Abstract
We have previously demonstrated that bilirubin administration to the recipient induces tolerance towards islet cell transplants across a complete MHC mismatch in a mouse model. Here we assess the mechanisms of such protection. Bilirubin treatment of recipients improved function of islet allografts by suppressing expressions of proinflammatory and proapoptotic genes in those islets and by increasing Foxp3+ T regulatory (Treg) cells at the site of transplanted islets at various days after transplantation. No prolongation of graft survival was observed in recipients treated with bilirubin when CD4+CD25+ T cells were predepleted from those recipients, indicating that Treg cells are necessary for the protective effect of bilirubin. Adoptive transfer of Treg cells from tolerant mice into Rag1-/- recipients resulted in long-term acceptance of skin allografts in an alloantigen-specific manner, suggesting that Treg cells are sufficient to induce tolerance. In addition, bilirubin treatment promoted de novo generation of Treg cells in Rag1-/- recipients. Thus, bilirubin treatment to the recipients prolongs islet allograft survival via a Treg-dependent manner in which CD4+CD25+ Treg cells are both necessary and sufficient for tolerance induction and graft acceptance. Bilirubin treatment promotes de novo generation of Treg cells that might account for the protective effects of bilirubin given to recipients.
Introduction
Transplantation of islets is an important approach to the treatment of type 1 diabetes (16). However, immunosuppressive agents used after islet transplantation can cause many toxic side effects to the recipients (2). Our novel findings demonstrate that administering bilirubin to donor and/or recipient mice leads to long-term (>100 days) survival and antigen-specific tolerance to islet allografts in the recipients without any detectable toxicity (20, 39). Further studies indicate that donor treatment with bilirubin leads to a decreased inflammatory response in the islets after transplantation that may be responsible for the survival of those islets (39). What remain unclear are the mechanisms by which bilirubin treatment of the recipient manifests the protective effects. Thus, the focus of this study was to determine how treatment of the recipient with bilirubin induced protection against rejection and promoted long-term survival and tolerance of islet allografts following transplantation.
Heme oxygenase-1 (HO-1) is the rate-limiting enzyme that degrades heme to generate equal molar amounts of biliverdin, carbon monoxide (CO), and ferrous iron (29). Biliverdin can be rapidly converted into bilirubin, a potent antioxidant, by biliverdin reductase (1). In vitro, bilirubin scavenges peroxyl radicals as efficiently as α-tocopherol, the most potent antioxidant of lipid peroxidation (34). In vivo, bilirubin is the most abundant endogenous antioxidant in mammalian tissues, accounting for the majority of the antioxidant activity of human serum (24, 35). Bilirubin at concentrations found in normal human plasma protects albumin from oxidation (35). Bilirubin at nanomolar concentrations is neuroprotective against hydrogen peroxide in hippocampal and cortical neuronal cultures (7). Bilirubin biosynthesis can be induced by oxidative stress in the ischemia-reperfusion model of rat liver and acts as a physiological cytoprotective antioxidant (42).
Although other functions of bilirubin besides its extraordinary antioxidant effects are not well documented, there is some evidence indicating that bilirubin might play an important role in the immune system (28). In 1970, Najedla showed that bilirubin significantly suppresses antibody levels against diphtheria, tetanus, and pertussis in infants with hyperbilirubinemia, suggesting a role of bilirubin in the development of immune system (26). In addition, bilirubin was shown to be able to decrease IL-2 production in human lymphocytes (12) and influence Fc receptors in macrophages (38). Recent study suggests that bilirubin acts as a powerful immunomodulatory agent in an experimental autoimmune encephalomyelitis model by promoting apoptosis of reactive T cells and inhibiting activation of immune transcription factors and expression of inducible MHC class II (23).
We have shown that bilirubin treatment to the donor induces islet allograft survival and tolerance by suppressing expressions of proinflammatory cytokines, yet we do not know how bilirubin given to the recipient protects islet allografts from rejection after transplantation. We hypothesized that bilirubin exerts its protective effects via modulating Treg cells in our islet transplantation model. Namely, bilirubin administered to the recipient promotes function of Treg cells to maintain immunologic unresponsiveness and tolerance. Although little is known of the effect of bilirubin on T cells, studies have shown that HO-1 expression is indispensable for the development and function of CD4+CD25+ Treg cells (3, 4). Our own findings demonstrated that the tolerogenic effect of HO-1 plus donor-specific transfusion in a cardiac transplantation model was dependent on CD4+CD25+ Treg cells (20, 43). As a product of HO-1 enzyme activity, bilirubin in many situations mimics the effect of HO-1 and we anticipate that this will also be the case in our islet transplantation model. Such information is critical for us to develop therapies that can enhance islet allograft survival after transplantation.
Materials and Methods
Animals
Male C57BL/6 (H-2b), BALB/c (H-2d), DBA/1 (H-2q), and Rag1-deficient (Rag1-/-) mice at 6–8 weeks of age were purchased from the Jackson Laboratory (Bar Harbor, ME). Foxp3gfp knockin mice at the C57BL/6 background in which expression of Foxp3 can be identified by green fluorescent protein (GFP) were also purchased from the Jackson Lab. All animals were raised under standard conditions. The animal protocol was approved by the Animal Care Committee of the Beth Israel Deaconess Medical Center.
Islet Isolation and Transplantation
The pancreas was perfused with collagenase V at 0.8 mg/ml and digested for 14 min at 37°C. Islets were separated using density gradient centrifugation. Islet purity was assessed by dithizone (Sigma-Aldrich, St. Louis, MO) staining after isolation. An algorithm was used for the calculation of the 150-μm diameter islet equivalent (IEQ) number. Islet cell viability was assessed using fluorescence staining with acridine orange and propidium iodide (18). Our isolation protocol usually yields 90–95% of viable cells before transplantation. Recipients were rendered diabetic using streptozotocin (STZ, 225 mg/kg, IP, Sigma). Five days after STZ administration, mice with two consecutive blood glucose levels exceeding 350 mg/dl were used as recipients. Islets (500–550 IEQ) were transplanted under the kidney capsule of recipients. Blood glucose levels of the recipients were measured twice weekly with a glucometer (Roche, Basel, Switzerland) following transplantation. Recipients with a blood glucose <200 mg/dl were considered normoglycemic. Grafts were deemed rejected when two consecutive blood glucose levels were >300 mg/dl after a period of primary graft function.
Glucose Tolerance Test
Mice that had been fasted overnight were injected with 2 g/kg glucose intraperitoneally. Serum blood glucose levels were measured at 0, 30, 60, and 90 min after glucose injection using a glucometer.
Real-Time RT-PCR Analysis
Total RNA was extracted using Qiagen RNA kit (Qiagen Inc, Charworth, CA). DNase treatment was performed to avoid the contamination by genomic DNA. Realtime RT-PCR was performed to quantify the amount of target gene in each sample at mRNA level using the ABI PRISM® 7700 Sequence Detection Systems as described previously (40).
Bilirubin and Carbon Monoxide (CO) Treatments
Bilirubin (Frontier Scientific, Logan, UT) was dissolved in a 0.2 N sodium hydroxide, subsequently adjusted to a pH of 7.4 with 1 N hydrochloride acid, diluted with 1× PBS to a final volume and stored at −80°C until use. Light exposure was limited as much as possible. Bilirubin at 20 μmol/kg was given to the recipient (IP) twice per day from day -1 until day 13 posttransplantation. CO exposure to the recipient was performed in a chamber containing 250 parts per million (ppm) CO for 1 h per day from day -1 until day 13.
Immunohistochemistry
Islet grafts including a portion of the kidney were harvested and snap frozen in liquid nitrogen for immunohistological staining. Mouse anti-Foxp3 or rabbit anti-GFP antibodies (eBioscience, San Diego, CA) were used to detect the expression of Foxp3 and GFP. Secondary antibody was FITC-labeled anti-mouse or anti-rabbit antibodies (Vector Labs, Burlingame, CA). Costaining with an anti-insulin antibody on the same section was performed to localize the transplanted grafts. Six random areas of interest (400 × 400 pixel area) were selected from each tissue section, total numbers of cells as well as Foxp3- or GFP-positive cells were counted and the percentage of positive cells was calculated.
Depletion of CD4+CD25+ Cells and FACS Analysis
CD4+CD25+ T-cell depletion was carried out in the recipient by using an anti-CD25 antibody given at day -6 and -1 before islet transplantation (250 μg/mouse, IP; Bioexpress Inc., West Lebanon, NH). Depletion of CD4+CD25+ cells was confirmed by FACS analysis at day 1 of transplantation using the anti-CD25-PE antibody. Briefly, single cell suppressions were prepared from splenocytes and lymphocytes of mice treated with the anti-CD25 antibody or nontreated controls. Cells were stained with the anti-CD25-PE antibody (BD Bioscience, San Jose, CA) for 15 min at room temperature. Percentage of CD4+CD25+ cells in whole splenocytes and lymphocytes was analyzed by FACSort with Cell-quest software (BD Bioscience).
Adoptive Transfer and Skin Transplantation
CD4+CD25+ Treg cells (1 × 105) from naive or tolerant mice were cotransferred with CD4+CD25- T effector (Teff) cells from naive mice into Rag1-/- recipients by tail vein injection. Twenty-four hours after adoptive transfer, a full-thickness donor (BALB/c or DBA/1) tail skin (6×8 mm) was transplanted onto the dorsum of the Rag1-/- recipients. The wound was secured with gauze and adhesive strips. Primary function of transplanted graft was evaluated 7 days posttransplantation after removing strips and gauze and monitored daily after until graft rejection.
Statistical Analyses
Kaplan-Meier survival curves were performed using the Statview software and the statistical differences were assessed by the Log-rank test. Values of p < 0.05 were considered significant. Survival data are expressed as mean survival time (MST) ± SD. Differences between cytokine expressions were compared for statistical significance by the Student's t-test.
Results
Administration of Bilirubin and/or CO to the Recipients Prolongs Islet Allograft Survival
To confirm that bilirubin given to the recipient prolongs islet allograft survival following transplantation, BALB/c (H-2d) islets were transplanted into C57BL/6 (H-2b) recipients that had been rendered diabetic by STZ injection as previously described (20). Without any treatment, BALB/c islets were rejected in 19.3 ± 3.4 days when 500–550 IEQs were transplanted (n = 11) (Fig. 1). Administering bilirubin to the recipient (20 μmol/kg, IP, twice per day, from day -1 to day 13) significantly prolonged islet allograft survival, with a mean survival time of 40.6 ±18.3 days (n = 6, p = 0.0024 vs. control). Exposing CO to recipients at 250 ppm for 1 h per day from day -1 until day 13 prolonged islet allograft survival to 30.1 ±8.1 days (n = 5, p = 0.0035 vs. control). In contrast, combining both treatments led to long-term survival of 50% of transplanted islets (n = 6) (Fig. 1). Antigen-specific tolerance was confirmed in recipients bearing long-term surviving grafts as challenged by transplanting a second graft either from a same donor strain (BALB/c) or a third party (DBA/1).
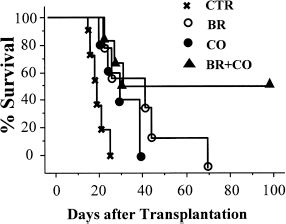
Survival of BALB/c islets in C57BL/6 recipients treated with bilirubin or the combination of bilirubin and CO. BALB/c islets were rejected in 19.3 ± 3.4 days in C57BL/6 recipients that received vehicle alone (X, n = 11). Treatments of recipients with bilirubin (BR, open circles, n = 6) or CO (filled circles, n = 5) significantly prolonged islet allograft survival to 40.6 ± 18.3 and 30.1 ±8.1 days, respectively. Combination of bilirubin and CO treatments led to 50% of grafts surviving >100 days (filled trianges, n = 6, p = 0.007 vs. control).
Recipient Treatment with Bilirubin Improves the Function of Transplanted Grafts
To compare the functional difference of islet allografts transplanted into bilirubin-treated recipients or controls, a glucose tolerance test was performed in recipient mice 6 days following islet transplantation. Glucose at 2 g/kg (IP) was given to recipients that had been fasted overnight and blood glucose levels of those mice were measured 30, 90, and 120 min after glucose challenge. As shown in Figure 2, grafts transplanted into bilirubin-treated recipients responded significantly better to glucose stimulation at 120 min after glucose injection compared to grafts transplanted into vehicle-treated controls, suggesting that bilirubin given to recipients improved function of transplanted islets.

Islet allografts from bilirubin-treated recipients responded better than grafts from vehicle-treated controls. Glucose tolerance tests were performed in C57BL/6 recipients receiving BALB/c islets 6 days after islet transplantation. Glucose at 2 g/kg was given to recipient mice and blood glucose levels were measured at 30, 90, and 120 min after glucose challenge. Mice received bilirubin (solid line) responded much better than controls (dotted line). At least three animals were included in each group. *p < 0.05 as analyzed by Student's t-test.
Expressions of Proinflammatory and Proapoptotic Genes in Transplanted Islets
Donor treatment with bilirubin suppressed expression of proinflammatory and proapoptotic genes at various days following transplantation (39). We evaluated whether recipient treatment with bilirubin would exert similar effects by measuring expressions of tumor necrosis factor-α (TNF-α), inducible nitric oxide synthase (iNOS), and caspase 12 in transplanted grafts harvested at various days following transplantation at mRNA level by realtime RT-PCR. Administering bilirubin to recipients suppressed expressions of TNF-α (Fig. 3A), caspase 12 (Fig. 3B), and iNOS (data not shown) in transplanted islets at 1 and 3 days following transplantation compared to grafts harvested from control recipients. No difference in the expression levels of cytokines was observed at 7 days after transplantation. It thus seems that bilirubin administration to the recipient led to a better function of transplanted grafts associated with decreased inflammation and apoptosis of β-cells.
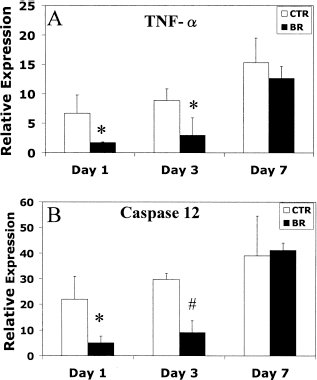
Bilirubin given to recipients suppressed expressions of TNF-α and caspase 12 in transplanted grafts at various days following transplantation. Grafts from bilirubin-treated recipients (black bars) showed much less expressions of TNF-α (A) and caspase 12 (B) at 1 and 3 days following transplantation compared to grafts from vehicle-treated controls (white bars). *p < 0.05 and #p < 0.01 versus control as analyzed by Student's t-test. Values were means of 3–4 grafts per group.
Identification of Treg Cells in the Recipients
To evaluate whether bilirubin administration prolonged islet allograft survival via regulation of Treg cells in the recipient, islet allografts harvested from bilirubin-treated or control recipients at 1, 7, and 10 days following transplantation were stained with anti-Foxp3 as well as anti-insulin antibodies. No Foxp3+ cells were observed in tissue sections from either bilirubin-treated or control recipients at 1 day following transplantation (Fig. 4A and B). In contrast, at later times many more Foxp3+ cells were observed surrounding islet grafts from bilirubin-treated recipients (300 in 1,800 cells counted at day 7, and 150 in 2,000 cells counted at day 10, n = 3) (Fig. 4D and F) compared to those from controls (40 in 1,100 cells counted at day 7, and 80 in 1,500 cells counted at day 10, n = 3 (Fig. 4C and E), indicating that recipient treatment with bilirubin increased the numbers of Treg cells at the site where islets were transplanted.

The presence of Foxp3+Treg cells in islet grafts at 1, 7, and 10 days following transplantation as analyzed by immunohistochemistry. Tissue sections of islet grafts harvested from control or bilirubin-treated recipients at 1, 7, and 10 days following transplantation were stained with anti-foxp3 antibody. Much more Foxp3+ cells (green) were observed surrounding islets (red) from harvested from bilirubin-treated recipients (B, D, and F) compared to those from controls (A, C, and E) at 7 and 10 days following transplantation. Magnification: 200×. Data are representative of three grafts in each group.
Treg Cells Are Essential for the Protective Effects of Bilirubin
To assess whether CD4+CD25+ Treg cells were essential for the survival of islet allografts after being transplanted into bilirubin-treated recipients, we predepleted CD4+CD25+ Treg cells from the recipient by giving them anti-CD25 antibody before islet transplantation. No CD4+CD25+ cells were detected as confirmed by FACS analysis on day 1 posttransplantation, as described in our previous study (20). BALB/c islets were rejected in 20.4 ±3.4 days after being transplanted into C57BL/6 recipients in which CD4+CD25+ Tregs were predepleted (n = 5). Similarly, grafts that had been transplanted into Treg cell-depleted recipients treated with bilirubin were also rejected in 20.1 ±4.1 days (n = 5, p = 0.9 vs. control) (Fig. 5); that is, no prolongation of graft survival was observed, suggesting that the presence of CD4+CD25+ Treg cells was essential for bilirubin to prolong islet allograft survival.

Survival of BALB/c islets in C57BL/6 recipients in which the CD4+CD25+ T cell population was predepleted. CD25+ cells were depleted in the recipients using an anti-CD25 antibody. BALB/c islets (500–600 IEQ) were rejected spontaneously in CD4+CD25+ cell-depleted recipients treated with bilirubin (filled circles, n = 5) or not treated (open circles, n = 5, p = 0.6 vs. control).
Treg Cells Are Sufficient for Tolerance Induction
To evaluate whether Treg cells from tolerant recipients that received both bilirubin and CO treatment were sufficient for tolerance induction, we performed skin transplantation together with adoptive transfer of various cell populations into Rag1-/- recipients. CD4+CD25- Teff cells (1 × 105) from naive mice and CD4+CD25+ Treg cells (1 × 105) from either naive or tolerant mice were coinjected into Rag1-/- recipients. Skin grafts from either an original islet donor strain (BALB/c) or a third-party strain (DBA/1) were transplanted side by side into recipients 24 h after adoptive transfer. Transfer of CD4+CD25- Teff cells alone induced rapid rejection of both DBA/1 and BALB/c skin grafts (11.6 ±0.6 and 15 ± 1.0 days, respectively, n = 3) (Fig. 6). Cotransfer of CD4+CD25+ Treg cells from naive animals slightly prolonged survival of skin grafts: mean survival times were 15.4 ±7.1 days for DBA/1 grafts and 19.1 ±5.1 days for BALB/c grafts (n = 7). In striking contrast, co-transfer of Teff cells from naive and Treg cells from tolerant mice led to long-term survival of all BALB/c grafts while all DBA/1 grafts were rejected promptly (14.1 ±2.9 days, n = 7), indicating that Treg cells from tolerant mice blocked the ability of naive Teff cells to reject skin graft and that Treg cells generated by bilirubin and CO treatment were sufficient for the tolerance induction in the recipients.

Survival of skin grafts in Rag1-/- recipients that received adoptive transfer. CD4+CD25- cells from naive or tolerant mice were adoptively transferred into Rag1-/- recipients. Skin grafts from BALB/c or DBA/1 mice were transferred to Rag1-/- recipients 24 h after adoptive transfer. Mice that received naive Teff cells alone or the combination of naive Teff and Treg cells rejected their grafts promptly while mice that received Teff cells from naive mice and Treg cells from tolerant mice accepted grafts from BALB/c donors permanently but rejected grafts from DBA/1 donors promptly.
De Novo Generation of Treg Cells in Rag1-/- Recipients
To question whether increased Treg cells surrounding islet allografts are generated from Teff cells, we adoptively transferred CD4+GFP- Teff cells (1 × 106) from Foxp3gfp knockin mice into diabetic Rag1-/- recipients 24 h before islet transplantation. Recipients were treated with bilirubin alone, bilirubin plus CO, or vehicle. Presence of CD4+GFP+ Treg cells was evaluated by immunohistochemistry in transplanted grafts 7 days posttransplantation. No GFP+ cells were observed in grafts harvested from control recipients. In contrast, many GFP+ cells were observed surrounding transplanted islets from recipients that had received bilirubin (95/900 cells counted, n = 3) or the combination of bilirubin and CO (165/867 cells counted, n = 3), indicating that recipient treatment with either bilirubin or bilirubin plus CO promoted conversion of Teff cells into Treg cells, which might be responsible for the prolongation and tolerance induction by bilirubin or bilirubin and CO. We also collected total splenocytes and lymphocytes from Rag1-/- recipients treated with bilirubin, bilirubin and CO, or vehicle and analyzed numbers of CD4+GFP+ cells by FACS analysis. The percentage of CD4+GFP+ cells ranged from 0.2% to 1%, with no significant difference among different treatment groups (data not shown).
Discussion
One of the major problems of islet transplantation is the rejection of transplanted grafts by the immune response mounted against the graft by the recipient (8, 33, 36). Immunosuppressive agents used so far have many toxic side effects (2, 9, 10, 19). We and others have shown that induction of HO-1 in the recipient significantly suppresses islet allograft rejection and leads to antigen-specific tolerance to transplanted islets in a mouse model (30, 31, 40). Administration of one product of heme degradation, bilirubin, at least in part accounts for the protective effect of HO-1: bilirubin when given to the recipient, prolonged islet allograft survival in a major mismatch islet transplantation model (20). Most importantly, bilirubin, a molecule present in all cells of the body, appears to have little if any toxicity in humans (except in the immediate neonatal period) at the levels used in our treatments. It thus seems that administration of bilirubin would be useful clinically for islet transplantation. We undertook this study to understand the mechanisms of how bilirubin given to the recipient induces islet allograft survival and tolerance to transplanted grafts.
Native islets are richly supplied with blood but lose their vasculature during islet harvesting (32). Revascularization of transplanted islets that requires several days exposes those islets to hypoxia/reoxygenation injuries (11, 21, 37). Hypoxia/reoxygenation generates ROS that can directly injure islet cells by inducing DNA strand breakage and peroxidation of proteins and lipid, and at the same time activates inflammation and apoptosis (9). Pancreatic β-cells are particularly susceptible to destruction by ROS because they contain low levels and activities of endogenous enzymes protecting against oxygen reduction products (13). Our data show that islet allografts harvested from recipients receiving bilirubin treatment functioned significantly better when compared to grafts collected from recipients receiving vehicle alone. In addition, recipient treatment with bilirubin suppresses expression of proinflammatory and proapoptotic genes in islet allografts at various days following transplantation. We envisage that the antioxidant property of bilirubin contributed to its protective action (i.e., bilirubin treatment suppresses generation of ROS and the consequent production of proinflammatory and proapoptotic genes), thus improving survival and function of transplanted islets. Data from our previous study that in vitro culture of βTC3 cells, an insulinoma cell line, with bilirubin, protected those cells from lipid peroxidation induced by hydroxyl radicals lends support to this view (39).
We demonstrate that CD4+CD25+ Treg cells are required for bilirubin to improve islet allograft survival; that is, bilirubin failed to prolong islet allograft survival when CD4+CD25+ Treg cells were depleted from the recipient prior to transplantation. In addition, we show that CD4+CD25+ Treg cells are sufficient for the tolerance induction of bilirubin and CO treatment to the recipient. Cotransfer of CD4+CD25- Teff cells from naive mice together with CD4+CD25+ Treg cells from tolerant mice into Rag1-/- recipients led to long-term survival of grafts from the original donor strain and rapid rejection of third-party grafts. Thus, Treg cells from tolerant mice blocked the ability of naive Teff cells to reject skin graft in an antigen-specific manner and those Treg cells are sufficient for tolerance induction in our treatment protocol. In this islet transplantation model, administering bilirubin or CO alone to the recipient only prolonged islet allograft survival to periods of less than 100 days, while combining bilirubin and CO treatments led to 50% of islet allografts surviving >100 days (long-term survival). It thus seems that better results could be obtained with dual therapy (bilirubin and CO) used in our islet transplantation model, a phenomena also observed in the heart and kidney transplantation models by another group (25).
Our finding that bilirubin given to the recipient during the peritransplantation period promotes de novo generation of Treg cells is novel and significant. Foxp3+ Treg cells are mainly generated from naive CD4+ T cells in the thymus. Recent studies indicate that Treg cells can be generated from naive CD4+ T cells in the peripheral lymphoid tissues in the presence of TGF-β (5, 15, 17, 27). The majority of naive CD4+ T cells is capable of activating Foxp3 expression and differentiating into Treg cells (22). Whether a cell is induced to express Foxp3 and become a Treg fate is profoundly inhibited by Th1/Th2-polarizing/effector cytokines including IL-12, INF-γ, or IL-4 (41). Most importantly, CD4+CD25+ Treg cells converted from donor CD4+CD25- T cells were shown to be phenotypically and functionally equivalent to naturally occurring Treg cells (6). In our study, bilirubin or the combination of bilirubin and CO given to the recipient promoted de novo generation of Treg cells from Teff cells adoptively transferred into Rag1-/- recipients before islet transplantation. Those treatments might convert the whole body into an environment that favors Treg cell generation. Although unlikely, there is the possibility that the Treg cells we observed in Rag1-/- recipients resulted from proliferation of a few Treg cells that contaminated the Teff cells during FACS sorting (the purity of CD4+GFP- cells we adoptively transferred was approximately 98%). Treatment with bilirubin might favor such proliferation in Rag1-/- recipients. More studies are required to delineate the sources of de novo generated Treg cells and the mechanisms of how bilirubin favors such generation.
The suppressive capacity of Treg cells has been shown to depend on direct cell-cell contact (14). In our study, increased numbers of Treg cells were mainly observed locally surrounding transplanted islet. Even Treg cells generated de novo from Teff cells were only observed around transplanted islets, not in spleen or lymph nodes, suggesting that Treg cells have to be closely localized to β-cells to protect them from being attacked by aggressive T cells.
Administering bilirubin to recipients has potential ad-vantages over other treatment protocols (e.g., costimulatory blockade). Bilirubin can profoundly regulate the immune system through multiple pathways. Bilirubin induces apoptosis of reactive T cells and inhibits activation of immune transcriptional factors and expression of inducible MHC class II (42). In the mean time, bilirubin significantly suppresses activation-induced CD28, B7–1, and B7–2 expression. These capabilities of bilirubin make it an interesting choice for the tolerance induction as compared with other conventional treatment protocols.
In conclusion, the present study demonstrates that bilirubin given to recipients improves both survival and function of transplanted islets. Treg cells are essential and sufficient for the tolerance induction of bilirubin treatment. Bilirubin treatment of mouse recipients promotes de novo generation of Treg cells that may account for the tolerance induction of such treatment.
Footnotes
Acknowledgments
This work was supported in part by JDRF grants 5-2006-911, 5-2007-989, and 1-2007-629 to H.W. and the Julie Henry Fund of the Division of Transplantation, Department of Surgery, BIDMC, Harvard Medical School.
