Abstract
Transplantation of islet allografts into type 1 diabetic recipients usually requires multiple pancreas donors to achieve insulin independence. This adds to the challenges of immunological monitoring of islet transplantation currently relying on surrogate immune markers in peripheral blood. We investigated donor origin and infiltration of islets transplanted in the liver of a T1D patient who died of hemorrhagic stroke 4 months after successful transplantation with two intraportal islet grafts combining six donors. Immunohistological staining for donor HLA using a unique panel of human monoclonal HLA-specific alloantibodies was performed on liver cryosections after validation on cryopreserved kidney, liver, and pancreas and compared with auto- and alloreactive T-cell immunity in peripheral blood. HLA-specific staining intensity and signal-to-noise ratio varied between tissues from very strong on kidney glomeruli, less in liver, kidney tubuli, and endocrine pancreas to least in exocrine pancreas, complicating the staining of inflamed islets in an HLA-disparate liver. Nonetheless, five islets from different liver lobes could be attributed to donors 1, 2, and 5 by staining patterns with multiple HLA types. All islets showed infiltration with CD8+ cytotoxic T cells that was mirrored by progressive alloreactive responses in peripheral blood mononuclear cells (PBMCs) to donors 1, 2, and 5 after transplantation. Stably low rates of peripheral islet autoreactive T-cell responses after islet infusion fit with a complete HLA mismatch between grafts and recipient and exclude the possibility that the islet-infiltrating CD8 T cells were autoreactive. HLA-specific immunohistochemistry can identify donor origin in situ and differentiate graft dysfunction and immunological destruction.
Introducton
Islet transplantation is an effective treatment for brittle type 1 diabetes, and it allows most patients to achieve insulin independence. Transplanted β-cell mass is an important determinant of transplantation success. Single-donor transplantation is preferred, but islets from multiple donor organs and repeated transplantations are often required to achieve optimal function 1 . Although multidonor transplantation has improved transplantation outcome, it complicates understanding of improvements in isolation, transplantation, and immunosuppressive strategies. Identifying the fate of individual donor grafts is necessary to interpret changes in outcome with novel transplantation strategies. We previously reported on donor-specific alloreactive responses and recurrent autoimmunity in multidonor islet transplants by investigating circulating immune cells2–5. However, it remains to be determined how immunity measured in peripheral blood relates to local immunity in islet transplantation.
Opportunities to investigate transplanted islets in situ are rare. Percutaneous techniques have reduced side effects of islet transplantation, while investigating an intraportal graft by transcutaneous liver biopsy has proven infeasible 6 . Risk of complications precludes repeated liver biopsies or surgical major biopsies to access transplanted islets. Therefore, in situ studies can only be performed postmortem or on incidental patients who would require liver surgery. Identification of islet material in situ is necessary to investigate donor-specific effects. Donor and recipient human leukocyte antigen (HLA) typing are usually known and differ in unmatched cases. We previously established a bank of human HLA-specific monoclonal antibodies (mAbs) to study humoral rejection in transplantation 7 . The ubiquitous expression of HLA class I would allow for employment of these antibodies to differentiate between recipient and individual donors by immunohistochemistry.
We investigated islet donor origin in the case of a 61-year-old woman treated with islet transplantation for her brittle type 1 diabetes, who died of cerebral hemorrhage 4 months after receiving two intraportal islet grafts. Immunosuppression consisted of anti-thymoglobulin and methylprednisolone induction therapy and tacrolimus and mycophenolate mofetil maintenance therapy. She received islets from four donors in the first transplantation and from two donors in a second transplantation after 6 weeks. All donors had complete HLA-A, -B, and -DR mismatch with the recipient. At time of death she had a functioning graft with nonfasting C-peptide of 2.02 ng/ml at 220 mg/dl glycemia while using 13 units of insulin per day. Auto- and alloreactive immune responses of T cells and antibodies were monitored per protocol before and after transplantation.
Materials and Methods
Samples and Tissues
Blood samples were collected in sodium heparin tubes and serum tubes (BD Vacutainer, Breda, The Netherlands) containing silicate granulate for immune monitoring before and at 4, 6, 9, and 12 weeks after transplantation with signed informed consent of the patient and according to the approved protocol 2 . Autopsies and studies on organ specimens were performed after obtaining oral informed consent from the patient's family. For antibody optimization, cryopreserved kidney, liver, and pancreas tissue was obtained from leftover specimen selected to match allo-antibody HLA specificity. All materials were treated according to local and institutional regulations with approval from the Medical Ethical Committee of the Free University Brussels, Belgium, and in accordance with the 2008 revised principles of the Declaration of Helsinki.
Peripheral Blood Immune Analysis
Autoimmune responses were determined blinded from clinical results. Lymphocyte stimulation tests were performed before and at regular intervals after transplantation and on lymphocytes derived from different organs upon autopsy, as described before 8 . Briefly, 150,000 fresh peripheral blood mononuclear cells (PBMCs) per well or tissue-derived lymphocytes per well were cultured in 96-well round-bottom plates in Iscove's modified Dulbecco's medium (IMDM) with 2 mmol/L glutamine (Gibco, Paisley, Scotland) and 10% pooled human serum in the presence of antigen, interleukin-2 (IL-2; 35 U/ml; Novartis, Arnhem, The Netherlands), or medium alone in triplicates. After 5 days [3H]thymidine (0.5 μCi per well; DuPont NEN, Boston, MA, USA) was added, and [3H]thymidine incorporation was measured after 16 h on a betaplate counter. Antigens analyzed included islet autoantibody-2 (IA-2; 10 μg/ml), glutamate decarboxylase-65 (GAD65; 10 μg/ml), insulin (25 μg/ml), and tetanus toxoid (“recall” antigen; 1.5 LF/ml). Results were interpreted as stimulation index (SI) compared to medium value and with a cutoff value of SI >3.
Alloreactive T-cell responses were determined by cytotoxic T-lymphocyte precursor (CTLp) assay and mixed lymphocyte reaction (MLR). The CTLp assay to determine cytotoxic T-cell alloreactivity was described previously 9 . Briefly, PBMCs were cultured in a limiting dilution assay (40,000 to 625 cells/well, 24 wells per concentration) with different irradiated stimulator PBMCs expressing HLA class I antigens matching β-cell grafts (50,000 cells/well). Cells were cultured for 7 days at 37°C in 96-well round-bottom plates in RPMI-1640 medium (Gibco BRL, Paisely, UK) with 3 mmol/L L-glutamine, 20 U/ml IL-2, and 10% pooled human serum. Then Europium-labeled (Fluka, Buchs, Switzerland) graft HLA-specific target cells (5,000 cells/well) were added for 4 h. Wells were scored positive if the Europium release through target cell lysis exceeded spontaneous release +3× standard deviation. Quantification of CTLp frequencies was performed by computer software 10 . In parallel, one-way mixed lymphocyte cultures were set up in triplicates in 96-well V-bottom plates (Costar, Cambridge, MA, USA) in 150 μl RPMI-1640 with 2 mmol/L L-glutamine (Gibco) and 10% pooled human serum. Responder cells (40,000) were incubated with 50,000 irradiated (3,000 rad) stimulator cells per well at 37°C/5% CO2. After 5 days, [3H]thymidine (1.0 μCi per well) was added, and [3H]thymidine incorporation was measured on a betaplate counter (Wallac-LKB Betaplate 1205; Wallac, Turku, Finland) after 16 h. Proliferation in response to phytohemagglutinin was used as positive control. Results were interpreted as SI compared to background value (responder only + stimulator only). Production of different cytokines was measured with Luminex technology using a human Th1/Th2 Bioplex cytokine kit (Bio-Rad, Veenendaal, The Netherlands), including IL-2, IL-4, IL-5, IL-10, IL-12p70, IL-13, granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon-γ (IFN-γ), and tumor necrosis factor (TNF), according to the manufacturer's protocol.
Screening for the presence of HLA class I- and class II-specific antibodies was performed on all available samples by enzyme-linked immunosorbent assay (ELISA; LAT class Iⅈ One Lambda, Canoga Park, CA, USA). Islet cell autoantibodies (ICA), autoantibodies against IA-2 protein (IA-2A), and GAD were measured as described previously 11 . Briefly, ICA were determined by indirect immunofluorescence. IA-2A and GAD were determined by liquid phase radiobinding assays.
HLA-Specific Antibody Staining
Islet Donor Identification by HLA Antibodies

inconclusive; D, donor; LL, left liver lobe; LR, right liver lobe.
Donor and HLA Antibody Staining Combinations
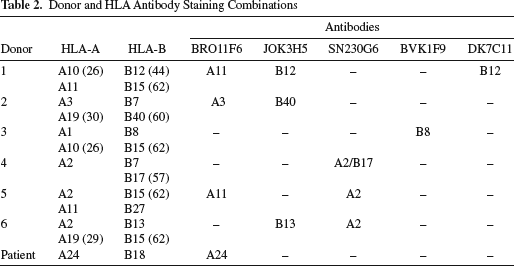
Results
Immune Response Analysis
Immune responses were determined in peripheral blood before and after transplantation. Autoreactive T-cell responses were tested by lymphocyte stimulation test to autoantigens and showed response to GAD65 (SI 13.2) before transplantation, but very low autoantigen-specific responses were shown after transplantation. Insulin auto-antibodies were present before transplantation and remained unchanged thereafter. Alloreactive cytotoxic T-cell frequency was reduced in the first 6 weeks after transplantation. After 8 weeks, responses increased to HLA-A11 (donors 1 and 5) and/or HLA-B62 (donors 1, 3, 5, and 6). By week 12, additional responses emerged to donor 1 (HLA-B44) and donors 2, 3, and/or 4 (HLA-A1, -A3, -A26, -B7, -B8, and/or HLA-B60) (Fig. 1). Alloreactive T-helper cell responses in MLR decreased after transplantation correlating with increased IL-10 levels and reduced IFN-γ/IL-10 ratio (Fig. 1). No alloreactive antibodies were measured before or after treatment (Tx).
Alloreactive T-cell responses around transplantation. Immune suppression, graft function, and alloreactive T-cell responses monitored before and after transplantation. (A) Timing of immune suppression: thymoglobulin (horizontal bar), methylprednisolone pulses (X), mycophenolate mofetil daily dose (dashed line), and tacrolimus trough levels (squares). (B) Random C-peptide measurements. (C) Alloreactive cytotoxic T-cell precursor (CTLp) frequency to six human leukocyte antigen (HLA) mismatched stimulator target combinations was measured. Reactivity 8 weeks after transplantation (filled triangle and square) suggests reactivity to HLA-A11 (donors 1 and 5) and/or B62 (donors 1, 3, 5, and 6). After 12 weeks, reactivity to stimulators depicted with open circles and diamonds suggests HLA-A3 or -B7 (both donors 2 and 4) reactivity, although A1, A26, B8, and/or B60 (donors 1, 2, and/or 3) is also possible. Further, changing reactivity to filled symbols suggests upcoming HLA-B44 reactivity matching donor 1. (D) Alloreactive T-helper proliferation was suppressed after transplantation. (E) Mixed lymphocyte reaction (MLR) cytokine response showed increasing interleukin-10 (IL-10) production, but stable interferon-γ (IFN-γ) response leading to sharp decrease in IFN-γ/IL-10 ratio. Symbols represent matched stimulators. Tx2, second transplant; TAC, tacrolimus; MMF, mycophenolate mofetil; PBMC, peripheral blood mononuclear cells; SI, stimulation index.
Optimization of HLA-Specific Antibody Staining for Donor Determination
HLA-Specific Antibody Staining on Validation Tissues

Data presented as “specific stain”/“background nonspecific staining.” nd, no data. 0, no detectable staining; +, detectable staining; ++, intermediate staining; ++(+), good to strong staining; +++, strong staining.
Postmortem Analysis
Postmortem, islet cell clusters were identified in the right and left liver lobes with varying mononuclear infiltrates. Consecutive sections of four cryopreserved blocks scattered throughout the liver were used for in-depth analysis. First two to four sections of each series were used for negative control staining, leaving out individual primary antibodies and revealing low (AF488) and negligible (Cy3 and AF647) background fluorescence by secondary antibodies. Five consecutive sections were stained for specific HLA and insulin. From the left liver lobe, one of two assessed blocks revealed a single insulin+ islet (Fig. 2 and Table 1, islet 1). An additional isletlike area in slides used to optimize staining has been left out of further analysis since endocrine origin could not be confirmed by insulin or glucagon staining. Sections from the right lobe revealed an area with five islet cell clusters of which one cluster of only six cells was identified in a single section and could not be analyzed in detail (Table 1, islets 2–5). Thus, in total five pseudoislets were revealed and assessed.
Identification of islet origin. Identification of donor 5 as source of islet 1 by staining with HLA-specific antibodies. Contrast-optimized pictures showing multifluorescent staining of consecutive slides of islet 1 to insulin (green) and different HLA specificities (red). 4′,6-Diamidino-2-phenylindole (DAPI) is shown in blue. Donors expressing the antibodies HLA specificity are listed below antibody clone names. Colors are pseudocolors. MoAb, monoclonal antibody.
Donor Characteristics

Relative contribution of each donor to the transplants is calculated from β-cell counting 2–6 days before transplantation and predicted cell loss between β-cell counting and transplantation. Viability was >90% on the day of transplantation in all preparations; combined Transplant 1 (Tx1, donors 1 to 4) contained 863 million cells with 27% β-cells; combined Tx2 (donors 5 and 6) contained 282 million cells with 26% β-cells. F, female; M, male; CMV, cytomegalovirus.
Islet Composition and Infiltration
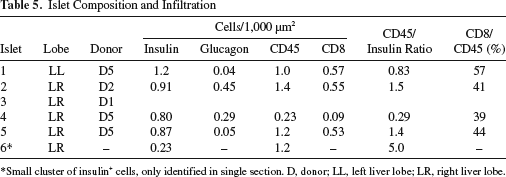
Small cluster of insulin+ cells, only identified in single section. D, donor; LL, left liver lobe; LR, right liver lobe.
Infiltrate by CD45+ cells was observed in all islet cell clusters and CD8+ cells could account for 39%–57% of these infiltrates (Fig. 3 and Table 5). Most infiltrating cells surrounded the insulin+ cells, while insulin+ cells seemed to have disappeared around immune cells infiltrating the islets. Occasionally, CD45+ or CD8+ cells were in direct contact with insulin+ cells. Although the rate of infiltration varied between islet clusters, there was no apparent relation to donor origin.
α-Cells and islet-infiltrating lymphocytes. Identification of α-cells and islet infiltrate around islet 1. Slides adjacent to those used for HLA staining were stained for glucagon (yellow), CD45 (red), and CD8 (red). Pictures are contrast optimized and show pseudocolors. CTLs, cytotoxic T-lymphocytes; Ins, insulin.
Discussion
We investigated the origin of identified islets in the liver of a patient with type 1 diabetes, who died 4 months after receiving islet transplants. We established HLA-specific immunohistochemical staining on tissues of various organs observing tissue-specific variation in HLA expression and background fluorescence. Nonetheless, several transplanted islets were identified in the patient's liver to originate from donors 1, 2, and 5. These islets were infiltrated with leukocytes including many cytotoxic T cells, which matches increasing frequency of CTLs specific to these donors measured in peripheral blood after transplantation.
We set up an immunohistochemical staining using HLA class I to evaluate donor origin of cells. Hereby, a broad range of chimeric combinations can be evaluated including transplants from multiple donors, as presented in this article. Previously, islets from two donors were identified in a patient who died with longstanding graft function using laser dissection of islets retrieving DNA for HLA class II-based polymerase chain reaction (PCR) 15 . With our current method, we investigated a more complex case receiving islets from six donors. Advantages of our method are that individual cells can be studied in combination with other markers and, despite the complexity, fewer sections of an islet are required for identification. The major limitation of our method is restriction to cryopreserved material likely to be related to the superior preservation of the tertiary and quaternary structure of HLA molecules recognized by the mAbs.
Significant variation for the HLA staining was noted when optimizing HLA-specific staining in different organs. This was especially relevant since HLA expression was studied on transplanted pancreatic endocrine tissue with relatively low HLA expression in liver tissue with relatively high autofluorescence, especially in the portal triad. Signal strength was optimized using secondary and tertiary staining to directly labeled human antibodies. This proved to be the most effective strategy to enhance staining with human antibodies in biotin-rich human tissue. The exceptional combination of tissues did not allow optimal positive control slides; therefore, only negative control slides were taken along for secondary and tertiary staining. To improve judgment, identified islets were classified by three independent observers.
To our knowledge, this is the first time human alloreactive antibodies have shown to bind β-cells. Staining intensity was not particularly strong compared to kidney or liver tissue, but was stronger than exocrine pancreatic tissue in donor organ slides. Further, HLA staining in the identified transplanted islets varied between individual cells, which may result from inflammation. In inflamed islets of type 1 diabetes patients, hyperexpression of HLA has been described previously16,17. The limited number of identified islets did not allow comparison of HLA staining strength to lymphocytic infiltration in this patient. Nonetheless, our findings demonstrate that alloreactive human antibodies can bind β-cells and may contribute to graft demise through antibody-dependent cellular cytotoxicity or complement-dependent cytotoxicity 18 .
We were able to identify donor origin of most islets identified in the cryopreserved patient's liver sections. Identified islets came from both left and right liver lobes. Insulin+ cells of three out of six donors were traced in the liver, suggesting contribution of multiple donors as well as first and second transplants to graft function. Although identified islets were predominantly of donor 5, the number of identified islets is insufficient to prove selective donor survival.
All identified islets were surrounded by considerable numbers of immune cells including numerous cytotoxic CD8 T cells. Identification of T-cell specificity was not possible, since this requires prior knowledge of the HLA–peptide combination involved 17 . The inverse pattern of leukocytes and insulin+ cells suggests these leukocytes may have destroyed parts of the graft. This would fit with rising donor-specific CTLs in peripheral blood after transplantation. The partial destruction of the grafts may imply that these CTLs are hampered in their efficiency. This may be through the immune suppressive therapy, on which we previously reported upcoming alloreactive CTL frequency after transplantation without correlation to graft failure 2 . We further reported that tapering the immune suppression was accompanied by increasing avidity CTLs and loss of graft function 19 . Also, the infiltrating cells may include alloreactive CD8 Tregs, which were previously identified to protect islets in an explanted pancreas graft 20 .
Autoimmunity was not observed in peripheral blood of our patient after transplantation. Probably recurrent auto-immunity has been prevented or delayed by HLA class I mismatch between the donors and the recipient, which precludes direct recognition of islet autoantigens on the allograft. Indeed, this is accordant with the observation that all identified islets contained β-cells.
In conclusion, we established histological identification of donor islet origin with human HLA-specific antibodies that specifically target transplanted human β-cells. This method was employed to investigate a case with multidonor islet transplantation to show identity and fate of multiple donor islets. We validated this method for multiple organs, suggesting it may also be effective to study other settings with multidonor chimerism.
Footnotes
Acknowledgments
The authors thank Dr. John Scharenberg of Sanquin Blood Supply for Alexa Fluor 488 labeling of human mAbs. This work was supported by the European Commission (BetaCellTherapy, No. 241883 in the FP7 program). The authors declare no conflicts of interest.
