divided in basal, medial and apical segments. Amplification of the SRY chromosome was heterogeneous between experimental groups, in different segments of the ventricles of the same animal, and also in triplicates of the same tissue sample. Table 1 describes a qualitative analysis to demonstrate late engraftment of transplanted cells, according to each experimental group of donors. Unfortunately, data from relative quantification of the SRY gene was not reliable to allow statistical analysis, as amplification in individual animals was greatly heterogeneous in triplicates.
SRY Chromosome Analysis by Real-Time PCR

Values are expressed as number of positive SRY amplification per analyzed animals. Amplification was considered qualitatively positive whenever at least one of the triplicates amplified.
Left Ventricular Function (Female Recipients)
Left ventricular systolic fractional area change in female recipients that were transplanted after induction of an acute MI with BM-MNCs derived from the 8 experimental groups is described in Table 2. We observed a decrease in left ventricular function recovery in transplanted animals with donor cells from young animals 28 days post-MI (ANOVA p = 0.004, post hoc difference between donor controls). In addition, left ventricular systolic fractional area change tended to be lower in transplanted animals with donor cells from aged animals (p = 0.06 for GLM interaction term between age and experimental groups).
Left Ventricular Systolic Fractional Area Change in Female Rats 28 Days After Cell Transplantation According to Experimental Donor Groups

Post hoc analysis showing difference between BM-MNCs from control versus 28-days postinfarcted animals. GLM interaction term between age and experimental groups: p = 0.06.
Discussion
Efficacy of different cell therapies to improve LV systolic function has been limited in most clinical studies (1,13,16,28), despite consistent encouraging results in experimental models of heart injury and regeneration (7,12,17,30,37). In the present protocol, we analyzed the effect and interaction of two central factors that could interfere with the effectiveness of cell therapy in the setting of ischemic heart disease: duration of heart injury and aging. Our findings suggest that there is substantial variability on stem cells' absolute number and functionality related to these factors for all stem cell phenotypes that we evaluated, leading to a diverse capacity to form colonies and migrate. In particular, the interaction of an aged animal with chronic LV injury (28 days post-MI) caused profound changes on these features. Such variability translated in significant changes in left ventricular systolic function assessed by echocardiography in transplanted animals. We believe that such factors should be carefully considered when planning new cell therapy strategies for patients with acute and chronic ischemic heart disease.
Our data suggests that the overall number of hematopoietic stem cells (HSCs) (CD34+/CD45+low) was similar in both age strata, but was somewhat increased late after the ischemic injury, particularly in aged animals. In this regard, the effect of aging on the absolute number of stem cells is not consensual, but there is solid experimental evidence in murines that HSCs within the bone marrow is increased in concentration and absolute number as the animals ages, paralleled with increased homogeneity of stem cells marker expression. The cause of the increased number of HMCs remains unclear, but a feedback compensatory mechanism could be present in response to a decline in functionality on a per cell basis (2). In fact, several studies have clearly shown that aged HSCs demonstrate a significant deficit in hematopoietic output when compared to HSC from young mice (18), besides having a higher myeloid progenitor activity. In addition, recent evidences suggest that cell–cell interactions with stroma in the stem cell niche are a central aspect for self-renewal and differentiation, as HSCs are extremely sensitive to these interactions and to secreted factors in this microenvironment. Loss of the osteoblastic niche with aging, associated with a proinflammatory state may participate in this complex interplay that cause reduced self-renewal and differentiation ability (18). We can speculate that the somewhat unexpected increase in CD34+/CD45+low cells in aged animals late after MI could be, at least in part, secondary to compensatory mechanisms of feedback associated to impaired autonomous stem cell mobilization from the bone marrow. This absolute increase found in our analysis, however, was accompanied by the “expected” reduced cytokine-induced migration and ability to form granulocytic/monocytic units.
Studies that have evaluated these factors in experimental models of cardiovascular disease are scarce. Heeschen et al. have demonstrated that the number of HSCs remains the same in chronic ischemic heart failure in humans, but the functional capacity of these cells was impaired in a murine model of hind limb ischemia (9). The observed HSCs dysfunction, however, could be secondary to several factors associated to chronic ischemic heart failure, such as risk factors that lead to atherosclerosis, other processes related to coronary artery disease itself, and aging, as heart failure is a disease of the elderly. In this scenario, our findings suggest that stem cells dysfunction may be to a great extent dependent on the interaction between aging and duration of myocardial injury. For instance, we have demonstrated that KDR+/CD34+ cells were greatly increased 48 h after an MI in young rats, an effect that was not observed in aged animals. Previous reports have shown that acute MI is associated with a rapid increased of EPCs in circulation due to a transient mobilization in response to plasma levels of VEGF (10,11). The detailed temporal dynamic of EPCs mobilization after an acute myocardial injury is not clearly defined, but our data suggest that in the setting of chronic heart failure the absolute number of KDR+ cells is no longer increased. Moreover, we have also demonstrated that the number of EPCs in the bone marrow was profoundly reduced in the aged group, even after an acute MI. Indeed, in this specific subgroup we have identified a 62-fold reduction on KDR+/CD34+ cells (from 2.5 ± 1.3% in young animals 48 h post-MI to 0.04 ± 0.03% in aged animals 48h post-MI, p < 0.0001 in post hoc analysis) (Fig. 4C). Our findings concur with recent data demonstrating that atherosclerotic risk factors and chronic heart failure are associated to reduced number and function of cultured EPCs, circulating CD34+/KDR+ cells and granulocyte macrophage colony-forming units in the bone marrow (14,36,38). A recent report has found similar absolute numbers of EPCs in peripheral blood in older patients with coronary artery disease when compared to younger subjects. These patients, however, apparently did not have significant LV systolic dysfunction (22). This latter finding reinforces the importance of the interaction of aging and presence of myocardial dysfunction as intrinsically dependent factors that might determine peripheral availability of stem/progenitor cells.
The importance of EPCs in the clinical arena have been recently highlighted by data demonstrating that reduced levels of circulating EPCs were an independent predictor of poor prognosis in a heterogeneous group of patients with stable and unstable coronary artery disease (29). Multiple factors determine the number of circulating EPCs, depending on a delicate and poorly understood balance between speed of bone marrow mobilization, engraftment, and turnover of EPCs at sites of vascular injury, inhibition of differentiation, and rates of apoptosis (25). Our data indicate that both aging and timing of myocardial injury act synergistically, decreasing their number in the bone marrow and their ability to migrate and form colonies.
There is no consensus about markers to identify mesenchymal stem cells in vivo (3). Although controversial as surface markers for mesenchymal stem cells, triple staining for CD44+/CD71+/CD29+ was significantly increased 48 h post-MI and attenuated 28 days post-MI in young rats. Disparate results have been reported with regard to mesenchymal stem cells and aging, with some groups finding an age-related decline in availability and function whereas others observing no change at all (5,6,26). Initial studies in rats suggest that mesenchymal stem cells from aged rats depict a reduced capacity to generate chondrogenic matrix in vitro (39).
There is an increasing body of evidence supporting that alteration in the VEGFR2 and SDF-1/CXC chemokine receptor-4 (CXCR4) axis is a crucial regulator of progenitor cell function, recruitment, and homing. Van Weel et al. demonstrated that VEGF-A, SDF-1, and CXCR4 expressions were generally decreased in ischemic muscle compared with nonischemic muscle in patients with chronic ischemia, whereas they were substantially increased in patients with acute-on-chronic ischemia (35). Recently, Seeger et al. have elegantly demonstrated that CXCR4+ bone marrow cells exhibit an increased therapeutic potential for blood flow recovery after acute ischemia. These investigators have demonstrated that CXCR4+ bone marrow cells have higher migratory capacity secondary in part to an increased release of paracrine factors (31). All these data concur with the concept that hypoxic preconditioning may be crucial to enhance cell therapy effectiveness. In this context, Tang et al. have found that hypoxic preconditioning increased CXCR4 expression in cardiosphere-derived, Lin–c-kit+ progenitor cells and markedly augmented their in vitro cell migration and in vivo recruitment to the ischemic myocardium. These effects were largely abolished by the addition of a CXCR4 inhibitor, indicating that the benefits of hypoxic preconditioning are in fact mediated by the SDF-1/CXCR4 axis (34).
Some aspects of our study design and methodology deserve consideration. We acknowledge that there is no consensual definition of markers for the different populations of stem cells in vivo (3). In the current protocol, we chose to use the most widely accepted expression markers both for mesenchymal and endothelial progenitor cells. Also, as we did not include a non-BM-MNC-treated group, we cannot assure that BM-MNC per se led to any substantial treatment benefit on LV function, particularly in aged group. Real-time PCR analysis for the SRY gene was particularly heterogeneous in different experimental groups and in specimens from the same animals, precluding definitive conclusions and comparative analysis of late engraftment. In this regard, engraftment heterogeneity may occur in part because of the intrinsic variability of intramyocardial administration of stem cells in rodent models and differences in cell viability related to microenvironment aspects of the myocardial tissue.
In conclusion, we have demonstrated that aging and heart disease are factors that may act synergistically determining stem cells availability and function. These and other factors that might influence stem cell mobilization, homing, engraftment, and differentiation are vital when planning new strategies to treat acute and chronic ischemic heart disease with novel cell regenerative therapies.
Footnotes
Acknowledgments
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundo de Incentivo a Pesquisa (FIPE-HCPA). Ana Ayala-Lugo was supported by an international exchange scholarship from CAPES/CNPq. We thank Maria Luiza Saraiva Pereira for the insightful comments and help on real-time PCR analysis.
