Abstract
The role of the novel costimulatory molecule TIM4 in anti-islet response is unknown. We explored TIM4 expression and targeting in Th1 (BALB/c islets into C57BL/6 mice) and Th2 (BALB/c islets into Tbet-/- C57BL/6 mice) models of anti-islet alloimmune response and in a model of anti-islet autoimmune response (diabetes onset in NOD mice). The targeting of TIM4, using the monoclonal antibody RMT4-53, promotes islet graft survival in a Th1 model, with 30% of the graft surviving in the long term; islet graft protection appears to be mediated by a Th1 to Th2 skewing of the immune response. Differently, in the Th2 model, TIM4 targeting precipitates graft rejection by further enhancing the Th2 response. The effect of anti-TIM4 treatment in preventing autoimmune diabetes was marginal with only minor Th1 to Th2 skewing. B-Cell depletion abolished the effect of TIM4 targeting. TIM4 is expressed on human B-cells and is upregulated in diabetic and islet-transplanted patients. Our data suggest a model in which TIM4 targeting promotes Th2 response over Th1 via B-cells. The targeting of TIM4 could become a component of an immunoregulatory protocol in clinical islet transplantation, aiming at redirecting the immune system toward a Th2 response.
Introduction
Islet transplantation has been shown to be a potentially effective cure for type 1 diabetes (T1D) and has been demonstrated to normalize glucose metabolism and halt the development of diabetic complications (9,38). Unfortunately, islet graft survival rates remain below those of other types of allografts (9,28). Considering the potential advantages of islet transplantation on diabetic complications (6,34,39) and the relatively low invasive nature of the procedure, much research has focused on increasing the success of islet cell transplantation (9,12,29). Although multiple factors have been deemed responsible for the failure of islet grafts, the development of the alloimmune response and the recurrence of autoimmune diabetes play primary roles. In the allo- and autoimmune responses, the reciprocal interaction between costimulatory molecules on T-cells and antigen-presenting cells (APCs) (e.g., CD40L/CD40 or CD28/B7.1-2) determines, together with the cytokine environment, the differentiation of naive Th0 cells into Th1, Th2, Th17, and Tregs predominantly (13,18,43), thus resulting in acceptance or rejection of the graft and in the onset or avoidance of autoimmunity. While the role of Th1 and Th17 cells in promoting allo- and autoimmune responses and of Tregs in promoting graft and self-tolerance has been clearly assessed (30,42,43), the role of Th2 cells remains controversial. Although Th1 to Th2 skewing is considered a pivotal marker of graft acceptance, the Th2 response per se has been shown to be capable of causing graft rejection (17,25,33,41), and the exact role of the Th2 response in islet graft rejection remains unclear. TIM4 is a novel costimulatory molecule and a member of the T-cell immunoglobulin mucin (TIM) family, and it was recently shown to contribute to the development of the Th1 and Th2 responses (16). The TIM family consists of eight members in mice (TIM1-TIM8) and three in humans (TIM1, TIM3, and TIM4) (16). TIM1 and TIM3 are primarily expressed on T-cells, while TIM4 is localized on APCs (20) and serves as a ligand for TIM1 (1,22). The TIM1-TIM4 axis appears to provide a positive activation signal (20,37), leading to T-cell differentiation and activation, at least in models of autoimmunity, allergy, and asthma (19). In allotransplantation, the targeting of TIM1 using an anti-TIM1 monoclonal antibody (mAb) has been shown to prolong allograft survival in a murine model of cardiac allograft rejection by reducing the Th1 and enhancing the Th2 response (35). Furthermore, anti-TIM1 mAb treatment was able to abrogate the Th17 response and to prolong allograft survival in a model of Th2/Th17 rejection (46). A recent study suggests that the TIM1-TIM4 costimulatory pathway may promote tolerance by expanding a population of regulatory B-cells (5). However, the specific role of TIM4 in immune activation and in anti-islet allo- and autoimmune response is not clearly defined (26,45), and TIM4 has been implicated as both an inhibitor (21) and enhancer (23) of the immune response. In our study, we investigate the role of TIM4 and its targeting during the allo- and autoimmune anti-islet immune responses, with the aim of developing therapeutic tools to prolong the life span of exogenous (in the context of islet transplantation) and endogenous (in the pathogenesis of autoimmune diabetes) islets.
Materials and Methods
Patients
Ten islet-transplanted patients, 10 patients with T1D, and 10 healthy controls were enrolled at the San Raffaele Scientific Institute with Institutional Review Board approval (Table 1. All subjects provided informed consent before study enrollment.
Characteristics of Islet-Transplanted Patients and Healthy Volunteers

Data are expressed as mean± SD. T1D, type 1 diabetes; EIR, exogenous insulin requirement; HbA1c, glycated hemoglobin.
Human Islet Transplantation and Immunosuppression
Islets were isolated from pancreata obtained from multiorgan donors, using a modified automated method, and were then purified by centrifugation on a discontinuous gradient as previously described (14). Islets were then transplanted intrahepatically according to ABO matching. Islet-transplanted patients received the standard triple immunosuppressive regimen: anti-thymoglobulin (Thymoglobulin, Genzyme, Framingham, MA, USA) as induction, followed by treatment with FK506 (target blood levels between 6 and 8 ng/ml; Astellas, Deerfield, IL, USA) and/or cyclosporine (target blood level 100 ng/ml; Novartis, Basel, Switzerland) and/or rapamycin (8-15 ng/ml; Pfizer, New York, NY, USA) and/or mycophenolate (2 g/day; Roche, Basel, Switzerland) and prednisone (5-10 mg/day; Bruno, Farmaceutici, Italy); cyclosporine drug level was assessed by immunocolorimetric assay (Siemens, Munich, Germany), FK506 by liquid chromatography-mass spectrometry (Thermo Scientific, Waltham, MA, USA). Steroids were tapered and then withdrawn within 6 months posttransplant. C-peptide level was assessed by immunofluorimetric assay (Tosoh, Tokyo, Japan); HbA1c level was assessed by high-performance liquid chromatography (Biorad, Hercules, CA, USA); exogenous insulin requirement was collected through patient interview.
PBMC From Human Patients
Peripheral blood mononuclear cell (PBMC) fractions were isolated from 20 ml of whole blood by Ficoll (GE Healthcare, Piscataway, NJ, USA) density gradient centrifugation.
Mice
C57BL/6J, BALB/cJ, NOD/ShiLtJ, and Tbet-/- mice (on a C57BL/6 background) were obtained from Jackson Laboratory (Bar Harbor, ME, USA) and maintained as a breeding colony in our animal facility. All mice were cared for and used in accordance with institutional guidelines. Protocols were approved by the Harvard Medical School Animal Care and Use Committee.
Islet Isolation and Transplantation
Pancreatic islets derived from BALB/c donor mice were isolated by collagenase (Sigma-Aldrich, St. Louis, MO, USA) digestion followed by density gradient purification and were handpicked. Islets were transplanted under the renal capsule of chemically induced 8-week-old mice [streptozotocin, 250 mg/kg, administered intraperitoneally (IP)]. Rejection of islet allografts was defined by blood glucose measurements >250 mg/dl on 2 consecutive days.
In Vivo Treatment Protocols
Islet-transplanted mice were treated with the anti-TIM4 mAb RMT4-53 (rat Ig2a; Bio X Cell; West Lebanon, NH, USA) by IP injection at doses of 500 μg on day 0 and 250 μg on days 2, 4, 6, 8, and 10. For B-cell depletion, C57BL/6 mice were treated with an anti-CD22 monoclonal antibody conjugated with calicheamicin (anti-CD22/cal; Wyeth, Madison, NJ, USA) at a dose of 160 μg IP at days 0 and 5 (3). In the diabetes prevention study, 10-week-old female nonobese diabetic (NOD) mice were treated with RMT4-53 at doses of 500 μg on day 0 and 250 μg on days 2, 4, 6, 8, and 10 or were left untreated. Treated mice were followed for diabetes onset, as defined by 2 consecutive days of blood glucose measurements >250 mg/dl (BD Logic glucose meter; BD Biosciences, San Jose, CA, USA).
Islet Pathology
Transplanted mice were sacrificed at various time points to obtain histology specimens. Kidney sections were stained with hematoxylin (Sigma-Aldrich) and eosin (Sigma-Aldrich) (3).
ELISPOT and Stimulation Assays
In vitro and ex vivo interferon-γ (IFNγ) and interleukin-4 (IL-4) production measurements using ELISPOT assays were performed to monitor the alloimmune response (BD Biosciences). C57BL/6 splenocytes (1 χ 106) were challenged in the presence of 1 χ 106 BALB/c irradiated splenocytes or 0.5 μg/ml anti-CD3 Ig/anti-CD28 Ig (BD Biosciences) and were cultured for 24 h (UFN-γ) or 48 h (IL-4) as previously described (41). Spots were counted on an Immunospot analyzer (Cellular Technology Ltd., Cleveland, OH, USA). In the autoimmune setting, splenocytes extracted from NOD mice were rechallenged with 150 μg/ml of the CD4-restricted peptide BDC2.5 (Chi Scientific, Maynard, MA, USA) and 50 μg/ml of the CD8-restricted peptide IGRP (Abcam, Cambridge, MA, USA). Bone marrow-derived dendritic cells (DCs) were generated as previously described (7,36). Lipopolysaccharide (LPS; 10 μg/ml) and PMA (50 ng/ml) used in the in vitro culture were obtained from Sigma-Aldrich.
Flow Cytometry and Intracellular Cytokine Staining
Anti-human CD19 (BD Bioscience) and anti-mouse B220 (BD Bioscience), CD11c (eBioscience, San Diego, CA, USA), TIM4 (Biolegend, San Diego, CA, USA), CD4 (BD Bioscience), CD25 (eBioscience), CD44 (BD Bioscience), CD40 (BD Bioscience), CD69 (BD Bioscience), CD80 (BD Bioscience), CD86 (BD Bioscience), CD62L (BD Bioscience), annexin V (BD Bioscience), 7-AAD (BD Bioscience), IL-17 (eBioscience), and FoxP3 (eBioscience) were used according to the manufacturer's recommendations (8). Antibodies were used at 0.5 μg/ml for receptor staining. Anti-human TIM4 was obtained from Novus Biologicals (Littleton, CO, USA): rabbit polyclonal conjugated to FITC for flow application (NBP1-76702F). Flow acquisition was performed on a FACSCalibur™ analyzer (Becton Dickinson, San Jose, CA, USA), and data were analyzed using FlowJo software version 6.3.2 (Tree Star, Ashland, OR, USA).
Th2 Differentiation Assay
Naive sorted ABM TCR-Tg CD4+CD25- T-cells (Th0) were isolated (CD4+CD25- isolation kit; Milteny Biotec, Auburn, CA, USA) and activated for 5 days with 1 μg/ml of plate-bound anti-CD3 and anti-CD28 antibodies (BD Biosciences) in the presence of the same number of isolated (CD19+ isolation kit; Milteny Biotec) BM12 CD19+ B-cells (which are specifically recognized by the ABM TCR-Tg T-cells). Cultures were supplemented with 5 ng/ml mIL-4 (R&D Systems, Minneapolis, MN, USA) and 1 μg/ml anti-IFN-γ-Ig (BD Bioscience) for Th2 differentiation. T-cells were then harvested and assessed by RT-PCR (40) for GATA3 expression (Th2-specific marker) to quantify level of differentiation (Mm00484683_m1; Life Technologies, Grand Island, NY, USA) (43).
Statistical Analyses
Data are expressed as mean ± standard error for parametric data and median ±95% confidence interval for nonparametric data. Kaplan-Meier analysis was used for survival analysis. When groups were compared cross-sectionally, the two-sided unpaired Student's t-test (for parametric data) or the Mann-Whitney test (for nonparametric data) was used according to value distribution. In the comparison of three or more groups, after assessing normal distribution, a one-way ANOVA test was performed followed by post hoc t test with Bonferroni correction. A value of p <0.05 (by two-tailed testing) was considered an indicator of statistical significance. Data were analyzed with a SPSS statistical package for Windows (IBM, Armonk, NY, USA). Graphs were generated using GraphPad Prism version 5.0 (GraphPad Software, San Diego, CA, USA).
Results
TIM4 Expression In Vitro: The Percentage of TIM4+ B-Cells Decreases During T-Cell Stimulation in the Allo- and Autoimmune Settings
We first analyzed TIM4 expression on APCs (B-cells and DCs) during polyclonal T-cell stimulation in both a nonautoimmune setting to mimic the alloimmune response and during islet peptide-mediated T-cell stimulation to mimic the anti-islet autoimmune response. During polyclonal T-cell stimulation, splenocytes from C57BL/6 mice were challenged with 0.5 μg/ml anti-CD3 Ig and anti-CD28 Ig for 1 or 2 days, and FACS analysis was performed; a reduced percentage of B-cells positive for TTM4 (Fig. 1A) and of DCs positive for TIM4 (Fig. 1B) was observed. The expression level of the other costimulatory molecules assessed was also modified on B-cells and DCs by anti-CD3-Ig and anti-CD28-Ig-mediated stimulation, confirming active modulation of these cells. In particular, a reduced percentage of B-cells positive for CD40 or CD80 was observed after stimulation (Fig. 1C), while the percentage of B-cells positive for CD86 or CD69 was increased (Fig. 1C). To assess if the reduced percentage of TIM4+ B-cells was due to a relatively higher level of apoptosis in TIM4+ cells, we compared the level of apoptosis in TIM4+ and TIM4- cells after the stimulation. No difference was observed (Fig. 1D). We thus hypothesize that the reduced percentage of TIM4+ cells after stimulation was due to the downregulation of TIM4 following T-cell-mediatedtransactivation. To assess that, we purified B-cells from splenocytes, and we cultured them in the presence of anti-CD40 for 1 day to mimic an interaction with activated CD40L+ T-cells. A reduction in TIM4+ B-cells was observed (Fig. 1E). To assess if TIM4 downregulation is a common feature following B-cell activation, we cultured B-cells in the presence of LPS. A consistent reduction in TIM4 expression was observed (Fig. 1E). PMA-mediated stimulation of B-cells was also associated with TIM4 downregulation (data not shown).

During anti-CD3/-CD28 Ig stimulation of splenocytes extracted from C57BL/6 mice, the percentage of B-cells positive for TIM4 (B220+TIM4+) (n = 3, *p <0.05 and **p> 0.01 vs. D0; A) and of DCs positive for TIM4 (CD11c+TIM4+) decreases over time (n = 3, *p <0.05 vs. D0; B). A reduction in B-cells positive for CD40 and CD80 and an increase in B-cells positive for CD86 and CD69 was also observed at day 2 after stimulation (n = 3, *p <0.05, **p <0.01, and ***p <0.001 vs. D0; C). No difference in terms of apoptosis was observed in TIM4+ and TIM4- B-cells at day 2 after stimulation (D). A reduction in the percentage of TIM4+ B-cells was observed after anti-CD40-Ig or LPS stimulation of isolated cells (n = 3, *p <0.05 vs. D0; E). An increase in DCs positive for CD86 and CD69 was also observed at day 2 after stimulation (n> = 3, **p <0.01 vs. D0; F). No difference in terms of apoptosis was observed in TIM4+ and TIM4- DCs at day 2 after stimulation (G). A reduction in the percentage of TIM4+ DCs was observed after anti-CD40-Ig or LPS stimulation of bone marrow-derived DCs (n = 3, *p <0.05 and **p <0.01 vs. D0; H). The percentage of B-cells positive for TIM4 decreases over time in splenocytes extracted from NOD mice and stimulated with the islet peptides BDC2.5 (n = 3, *p <0.05 and **p <0.01 vs. D0; I). The percentage of DCs positive for TIM4 remained unchanged in splenocytes extracted from NOD mice stimulated with BDC2.5 (J).
No variation in the percentage of CD40 and CD80-positive DCs was observed by anti-CD3-Ig and anti-CD28-Ig-mediated stimulation, while CD86 and CD69 were found to be increased (Fig. 1F). No difference in DC apoptosis was observed (Fig. 1G). A reduction in the expression of TIM4 in in vitro generated bone marrow-derived DCs cultured in the presence of LPS or anti-CD40-Ig was observed (Fig. 1H). For islet peptide-mediated T-cell stimulation, splenocytes extracted from normoglycemic NOD mice were stimulated with 150 μg/ml BDC2.5 (CD4-restricted) or 50 μg/ml islet-specific glucose-6-phosphatase catalytic subunit-related protein (IGRP) (CD8-restricted) peptides (4,15). A decrease in the percentage of B-cells positive for TIM4 was evident during BDC2.5 stimulation (Fig. 1I) and IGRP stimulation (data not shown). The percentage of CD40+ and CD80+ B-cells was also reduced, while stable levels of CD86 and CD69 were observed (data not shown). The percentage of DCs positive for TIM4 in the BDC2.5- and IGRP-stimulated settings remained stable (Fig. 1J and data not shown, respectively). Stable levels of CD86 and CD80 were observed, while CD40 and CD69 were increased, confirming that DCs were activated by peptide stimulus (data not shown). The activation of NOD B-cells with anti-CD40-Ig or LPS caused the downregulation of TIM4 expression, which was different in C57BL/6 mice (data not shown) where no reduction in TIM4 expression in DCs was observed. Thus, during both polyclonal antibody and islet peptide stimulation, a decrease in the percentage of TIM4+ B-cells is evident. The reduction in TIM4+ B-cells looks to be related to downregulation of the receptor following transactivation of B-cells mediated by activated T-cells. TIM4 appeared to be also downregulated in C57BL/6-derived DCs, while it was more stable in NOD-derived DCs.
TIM4 Expression In Vivo: The Percentage of TIM4+ B-Cells and DCs Decreases in the Anti-Islet Allo-and Autoimmune Responses
We then analyzed TIM4 expression in vivo during the alloimmune or autoimmune anti-islet response. To evaluate TIM4 expression on APCs during the alloimmune anti-islet response, we transplanted BALB/c mice islets into streptozotocin-treated C57BL/6 mice (fully mismatched), and recipient splenocytes were then harvested at 7 and 14 days after transplantation and assessed by flow cytometric analysis. A reduced percentage of B-cells positive for TIM4 (Fig. 2A) and DCs positive for TIM4 (Fig. 2B) was observed during the alloimmune response. To evaluate TIM4 expression on APCs during the autoimmune anti-islet response, we extracted splenocytes from NOD mice at different stages of disease (10-week-old prediabetic mice and >14-week-old hyperglycemic mice), and we then analyzed TIM4 expression. The percentage of B-cells positive for TIM4 was found reduced in hyperglycemic NOD mice compared to 10-week-old prediabetic mice (Fig. 2C); an increase in the percentage of B-cells positive for TIM4 was observed between 4- and 10-week-old mice. A similar pattern was observed for DCs (Fig. 2D). A reduction in TIM4 expression on B-cells and DCs occurs during the anti-islet alloimmune and, after a temporary increase, autoimmune responses.

TIM4 expression on APCs was evaluated by flow cytometry during the allo- (A, B) and autoimmune (C, D) anti-islet response in vivo. A decrease in the percentage of B-cells positive for TIM4 (n = 3, **p <0.01 vs. D0; A) and DCs positive for TIM4 (n = 3, **p <0.01 vs. D0; B) was evident after fully mismatched islet transplantation of BALB/c islets into C57BL/6 recipients. In diabetes prevention studies, a reduction in the percentage of B-cells (n = 3, **p <0.01 vs. 10 weeks; C) and DCs (n = 3, *p<0.05 vs. 10 weeks; D) positive for TIM4 was observed in hyperglycemic mice.
Targeting TIM4 In Vitro: RMT4-53 Reduces IFN-γ Production in the Allo- but Not in the Autoimmune Setting
To investigate the effect of TIM4 targeting in islet graft rejection and in the onset of autoimmune diabetes, we first tested the effect of RMT4-53 (an anti-TIM4 mAb) during polyclonal or islet peptide-specific stimulation. Splenocytes obtained from C57BL/6 mice were stimulated with anti-CD3/-CD28 Ig in the presence of RMT4-53 (1, 10, or 100 μg/ml) for 24 h. ELISPOT analysis revealed that 100 μg/ml RMT4-53 reduced the number of IFN-γ-producing cells (Th1) (number of IFN-γ-producing spots: RMT4-53 treated= 111 ± 15, untreated= 349 ± 16; n = 3; p = 0.009; data not shown) and skews the immune response toward a Th2 profile (IL-4/TFN-γ ratio: RMT4-53 treated = 0.7 ±0.2, untreated=0.2 ±0.03; n = 3; p = 0.02; data not shown). In autoimmune-relevant assays (BDC2.5 and IGRP stimulation for 24 h), the use of RMT4-53 did not significantly affect the number of IFN-γ- or IL-4-producing cells nor did it change the IL-4/IFN-γ ratio. TIM4 targeting in vitro with RMT4-53 therefore reduces the Th1 and favors the Th2 response during polyclonal but not islet peptide-mediated T-cell stimulation.
Targeting TIM4 In Vivo in a Model of the Th1-Mediated Alloimmune Anti-Islet Response: RMT4-53 Promotes a Th2 over a Th1 Response and Prolongs Islet Graft Survival
We tested the effect of TIM4 targeting in vivo in a model of the Th1-mediated alloimmune anti-islet response. BALB/c islets were transplanted into hyperglycemic C57BL/6 mice, and graft survival was evaluated in untreated and RMT4-53-treated mice (500 μg IP day 0 and 250 μg IP at days 2, 4, 6, 8, and 10). A significant prolongation of graft survival was observed in treated mice compared to untreated mice [mean survival time (MST), days: RMT4-53 treated= 19, untreated= 13; n =10; p = 0.01 (Fig. 3A)]. Flow cytometric analysis of splenocytes extracted from treated and untreated mice at 14 days after transplantation revealed a reduced percentage of B-cells positive for TIM4 (Fig. 3B) and DCs positive for TIM4 (Fig. 3C) in RMT4-53-treated mice. The reduction in TIM4+ B-cells and DCs was not evident when transplanted mice were treated with anti-TIM1 antibodies. We hypothesize that RMT4-53 may have a depleting effect on TIM4+ cells during transplantation. We then phenotypically and functionally analyzed the immune system profile of treated and untreated mice. ELISPOT analysis of recipient splenocytes challenged with donor antigens revealed no significant differences in terms of number of IFN-γ-producing cells between RMT4-53-treated and untreated mice (Fig. 3D), while on the contrary, an increase in IL-4-producing cells was observed in RMT4-53-treated mice (Fig. 3E). The Th2/Th1 ratio (IL-4-/TFN-γ-producing cells) increased significantly in RMT4-53-treated mice compared to controls (Fig. 3F). We did not observe any significant differences in the percentages of CD4+IL-17+ T-cells and CD4 T effector cells (CD4+CD44highCD62Llow) between RMT4-53-treated and untreated mice (Fig. 3G, H, respectively), while a slight reduction in Tregs (CD4+CD25+FoxP3+) was observed in the RMT4-53-treated group (Fig. 3I). TIM4 targeting in vivo, in a model of the Th1-mediated anti-islet response, promotes islet graft survival and is associated with a Th1 to Th2 skewing of the immune response.
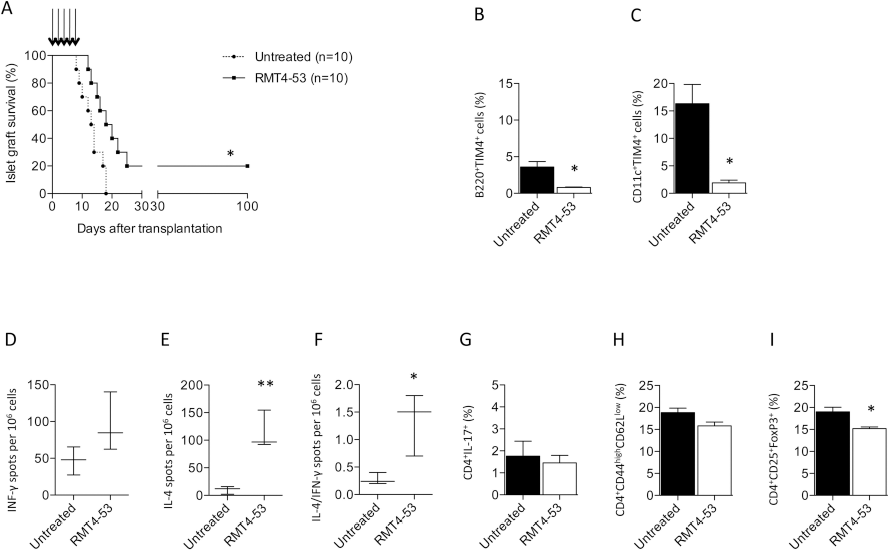
The effect of TIM4 targeting with RMT4-53 was evaluated during the alloimmune anti-islet response; RMT4-53 treatment induced a prolongation of islet graft survival compared to untreated recipients (n = 10, *p <0.05 vs. untreated; A). A decrease in the percentage of B-cells (n = 3, *p <0.0 vs. untreated; B) and DCs (n = 3, *p <0.05 vs. untreated; C) positive for TIM4 was observed 14 days after transplantation in the splenocytes obtained from RMT4-53-treated compared to untreated mice. In ELISPOT assays, an increase in the number of anti-donor-specific IL-4- (n = 3, **p <0.01 vs. untreated; E), but not IFN-γ-producing cells (D), was observed in splenocytes obtained from RMT4-53-treated compared to untreated recipients. The Th2/Th1 ratio shows a significant Th2 deviation in RMT4-53-treated mice (n = 3, *p <0.05 vs. untreated; F). The percentage of Th17 (G) and CD4+ T effector cells (H) was similar in RMT4-53-treated and untreated mice, while a reduction in the percentage of Tregs was observed in RMT4-53-treated compared to untreated mice (n = 3, *p <0.05 vs. untreated; I).
Targeting TIM4 In Vivo in a Model of the Th2-Mediated Alloimmune Anti-Islet Response: RMT4-53 Exacerbates the Th2 Response and Precipitates Islet Graft Rejection
We then analyzed the effect of targeting TIM4 with RMT4-53 in a model of the Th2-mediated alloimmune anti-islet response: BALB/c islets were transplanted into streptozotocin-treated Tbet-/- C57BL/6 mice, which are characterized by high Th2 (and Th17) and virtually absent Th1 responses (31). Delayed islet graft rejection was observed in Tber-/- mice compared to wild-type (WT) recipients, with 40% of mice displaying long-term islet graft function [MST: Tber/- untreated = 19 days; n = 10; p = 0.008 vs. WT untreated (Fig. 4A)] and preserved islet morphology (Fig. 4B). Splenocytes from Tbet-/- or WT mice were extracted at 14 days after transplantation and were stimulated with donor-derived antigens in an ELISPOT assay; an augmented Th2/Th1 ratio was observed in Tbet-/- compared to WT mice (Fig. 4C). We then analyzed the effect of RMT4-53 treatment in Tber/- recipients. RMT4-53 treatment accelerated islet rejection compared to untreated Tber-/- recipients [MST: Tber-/- RMT4-53 treated = 9 days, n = 7; p = 0.0002 vs. Tbet-/- untreated (Fig. 4A)]. In our ELISPOT assay, a further increase in the Th2 anti-islet response was observed (Fig. 4C). TIM4 targeting in a model of the Th2-mediated alloimmune anti-islet response thus precipitates graft rejection and further enhances the Th2 response.

The effect of TIM4 targeting with RMT4-53 was tested in a model of the Th2 (and Th17) immune response (BALB/c islets into Tbet-/- C57BL/6 mice). A prolongation of islet allograft survival was observed in Tbet-/- C57BL/6 compared to WT recipients (##p <0.01 vs. WT untreated; A). RMT4-53 treatment in Tbet-/- mice induced an acceleration of islet allograft rejection (Tbet-/- RMT4-53 treated ***p <0.001 vs. Tbet-/- untreated; A). Pathology of the graft revealed that islets were still preserved at 100 days after transplantation in normoglycemic untreated Tbet-/- C57BL/6 mice (B) with only a mild infiltrate within the islet graft. An increased Th2/Th1 ratio was demonstrated in Tbet-/- recipients challenged with donor-derived splenocytes compared to WT recipients (n = 3, ***p <0.001 vs. WT untreated; C); a further increase was observed in RMT4-53-treated Tber-/- C57BL/6 compared to untreated Tber-/- C57BL/6 (n = 3, #p <0.05 vs. Tber-/- untreated; C). When BALB/c islets were transplanted into B-cell-depleted C57BL/6 mice, islet graft survival was prolonged compared to untreated mice (##p <0.01 vs. untreated; D); RMT4-53 treatment in B-cell depleted recipients promoted islet rejection (**p <0.01 vs. B-cell depleted; D). In B-cell-depleted mice 14 days after transplantation, graft histology still displayed partially preserved islet morphology (E). An increased Th2/Th1 ratio was observed in B-cell-depleted recipients compared to undepleted recipients, while no further Th2/Th1 increase was observed after RMT4-53 treatment (n = 3, *p <0.05 vs. untreated; F).
Targeting TIM4 In Vivo Does Not Promote the Th2-Mediated Anti-Islet Response in the Absence of B-Cells
We have previously demonstrated that the presence or absence of B-cells during allogeneic islet transplantation respectively favors a Th1 or Th2 anti-islet response (3,10). We thus evaluated whether the Th2 shift observed with RMT4-53 treatment was dependent upon B-cells. Depletion of B-cells was achieved with an anti-CD22/cal antibody, and graft survival and immune profiles were analyzed (3). B-cell-depleted mice experienced a slight delay in islet graft rejection [MST: B-cell depleted = 17 days, n = 6, p = 0.004 vs. untreated (Fig. 4D)] with islet grafts that remained mildly infiltrated at 14 days after transplantation (Fig. 4E); B-cell-depleted mice also displayed an increased Th2/Th1 ratio (Fig. 4F). When B-cell-depleted mice were treated with RMT4-53, no further enhancement of the Th2/Th1 ratio was obtained by RMT4-53 treatment (Fig. 4F) nor was any prolongation of graft rejection observed (on the contrary, an acceleration was evident) [MST: B-cell depleted RMT4-53 treated = 11 days, n = 3; p = 0.002 vs. B-cell depleted (Fig. 4D)]. These data demonstrate that the effect of anti-TIM4 treatment on Th2/Th1 ratio and islet graft protection is dependent upon the presence of B-cells.
TIM4+ B-Cells Are Characterized by a Low Costimulatory Molecule Expression Profile and Reduced IL-4 Production
We investigated if the redirection toward a Th2 response obtained by the blockade of TIM4+ B-cells may be justified by the specific differences between TIM4+ and TIM4- B-cells. We first analyzed the costimulatory molecule profile in TIM4+ and TIM4- B-cells extracted from C57BL/6 splenocytes. Lower expression in CD40 (Fig. 5A), CD80 (Fig. 5B), CD86 (Fig. 5C), and CD69 (Fig. 5D) was observed in TIM4+ B-cells compared to TIM4- B-cells. Cytokine profiles are also different in TIM4+ and TIM4- B-cells. The percentage of IL-4+ B-cells is severely reduced in TIM4+ B-cells compared to TIM4- B-cells (Fig. 5F), while no difference in IFN-γ production was observed (Fig. 5E). The IL-4/IFN-γ ratio is thus consequently higher in the TIM4- B-cell population than in TIM4+ (Fig. 5G). These results indicate that TTM4- B-cells may be more functional than TIM4+ B-cells for Th2 differentiation. To further investigate the role of TIM4+ B-cells, we differentiated naive Th0 cells toward Th2 in an appropriate cytokine milieu (43) in the presence of B-cells and presence or absence of RMT4-53. T-cells were then harvested and assessed for level of differentiation by the analysis of the Th2-specific marker GATA3. RT-PCR analysis showed higher GATA3 expression levels when RMT4-53 was added to the differentiation media (Fig. 5H). These data indicate a better Th2 differentiation potential for TIM4- over TIM4+ B-cells.

The different phenotype of TIM4+ and TIM4- B-cells was evaluated by flow cytometry. Reduced CD40 (A), CD80 (B), CD86 (C), and CD69 (D) expression was observed in TIM4+ B-cells. (n = 3, *p <0.05, and ***p <0.001). No difference in term of IFN-γ positivity was observed (E), while the IL-4 and IL-4/IFN-γ ratio is highly reduced in TIM4+ B-cells (n = 3, *p <0.05; **p <0.01; F, G). In an in vitro assay, the Th2 generation, assessed by RT-PCR as increase in GATA3 expression, was more efficient when Th0 were cultured in the presence of B-cells and RMT4-53 (n = 3, *p <0.05 and **p <0.01 vs. Th0; H).
Targeting TIM4 In Vivo in a Model of the Autoimmune Anti-Islet Response: RMT4-53 Does Not Promote the Th2 Response or Delay Diabetes Onset
The efficacy of TIM4 targeting was then tested in the prevention of diabetes in NOD mice. RMT4-53 treatment (500 μg IP at day 0 and 250 μg IP at days 2, 4, 6, 8, and 10 beginning at 10 weeks of age) was ineffective in delaying diabetes onset (Fig. 6A). An increased percentage of TIM4-expressing B-cells (Fig. 6B), but not of TIM4-expressing DCs (Fig. 6C), was observed in the spleens of RMT4-53-treated mice compared to untreated mice at 14 weeks of age. Analysis using an ELISPOT assay revealed a higher number of both IFN-γ- and IL-4-producing cells in splenocytes obtained from RMT4-53-treated mice challenged with BDC2.5 peptide (Fig. 6D, E) and IGRP peptide (Fig. 6D, E). An increased Th2/Th1 ratio in RMT4-53-treated mice was observed in response to IGRP, but not to BDC2.5 peptide (Fig. 6F). No significant differences were noted with regard to CD4+ IL17+ T-cells or Tregs between RMT4-53-treated and untreated mice (Fig. 6G, I), while a slight reduction in CD4 T effector cells was observed in RMT4-53-treated mice (Fig. 6H). In the autoimmune diabetes model, targeting TIM4 did not appear to prevent or delay diabetes onset or to induce a clear Th2 skewing of the immune response.

RMT4-53 treatment was evaluated in diabetes prevention in NOD mice. No effect was observed on the onset of diabetes after RMT4-53 treatment (A). An increase in the percentage of B-cells (but not of DCs) positive for TIM4 cells was observed in RMT4-53-treated mice compared to untreated mice (n = 3, *p <0.05; B, C). An increase in the number of IFN-γ-producing cells was observed in RMT4-53-treated mice during BDC2.5 (n = 5, *p <0.05 vs. untreated; D) and IGRP challenge (n = 5, *p <0.05 vs. untreated; D) compared to untreated mice. An increase in the number of IL-4-producing cells was observed as well (BDC2.5 peptide: n = 5, *p <0.05 vs. untreated; IGRP: n = 5, *p <0.05 vs. untreated; E). The ratio of Th2/Th1 shows a significant skewing toward a Th2 response only with IGRP peptide challenge (n = 5, *p <0.05 vs. untreated; F). No effect was observed on the percentage of Th17 cells (G), while a slight decrease in the percentage of T effector cells in RMT4-53-treated compared to untreated mice was observed (n = 3, *p <0.05 vs. untreated; H). The percentage of Tregs was unchanged (I).
The TIM4+ B-Cell Population Is Increased in T1D and Islet-Transplanted Patients
We then sought to examine the TIM4 expression profile by flow cytometry in B-cells obtained from islet-transplanted patients and from patients with T1D to determine the relevance of TIM4 in humans (Table 1. In healthy controls, TIM4+CD19+ B-cells represented approximately 6% of PBMCs (Fig. 7A, E) and 54% of all B-cells. An increase in the percentage of TIM4+ B-cells was seen in T1D patients (Fig. 7B, E); a less pronounced increase was also observed in islet-transplanted patients (Fig. 7C, E). The percentage of TIM4+ cells was gated according to isotype control staining (Fig. 7D).

TIM4 expression in B-cells obtained from T1D patients and islet-transplanted patients was evaluated using flow cytometry. An increase of TIM4+ B-cells was observed in T1D patients (n = 6; *p <0.05 vs. healthy volunteers; B, E) and in islet-transplanted patients (n = 10; *p <0.05 vs. healthy volunteers; ##p <0.01 vs. T1D; C, E) compared to healthy controls (A). TIM4+ staining was determined according to isotype control staining (D).
Discussion
The finely tuned interactions between costimulatory molecules on APCs and T-cells determine the activation or the suppression of allo- and autoimmune responses (11,24). The targeting of costimulatory pathways represents an emerging therapeutic opportunity for the prevention of graft rejection and development of autoimmunity (18). The most well-studied and well-characterized pathways thus far are the CD28/B7.1-2 and the CD40L/CD40 costimulatory pathways, and the targeting of these has been shown to be efficient in preventing both graft rejection and autoimmunity in several animal models (41) and in clinical settings (27,44). More recently, additional costimulatory pathways have been described (e.g., OX40/OX40L and the ICOS/B7h) (2). In our work, we evaluated the possible efficacy of TIM1-TIM4 pathway targeting, using the monoclonal anti-TIM4-Ig RMT4-53 antibody, in preventing anti-islet allo- and autoimmunity.
TIM4 is expressed on B-cells and DCs and appears to be downregulated following alloimmune and autoimmune anti-islet responses. The downregulation of TIM4 seems to be a common pattern after B-cell activation and was also evident in cells cultured with LPS or anti-CD40-Ig. The use of RMT4-53 (a mAb targeting TIM4) alleviates the anti-islet alloimmune response in C57BL/6 mice and delayed islet graft rejection; the effect seems to be associated with a skewing toward a Th2 immune profile. On the contrary, treatment with RMT4-53 was not shown to prevent diabetes onset in the NOD autoimmune model.
The redirection of the immune system toward a Th2 immune profile appears to be mediated by B-cells, as no increase in the Th2 response was observed after B-cell depletion in RMT4-53-treated or untreated recipients. We have previously demonstrated that the presence of B-cells is fundamental for directing the anti-islet alloimmune response toward a Th1 profile (3). Our data here demonstrate (i) B-cell depletion causes Th2 deviation of the anti-islet response, (ii) RMT4-53 treatment causes a similar deviation of the anti-islet response, (iii) no synergistic or additive effect is observed on the Th2 response when combining the two treatments; these observations are compatible with an operative model in which the Th2 anti-islet alloimmune response is kept under control by a population of TIM4+ B-cells, and the function of these cells is suppressed by either anti-TIM4-Ig or B-cell depletion (Fig. 8). Compared to TIM4- B-cells, in fact, TIM4+ B-cells appear less efficient in priming the Th2 response because of a highly reduced expression of IL-4, which is fundamental for Th2 response development. Consistently with this model, the reduction of TIM4 expression on B-cells after anti-TIM4 treatment in the alloimmune, but not the autoimmune, setting can also contribute to the more evident redirection toward a Th2 profile observed in the alloimmune setting. The relative contribution in this model of the blockade of the phagocytosis of apoptotic bodies that could be obtained by RMT4-53 needs to be further investigated. However, it is likely that TIM4 has a role in the redirection of the immune system, which is separate and independent by its role as a phagocytic receptor. In fact, we do not observe a Th2 redirection of the immune response with RMT4-53 after the depletion of B-cells, which have little function in the scavenging of apoptotic bodies. Our data also clarify the role of the Th2 response in anti-islet rejection. The switching from a Th1 to Th2 response in a model of the Th1-mediated anti-islet response functions in the delay of islet graft rejection, possibly due to a reduced rejection capacity of the Th2 response. Interestingly, the rejection kinetics observed after TIM4 treatment in the Th1 model is similar to that which is observed in the genetic model of Th2 islet rejection, confirming the mechanism of action of RMT4-53. Conversely, in a model in which the Th2 response is already prominent, further stimulation of the Th2 response by RMT4-53 treatment precipitates graft rejection, confirming the potential capacity of Th2 cells in mediating islet rejection.

Model of the role of TIM4+ B-cells in the alloimmune response in islet transplantation. TIM4 B-cells in normal conditions inhibits the Th2 immune response (A). TIM4 targeting with RMT4-53 enhances the Th2 response (B). In the absence of a Th1 response (as in Tbet-/- mice), TIM4 still exerts a Th2 inhibitory effect (C), and TIM4 targeting with RMT4-53 enhances the Th2 response (D). In the absence of B-cells, an increased Th2 response is evident due to the lack of TIM4 inhibition (E), and TIM4 targeting with RMT4-53 does not further increase Th2 response (F).
The increase in TIM4+ B-cells observed in islet-transplanted patients and T1D patients compared to healthy controls could contribute, according to our model, to the decreased Th2/Th1 ratio demonstrated in these patients (32). Consistent with the murine model of islet transplantation, a reduction in TIM4 expression on B-cells is observed in T1D patients after transplantation.
In conclusion, our work demonstrates a significant role for TIM4 and TIM4 targeting in the anti-islet alloimmune response, while the role of TIM4 or its blockade in the autoimmune response appears to be marginal. TIM4 inhibition may favor the Th2 over the Th1 response, and, although in the context of islet transplantation a high Th2 response can still be deleterious for the graft, given its reduced anti-graft-rejecting capacity, we hypothesize that anti-TIM4 treatment could serve as a component of a combination therapy to promote islet graft survival.
Footnotes
Acknowledgment
Paolo Fiorina is the recipient of a JDRF Career Development Award, an ASN Career Development Award, and an ADA Mentor-based Fellowship grant. P.F. is also supported by a Translational Research Program (TRP) grant from Boston Children's Hospital; Harvard Stem Cell Institute grant (“Diabetes Program” DP-0123-12-00); Italian Ministry of Health grant RF-2010-2303119. P.F. and Andrea Vergani are supported by an Italian Ministry of Health grant: (“Staminali” RF-FSR-2008-1213704). A.V. has been supported by an NIH-Research Training grant to Boston Children's Hospital in Pediatric Nephrology (T32DK007726-28). A.V. is supported by the “AMD-SID Pasquale di Coste Scolarship.” Francesca D'Addio is a recipient of Italian Scientists and Scholars of North America Foundation (ISSNAF)-Fondazione Marche Fellowship. Roberto Bassi is supported by an ADA Mentor-based Fellowship grant to P.F. and by an AST Genentech Clinical Science Fellowship Laurence A. Turka is funded by R01 AI-037691. A.V. conducted this study as partial fulfillment of his Ph.D. in molecular medicine, San Raffaele University, Milan, Italy. The authors declare no conflicts of interest.
